Abstract
BACKGROUND:
To date, a few studies indicated that probiotics are beneficial to pouchitis, but no meta-analyses summarized the outcomes of probiotics in pouchitis in detail.
OBJECTIVE:
This meta-analysis discusses probiotics in the prevention of pouchitis for patients after ileal pouch-anal anastomosis (IPAA) and the relationship between probiotics preventive effect and the duration of therapy and history.
METHODS:
PubMed, EMBASE and Cochrane Library databases were searched from inception until February 2022. Risk ratio (RR), mean difference (MD) and their 95% confidence interval (CI) were analyzed by Review Manager 5.3. The subgroup analysis was also performed to explore the agent for influencing outcomes.
RESULTS:
A total of 8 studies were included in this meta-analysis. The incidence of pouchitis in probiotics was significantly lower than that in the control (RR
CONCLUSIONS:
Probiotics are beneficial in the prevention of pouchitis after IPAA, especially in the short-term.
Introduction
Pouchitis is a common complication after restorative proctocolectomy with ileal pouch-anal anastomosis (IPAA) seen in patients with ulcerative colitis (UC) and is a nonspecific inflammatory in the ileal pouch [1]. Over 50% of UC patients after IPAA experience pouchitis and preventive strategies are therefore of crucial importance. The pathogenesis of pouchitis is complicated. The dysbacteriosis of ileal pouch is one of the most important mechanisms [2]. An analysis of the microflora indicated that there is a great difference between pouchitis and non-pouchitis patients [3]. During pouchitis, the reduction of microflora diverse and the anaerobic to aerobic ratio are seen in pouchitis patients [4]. Some studies also indicated that sulfate-reducing bacteria, enterobacteriaceae are common bacteria associated with pouchitis [5, 6]. On the other hand, the anti-microbial treatment is an effective method for pouchitis and is superior to anti-inflammatory therapy in inducing remission in pouchitis patients [7, 8, 9]. Therefore, it is obvious that the microflora is closely related to pouchitis.
Probiotics are living microorganisms that are beneficial to host. They can regulate the tight junctions, properties of the mucus layer to maintain the intestinal homeostasis [10, 11, 12]. Laval et al. indicated that Lactobacillus rhamnosus CNCM I-3690 maintains the epithelial barrier through modulating occludin and E-cadherin in the murine model [12]. Probiotics also have an anti-microbial function to maintain intestinal balance [13]. Furthermore, some systematic reviews and meta-analyses indicated that probiotics are beneficial to the prevention and treatment of gastrointestinal disease, including the inflammatory bowel disease (IBD), irritable bowel syndrome (IBS), antibiotic associated diarrhea (AAD) [14, 15, 16].
Some meta-analyses also mentioned that probiotics are beneficial to patients after IPAA, but they did not summarize the outcomes of probiotics for them in detail [14, 17, 18]. In this meta-analysis, we discuss the efficacy of probiotics in preventing pouchitis for patients after IPAA, the short-term and long-term preventive effects, and the pouch disease activity index (PDAI) after the administration. The agents that may influence the outcomes are also discussed.
Methods
Search strategy
The MeSH terms “proctocolectomy, restorative”, “pouchitis”, “probiotics”, “escherichia coli”, “VSL3”, “streptococcus”, “saccharomyces”, “lactobacillus”, “bifidobacterium”, “enterococcus” and their entry terms were searched in PubMed, EMBASE and Cochrane Library databases from inception to February 2022. The study also gained from reference of relevant reviews.
Study selection
We included studies that met the following criteria. Inclusion criteria: (1) All studies reported administration of probiotics for patients after restorative proctocolectomy with IPAA. (2) All patients were without pouchitis at the study entry (PDAI
The study selection was completed by two researchers. Any contradictions between the two researchers were solved by discussion or decided by a third reviewer.
Data extraction
The following data were extracted: type of study; type of probiotics; the diagnostic criteria; the start time of probiotics administration; the population of the control and probiotics; the number of patients with pouchitis in different time periods; the population of pouchitis; the PDAI scores of pouchitis-free population after treatment. The data extraction was completed by two researchers. Any contradictions between the two researchers were solved by discussion or decided by a third reviewer.
