Abstract
PURPOSE:
Previous studies have found motor function to correlate with spinal motor level and, accordingly, individuals with spina bifida are frequently categorized clinically in this manner. The aim of the current study was to describe how lower extremity functions including strength, selective motor control, and mirror movements vary by motor level in children and young adults with spina bifida.
METHODS:
A single center, retrospective, cross-sectional, descriptive study using data collected in the National Spina Bifida Patient Registry and by a gait laboratory was performed.
RESULTS:
Seventy-seven individuals with spina bifida were included with the majority having myelomeningocele (59 lumbar, 18 sacral motor level). Lower extremity strength and selective motor control varied to a certain extent with motor level. However, 90% of individuals showed strength or weakness in at least one muscle group that was unexpected based on their motor level. Mirror movements did not clearly vary with motor level.
CONCLUSION:
Lower extremity strength, selective motor control, and mirror movements in individuals with spina bifida were not entirely predicted by motor level. This highlights the possible need for an improved spina bifida classification system that describes not only spinal motor level but more clearly defines a particular individual’s functional motor abilities.
Introduction
Spina bifida is one of the most common neural tube defects, affecting an estimated one in 2,758 births in the United States annually [1]. Less severe causes of spina bifida include spinal lipoma, in which a lipoma grows attached to the spinal cord [2]. Other subtypes, in ascending order of severity, include meningocele, which is characterized by failure of vertebral closure with resulting herniation of the meninges through the spinal defect, and myelomeningocele, in which the spinal cord, nerves and the meninges herniate through the defect [3]. Regardless of subtype, the condition frequently causes lower motor neuron and sometimes upper motor neuron injury [4]. Individuals with spina bifida commonly have associated conditions including Chiari II malformation, hydrocephalus, spinal cord tethering, musculoskeletal deformities such as hip subluxation or dislocation, as well as neurogenic bowel and bladder [3].
Several authors have asserted that the level of an individual’s neurologic lesion can help explain the interpersonal variability in rate of complications, including variable motor outcomes, seen in the spina bifida population [5–11]. Specifically, more caudal spinal lesions have been associated with better lower extremity strength [6, 8–10]. This association is partly responsible for the historical categorization of individuals with spina bifida by spinal motor level through various systems such as the Sharrard Classification System or the International Myelodysplasia Study Group Criteria [12, 13]. However, this historical understanding of spinal cord motor level correlation with motor function has been increasingly questioned by authors who have observed variable motor outcomes among children with similar motor levels [14–16]. As such, it remains challenging to accurately predict a child’s expected strength or gross motor ability based solely on their motor level. Better quantification of lower extremity motor function may therefore assist clinicians in providing improved counseling to patients as well as their families.
One factor that may contribute to the variability in motor outcomes in spina bifida is selective motor control. This is the ability of an individual to adequately isolate muscle activation in order to perform volitional movement [17]. Selective motor control has been identified as one of the impairments most strongly associated with impaired gross motor function in individuals with cerebral palsy [18]. However, selective motor control has not yet been systematically documented in the spina bifida population [19, 20].
Another factor that may contribute to the variability in motor outcomes in spina bifida are mirror movements, which are involuntary movements that appear in contralateral homologous muscles during voluntary ipsilateral movement. Mirror movements can be seen in the neurotypical population, especially before an individual is 10 years old, and during movements such as blinking or extreme exertion [21–23]. In contrast, pathologic mirror movements can be seen in a variety of congenital and acquired conditions such as Parkinson’s disease, hemiplegic cerebral palsy, callosal agenesis, stroke, and craniovertebral anomalies [24–27]. Several case reports and case series have noted mirror movements in spina bifida, most focusing on the upper extremities of individuals with cervical level spinal lesions [28–30]. Although lower extremity mirror movements have the potential to impair lower extremity function, no systematic assessment has explored the prevalence of lower extremity mirror movements in the spina bifida population.
The present study sought to systematically describe lower extremity strength, selective motor control, and mirror movements by motor level in children and young adults with spina bifida.
