Abstract

Florian Heinen, Petr Kanovský, A. Sebastian Schroeder, Henry G. Chambers, Edward Dabrowski, Thorin L. Geister, Angelika Hanschmann, Francisco J. Martinez-Torres, Irena Pulte, Marta Banach and Deborah Gaebler-Spira
[Journal of Pediatric Rehabilitation Medicine,
“Patients in all three incobotulinumtoxinA dose groups (total body doses of 4, 8, and 16 U/kg [maximum 100, 300, and 400 U]) experienced improvements in AS-PF scores on the primary body side chosen for treatment 4 weeks post-injection in injection cycle 1 versus the study baseline (Fig. 3) that were significant in MMRM analyses (p < 0.0001 for all).”
“Likewise, investigator’s GICS-PF scores indicated a clinically meaningful response to incobotulinumtoxinA treatment in all dose groups 4 weeks post-injection in injection cycle 1 (Fig. 4) that were significant in ANCOVA analyses (p < 0.0001 for all doses in injection cycle 1, and also in injection cycle 2).”
“AS-PF scores of the primary body side further improved from baseline to week 4 of the second injection cycle in all three dose groups (Fig. 3); improvements were significant versus baseline in MMRM analyses (p < 0.0001 for all doses).”
“Following unilateral LL treatment, all doses of incobotulinumtoxinA resulted in improvements from baseline in AS scores of the knee flexors (Fig. 5A) and the thigh adductors (Fig. 5B) 4 weeks following each injection; improvements in knee flexors were significant in MMRM analyses for the high dose group in injection cycle 1 and 2 and for the low dose group in injection cycle 2 (p < 0.01).”
“Whether from the perspective of the investigator, the parent/caregiver, or the patient, GICS scores confirmed a consistent and global improvement in LL spasticity at 4 weeks post-injection for all three incobotulinumtoxinA dose groups and across both injection cycles (Fig. 6A–C); all scores were significant in ANCOVA analyses (p < 0.0001 for all doses in both injection cycles).”
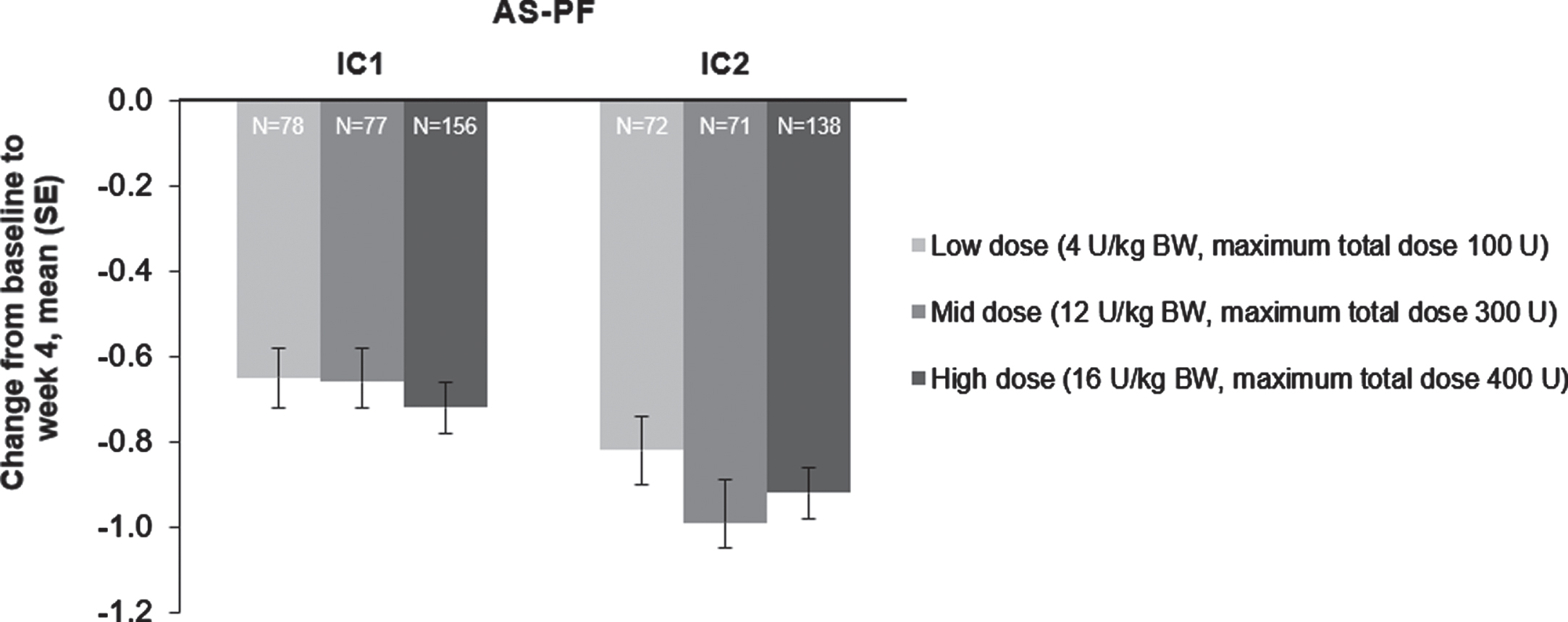
The effect of incobotulinumtoxinA on mean change from baseline at week 4 on the AS-PF on the primary body side, FAS, OC. AS score: 5-point scale from 0 (no increase in muscle tone) to 4 (limb rigid in flexion or extension). The change in the AS-PF from baseline to week 4 was the primary efficacy variable. AS = Ashworth Scale; AS-PF = Ashworth Scale of the plantar flexors; BW = body weight; FAS = full analysis set; IC = injection cycle; kg = kilogram; OC = observed cases; SE = standard error; U = unit.