Assessing quality of included studies
The assessment quality was performed by the Cochrane Collaboration’s Tool for Assessing Risk of Bias. The quality was assessed according to the aspects as follows: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias). The assessing quality was completed by two researchers. Any contradictions between the two researchers were solved by discussion or decided by a third reviewer.
Statistical analysis
All data were analyzed by Review Manager 5.3. The risk ratio (RR) and its 95% confidence interval (CI) were estimated by the Mantel-Haenszel analysis method. The mean difference (MD) and its 95% CI were estimated by the inverse variance analysis method. The heterogeneity was evaluated by Cochrane Q test and Quantity
Results
Literature search results
The screening process and results are shown in Fig. 1. 1163 studies were searched from PubMed, EMBASE, Cochrane Library databases and other sources. 184 studies were removed due to duplication. 960 studies were removed according to the title and abstract. 11 studies were removed after screening through full-text based on the selection criteria. Finally, 8 studies were included in this meta-analysis [21, 22, 23, 24, 25, 26, 27, 28] (Fig. 1).
Characteristics of included studies and patients
The characteristics of the studies are shown in Table 1. 3 studies recorded the patients with past history of recurrent or chronic pouchitis and 5 studies recorded the patients without past history. The bias of the included studies is shown in Fig. 2.
The incidence of pouchitis after taking probiotics
The incidence in probiotics was significantly lower than the control (RR
The short-term and long-term preventive effects of probiotics
The probiotic group was compared to the placebo group at two time periods: 0–6 months (Fig. 5A) and
Characteristics of the included studies
Characteristics of the included studies
RCT: random-controlled trial; NA: Not available; IPAA: Ileal Pouch-Anal Anastomosis; PDAI: Pouchitis Disease Activity Index; mPDAI: modify Pouchitis Disease Activity Index; 1. Data represent mean
Flowchart for study selection.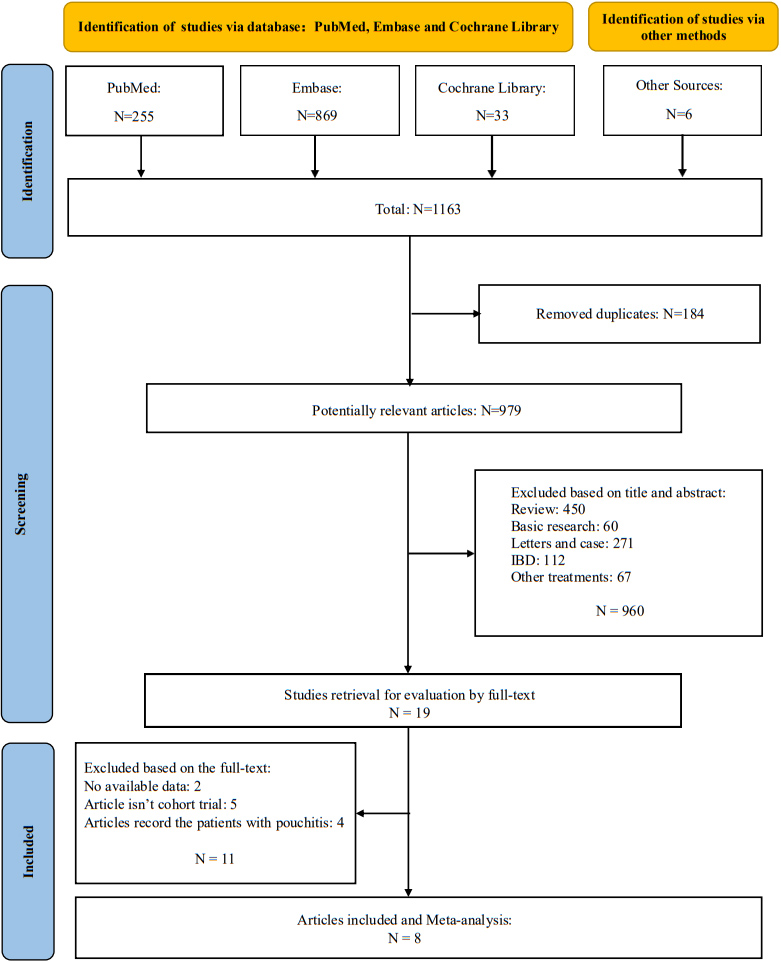
Risk of bias in the included studies.
