Abstract
BACKGROUND:
Pulse-inversion-based tissue harmonic imaging has been utilized for many years because it can effectively eliminate the harmonic leakage and produce low side-lobe. However, the pulse inversion method is sensitive to imaging object movements, which may result in motion artifacts. Spatial resolution and contrast were limited.
OBJECTIVE:
To improve ultrasound image quality by a new pulse-inversion-based tissue harmonic imaging technique.
METHODS:
Continuous wavelet transform is applied to investigate the correlation between mother wavelet and the received echoes from two opposite pulses. To get a better correlation, a novel mother wavelet named ‘tissue wavelet’ is designed based on the Khokhlov-Zabolotskaya- Kuznetsov (KZK) wave equation. Radio frequency data were obtained from open Ultrasonix SonixTouch imaging system. Experiments were carried on ultrasonic tissue phantom, human carotid artery and human liver.
RESULTS:
The average improvement of lateral spatial resolution is 49.52% compared to pulse-inversion-based tissue second-harmonic Imaging (PIHI). Contrast ratio (CR) and contrast-to-noise ratio (CNR) increased by 5.55 dB and 1.40 dB over PIHI. Tissue wavelet performs better than Mexh and Morl wavelet in lateral spatial resolution, CR, and CNR.
CONCLUSION:
The proposed technique effectively improves the imaging quality in lateral spatial resolution, CR, and CNR.
Keywords
Introduction
Correlative techniques on tissue harmonic imaging
Medical ultrasound diagnosis has become an effective technique in clinical medical diagnosis because of its non-destructive, simple and economical advantages [1, 2, 3]. In a variety of imaging methods, tissue harmonic imaging (THI) has been investigated and utilized for many years [4, 5, 6, 7]. THI technique, which uses the characteristics of nonlinear echo components produced by ultrasonic propagation in biological tissues, can effectively improve the spatial resolution of the image, reduce artifacts, and improve the quality of deep tissue imaging. However, because the attenuation and the square of the frequency are in a direct relationship, the harmonic signal is usually weak. At the same time, background noise, harmonic leakage and echo signal aliasing caused by narrow-band ultrasonic transmission, lead to low spatial resolution and contrast. The application of tissue harmonic imaging in clinical diagnosis has been limited as a result.
Several techniques have been studied for the improvement of tissue ultrasound imaging quality. Conventional THI must consider the compromise between detection sensitivity and spatial resolution. The problem can be effectively solved through the proposition of pulse inversion. Pulse inversion technique can eliminate fundamental and other odd harmonics and retain even harmonics by transmitting two opposite pulses [8, 9]. The pulse inversion method can effectively eliminate the harmonic leakage and produce low side lobe. However, the multi-pulse excitation makes the imaging frame rate lower. At the same time, the pulse inversion method is sensitive to imaging object movement and probably causes motion artifacts.
In recent years, coded emission technique has been widely used in the detection of tissue ultrasound imaging. Coded emission technique can improve the emission energy and improve the signal-to-noise ratio (SNR) at a lower amplitude of transmitting sound pressure. The Che-chou Shen research group had studied varied Golay decoding methods and testified the effectiveness of SNR improvement [10, 11, 12]. Chirp coded excitation is a valid technique which can effectively improve the ultrasound penetration depth and imaging quality [13, 14, 15, 16]. Coded emission technique can effectively improve the imaging SNR, but the ultrasonic energy is limited because of the consideration of ultrasonic safety. At the same time, incomplete pulse compression leads to serious side lobes. Besides, coded emission technique requires complex excitation and decoding equipment, which brings difficulties to the engineering implementation.
Compared with the second harmonic imaging, the higher harmonic imaging has drawn much attention. Higher harmonic imaging has superior spatial resolution and is proved to improve the ultrasound image quality effectively [17, 18, 19]. However, higher harmonic imaging method requires advanced equipment and higher hardware costs. As a result, much researches had focused on the design of higher-order harmonic ultrasound transducers [20, 21].
Wavelet transform technology is a widely used time-frequency analysis technology [22]. It has characteristics of multi-spatial resolution analysis, so has important scientific significance and application value in e.g. EEG analysis, speech detection and computer vision [23, 24, 25, 26, 27]. In our previous work, we applied the continuous wavelet transform (CWT) to the field of ultrasound contrast imaging. The contrast to tissue ratio is increased significantly [28].
In this study, we combined the CWT process with pulse-inversion-based tissue ultrasound imaging technique. In addition, we designed a new mother wavelet named ‘tissue wavelet’ based on the Khokhlov-Zabolotskaya-Kuznetsov (KZK) wave equation, for the purpose of obtaining better correlations between mother wavelet and surrounding tissues. In order to verify the effectiveness of the proposed method, in-vitro experiments were done with an ultrasonic tissue phantom, whereas in-vivo experiments were done on human carotid artery and human liver, respectively. The results were compared with conventional pulse inversion harmonic imaging under the same experimental conditions. In addition, the effectiveness of tissue wavelet was studied compared with the other two commonly used mother wavelets. Results in B mode were also discussed as a comparison.
Contributions
Contributions of this paper to the ultrasound imaging field mainly focus on the following three aspects:
In this study, a new pulse-inversion-based tissue ultrasound imaging technique was proposed. CWT was applied to investigate the correlation between mother wavelet and the received ultrasonic echoes. For the purpose of getting a better correlation, a novel mother wavelet named tissue wavelet is designed based on the KZK wave equation. An adaptive imaging algorithm is proposed since tissue wavelet constructed did not satisfy the wavelet admissible condition.
Basic principle of the imaging algorithm
In this part, the basic principle of the proposed imaging technique is demonstrated concisely. In conventional pulse-inversion technique, two ultrasonic echoes received from opposite pulses are summed. We analyze the echoes from one single scan line. The equation can be written as follows:
Where
The formula of CWT is as follows:
with
where
CWT could be regarded as the correlation analysis of the original signal and a set of functions produced by the mother wavelet. This means that the size of transformed wavelet coefficients represents the degree of correlation. The more similar the mother wavelet and original signal are, the larger the transformed wavelet coefficient is. The larger transformed wavelet coefficient represents a stronger correlation between the original signal and the mother wavelet. This imply that the correlation could be enhanced by applying more suitable mother wavelets.
In this study, we utilize CWT to process the total ultrasonic echo signals at receive. Equation (1) can be written as follows:
Using the linear properties of CWT, we can write:
It can be seen from Eq. (5) that
In this paper, we proposed a new Pulse-Inversion based Wavelet analysis Imaging (PIWI) method in tissue ultrasound imaging field. Figure 1 demonstrate the flowchart of the imaging algorithm.
