Abstract
BACKGROUND:
OBJECTIVE:
To define the GI and GU toxicities in prostate cancer to prevent adverse events after treatment.
METHODS:
We searched published studies in PubMed, Cochrane, and Embase databases up to December 31, 2022. The endpoints were the RRs of GI and GU toxicities. Pooled data were assessed using a random-effects model.
RESULTS:
Fifteen eligible studies were included into this analysis. LDR-BT had significantly lower RRs than LDR-BT
CONCLUSION:
The results implied that BT with and without EBRT can result in both GI and GU toxicities in patients with prostate cancer, with LDR-BT leading to a poorer urinary function than EBRT.
Keywords
Introduction
Prostate cancer is malignant tumor worldwide that threatens the health of older men [1]. Currently, transperineal interstitial permanent (Iodine)
BT is a recommended monotherapy for low-malignant prostate cancer and boost for intermediate- and high-risk prostate cancer [5, 6, 7, 8]. The incidence of gastrointestinal (GI) and genitourinary (GU) toxic adverse events (AEs) varies with the choice of treatment. Given that the outcomes of BT, RP, and EBRT are similar, the prognosis accuracy, selection of a treatment regimen, and risks of GI and GU toxicities during and after radiotherapy (RT) are important. GI and GU toxicities from RT options should be considered because of their impact on patients’ health [9, 10]. The concern with BT is its toxicity to nearby organs and tissues, particularly with respect to GI and GU health [11].
An optimal therapeutic outcome would include maximal survival benefits with low GI and GU toxicities. Several clinical studies have indicated that BT, EBRT, and their combined therapy may be associated with a high prevalence of different GI and GU toxicities. However, there is no meta-analysis of recent clinical studies. Therefore, we assessed the risks of GI and GU toxicities from BT, EBRT, and combined-treatment use in men with prostate cancer.
Materials and methods
Literature search
This systematic review and meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement and was registered at PROSPERO (Number: CRD42021249602) [12].
The literature was searched in Cochrane Library, PubMed and Embase databases up to December 31, 2022. The following Mesh keywords were used during the search: “prostatic neoplasms,” “prostatic intraepithelial neoplasia,” “brachytherapy,” “Iodine-125,” “I-125,” and “125I.”
Inclusion and exclusion criteria
The inclusion criteria for clinical studies were as follows: only cohort studies in adult men with localized prostate cancer; no lymph node involvement; no distant metastases; comparisons between
Data extraction
Studies were extracted by two reviewers independently to identify eligible studies. A third reviewer arbitrated any disagreements. These data included year of publication, first author, country of origin, study design, enrollment period, type of the patients, sample sizes, type of interventions, follow-up time, and details of toxic outcomes. The endpoints of GI and GU toxic complications were defined as AEs based on the CTCAE or RTOG. Only events greater than grade 2 were considered.
Quality assessment
The Newcastle-Ottawa scale (NOS) to evaluate the quality of the clinic studies. Quality assessment included measurement of exposure factors, among-group similarity, and patient selection. Any disagreement was resolved by consensus. Studies with NOS scores
Statistical analysis
A pooled estimate of the differences in risk for the single studies was calculated using a random-effect model base on the Mantel-Haenszel method; the results of studies are illustrated using forest plots. Dichotomous parameters were expressed as risk ratios (RRs). All results of studies were presented with a 95% confidence interval (95% CI). Homogeneity within the data of each study was assessed using a chi-square test, setting the degrees of freedom to the number of analyzed studies minus one. Heterogeneity was observed with the Cochran Q test and I
Results
Study selection
We identified 16 213 potentially relevant studies. After removing 4407 duplicate studies, the title and abstract of the remaining 11 806 studies were assessed. Of these, 11 745 studies removed without inclusion criteria. Finally, 15 studies were included in the meta-analysis (Fig. 1): four prospective cohort studies and 11 retrospective cohort studies [16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30].