Risk ratio (RR) for the pouchitis rate after administration of probiotics.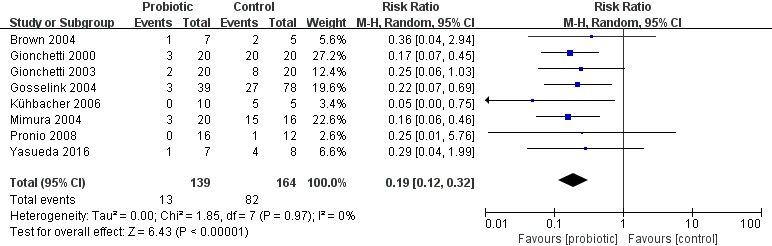
Risk ratio (RR) for the time to onset of pouchitis in patients in the probiotic and placebo groups.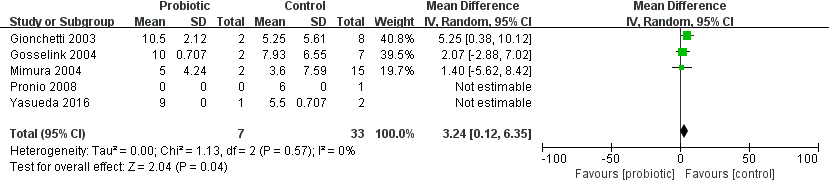
6–12 months (Fig. 5B). Due to the limitation of data, patients with pouchitis in different time periods were all diagnosed for the first time during the study period, and patients with recurrent or chronic pouchitis were not included in the number of patients in the next period. During 0–6 months, the probiotics group had a significant preventive effect on pouchitis compared with the placebo group, and the incidence of pouchitis was statistically significant between the two groups (RR
(A) Risk ratio (RR) for the pouchitis rate after administration of probiotics between 0 and 6 months; (B) RR for the pouchitis rate after administration of probiotics between 6 and 12 months.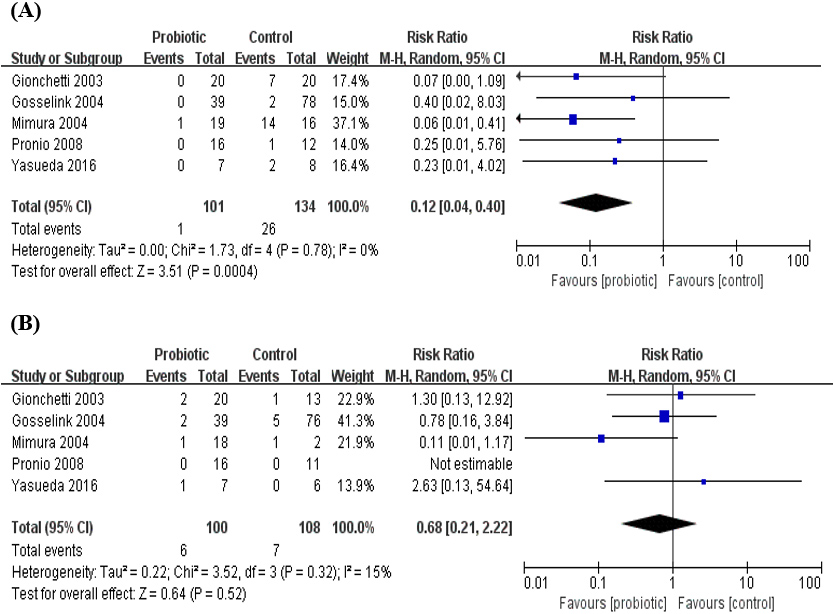
We performed a subgroup analysis based on past history (patient with or without chronic or recurrent pouchitis), duration of treatment, type of control (placebo or no treatment) and type of probiotics (Table 2). The results showed that the subgroup difference was significant after subgroup analysis according to the past history of chronic or recurrent pouchitis, but not significant based on the other agents. Subgroup analysis was performed on the number of patients with pouchitis in the probiotic group and the placebo group according to the presence or absence of previous history of chronic or recurrent pouchitis (Fig. 6). There was statistically significant difference between the probiotic group and placebo group regardless of prior history ((RR
Subgroup analysis of outcomes for probiotics in the prevention of pouchitis
Subgroup analysis of outcomes for probiotics in the prevention of pouchitis
RR: Risk ratio, CI: Confidence interval.