Flowchart of the proposed pulse-inversion based wavelet analysis imaging (PIWI) algorithm.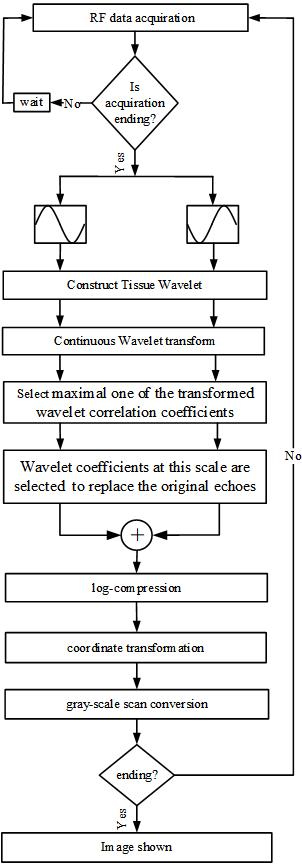
The imaging strategy starts with emitting a pair of pulses, which have the same amplitude and opposite phase. Then the corresponding echo signals were received. Radio frequency (RF) data can be acquired through an open ultrasonic platform.
Post-processing of the imaging algorithm includes five steps:
Select a suitable mother wavelet. The selection principle is that the power spectrum of the mother wavelet should be well-matched with the characteristic of tissues. The biological tissues and background noise can be differentiated consequently. CWT is implemented to process the ultrasonic echo signals from the two inverted pulses at receive. Select an appropriate scale, and get the maximal one of the transformed wavelet coefficients. Wavelet correlation coefficients at this scale are chosen to replace the original received echo signals. The echo signals replaced by the wavelet coefficients from the two inverted pulses are summed to obtain a beam.
Subsequent procedures include log-arithmetic compression, coordinate transformation and gray-scale scan conversion. The algorithm is finished after the image is shown.
Pulse-inversion-based tissue second-harmonic imaging (PIHI) technique and THI mode are implemented as a comparison, which were filtered at twice the fundamental frequency.
In the 1970s, Khokhlov, Zabolotskaya and Kuznetsov proposed a new wave equation for describing nonlinear ultrasonic field, which is named KZK equation. Subsequently, a large number of experimental, theoretical and numerical results testify its accuracy [29, 30, 31]. KZK equation has been widely utilized in the simulation and calculation of nonlinear sound fields. In this study, the model used to construct tissue wavelet is obtained from numerical solutions of the KZK nonlinear parabolic wave equation.
The expression of KZK equation is as follows, and the symbol is defined in Table 1.
Definition and values of symbols in KZK equation
Where
Tissue wavelet is constructed as follows:
Solve the KZK equation and obtain the echoes. The choice of parameters in the equation, such as the absorption coefficient, the speed of sound and the nonlinear coefficient, are set according to the biological characteristics of human soft tissue. Other parameters, such as the acoustic pressure and frequency are dependent on a specific experiment. The explicit values are shown in Table 1. The constructed tissue wavelet would be used in experiments of ultrasonic tissue phantom, human carotid artery and human liver. Filter the echo signals obtained in Step 1. The band-pass filter is devised on the basic of the bandwidth of ultrasonic sensor. Normalize the filtered echo signals obtained in Step 2. The normalized signal will be used as tissue wavelet.
The echoes are obtained by numerically solving KZK equation under different external sound field conditions. Based on the results calculated by the theoretical model, we can construct a mother wavelet suitable for the detection of biological tissue echoes. Utilizing the difference of wavelet coefficients after transformation, the biological tissue echo signals and noise signals can be effectively distinguished.
In order to investigate the effectiveness of the tissue wavelet, Morlet (Morl) wavelet and Mexican hat (Mexh) wavelet were employed as a comparison. These two mother wavelets are widely used and have superior performance in signal process. Figure 2 show images of tissue wavelet used in ultrasound tissue phantom and experiments in-vivo. Mexh wavelet, Morl wavelet and their corresponding spectrum are shown in Fig. 3.
Tissue wavelet at different phases and their corresponding spectrum.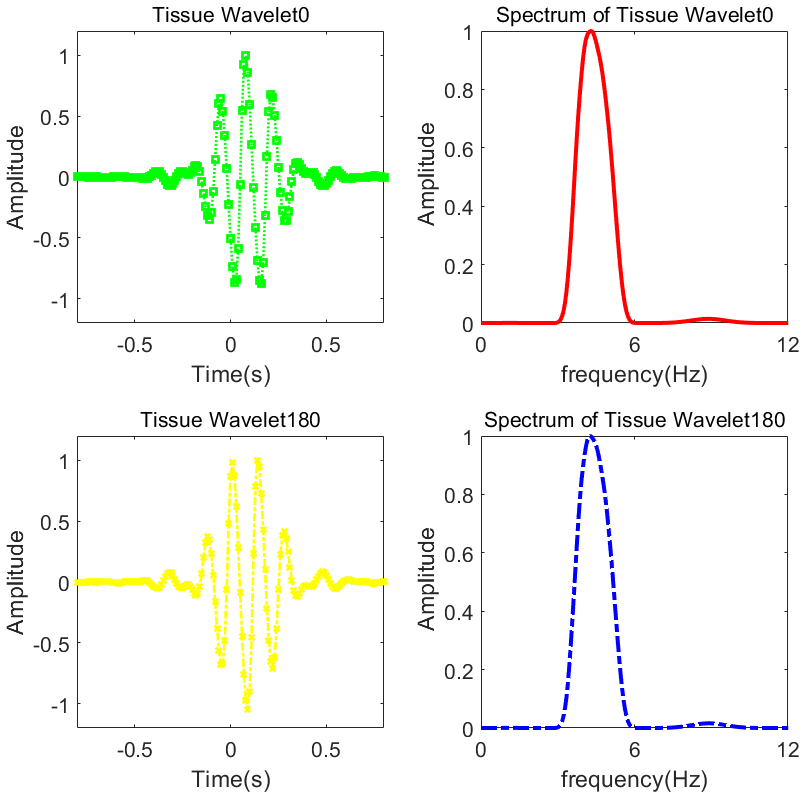
Mexican (Mexh) hat wavelet, Morlet (Morl) wavelet and their corresponding spectrum.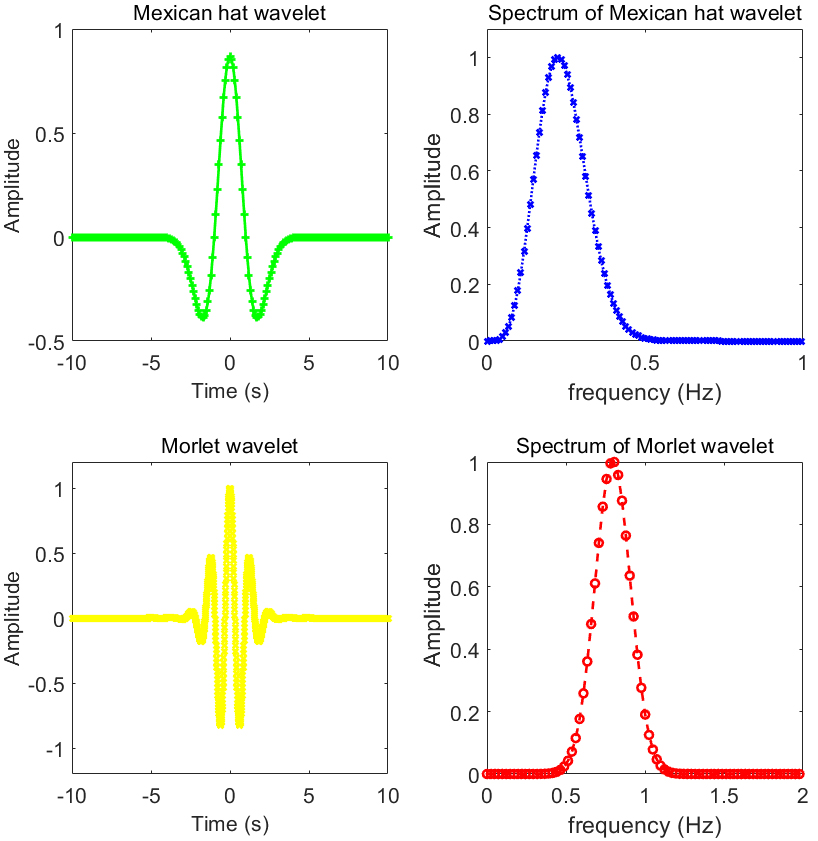
We select four commonly used indicators to evaluate the image quality: contrast ratio (CR), contrast-to-noise ratio (CNR), speckle signal-to-noise ratio (sSNR) and spatial resolution.