Methods
This was a retrospective, cross-sectional study that was approved by the University of Minnesota Institutional Review Board. After informed consent was obtained from participants or their caregivers, they were enrolled in the National Spina Bifida Patient Registry (NSBPR) at Gillette Children’s. The NSBPR is a collaborative study group associated with the Centers for Disease Control and Prevention (CDC) that collects data on individuals with spina bifida throughout the United States [31]. A subgroup of individuals participating in the NSBPR at Gillette Children’s who had undergone a physical examination during a clinical gait laboratory analysis were identified and included in the study if they had not previously opted out of the use of their records in research. Gait laboratory data were obtained by certified physical therapists. Gait laboratory data and NSBPR data could have been gathered asynchronously (i.e., years before or after each other). If a participant had more than one gait laboratory visit, data from the most recent visit was used.
Gait laboratory data extracted included strength, selective motor control, and mirror movements of the lower extremities. Strength was graded using a modified Medical Research Council scale as 0 (no contraction), 1 (trace muscle movement), 2 (full range of motion [ROM] with gravity eliminated), 3 (full ROM against gravity), 4 (full ROM against some resistance) or 5 (normal strength) [32]. Muscles graded included the hip flexors, hip adductors, knee extensors, ankle dorsiflexors, ankle invertors, hip extensors, hip abductors, knee flexors, ankle evertors, plantarflexors, hallux extensors, and hallux flexors. Lower extremity selective motor control was graded per the scale described by Gage et al. as 0 (no movement or completely patterned movement), 1 (partial isolation of movement), or 2 (complete isolation of movement) [33]. Only the left limb was analyzed for strength and selective motor control such that the statistical concept of independence of observations was not violated, ensuring that each data point was independent and a participant was not overrepresented in the dataset. Mirroring was evaluated in a subset of participants that had a gait laboratory evaluation video in which a physical therapist had seated participants demonstrate toe and foot movements including ankle inversion/eversion, dorsiflexion/plantarflexion, and toe flexion/extension. Mirroring was scored by two physiatrists (LEK and KFT) according to the Wood and Teuber scale: 0 (no imitative movements), 1 (barely detectable mirror movement), 2 (mild mirror movements), 3 (sustained, stronger mirror movements) and 4 (mirror movements sufficient to be comparable to movement on the volitional side) [34]. Disagreements were resolved through rater consensus. Individuals were not required to have complete data for inclusion in analysis for a given variable. Missing data were treated as missing and not imputed. For example, if an individual had missing mirror movements but had lower extremity strength and selective motor control, their data were included only in the strength and selective motor control summary statistics. Physical examination data across all strength and selectivity variables were missing for 0–13% of the sample, while mirroring data was missing for 44% of the sample.
The spina bifida motor level was extracted from the NSBPR. NSBPR motor levels were inputted into the data set by a research coordinator who obtained this information from clinical documentation. The NSBPR defines the motor level as being “the lowest level of independent movement which can be reproduced by the patient” –a definition similar to that of other authors who have published data from the NSBPR [35]. The outcome data used to determine the functional motor level was obtained through patient examination at the gait laboratory. The ability of an individual to move through at least full ROM with gravity eliminated (i.e., at least 2/5 strength) was operationally defined as reproducible, independent movement, which could be used in determining an individual’s motor level in a manner consistent with the NSBPR definition of functional level. In keeping with the NSBPR’s definition of spinal motor levels, individuals were graded as having the following levels: thoracic (flaccid lower extremities present), high-lumbar (hip flexion present), mid-lumbar (knee extension present), lower-lumbar (ankle dorsiflexion present), and sacral (ankle plantarflexion present) [35].
Unexpected strength was defined as at least 2/5 strength in muscles innervated caudal to the motor level, and unexpected weakness was defined as strength≤1/5 in muscles innervated at or cephalad to the motor level. Expected and unexpected innervation definitions were based on previous publications reporting lower extremity neurosegmental anatomy (Fig. 1) [13, 36]. Additionally, expected and unexpected innervation was based on an assumption that an individual with spina bifida may be more likely to have intact myotomes above versus below their spina bifida motor level, as is implied by many spina bifida classification systems [37].
