Abstract
BACKGROUND:
Hypertension is a well-recognized risk factor for cardiovascular, which is also a critical factor in causing myocardial fibrosis (MF).
OBJECTIVE:
The study aimed to explore the effect of Atractylenolide II (ATL-II) on MF and oxidative stress in spontaneous hypertension rats (SHR).
METHODS:
The body weight of rats after injection of ATL-II was quantitatively analyzed. The left ventricular function of SHR was evaluated by Echocardiographic. HE staining, Masson trichrome staining, left ventricular mass index (LVMI) and immunofluorescence was applied to investigate the effects of ATL-II on MF. RT qPCR was used to detect the Collagen I,
RESULTS:
ATL-II could dose-dependently improve the BW of SHRs (
CONCLUSION:
ATL-II could exert an inhibiting effect on MF and oxidative stress in SHRs. Hence, ATL-II may hold promise for the treatment of MF and oxidative stress in Spontaneous Hypertension.
Introduction
Hypertension is a well-recognized risk factor for cardiovascular, which is also a critical factor in causing MF [1]. MF, a serious adverse effect of hypertension on the heart, is a common pathological damage caused by various cardiovascular diseases [2, 3]. Especially, MF is irreversible, which becomes a difficult problem in clinical treatment [4].
MF is the result of excessive deposition of Collagen fibers in the extracellular matrix, which causes heart failure and ventricular hypertrophy [5, 6, 7]. MF markers include collagen I, fibronectin,
Oxidative stress is the result of the imbalance between reactive oxygen species (ROS) production and reduced antioxidant capacity in the organism [11]. Oxidative stress plays a critical role in the pathophysiology of cardiovascular diseases, such as hypertension, MF, and cardiomyocyte apoptosis [12]. Oxidative stress causes excessive production of reactive oxygen species (ROS), which is an important event in the development of cardiovascular diseases. Meanwhile, previous studies have found hypertension had a relationship with oxidative stress, oxidative stress could raise blood pressure [13]. Besides, some studies confirmed MF induced by hypertension may be closely related to the decrease of antioxidant enzymes [14, 15].
Atractylodes macrocephala (AM) is an important traditional herbal medicine in Asia, which is widely used to treat gastrointestinal, cardiovascular, and immune system diseases [16]. Many natural compounds were extracted from Atractylodes macrocephala, such as polysaccharides, sesquiterpenoids, and volatile oils. These extracts have various pharmacological effects, such as anti-tumor, anti-inflammatory, and regulating the digestive system [17]. Existing researches indicate that AT-II which is a major sesquiterpenoid extracted from AM exhibits anti-hyperglycemia anti-cancer and anti-oxidant activities [18, 19, 20]. However, few studies paid much attention to MF caused by hypertension.
Hence, the purpose of the research is to explore the efficiency of ATL-II on MF and oxidative stress in SHRs, which would provide valid data for the application of ATL-II to treat myocardial diseases associated with MF and oxidative stress in the future.
Methods
Animals and treatments
Spontaneous hypertension rats (SHRs) and Wistar Kyoto rats (WKYs) (male, 200–250 g, 6 weeks old) were purchased from the Model Animal Research Center of Nanjing University, China (SCXK2012-0005). The rats were randomly divided into 5 groups (
LVMI assay
The fed rats should be weighed in body weight (BW) every week. After 6 week’s treatment, the rats were sacrificed and the hearts were collected. Hearts and left ventricles were weighed, respectively. LVMI was obtained by calculating the ratio of BW to left ventricular mass (LVM).
Echocardiography assessment
After ATL-II injection for 6 weeks, the rats’ cardiac function was measured by an echocardiography system (EPIQ 7C, Jiangsu, China). The rats were anesthetized with 2% sodium pentobarbital, and their chest hairs were removed. In the M-mode curve, the relevant indexes were recorded: left ventricular end-systolic diameter (LVESD), left ventricular end-diastolic diameter (LVEDD), left ventricular end-systolic volume (LVSV), left ventricular end-diastolic volume (LVDV), left ventricular ejection fraction (LVEF) and left ventricular fractional shortening (LVFS). LVEF and LVFS were used as parameters to indicate cardiac function [23].