Study characteristics
Of the 12 773 patients in the included studies, 9405 were treated with LDR-BT, 2468 were treated with LDR-BT
Meta-analysis of GI toxicities between LDR-BT and LDR-BT
EBRT
Acute and late GI toxicity
A meta-analysis of six studies [20, 22, 23, 24, 26, 29] revealed that the RR of acute GI toxic complications from LDR-BT was significantly lower than that from LDR-BT
A meta-analysis of 11 studies [16, 19, 20, 21, 22, 23, 24, 25, 26, 27, 29] revealed that the RR of late GI toxic complications from LDR-BT was significantly lower than that from LDR-BT
Studies characteristics
Studies characteristics
RCS, Retrospective Cohort Study; PCS, Prospective Cohort Study; LDR-BT, Low-dose-rate brachytherapy; HDR-BT, High-dose-rate brachytherapy; 2D-CRT, 2-dimensional conformal radiation therapy; 3D-CRT, 3-dimensional conformal radiation therapy; IMRT, Intensity-modulated radiation therapy; VMAT, Volume-modulated arc therapy; CTCAE, Common Terminology Criteria for Adverse Events; RTOG, Radiation Therapy Oncology Group grading; GI toxicity, Gastrointestinal toxicity; GU toxicity, Genitourinary toxicity; NA, Not Available.