Point targets in the experimental data were used to assess the spatial resolution. The full width at half maximum (FWHM,
CR, CNR and sSNR were selected to evaluated the cyst targets in experimental data. They were defined as follows [33, 34, 35]:
Where
In our study, radio frequency data were obtained from open Ultrasonix SonixTouch imaging system, and experiments were carried on ultrasonic tissue phantom, human carotid artery and human liver.
An open ultrasound imaging platform Sonix Touch (Ultrasonix Medical Corp., Canada) was utilized to acquire RF data for offline analysis [36]. 128-element linear array sensors (L14-5/38 probe, 7.5 MHz center frequency, 65% bandwidth) were used in our experiments. The transmitted center frequency was set at 5.0 MHz both for experiments in phantom and in-vivo. The analysis tool employed for post-processing was Matlab (Mathworks, Natick, MA, USA).
In order to investigate the effectiveness of the imaging algorithm, experiments in-vitro were implemented on a standard tissue mimicking ultrasound phantom (KS107BG, manufactured by Institute of Acoustics, Chinese Academy of Science). The device is composed of ultrasonic tissue mimicking material and a variety of test targets are embedded in it, as well as acoustic windows and shells.
Two types of phantom data were chosen to assess the proposed technique. For the spatial resolution evaluation, isolated point targets were selected, which were arranged vertically and horizontally in the ultrasound tissue phantom. The second phantom dataset containing anechoic cysts, were used to estimate the CR, CNR and sSNR. The cysts employed were set at the depth of 15 mm and 30 mm, respectively. The corresponding diameter of the cysts were 2 mm and 4 mm.
Experiments in-vivo employed data of human carotid artery and liver for image algorithm assessment. The clinical study was approved by the Medical Ethics Committee at Tangdu Hospital (Xi’an, China). Written informed consent from the volunteer providing data was obtained. The real images of SonixTouch imaging platform and ultrasound tissue phantom are demonstrated in Fig. 4.
Experimental materials: (a) SonixTouch ultrasound imaging platform; (b) tissue ultrasound phantom.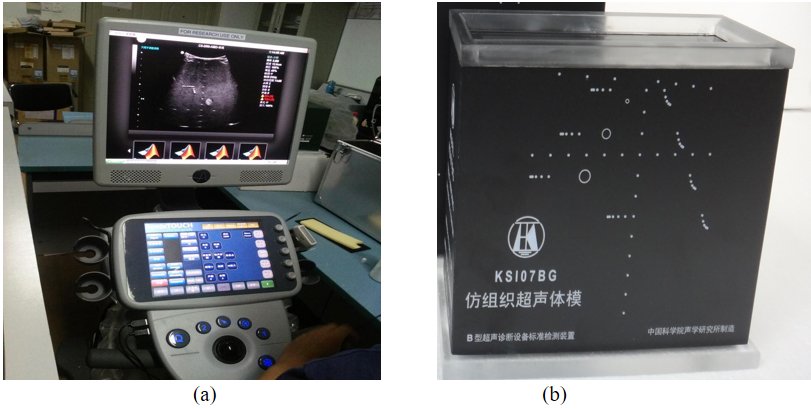
Experiments in B mode imaging were conducted in order to investigate the effectiveness of imaging algorithm and tissue wavelet. Performance of four different methods were compared on the same set of experimental data. The four methods are: Tissue ultrasound harmonic imaging (TUHI), tissue ultrasound imaging method based on Mexh wavelet (TUI-Mexh), tissue ultrasound imaging method based on Morl wavelet (TUI-Morl), and tissue ultrasound imaging method based on tissue wavelet (TUI-TW).
We also discuss the experimental results using pulse-inversion imaging mode. Pulse-inversion-based wavelet analysis imaging technique utilizing tissue wavelet (PIWI-TW) is proposed in our paper. We compared the performance of the introduced PIWI-TW method with the frequently-used PIHI technique. As a comparison, Mexh wavelet and Morl wavelet were employed to process the RF experimental data obtained under pulse-inversion imaging mode (PIWI-Mexh and PIWI-Morl).