Subgroup analysis of previous history of chronic or recurrent pouchitis.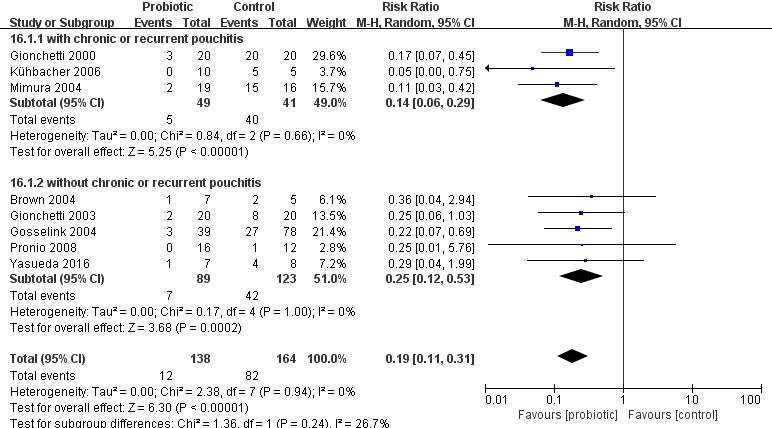
Pouchitis disease activity index (PDAI) after the administration of probiotics. A. PDAI; B. Clinical PDAI; C. Endoscopic PDAI; D. Histological PDAI.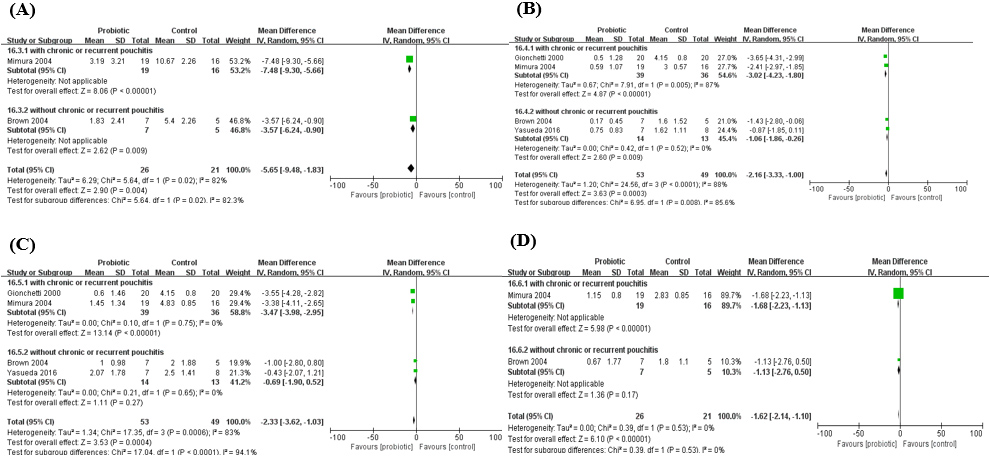
Dysbiosis of the ileal pouch microbiota is a hypothesis about pathogenesis of pouchitis [2]. During pouchitis, the abundance of Enterobacteriaceae is increased and the abundance of Bacteroides and F. prausnitzii, which have an anti-inammatory effect, were decreased [4, 29]. There are a few meta-analyses regarding the efficacy of probiotics in administration of pouchitis. Elahi et al. indicated that probiotics are beneficial to management in pouchitis [17]. Shen et al. showed that VSL#3, a common production used in UC patients containing four strains of lactobacilli, three strains of bifidobacteria, and one strain of streptococcus, is beneficial to maintaining remission for patients with pouchitis [14]. VSL#3 has been shown to be effective in the prevention of pouchitis onset [36]. Singh et al. also showed that VSL#3 is beneficial to preventative therapy for patients after IPAA [18]. However, they did not discuss the agents which may influence outcome of probiotics for pouchitis. In this meta-analysis, we discussed the efficacy of probiotics in preventive therapy for patients after IPAA based on the number of pouchitis rate and PDAI score after administration of probiotics, and the short-term and long-term effects of probiotics was also discussed.