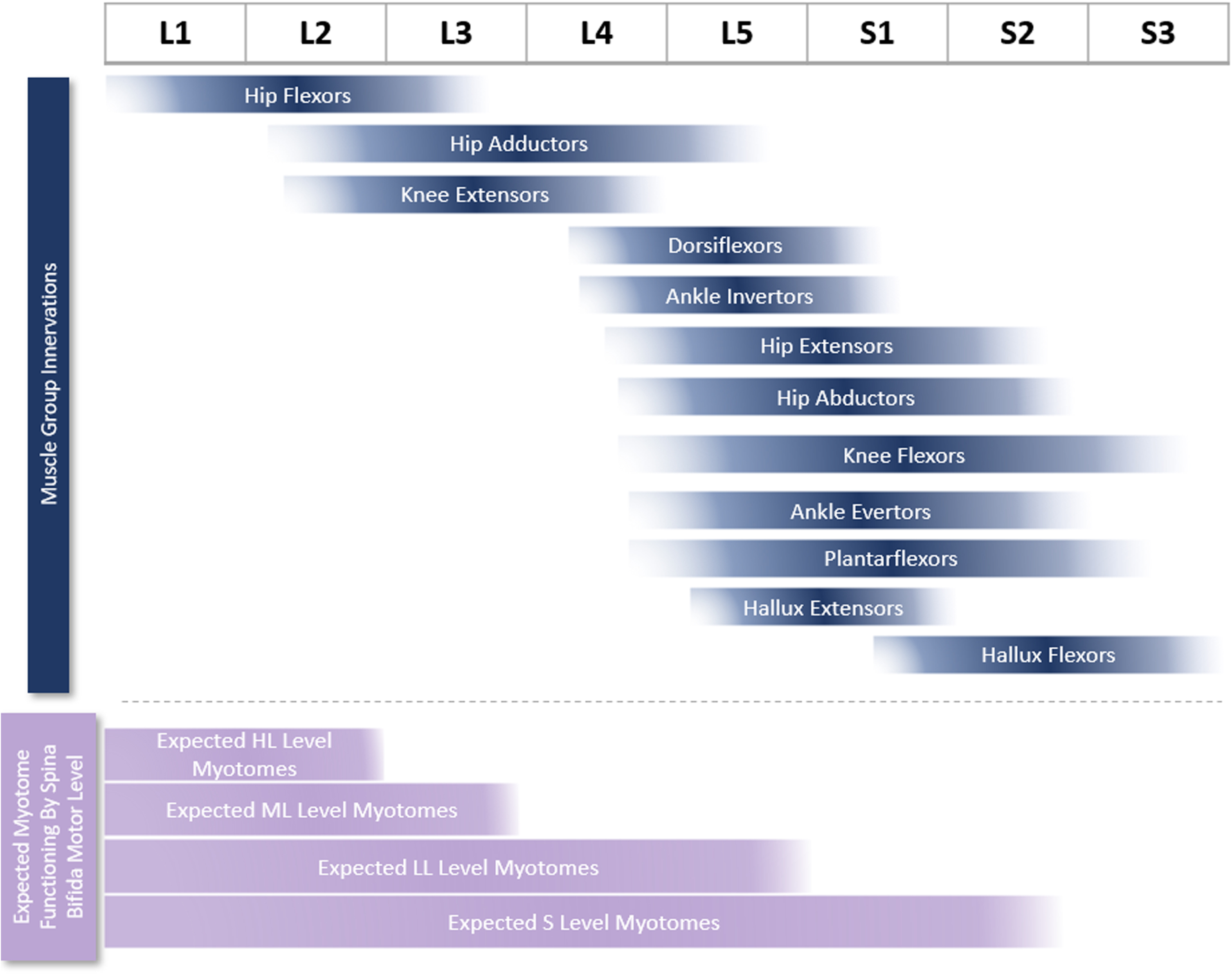
Lower extremity muscle group innervations [13, 36] and expected functioning myotomes for spina bifida patients with high-lumbar (HL), mid-lumbar (ML), lower-lumbar (LL), and sacral (S) motor levels. Myotome innervations were preferentially determined by a particular myotome showing an electrophysiologic response in > 50% of individuals stimulated at a particular ventral root level, as reported by Phillips et al. [36]. For muscle groups not studied by Phillips et al., myotome innervation was defined per Sharrard [13].