Results in B mode imaging
Phantom results in B mode imaging using different techniques: Tissue ultrasound harmonic imaging (TUHI), tissue ultrasound imaging method based on Mexh wavelet (TUI-Mexh), tissue ultrasound imaging method based on Morl wavelet (TUI-Morl), and tissue ultrasound imaging method based on tissue wavelet (TUI-TW). All images are demonstrated with a 70 dB dynamic range.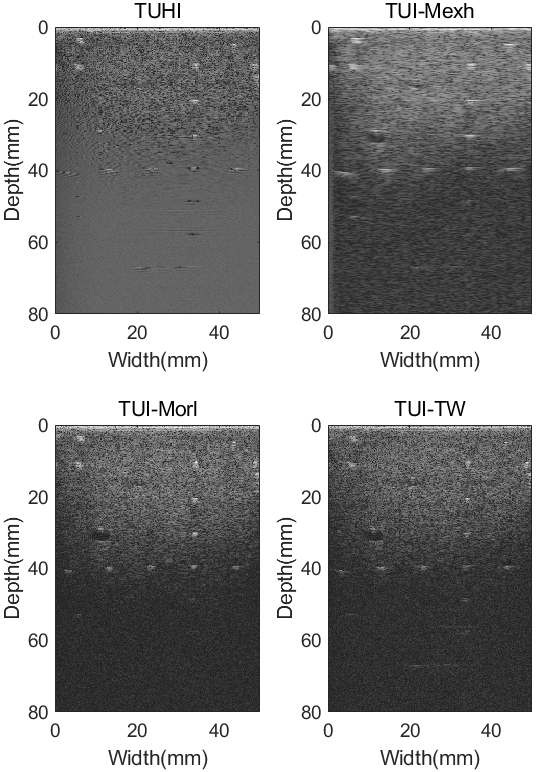
Figure 5 shows the results of the ultrasound tissue phantom in B mode using different methods with a dynamic range of 70 dB. The setting of the depth was 8.0 cm. In the axial direction, 8 targets were located from depths 10 mm to 80 mm, and the center distance between each two targets is 10 mm. In the lateral direction, 5 targets were arranged at depths 40 mm and the center distance between each two targets is 10 mm. The cysts used were set at the depth of 15 mm and 30 mm. The diameter of the cysts were 2 mm and 4 mm, respectively.
It can be observed from Fig. 5 that the background noise is apparent in the TUHI image, while the point targets in horizontal direction and cyst targets are indistinct. The noises?are partial attenuate, which can be observed from TUI-Mexh, TUI-Morl and TUI-TW images. The selection of Mexican hat wavelet makes the targets visible but causes more artifacts, which can be seen from TUI-Mexh image. It can also be observed that the horizontal and vertical point targets cannot display in TUHI, TUI-Mexh and TUI-Morl images when the imaging depth is deeper than 40 mm. However, the point targets are partially visible in TUI-TW image at the same depth.
Region of interest (ROI) selection for assessment of spatial resolution, contrast ratio (CR), contrast-to-noise ratio (CNR) and speckle signal-to-noise ratio (sSNR) in B mode phantom images.
In order to evaluate the proposed tissue wavelet, Regions of Interest (ROI) were chosen for quantitative analysis. Figure 6 shows the selected specific points target, cyst targets and line target. Point A (37 mm, 20 mm) and B (37 mm, 60 mm) were chosen for axial spatial resolution study, while point C (13 mm, 40 mm) and D (47 mm, 40 mm) were chosen for lateral spatial resolution study. We select Cyst C and Cyst D for discussion of CR, CNR and sSNR. The diameter of the cysts were 2 mm and 4 mm, respectively. Besides, five points, which were located at the depth of 70 mm, are chosen for assessment of deep tissue imaging quality. Among the five points, the center distance between each two targets were 1 mm, 2 mm, 3 mm and 4 mm, respectively.
We selected two points (red boxes in Fig. 6) located at A (37 mm, 20 mm) and B (37 mm, 60 mm) to estimate the axial spatial resolution. The axial variations of point A and B are shown in Fig. 7. It can be observed from Fig. 7a and b that, the main-lobe of TUI-TW are similar with TUHI, TUI-Mexh and TUI-Morl images (except for point B of TUHI image). It can be seen that TUI-Mexh has the highest side-lobe level.
Axial FWHM
values of the point targets in B mode phantom images
Axial FWHM
The axial variations of the point targets in B mode phantom images. (a) Point A, (b) Point B.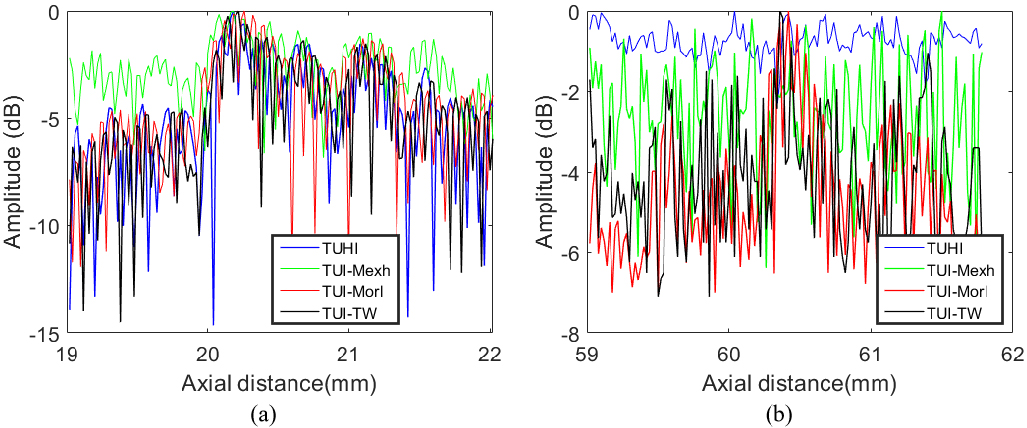
Table 2 shows the statistical results of axial FWHM. The Average value in Table 2 is the mean value of FWHM for all points in Fig. 6. As seen, the axial FWHM values in the B-mode experimental images of different techniques are almost the same. The specific reasons will be detailed in the Discussion section. The FWHM value of UTHI images positioned at point B is empty because the full width at half maximum of point B can not be calculated, which can be seen from Fig. 7b.
Two points (green boxes in Fig. 6) located at C (13 mm, 40 mm) and D (47 mm, 40 mm) were selected to estimate the lateral spatial resolution. The lateral variations of point C and D are shown in Fig. 8. It can be observed that the TUI-TW technique display clearly narrower main-lobe widths compared to TUI, TUI-Mexh and TUI-Morl methods. It can also be seen that UTHI has the highest side-lobe levels and TUI-Mexh has the widest main-lobe widths.
Lateral FWHM
CR
The lateral variations of the point targets in B mode phantom images. (a) Point C, (b) Point D.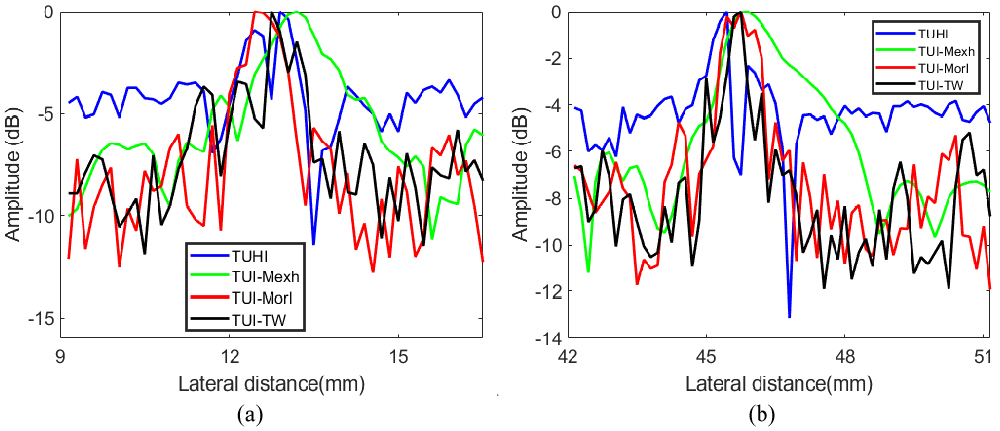
Table 3 demonstrates the statistical results of lateral FWHM. The Average value in Table 3 is the mean value of FWHM for all points in Fig. 6. As seen, lateral spatial resolution of TUI-TW is the smallest among all methods, The average lateral FWHM of TUI-TW can achieve an improvement of 2.40 mm compared with TUHI. TUI-Mexh has the biggest FWHM due to the artifacts.