Details of study quality evaluation via the Newcastle-Ottawa Scale
Study selection. Flow diagram summarising selection of studies that meet inclusion criteria.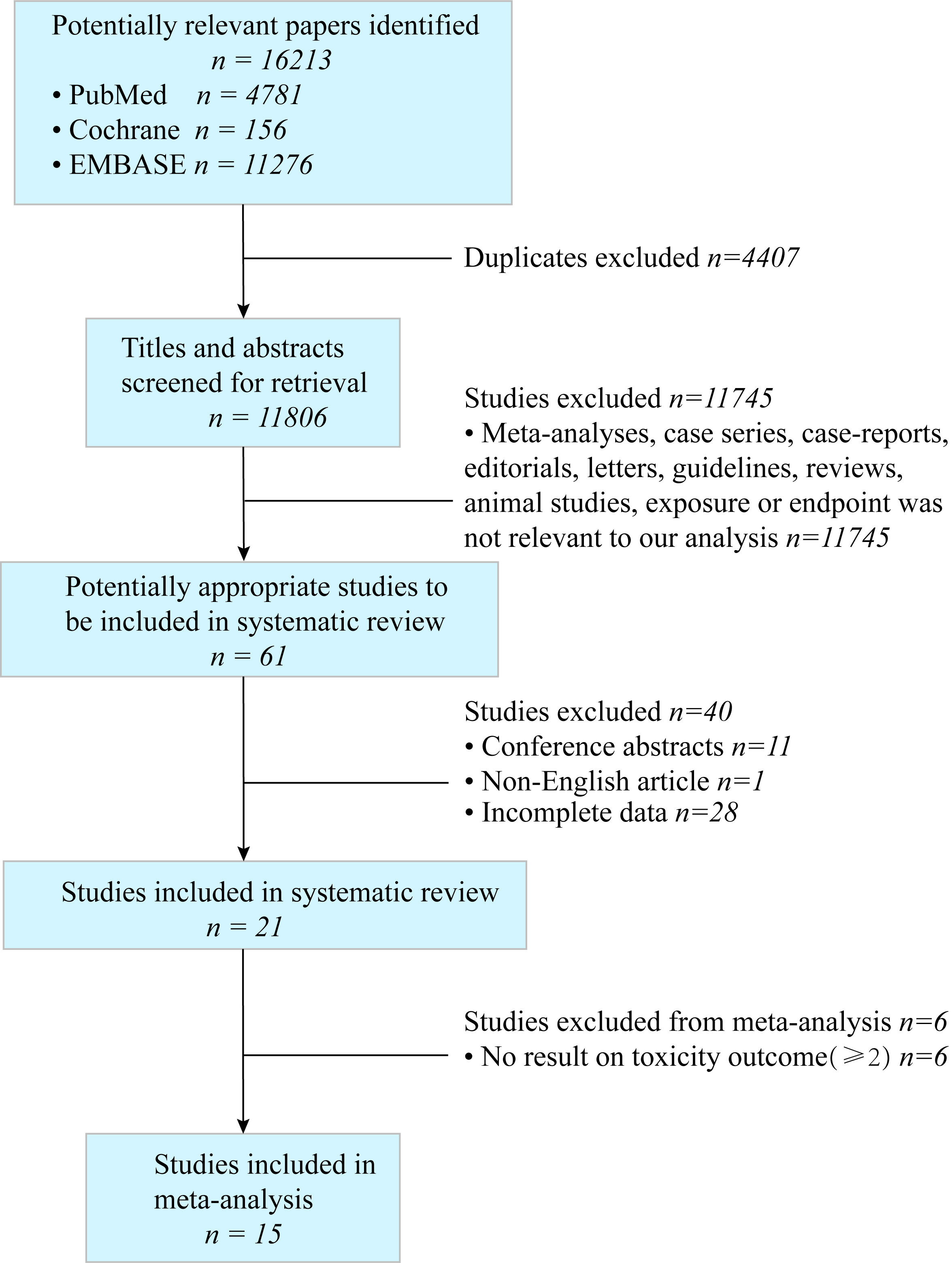
(a) Forest plot of RR for acute GI toxicity following LDR-BT 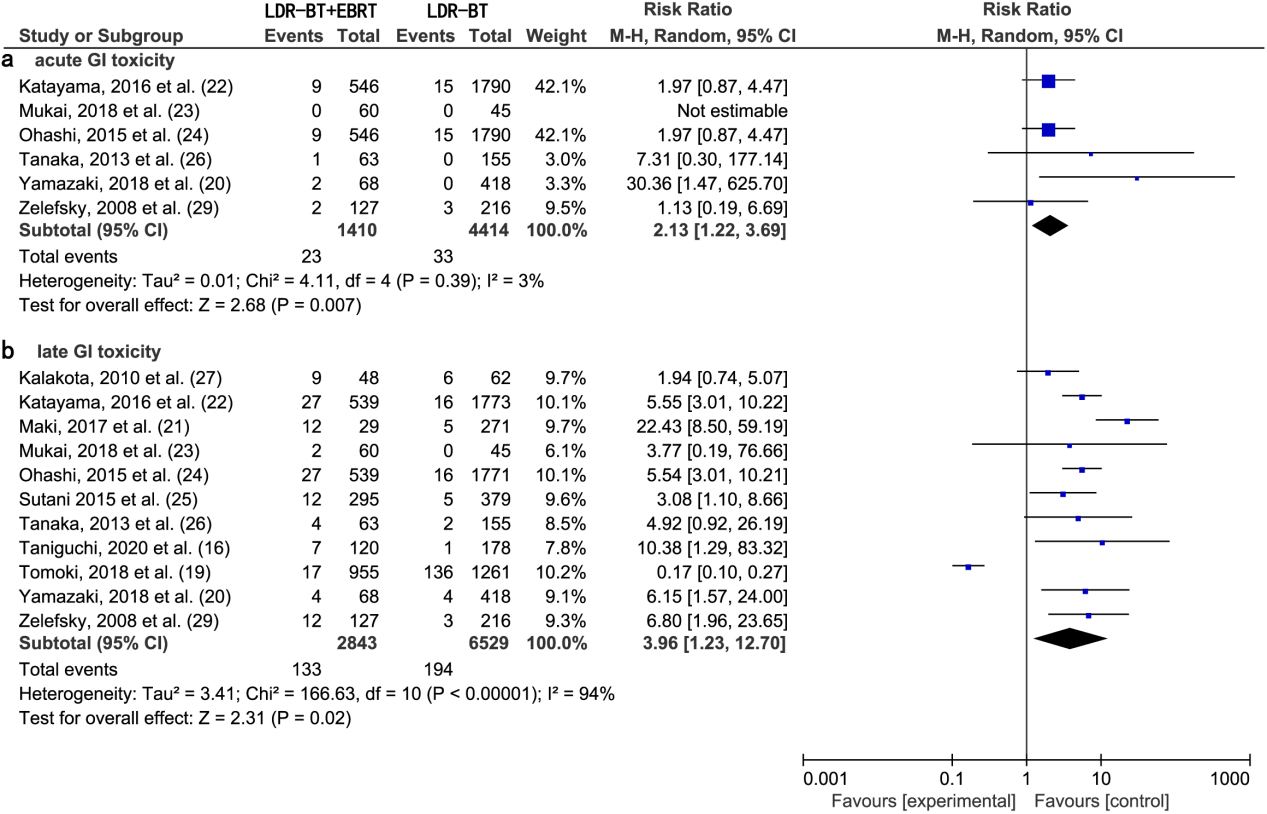
(a) Forest plot of RR for acute GU toxicity following LDR-BT 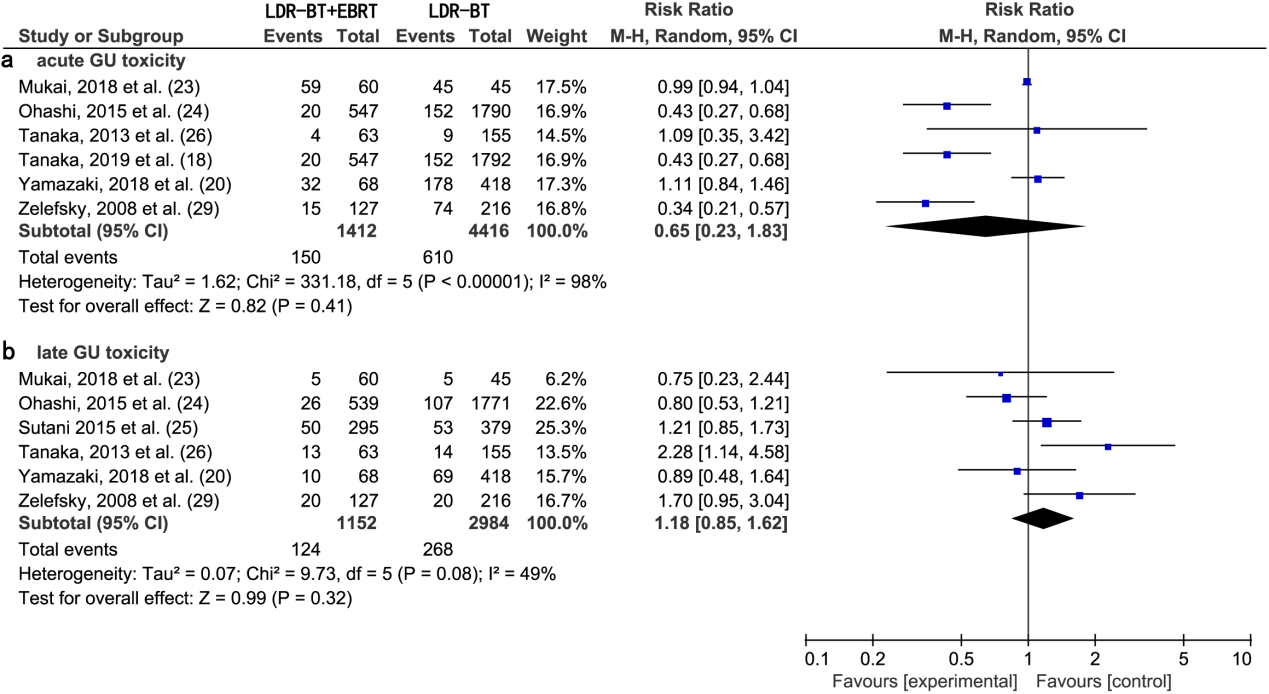
Acute and late GU toxicity
A meta-analysis of six studies [18, 20, 23, 24, 26, 29] revealed that there was no significant difference in the RR of acute GU toxic complications between LDR-BT and LDR-BT
A meta-analysis of six studies [20, 23, 24, 25, 26, 29] revealed that there was no significant difference in the RR of late GU toxic complications between LDR-BT and LDR-BT
Heterogeneity analysis of LDR-BT and LDR-BT
EBRT studies
Six studies reporting acute GI toxicity did not present heterogeneity (
Sensitivity analysis (acute GI toxicity)
Sensitivity analysis (acute GI toxicity)
Sensitivity analysis (late GI toxicity)
Acute and late GI toxicity