HE and Masson staining
Myocardial tissue samples were fixed with 4% formaldehyde solution (Aladdin, Shanghai, China), embedded in paraffin, and then cut down into slices. After deparaffinized, HE and Masson’s staining were performed to evaluate cardiac fibrosis following the manufacturer’s guidance of the HE and Masson staining kit (Jiancheng Bioengineering Institute, Nanjing, China). Lastly, the slices were inspected under a light microscope (SteREO Discovery.V20, OPTON, Germany).
Immunofluorescence assay
Cardiomyocytes from rats were fixed in 4% paraformaldehyde (Aladdin, Shanghai, China) at 25
Immunohistochemistry assay
The samples were digested with enzymes and dehydrated with alcohol. Then the samples were treated with rabbit polyclonal antibodies against
RT-qPCR assay
The TRIzol reagent (Invitrogen, Hudson, CA, USA) was applied to extract the total RNA of myocardial tissues. Then cDNA was synthesized using the RT-PCR kit (Invitrogen, Hudson, CA, USA). Moreover, RT-qPCR was performed with a CellsDirect
Western blot assay
The total protein of myocardial tissues was extracted with RIPA lysate (Beyotime, Shanghai, China), and total protein concentration was measured using the BCA protein assay kit (Beyotime, Shanghai, China). Protein samples were subjected to SDS/PAGE and then transferred onto polyvinylidene difluoride (PVDF) membranes (Millipore, MA, USA). Subsequently, the membranes were incubated for 1 hour in a blocking buffer with 5% bovine serum albumin. Moreover, the membranes were incubated with primary antibodies against Vimentin (Invitrogen, Hudson, CA, USA, 1:1000 dilution), Fibronectin (Invitrogen, Hudson, CA, USA, 1:100 dilution),