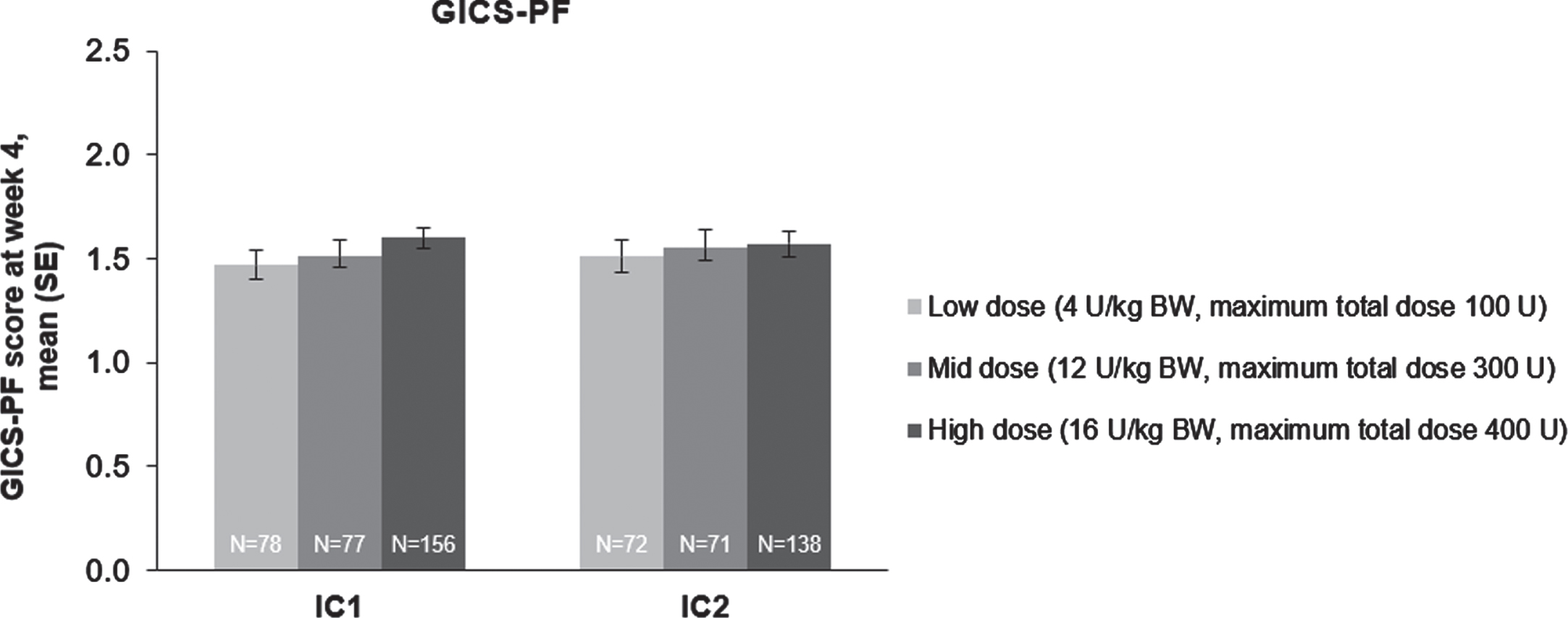
The effect of incobotulinumtoxinA on investigator’s GICS-PF score at week 4; FAS, OC. Investigators were asked to rate their overall impression of change in spasticity of the PFs compared with the condition before the last injection; positive values indicate better results. Investigator’s GICS-PF score at week 4 was the coprimary efficacy variable. BW = body weight; FAS = full analysis set; GICS-PF = Global Impression of Change of Plantar Flexor Spasticity Scale; IC = injection cycle; kg = kilogram; OC = observed cases; SE = standard error; U = unit.