A meta-analysis of two studies [28, 30] revealed that there was no significant difference in the RR of acute GI toxic complications between LDR-BT and EBRT alone (0.75; 95% CI, 0.21–2.74;
(a) Forest plot of RR for acute GI toxicity following LDR-BT and EBRT. (b) Forest plot of RR for late GI toxicity following LDR-BT and EBRT.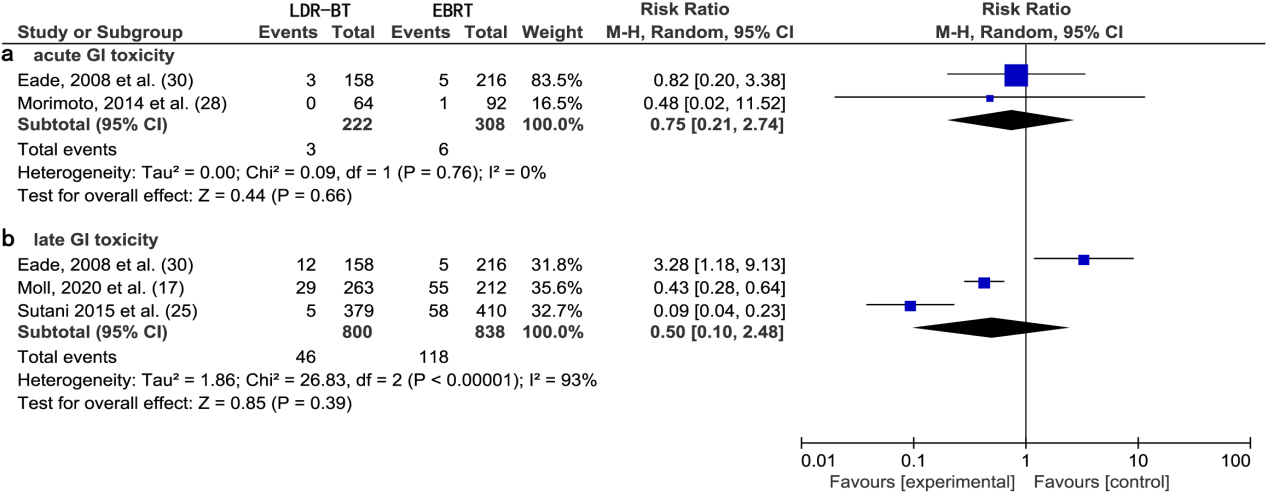
(a) Forest plot of RR for acute GU toxicity following LDR-BT and EBRT. (b) Forest plot of RR for late GU toxicity following LDR-BT and EBRT.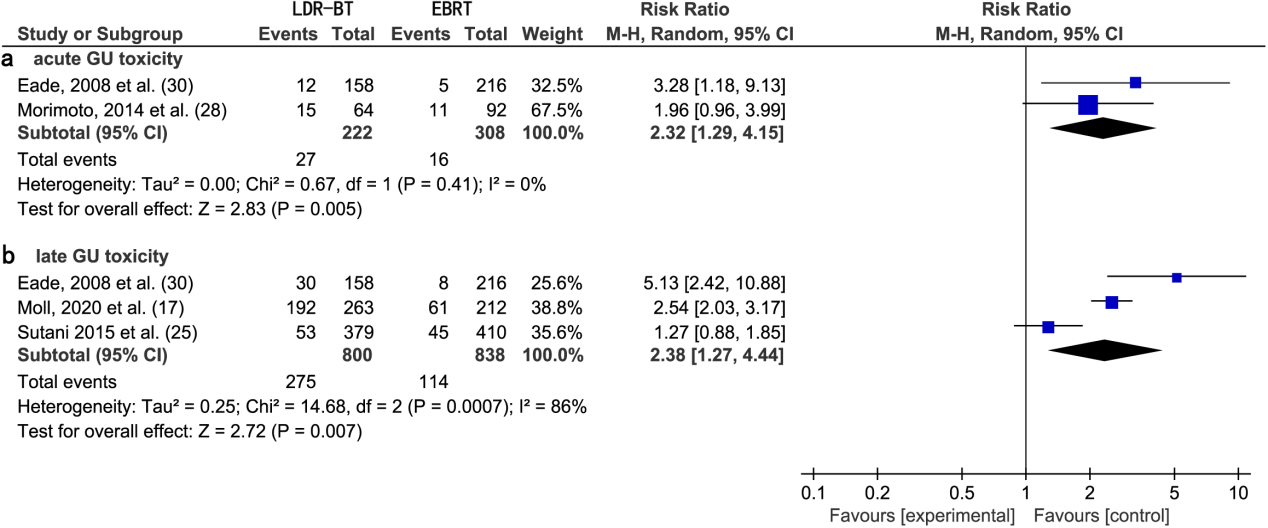
A meta-analysis of two studies [28, 30] revealed that the RR of acute GU toxic complications from EBRT was significantly lower than that from LDR-BT alone (2.32; 95% CI, 1.29–4.15;
Meta-analysis of GI and GU toxicities between HDR-BT and LDR-BT
Acute GI toxicity and GU toxicity
A meta-analysis of two studies [20, 28] revealed no significant difference in the RR of acute GI toxic complications between HDR-BT and LDR-BT alone (4.14; 95% CI, 0.84–20.40;
(a) Forest plot of RR for acute GI toxicity following HDR-BT and LDR-BT. (b) Forest plot of RR for acute GU toxicity following HDR-BT and LDR-B.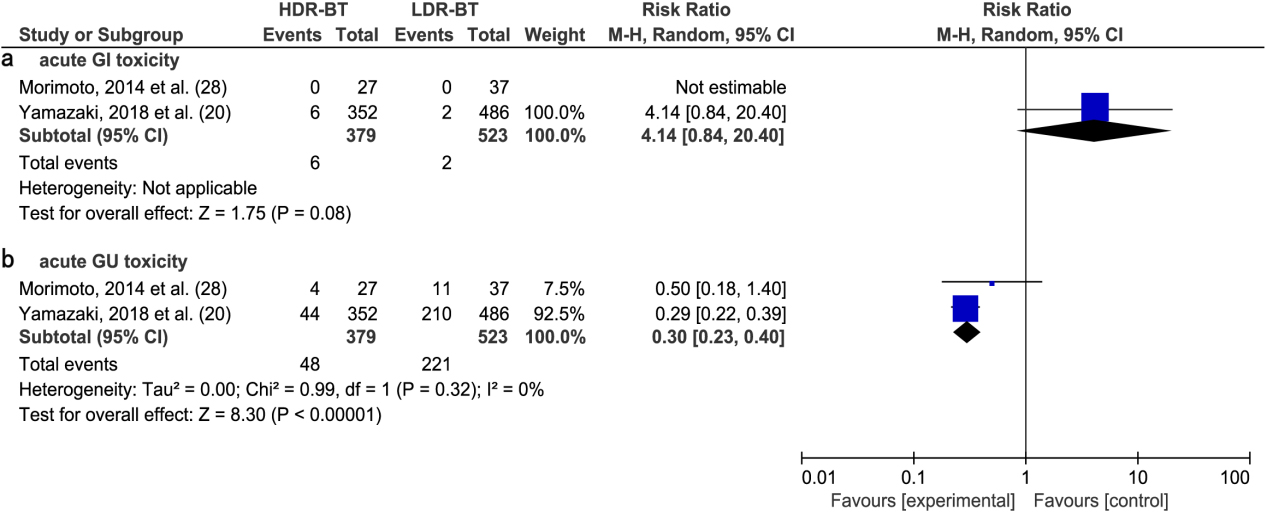