TUNEL staining
A one-step TUNEL Apoptosis Assay Kit (Beyotime, Shanghai, China) was applied to measure cardiomyocyte apoptosis according to the manufacturer’s instructions. Cardiomyocytes were fixed in 4% paraformaldehyde at room temperature for 15 minutes and then permeabilized with 0.01% Triton X-100 for 10 minutes at room temperature. Then Cardiomyocytes were incubated with TUNEL mix for 1 hour at 37
H
O
, GSH-PX, SOD, and MDA determination
After 10% chloral hydrate (Aladdin, Shanghai, China) anesthesia, rats’ blood was obtained from the abdominal aorta. Then blood was centrifuged at 3000 rpm for 15 min, the serum was separated and cryopreserved at
Data analysis
Statistical analysis was performed by SPSS 17.0 software (SPSS Inc., Chicago, USA). All experiments were repeated three times, and the data were presented as mean
Results
The effect of ATL-II on cardiac function in SHRs
The rats BW were weighed to verify whether ATL-II affected the BW of SHRs. As Fig. 1A shows that rats BW of the ML group was the lowest (
ATL-II enhances cardiac function in SHRs. (A) Quantitative analysis of rats BW after ATL-II injection. (B) Cardiac function was assessed by echocardiography in rats after ATL-II injection for 6 weeks. All data were presented by means 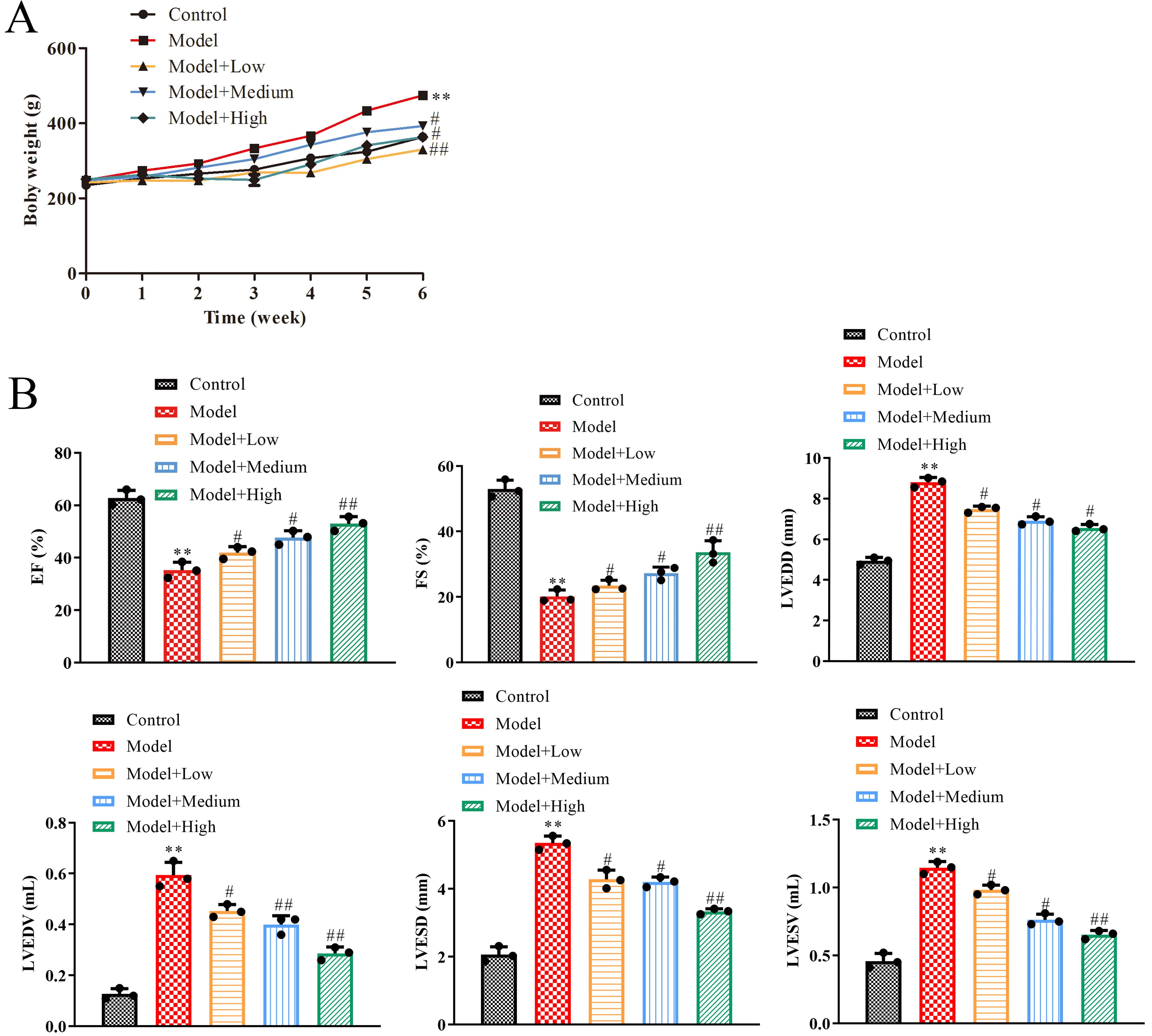