Treatments prior to the gait lab physical examination were also extracted from the NSBPR, including prenatal closure, as were treatments that may have occurred between NSBPR data collection and gait lab evaluation, including tethered cord release. Spina bifida type was categorized as myelomeningocele or other (including fibrolipoma of the filum terminale or lipoma of the spinal cord).
Summary statistics were calculated for each motor level group for this descriptive study.
There were 78 individuals who met the inclusion criteria, but since there was only one person with a thoracic motor level, aggregate data were only summarized for the 77 individuals in the high-lumbar, mid-lumbar, lower-lumbar, and sacral level groups (Table 1). Participants were 4.4–24.5 years old at the time of their physical examination, which occurred between 1995 and 2020. The majority did not have prenatal closure, had myelomeningocele, were community ambulators, and sometimes used a wheelchair. On average, the participants had the physical examination four years (standard deviation: 8) prior to NSBPR data entry. Therefore, participants were a mean of 15 years old (standard deviation: 8; range: 2–36) when ambulation status was documented in the NSBPR.
Characteristics of participants
Characteristics of participants
Δ Percentages are rounded up to the nearest whole number. ∈Spina bifida motor levels were determined per NSBPR diagnostic criteria: high-lumbar (hip flexion present), mid-lumbar (knee extension present), lower-lumbar (ankle dorsiflexion present), and sacral (ankle plantarflexion present). *Other types of spina bifida included fibrolipoma of filum terminale and lipoma of spinal cord.
There was a trend towards greater strength and selective motor control among those with more caudal motor levels (Figs. 2 & 3). However, 64% of individuals had at least one muscle group that showed unexpected strength based on their functional motor level and expected neurosegmental innervation (Fig. 4) [13, 36]. Specifically, there were 3–5 muscle groups for each level group (high-lumbar, mid-lumbar, and lower-lumbar) in which at least one-third of individuals had strength that would not have been anticipated based on their assigned motor level. For example, all three individuals with high-lumbar motor levels showed unexpected strength of the knee flexors, hip abductors, and knee extensors. There was also unexpected weakness in 36% of individuals from the lower-lumbar and sacral groups (Fig. 4). About one-quarter of individuals in the lower-lumbar group had weakness in the dorsiflexors, and almost two-thirds had ankle inverter and ankle evertor weakness. Additionally, over one-third of individuals in the sacral group had deficient strength in the ankle evertors, plantarflexors, hallux extensors, and hallux flexors. There was also a trend towards decreased lower extremity muscle strength in those with myelomeningocele versus participants with other causes of spina bifida (data not shown). For muscle groups with expected innervation for a given motor level, most individuals had at least partial selective motor control, though some unexpected deficits were again identified in the lower-lumbar and sacral groups (Fig. 3).