With the results of this meta-analysis, we evaluated the significance of RRs of GI toxicities and GU toxicities in studies by comparing the following therapies: (A) LDR-BT and LDR-BT
The varied RT doses in studies must be considered when comparing the RRs of GI and GU toxicities between different therapies [31]. BT
Testosterone levels may decline as a result of hormone therapy (HT) on urinary toxicity [42, 43]. HT itself may affect urinary symptoms: this must be considered when comparing both the tumor outcome and toxicity of BT and BT
Large, population-based cancer patient cohort studies show that BT
Previous studies reported a 15 years disease-free survival rate of 80.4% after treatment of men with localized prostate cancer with BT [57, 58]. Another studies reported a 5 years and 10 years OS rate of 94% and 84%, respectively, among similar patients [59]. These findings confirm the long-term efficacy of BT treatment for localized prostate cancer. Kee et al. [60]. Published a meta-analysis of clinical randomized control trials, which revealed a significantly higher 5 years biochemical progression-free survival with
The literature search and screen of this meta-analysis were strict, the included studies were comprehensive, and outcomes were highly credible. However, our analysis has several limitations: (A) the total number of included studies was small (
Conclusions
Our findings implied that BT with and without EBRT can result in both acute and late GI toxic and GU toxic complications in men with localized prostate cancer, with LDR-BT leading to a poorer urinary function than EBRT. The results of this study reveal the need to prevent GI and GU toxic complications after multiple forms of radiotherapy in the future. Prospective clinical studies are needed to verify and expand on our results. When making decisions for treating local prostate cancer, clinicians should balance the effectiveness of different radiotherapies with their safety depends on the actual clinical characteristics.
Footnotes
Acknowledgments
This research was supported by the Natural Science Foundation of Fujian Province (2022J011393).
Conflict of interest
None to report.