Moreover, cardiac function was assessed by echocardiography in rats after different dosages of ATL-II injection for 6 weeks. The results indicated that LVEDD, LVESD, LVEDV, and LVESV were significantly increased in the M group, and EF and FS were significantly decreased in the M group (
ATL-II attenuates MF in SHRs. (A) Representative images of HE Staining in myocardial sections. (B) Representative image of Masson staining in myocardial sections. (C) Representative images of 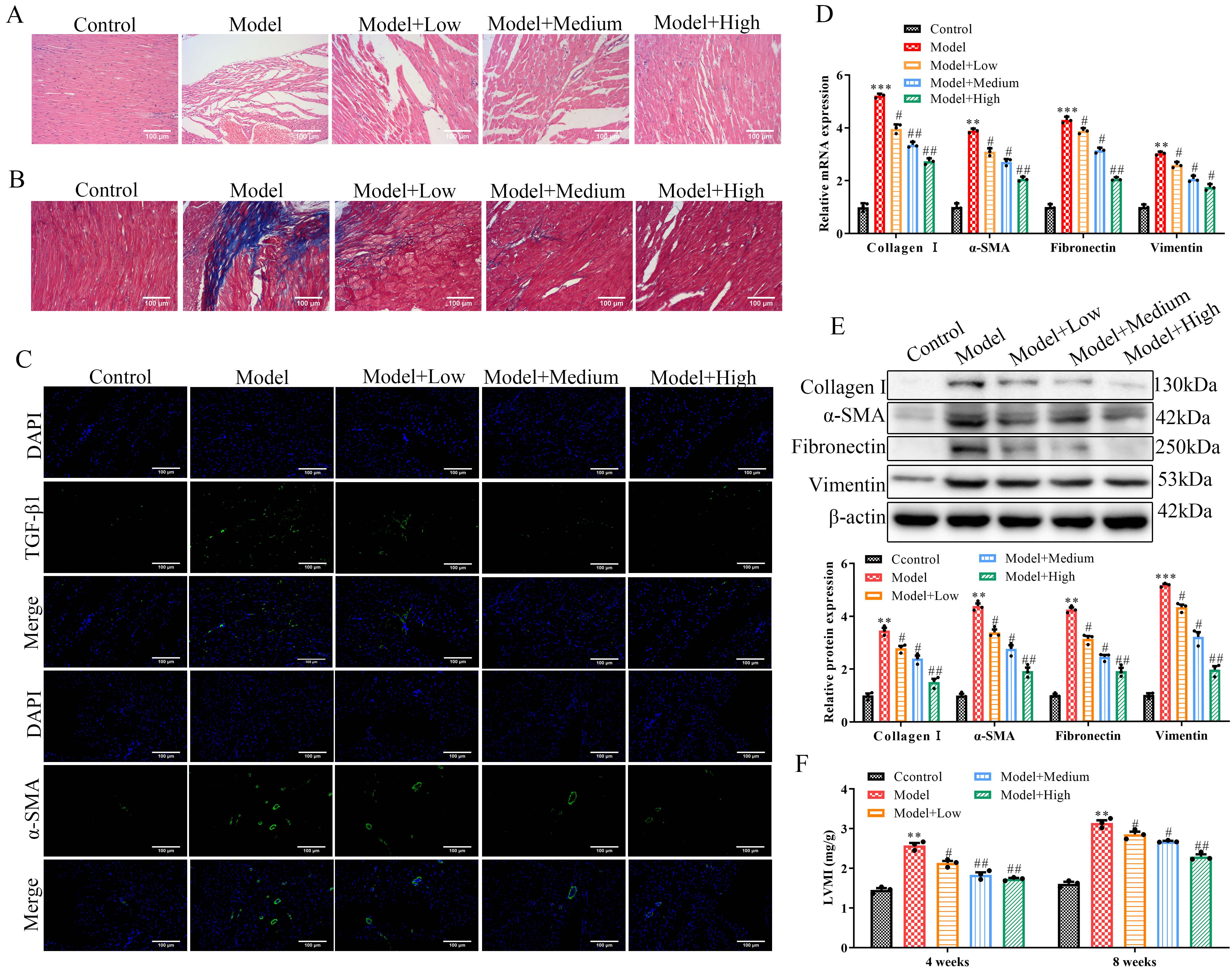
To clarify the effect of ATL-II on MF in the SHRs, relevant indexes were measured. As shown in Fig. 2A, the results of HE staining revealed that obvious myocardial tissue damage was observed in the M group compared to the C group (
Then, Masson staining was applied to observe the myocardial sections, which showed that large amounts of collagen deposition and disorder arrangement of myocardial fibers were observed in the M group compared to the C group (
Moreover, immunofluorescence assays for
Besides, RTqPCR and western blot assays showed that expression levels of Collagen I,
In addition, LVMI was calculated when ATL-II treatment was for 4 and 8 weeks. The results showed that compared with the C group, the LVMI of the M group was significantly improved (
The effects of ATL-II on cardiomyocyte apoptosis in SHRs
The results of TUNEL staining indicated that cardiomyocyte apoptosis was obviously inspected in the M group. The apoptosis rate of the M group significantly exceeded the C group, which showed significant differences (