We selected five points (yellow dotted line in Fig. 6) positioned at the depth of 68 mm to assess the imaging quality of deep tissue. The results are shown in Fig. 9. It can be observed that five points can be distinguished in TUI-Mexh and TUI-TW method. However, the curves of TUHI and TUI-Morl are irregular, thus the five points cannot be differentiated at all. The side-lobe level of TUI-Mexh is higher than TUI-TW. The reason will be demonstrated in the discussion section.
From Fig. 5, it can be seen that TUI-Mexh, TUI-Morl and TUI-TW have better performance in detecting anechoic cysts than the TUHI images. Besides, the cyst contrast of TUI-Morl and TUI-TW images are better than TUI-Mexh image. TUI-Morl and TUI-TW have the similar visual assessment for cyst images.
Table 4 shows the CR, CNR and sSNR values for the selected cysts of B mode phantom images. Cyst E and Cyst F are drawn in Fig. 6 using blue and purple circles, while the corresponding background regions are shown at the same depth. The subscripts of E and F represent the Cyst E and Cyst F, respectively. As seen from Table 4, TUI-TW can offer CR and CNR improvements by 9.16 dB and 1.55 dB over TUHI. The TUI-Morl method has a best sSNR than TUHI, TUI-Mexh and TUI-TW method. It can also be seen that CR, CNR and sSNR values for Cyst E is smaller than that for Cyst F.
In-vivo study of B mode: Human carotid artery
In order to study the applicability of the proposed imaging technique, experiments were conducted in-vivo. The experimental data were obtained from the carotid artery of a 40-year-old healthy female volunteer. The results of ultrasonic gray images are displayed in Fig. 10 with a dynamic range of 60 dB for the four different methods. As can be seen from Fig. 10, background noise and artifacts are obvious in TUHI image. In the TUI-Mexh, TUI-Morl and TUI-TW images, noise inside the artery can be reduced, while the artery walls are clearer, compared to that of TUHI. It is worth noting that, compared to TUI-Mexh and TUI-Morl images, TUI-TW can display more detailed biological tissues, for example, the areas shown in the green dashed box of Fig. 10.
CR
, CNR
and sSNR
of the carotid artery images for different methods
CR
Points at deep depth of 68 mm in B mode phantom images.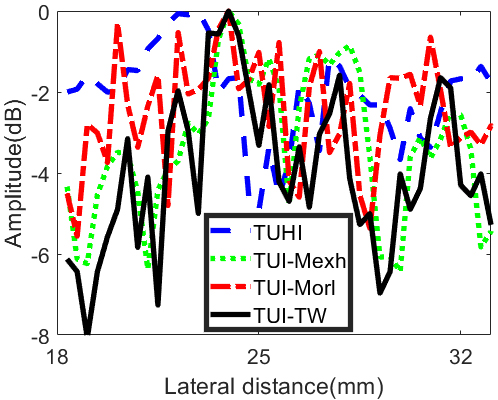
Carotid artery images using different techniques: Tissue ultrasound harmonic imaging (TUHI), tissue ultrasound imaging method based on Mexh wavelet (TUI-Mexh), tissue ultrasound imaging method based on Morl wavelet (TUI-Morl), and tissue ultrasound imaging method based on tissue wavelet (TUI-TW). All images are demonstrated in a 60 dB dynamic range.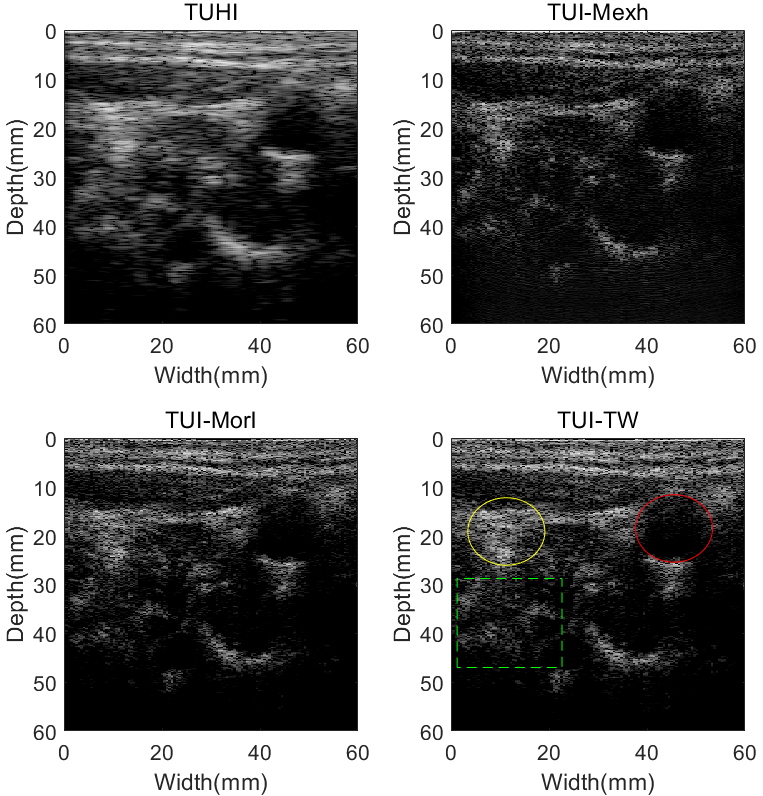
Table 5 demonstrated the CR, CNR and sSNR values for B mode carotid artery images. Artery and surrounding tissues were drawn in Fig. 10 using red and yellow circles, respectively. It can be observed from Table 5 that, TUI-TW can achieve CR and CNR increasements by 4.25 dB and 0.81 dB over TUHI. The TUI-Mexh has a better sSNR than TUHI, TUI-Morl and TUI-TW in B mode experiments in-vivo.
Results of phantom images using different imaging techniques: Pulse-inversion-based tissue second-harmonic imaging (PIHI), pulse-inversion-based wavelet imaging utilizing Mexh wavelet (PIWI-Mexh), pulse-inversion-based wavelet imaging utilizing Morl wavelet (PIWI-TW) and pulse-inversion-based wavelet imaging utilizing tissue wavelet (PIWI-TW). All images are illustrated with a 68 dB dynamic range.