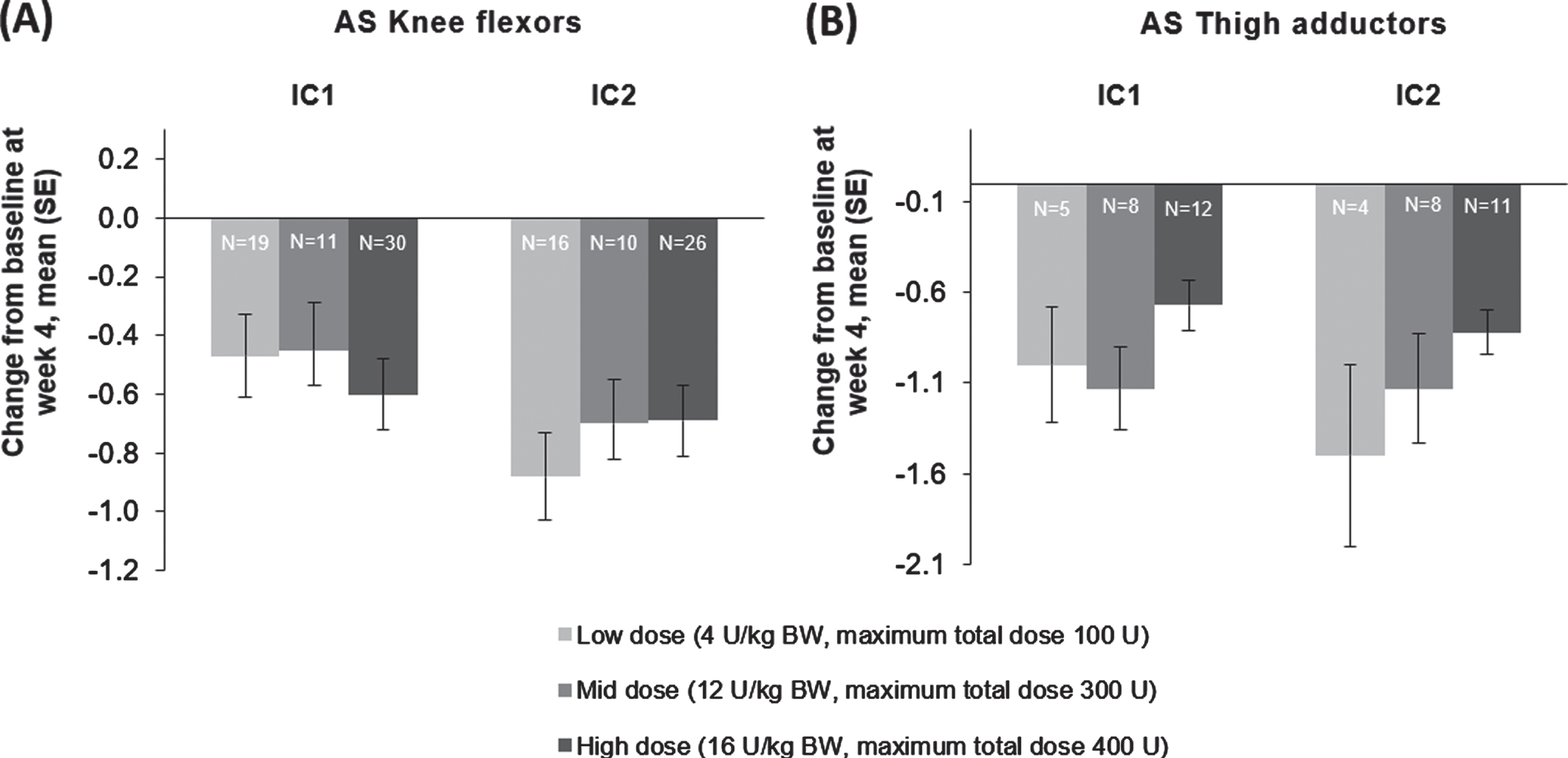
The effect of incobotulinumtoxinA on mean change from baseline at week 4 on AS as measured on the (A) knee flexors and (B) thigh adductor muscles, FAS, OC. AS = Ashworth Scale; BW = body weight; FAS = full analysis set; IC = injection cycle; kg = kilogram; OC = observed cases; SE = standard error; U = unit.