During the administration of probiotics, we found that patients after treatment are less likely to be attacked by pouchitis than the control group. The PDAI in probiotics group was also lower than the control. This indicates that probiotics prevent the episode of pouchitis for patients after IPAA, which is in line with previous meta-analyses. Then we performed the subgroup analysis based on type of probiotics, the duration of treatment, past history of chronic or recurrent pouchitis and type of control. We found that the past history of chronic or recurrent pouchitis was not a significant factor in the preventive effect of probiotics on pouchitis. However, we found that probiotics have a protective effect on pouchitis, but this prevention may differ in the short-term and long-term. Probiotics did not achieve the expected effect in the latter six months, but the reasons may be various. First, 6 months after surgery may be the peak period of pouchitis, and the incidence of pouchitis is higher than that after 6 months. However, the incidence of the two groups in the included study was not very high, leading to the possibility of bias error in the above data analysis results. Secondly, probiotics may be considered to have less effect in the long-term prevention of pouchitis. Long-term use of probiotics may reduce the effect on intestinal flora, or there is a possibility that long-term use may lead to intestinal adaptation to probiotics. Probiotics can promote the strengthening of the intestinal barrier, reduce inflammation, and improve intestinal barrier function by restoring mucus layer thickness, tight junction protein, and producing specific antimicrobial and bioactive lipids with anti-inflammatory properties [30]. It is not excluded that long-term use of probiotics may lead to a decrease in the effect of probiotics on the intestinal barrier. At present, the differences between the short-term and long-term effects of probiotics still need to be further discussed. However, it is undeniable that probiotics have preventive and therapeutic effects on pouchitis, and it is not certain whether the rebound phenomenon will occur after taking probiotics in the short-term, so whether patients should only take probiotics in a short period of time after IPAA has not been concluded.
Probiotics were also used in patients during pouchitis. However, we did not summarize these studies by meta-anlaysis because most studies on patients during pouchitis did not meet the criteria of meta-analyses. The efficacy of probiotics in patients during pouchitis was controversial. Gionchetti et al. indicated that VSL#3 effective for active pouchitis [31]. However, many studies indicated that patients cannot get clinical or endoscopic response after administration of probiotics [33, 34]. We think the successful colonization of probiotcs is a key to treatment. In Gionchetti’s study, S. thermophilus, lactobacilli, bifidobacteria was significantly increased in feces after administration of probiotics [31, 32]. Kuisma et al. indicated that the microbial flora did not have significant difference between before and after administration of probiotics, in which the patients did not have clinical response [34]. The oxidative stress often occurred in inflammatory response, which link to the dysbiosis in IBD [35, 36, 37]. Most probiotics belong to anaerobic bacteria. So inflammatory environment influences the colonization of probiotics, and the “warfare” between Reactive Oxygen Species (ROS) and probiotics may influence the efficacy of probiotics. In contrast, due to the less ROS produced in non-inflammatory pouch, colonization of probiotics in pouch is much easier. This provides a prerequisite for the good efficacy of probiotics in patients during no inflammation in pouch.
This meta-analysis is not without limitations. First, the number of included studies and patients was small, which limited further investigation into probiotics for pouch patients. Secondly, some continuous variables in the original studies did not represent in mean
Conclusion
Probiotics are beneficial to preventative therapy for patients after restorative proctocolectomy with ileal pouch-anal anastomosis. Long-term use of probiotics in the prevention of pouchitis is lower than short-term use, which may be difficult to achieve expectations, but there is no consensus on whether patients after IPAA should use probiotics only for the short-term.
Ethics statement
This study was exempt from ethics approval. Informed consent was obtained from all individual participants included in the study.
Availability of data and materials
The data used or analysed during the current study are available from the corresponding author on reasonable request.
Funding
None to report.
Author contributions
GL: Conceived and designed the study.
WX, XZ: Collected and analyzed the data and wrote the first draft of the paper.
CL, QH: Supervised the data collection process and assisted with writing the paper.
AH: Contributed to the revision of the paper and approved the final version.
Footnotes
Acknowledgments
None to report.
Conflict of interest
The authors have no competing interest to report.