Figure 11 show the point and cyst targets images of ultrasound tissue phantom under the imaging mode of pulse-inversion. The dynamic range and imaging depth of all images were set to 68 dB and 10 mm, respectively. Five point targets were scattered at depths 40 mm, while the center distance between each two targets is 10 mm in the lateral direction. There are two groups of 5 point targets at depths 10 mm and 30 mm, respectively. The cyst targets were located as the same as B mode imaging.
As seen from Fig. 11, the background noise is existent, while the horizontal point targets located at 40 mm depth are not clearly displayed in the PIHI image. The noise is decreased in the PIWI-Mexh, PIWI-Morl and PIWI-TW images. These results are similar to B mode imaging. In the PIWI-TW images, the background noise is significantly suppressed, while at the same time the point targets are clearly visible. It should be noted that the cost of noise suppression is the invisibility of cyst targets, which can be observed from Fig. 11.
ROI were also chosen for quantitative analysis of the proposed method, of which the image is shown in Fig. 12. Point A (20 mm, 40 mm), Point B (50 mm, 40 mm), group points in Line C and D were chosen for lateral spatial resolution discussion. Among each group of 5 point targets, the center distance between each two targets were 4 mm, 3 mm, 2 mm and 1 mm, respectively. The cysts used for assessment were set at the depth of 15 mm (Cyst E) and 30 mm (Cyst F), while the diameter of the cysts were 2 mm and 4 mm, respectively.
Selection of point and cyst targets in pulse inversion phantom images for quantitative analysis.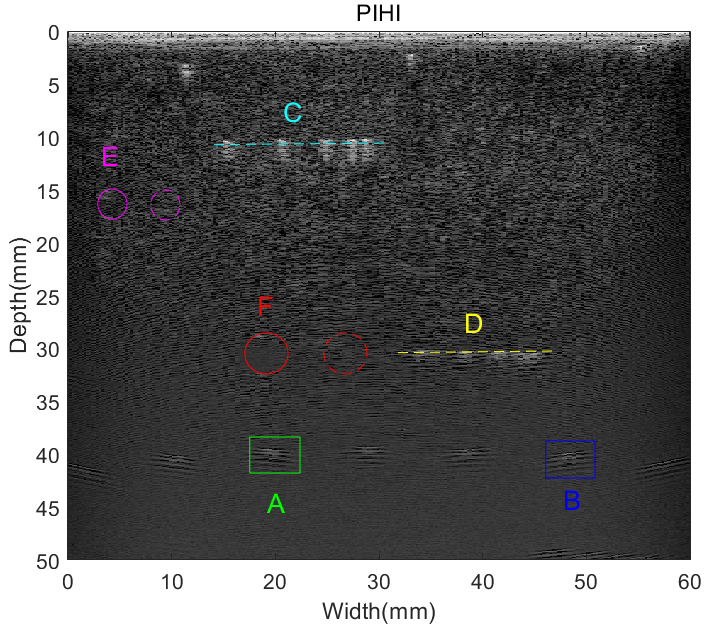
Two points positioned at A (18 mm, 40 mm) and B (48 mm, 40 mm) are selected to estimate the lateral spatial resolution with green and blue boxes, respectively. The lateral variations of point A and B are exhibited in Fig. 13. It can be observed that the utilization of tissue wavelet results in the narrowest main-lobe widths in PIWI-TW technique, compared to PIHI, PIWI-Mexh and PIWI-Morl methods. Besides, PIWI-TW has the smallest side-lobes level.
Lateral FWHM
values of the point targets in pulse-inversion mode phantom images
Lateral FWHM
The lateral variations of the point targets in pulse inversion phantom images. (a) Point A, (b) Point B.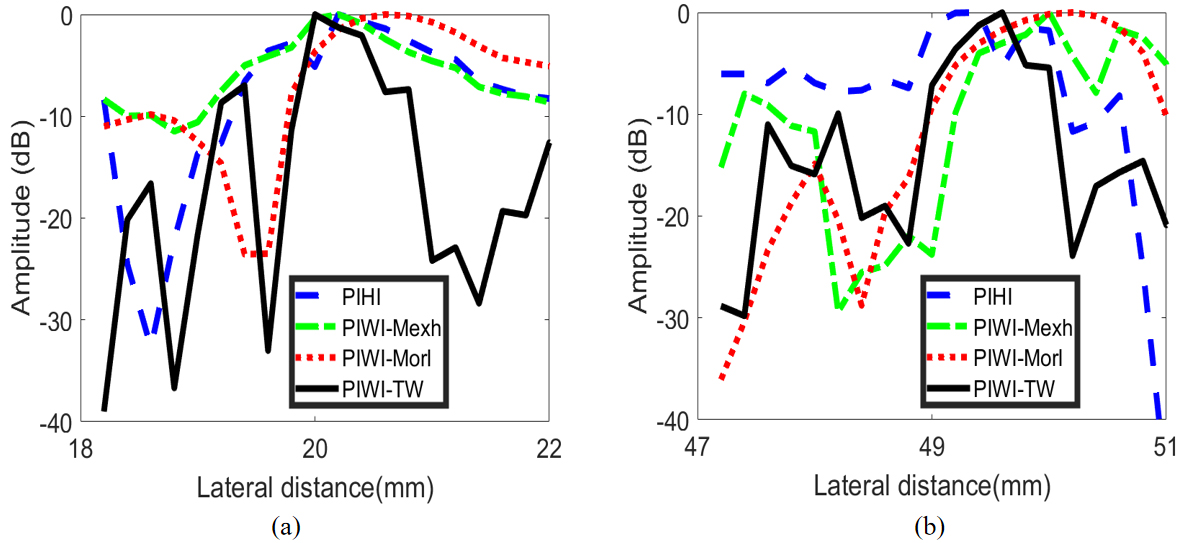
The statistical results of lateral FWHM for pulse inversion phantom images are exhibited in Table 6. The Average value in Table 6 is the mean value of FWHM for all points in Fig. 12. As seen, the lateral FWHM value of PIWI-TW technique is minimum among all four imaging methods. In comparison with PIHI, the average lateral FWHM of PIWI-TW can achieve an improvement of 1.03 mm. PIWI-Morl has the greatest FWHM among the four methods.
Noticing that the graphic results of axial FWHM are not given here. Because the values of axial FWHM calculated by different pulse-inversion-based imaging technique are almost the same, and the specific numerical values are about 0.52 mm. This situation is the same as that of B mode imaging.
We also selected two groups of point targets (cyan and yellow dotted line in Fig. 12) to evaluate the spatial resolution. Each group contains five points and the depth are located at 10 mm and 30 mm, respectively. The results are shown in Fig. 14. It can be observed from Fig. 14 that five points can be successfully distinguished using the four imaging methods both for group point targets in Line C and Line D. PIWI-TW can obtain the narrowest main-lobe width and the lowest side-lobe level. It can also be observed from Fig. 14 that lateral spatial resolution of group point targets located in Line C are better than that located in Line D.