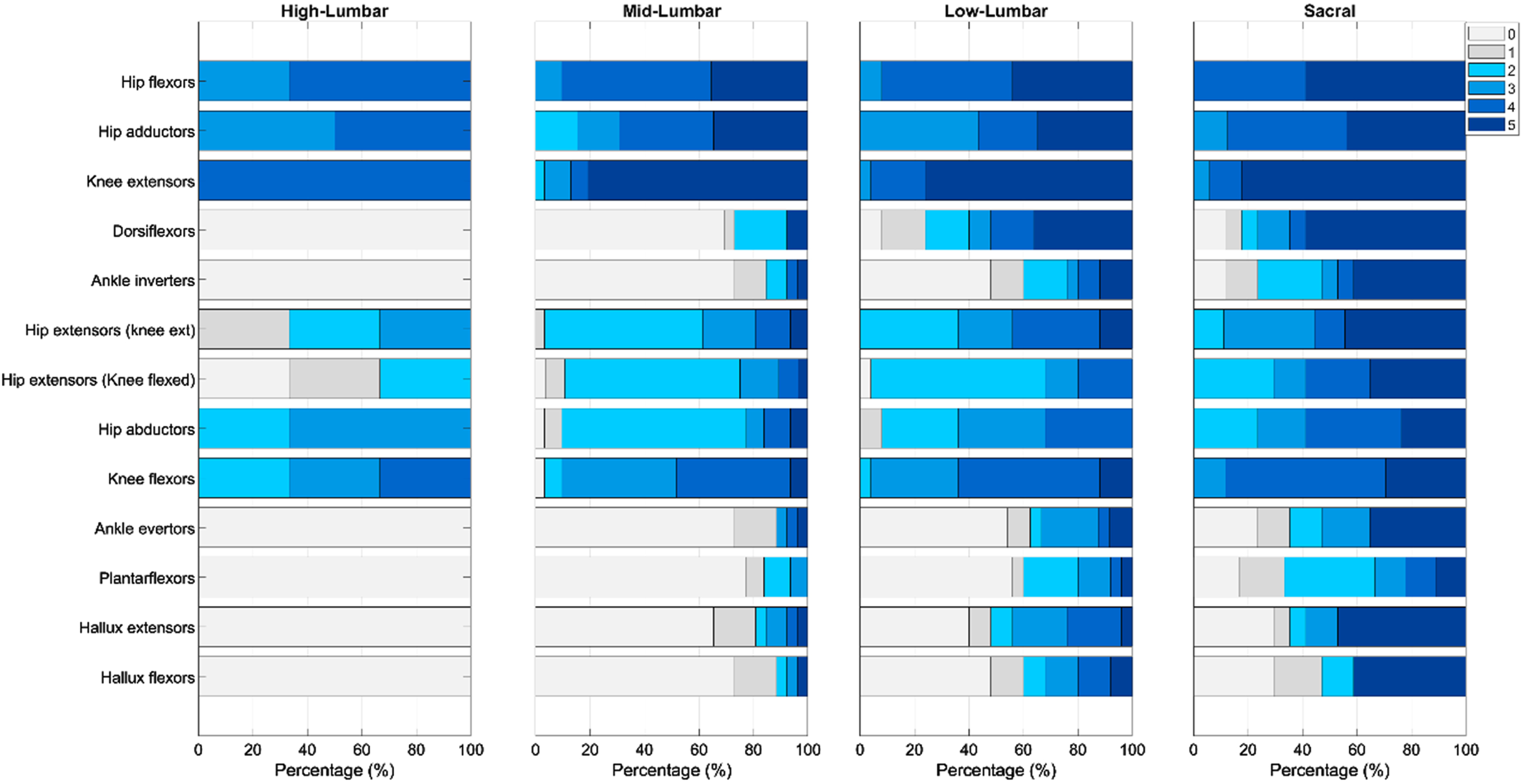
Strength of lower extremity muscles from cephalad to caudal level of expected innervation in spina bifida patients with high-lumbar, mid-lumbar, lower-lumbar, and sacral motor levels. Muscles were scored using the modified Medical Research Council scale as 0 (no contraction), 1 (trace muscle movement), 2 (full ROM with gravity eliminated), 3 (full ROM against gravity), 4 (full ROM against some resistance) or 5 (normal strength) [32].