Moreover, the results of RTqPCR presented that expression levels of Collagen I,
As Fig. 3C shows that few
ATL-II inhibits cardiomyocyte apoptosis in SHRs. (A) Representative image and quantitative analysis of TUNEL staining in myocardial sections. (B) RTqPCR for Collagen I, 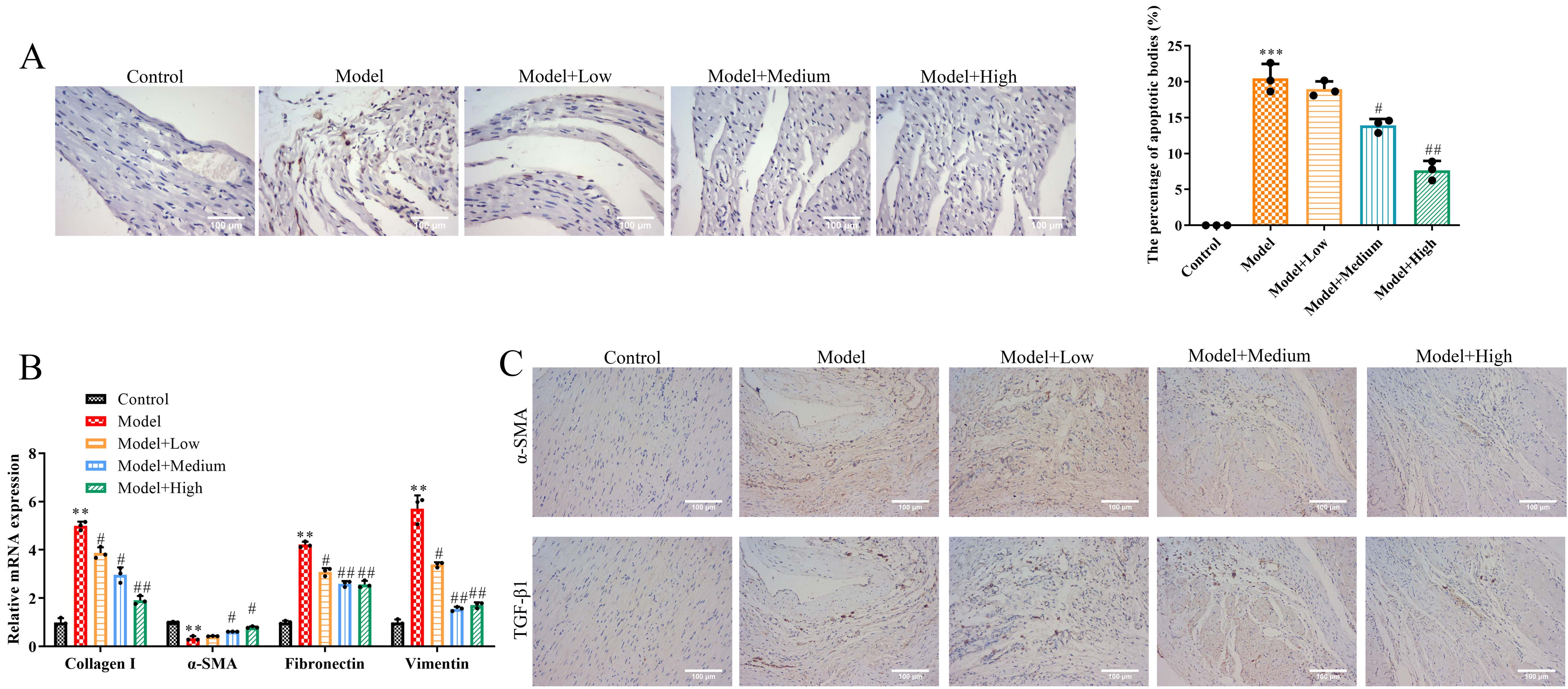
ATL-II alleviates oxidative stress in SHRs. (A) Quantitative analysis of GSH-PX, H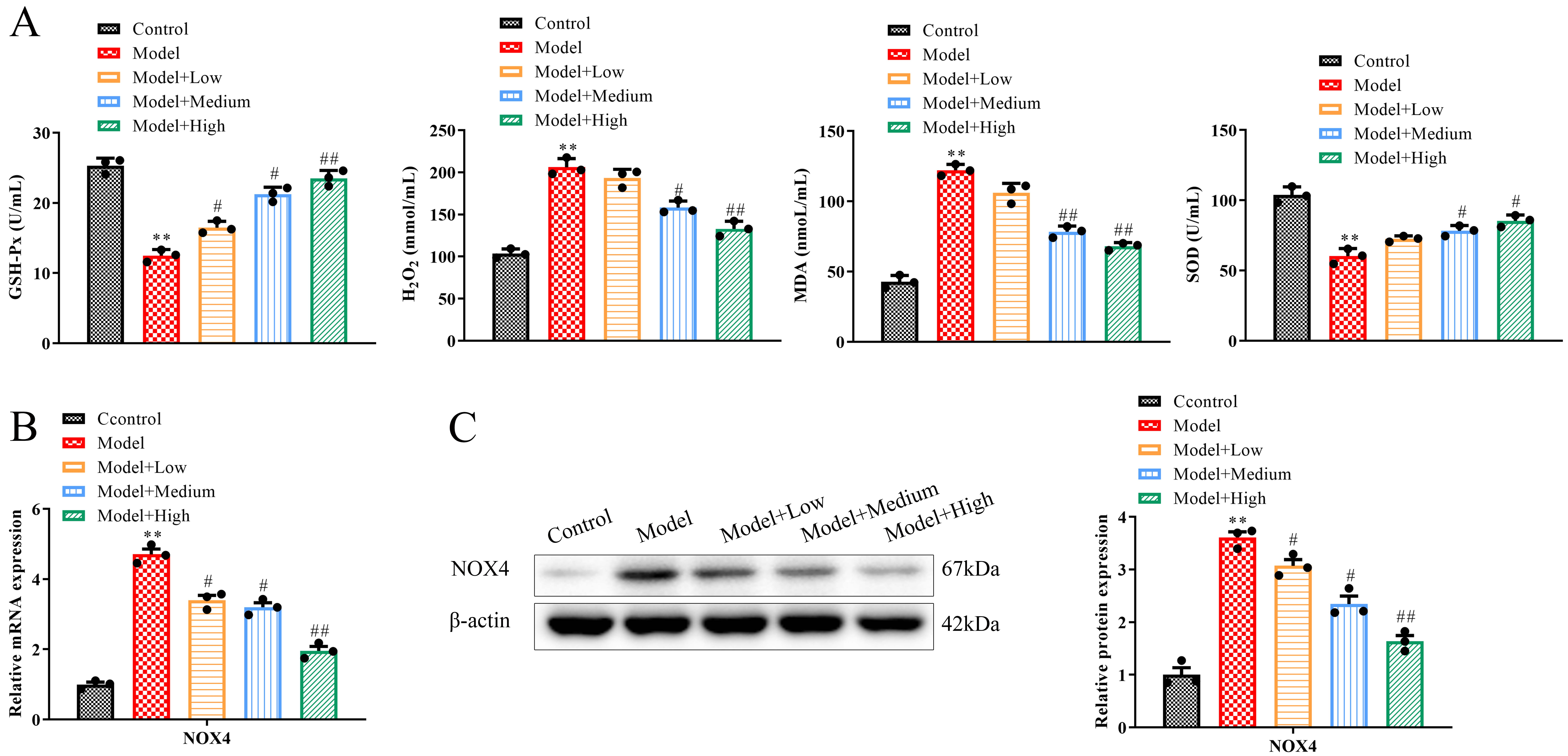
Activities of GSH-PX and SOD and contents of H
In addition, the mRNA expression level of NOX4 was up-regulated in the M group compared to the C group (
Discussion
Numerous studies showed that approximately 30% of the population died from cardiovascular diseases around the world, and more than half of the cardiovascular disease cases were caused by hypertension. Previous studies reported that MF was one of the key pathological features of myocardial remodeling in hypertensive. Meanwhile, MF also was an important reason for diastolic dysfunction, myocardial ischemia, and sudden cardiac death induced by hypertension. Furthermore, MF induced by hypertension was closely related to the decrease of antioxidant enzymes. In addition, Oxidative stress could increase peripheral vascular resistance and blood pressure.