In our study, the application of pulse-inversion technique and tissue wavelet lead to the decrease of background noise and artifact. The spatial resolution is thus improved. However, the suppression of noise results in the visual invisibility of anechoic cyst targets, which can be observed from Fig. 11. This will be discussed in Discussion Section.
CR
, CNR
and sSNR
of the cysts in pulse-inversion mode phantom images
CR
Results of group point targets in pulse inversion phantom images. (a) Group point targets in line C, (b) group point targets in line D.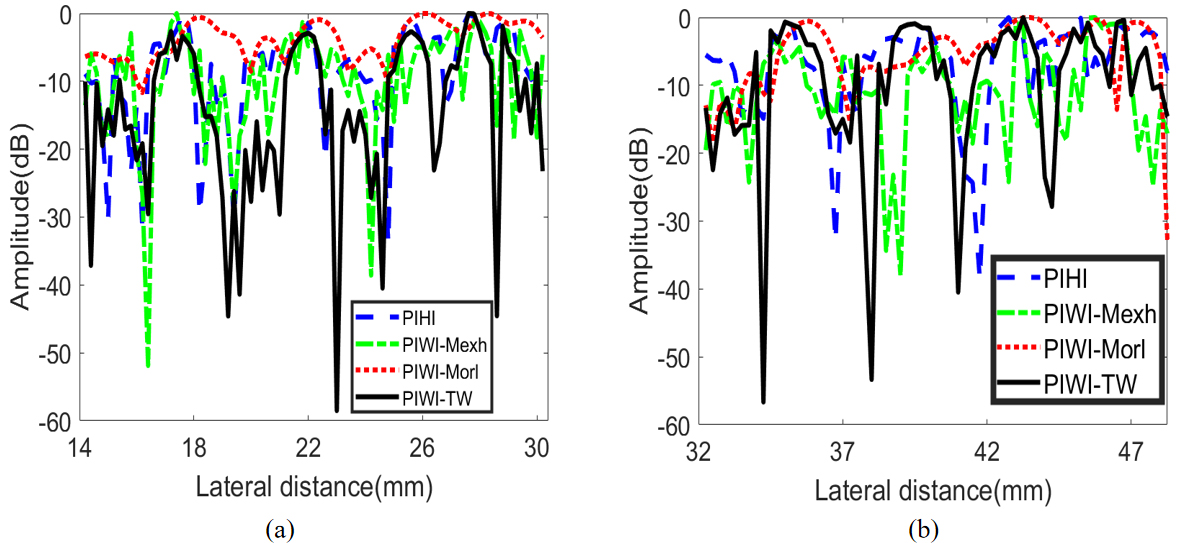
CR, CNR and sSNR for the cysts of phantom images using pulse inversion mode are exhibited in Table 7. Cyst E and Cyst F are drawn in Fig. 12 with purple and red circles, while the corresponding background regions are chosen at the same depth. The subscripts of E and F represent the Cyst E and Cyst F, respectively. As seen in Table 7, for a specific point, there are little differences among the CR, CNR and sSNR using different methods. Besides, the three parameters for Cyst E is also smaller than that for Cyst F, which is similar with B mode situation. However, the difference between them become diminished.
Experiments in-vivo were also implemented for the assessment of imaging algorithm. The experimental RF data were acquired from the liver of the aforementioned 40-year-old healthy female volunteer. Results of liver images under pulse inversion imaging mode are shown in Fig. 15. Comparing with PIHI image, noises inside the hepatic vein were suppressed in PIWI-TW image, and the contrast was enhanced. Just like B mode imaging, PIWI-TW can display more detailed biological tissues than PIWI-Mexh and PIWI-Morl, for example, the areas shown in the green dashed box of Fig. 15.
CR
, CNR
and sSNR
of the pulse-inversion liver images for different methods
CR
Pulse inversion images of a human liver using different imaging techniques: Pulse-inversion-based tissue second-harmonic imaging (PIHI), pulse-inversion-based wavelet imaging utilizing Mexh wavelet (PIWI-Mexh), pulse-inversion-based wavelet imaging utilizing Morl wavelet (PIWI-TW) and pulse-inversion-based wavelet imaging utilizing tissue wavelet (PIWI-TW). All images are illustrated with a 55 dB dynamic range.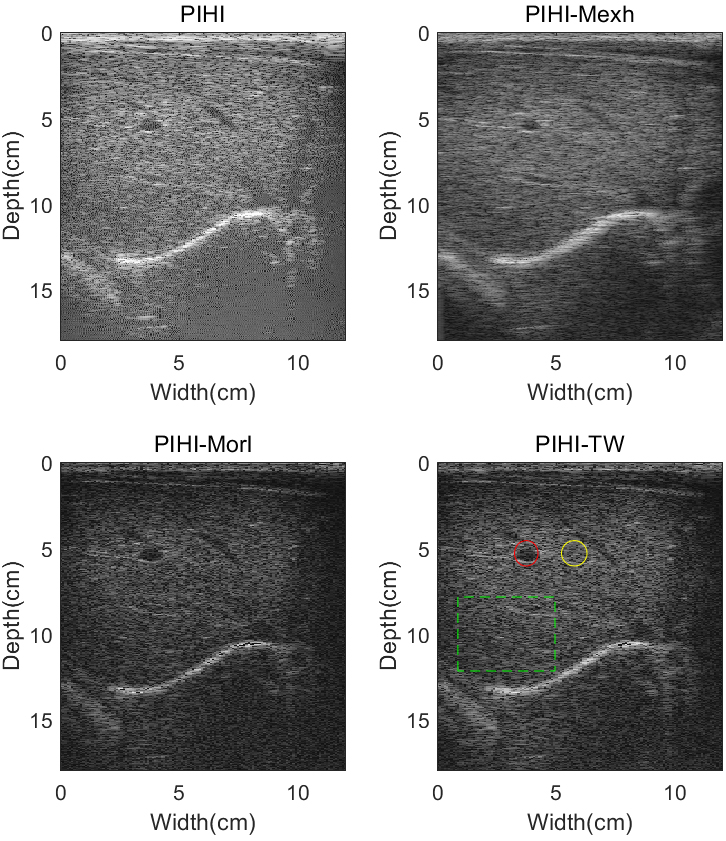
Table 8 shows the numerical values of CR, CNR and sSNR for liver images under pulse inversion imaging mode. Hepatic vein and surrounding tissues are shown in Fig. 15 with red and yellow circles, respectively. From Table 8, we can find out that PIWI-TW can obtain increase of CR and CNR by 5.55 dB and 1.40 dB over PIHI, respectively. The PIWI-Morl method has a better sSNR than PIHI, PIWI-Morl and PIWI-TW in pulse inversion images of human liver.