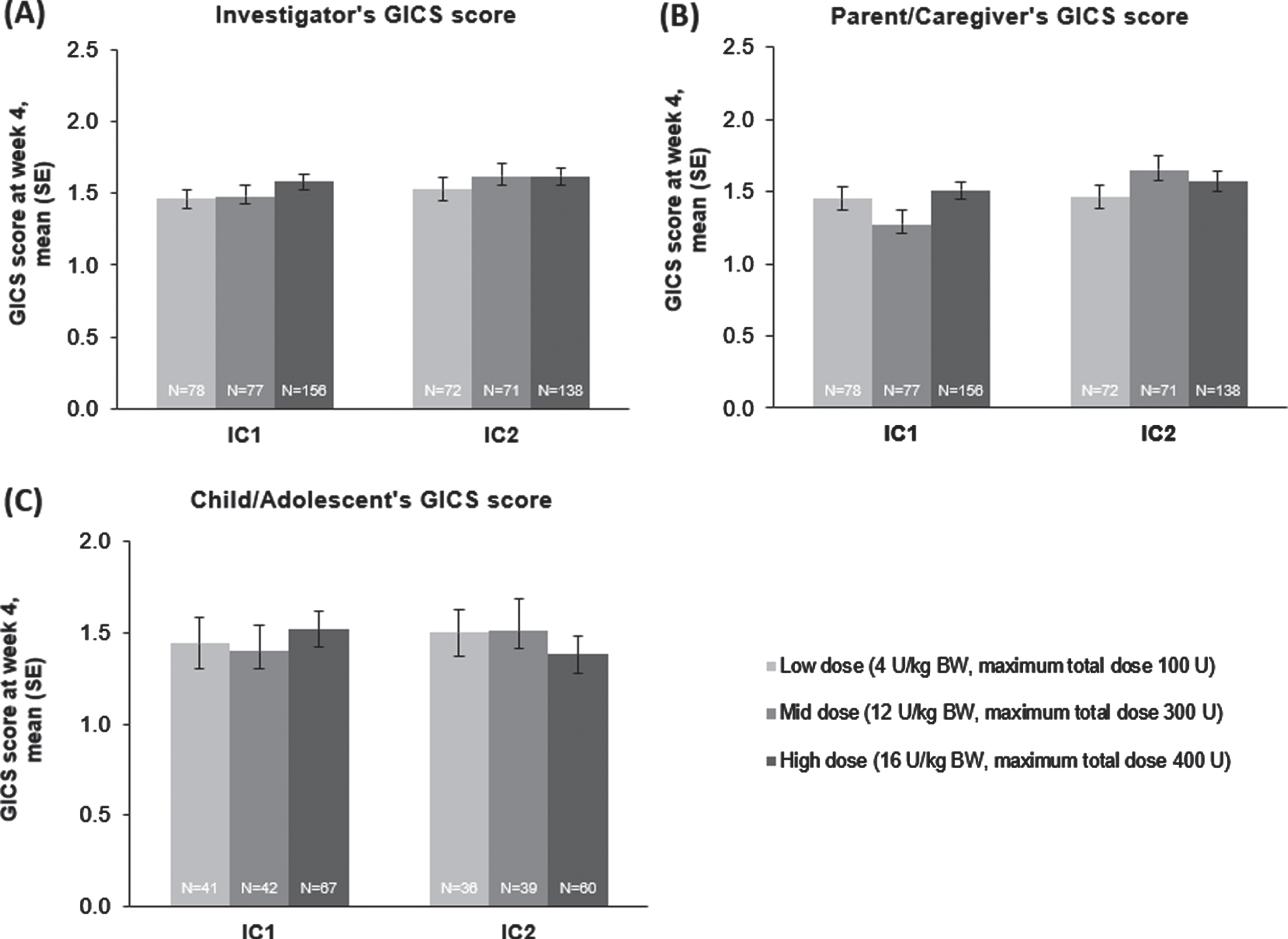
The effect of incobotulinumtoxinA on (A) investigator, (B) parent/caregiver, and (C) child/adolescent GICS scores†at week 4, FAS, OC. †GICS scores were available from 150 of 311 and 135 of 287 children/adolescents (48% and 47.0%) at IC1 and IC2. The proportion of children/adolescents responding was attributed to the respondents’ young age or their cognitive abilities. BW = body weight; FAS = full analysis set; GICS = Global Impression of Change Scale; IC = injection cycle; kg = kilogram; OC = observed cases; SE = standard error; U = unit.