In our study, M-mode echocardiography was carried out to assess myocardial function. Previous studies reported that MF caused by hypertension possessed a seriously bad effect on myocardial function Our research showed that EF and FS were increased after ATL-II treatment. ATL-II could improve myocardial function in SHRs.
Moreover, myocardial apoptosis and myocardial fibrosis were detected to assess the effect of ATL-II on MF in SHRs. Cardiac Fibroblasts (CFB) could differentiate as myofibroblasts (MFB), and then MFB participated in MF formation. In addition, some factors played a key role in the proliferation and differentiation of MF [24]. Collagen I,
Oxidative stress was defined as ROS increase and antioxidant activity reduction of vascular [29, 30, 31]. ROS comprised superoxide anion (O2), hydroxyl radical (OH), and hydrogen peroxide (H
However, we investigated the effects of ATL-II on myocardial fibrosis and oxidative stress in spontaneously hypertensive rats only at the histological level in animals. This is limited and challenging for the study of ATL-II function. Further studies investigating the safety of ATL-II at the cellular level and the molecular mechanisms underlying the effects on myocardial fibrosis and oxidative stress are also imperative. In addition, it has been recently reported that the mechanism by which hydrogen-rich saline (HRS) [39] and Asiatic acid (AA) [28] inhibit spontaneous hypertension and reduce myocardial fibrosis is also significantly related to the reduction of oxidative stress, and the mechanism may be achieved by inhibiting TGF-
Conclusion