In B mode imaging, the performance of TUI-TW is better than TUHI, TUI-Mexh and TUI-Morl. It can be seen from Fig. 5 that the wavelet transform process can reduce noise. Lateral spatial resolution of TUI-TW can achieve the optimal one among all methods. These results demonstrate the effectiveness of tissue wavelet in terms of lateral spatial resolution, CR and CNR. According to sound field theory, the energy of ultrasound wave decreases with the increase of penetration depth. Due to this acoustic attenuation, lateral spatial resolution in far field performs worse than the ones in near field. In Fig. 5, when the detection depth reaches 68 mm, point targets are difficult to distinguish (TUI-Mexh and TUI-TW images), and even cannot be displayed (TUHI and TUI-Morl images). Figure 9 shows the more accurate analysis of deep point targets and also testifies the effectiveness of tissue wavelet in terms of deep tissue imaging.
Experimental results in phantom and in-vivo under pulse inversion imaging modes, show that the PIWI-TW method can effectively improve the imaging quality than PIHI, PIWI-Mexh and PIWI-Morl technique. Results in Fig. 11 indicate the ability to reduce noise using wavelet transform, which is the same conclusion with B mode imaging. As seen from Fig. 12, Table 6 and Fig. 14, constructed tissue wavelet shows the effectiveness in terms of imaging lateral spatial resolution improvement. In-vivo results of Fig. 15 demonstrate that, compared with the other three imaging methods, PIWI-TW technique shows a better capability to suppress background noises and display more biological tissues at the same time. These results reveal the superiority of tissue wavelet in improving tissue ultrasound imaging quality. Results in Fig. 14 also verify the effectiveness of pulse inversion technology, comparing to B mode imaging. Due to the acoustic attenuation, lateral spatial resolution in near field performs better than the ones in far field, which could be seen from group point targets of Line C and Line D in Fig. 14.
Cyst targets in Fig. 11 are invisible. However, it should be noted that although images in pulse-inversion imaging mode are indemonstrable, aforementioned results of B mode can successfully prove the effectiveness of imaging algorithm and tissue wavelet.
Results of phantom under B imaging express that the TUI-Mexh image has the biggest Lateral FWHM of all four methods, while the sSNR of TUI-Morl image is biggest among all four methods. The reason might be the weaker artifacts noises in the TUI-Morl image. From the visual assessment of images in Fig. 5, the imaging quality in near field is worse than that in the far field. However, in B mode experiments in-vivo, the TUI-Mexh has a better sSNR than TUHI, TUI-Morl and TUI-TW. The results above show that Mexican hat wavelet performs better in in-vivo experiments, comparing to Morlet wavelet. The reason might be the better correlation between Mexican hat wavelet and the echo signals of tissues in carotid artery, compared to Morlet wavelet. While in pulse inversion imaging mode, the performance of the two mother wavelets mentioned above is just the opposite. This conclusion demonstrates the advantages of Morlet wavelet in detecting liver signals, comparing to Mexican hat wavelet. It should be noticed that the comparison above is just for Mexican hat wavelet and Morlet wavelet. Constructed tissue wavelet based on KZK equation has more superior performance, comparing to Mexh wavelet and Morl Wavelet.
As seen from Fig. 7 and Table 2, the axial spatial resolution of all methods has little difference. Results in pulse inversion imaging mode draw the same conclusion. It is because axial spatial resolution is mainly wave length dependent. The length of the wave is?invariant for a specific experiment.
Tissue wavelet performs better in pulse inversion imaging mode comparing with B imaging mode, which can be seen from Figs 5 and 11. The results confirm the superiority of pulse inversion imaging technique in tissue ultrasound imaging. The optimal imaging quality can be obtained by combining tissue wavelet and pulse inversion imaging technique.
For the mother wavelet utilized in wavelet process, the admissible condition is a very important feature [37, 38]. Theoretically, any arbitrary function can be used as a mother wavelet as long as it satisfies the admissible condition of wavelet transform. The expression of admissibility condition is as follows:
where
Mathematically, the admissible condition guarantees the mother wavelet preserves the zero mean value, which is essential for inverse wavelet transform. It means that the admissible condition is for the needs of inverse wavelet transform. If the mother wavelet does not meet the wavelet admissible condition, the reconstruction of the signal cannot be accomplished after wavelet decomposition.
Tissue wavelet designed in this paper do not conform to Eq. (11a) and (11b), which could also be seen from Fig. 2. However, tissue wavelet could be able to utilize in wavelet process because the inverse wavelet transform was not carried out in our imaging algorithm. Since the amplitude of the transformed wavelet coefficients were proportional to the energy of the echoes, the detection can be accomplished in the domain of the wavelet transform. Therefore, the reconstruction of signal is not necessary.
In our imaging algorithm, the maximal value of the wavelet coefficients was acquired at a suitable scale. Therefore, imaging quality is related to the selection of scales in wavelet process. The optimal scales were chosen by repeated cycles in our study. Besides, the variance of the wavelet coefficients and mean value could be considered besides the maximal value for a better performance. Further research could be focused on the adaptive optimized algorithm.
In this study, we designed tissue wavelet based on the KZK wave equation. Because of the limitation of calculation, tissue wavelets utilized in ultrasound phantom, human carotid artery and human liver are the same. Tissue wavelet library, which is constructed according to different biological characteristics of human tissue, will be our further study. Results in this paper confirm the efficiency of tissue wavelet and the suited imaging algorithm. However, the applicability and robustness of the proposed method would be enhanced if a library of tissue wavelet could be structured. In addition, with the development of computer software and hardware technology, our method will be able to be implemented in real-time in the ultrasonic imaging platform.
In this paper, a novel tissue ultrasound imaging technique, which is the combination of pulse inversion technique and wavelet transform, has been introduced. A new mother wavelet, named tissue wavelet, is designed based on the KZK wave equation. A suited tissue imaging algorithm is put forward.
The proposed method is applied on both experiments on phantom and in-vivo to evaluate its performance under pulse inversion and B imaging mode. Mexh wavelet, Morl wavelet and tissue wavelet has been comparative investigated under the same equal conditions.
Experimental results in phantom and in-vivo demonstrate that the proposed method can obtain better lateral spatial resolution, higher CR and CNR values than conventional PIHI technique. In addition, the superiority of tissue wavelet over Mexh and Morlet wavelet was proved in terms of lateral spatial resolution, CR and CNR. Experimental results verify the effectiveness of the corresponding imaging algorithm at the same time.
List of symbols
Funding
This research was supported by National Natural Science Foundations of China (grant numbers 62171177 and 82174166) and Doctoral Fund of Qingdao University of Science and Technology.
Footnotes
Acknowledgments
We thank the editors and reviewers for their hard work in the manuscript process. We also appreciate the female volunteer who provided the data.
Conflict of interest
The authors declare that they have no conflict of interest.