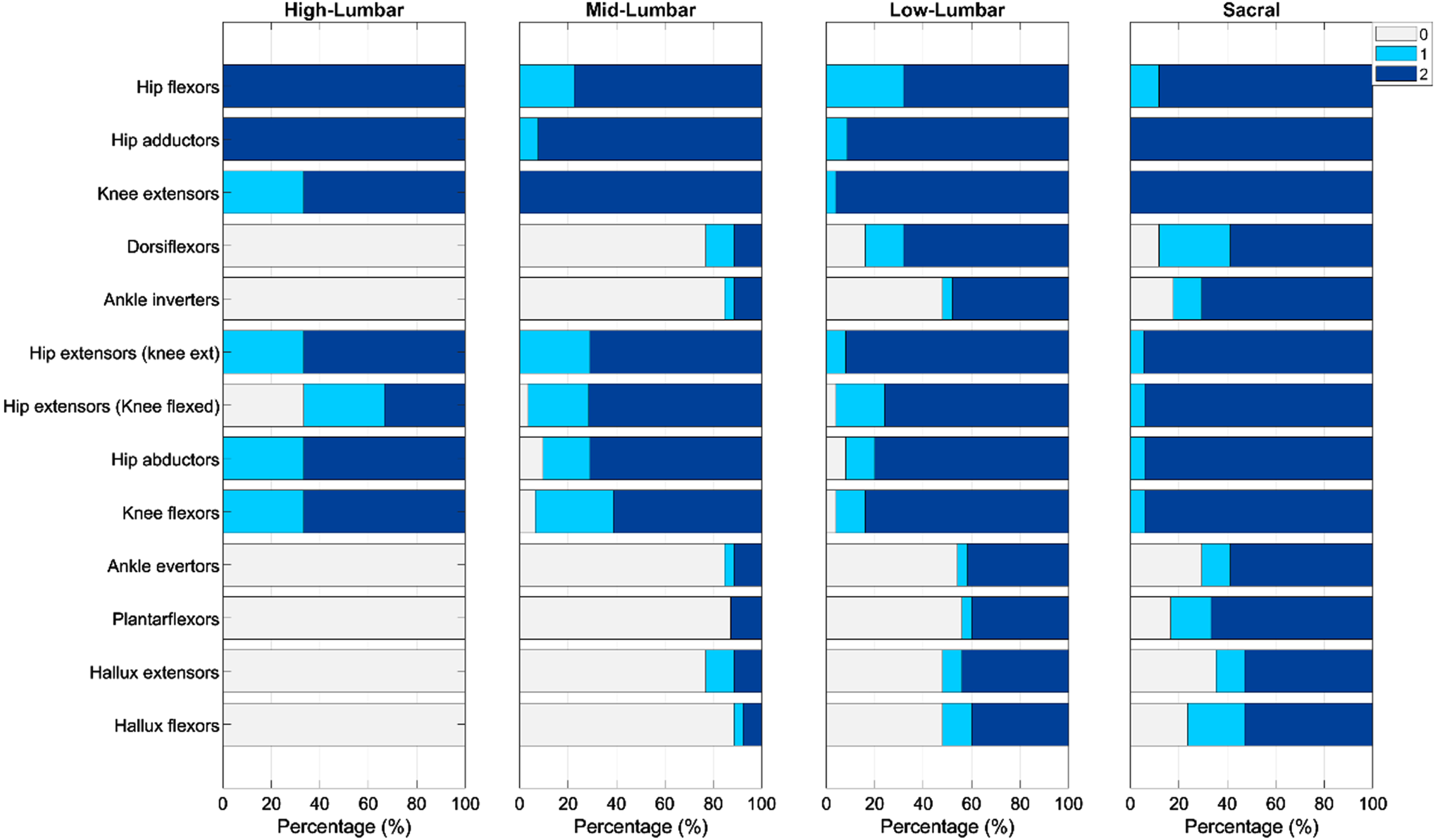
Selective motor control of lower extremity muscles from cephalad to caudal expected innervation as seen in spina bifida patients with high-lumbar, mid-lumbar, lower-lumbar, and sacral motor levels. Lower extremity selective motor control was graded according to Gage et al., where 0 = no movement or completely patterned, 1 = partial isolation, and 2 = complete isolation [33].