In conclusion, our study proved that ATL-II could alleviate MF and myocardial apoptosis in SHRs. Furthermore, our research also found ATL-II could protect cardiomyocytes against oxidative stress by enhancing the antioxidant activities and lowering the oxidation products, which more clearly and comprehensively pointed out the significant role of oxidative stress on MF than previous literature. We speculated that ATL-II alleviated MF by reducing oxidative stress in SHRs, but the potential molecular mechanism is still unclear. Hence, ATL-II could be a promising treatment for MF and oxidative stress caused by Spontaneous Hypertension.
Ethics statement
All animal experiments were approved by the Institutional Animal Care and Use Committee of the Nanjing University of Chinese Medicine, Jiangsu, China (IRB: XMLL-2021-880).
Funding
This work was supported by Jiangsu Province Traditional Chinese Medicine Technology Development Plan Project (No. YB201968).
Availability of data and material
All data generated or analyzed during this study are included in this published article.
Author contributions
XS conceived and designed the research, obtained financing, and revised the manuscript for intellect content. LW and JS acquired and analyzed the data. JK performed the statistical analysis. RP wrote the manuscript. All authors read and approved the final manuscript.
Footnotes
Acknowledgments
Not applicable.
Conflict of interest
The authors declare that they have no conflicts of interest.