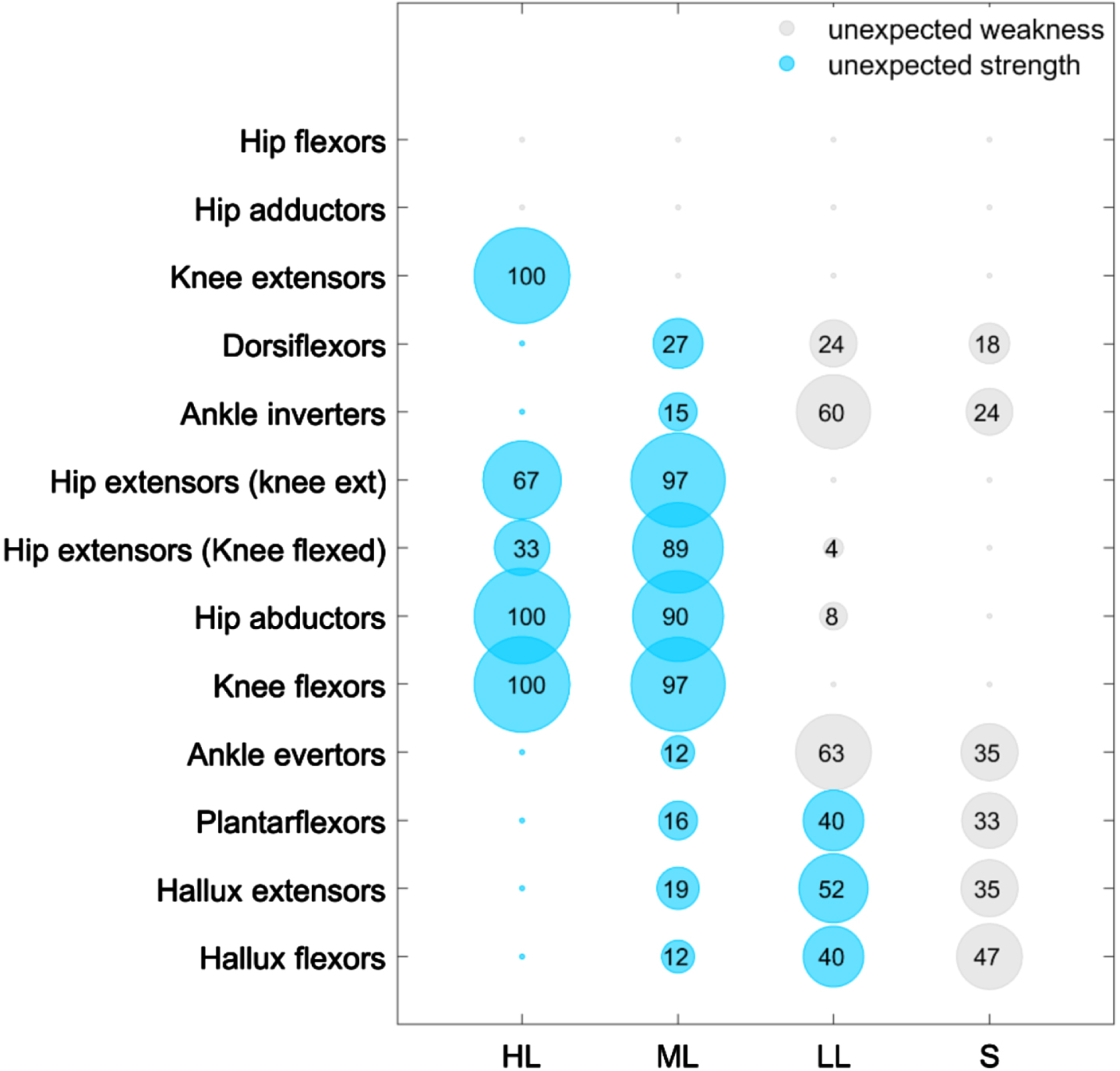
Unexpected strength and weakness in lower extremity musculature in spina bifida patients with high-lumbar (HL), mid-lumbar (ML), lower-lumbar (LL), and sacral (S) motor levels. Blue represents unexpected strength and gray represents unexpected weakness. The size of the circle and number indicate the percentage of participants for a given motor level having unexpected strength or weakness. The minute circles without numbers represent 0%. Unexpected strength was defined as at least 2/5 strength in muscles innervated caudal to the functional spinal level and unexpected weakness was defined as strength≤1/5 in muscles innervated at or cephalad to the functional spinal level.
A total of 44 individuals had gait laboratory functional foot videos available to analyze mirror movements (including the one individual with a thoracic motor level who scored a 3 but was not included in the summary data). Interestingly, mirroring occurred in the majority of these patients but did not differ much between mid-lumbar, lower-lumbar, and sacral groups (Fig. 5). A post-hoc analysis was performed to explore if mirror movements were related to participant age, but no significant trends were observed (data not shown).
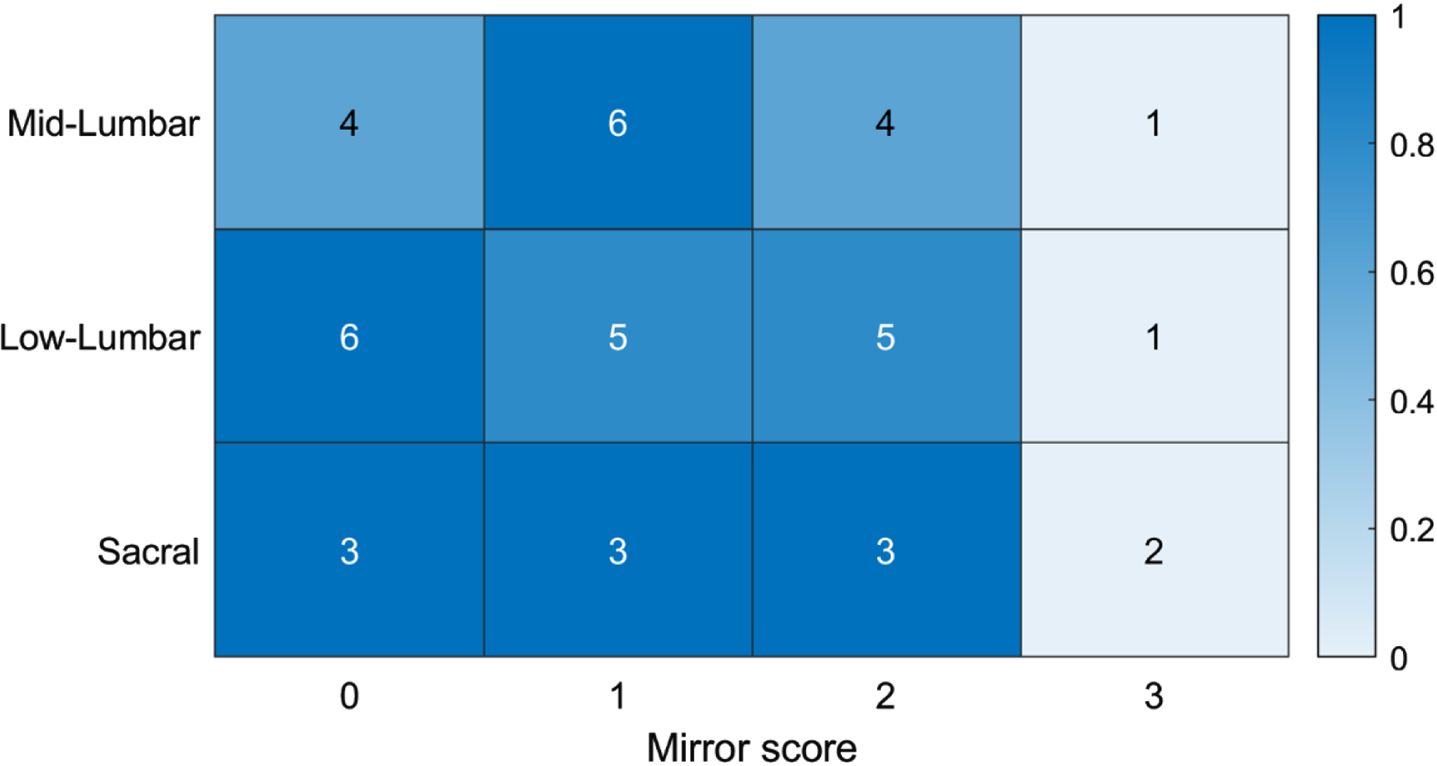
Lower extremity mirror movements of 43 individuals with mid-lumbar, lower-lumbar, and sacral spina bifida motor levels. The bar at the right and color coding represent the proportion of individuals within a certain functional spinal level that showed a specific mirroring score. Mirroring was scored according to the Wood and Teuber scale, as 0 (no imitative movements), 1 (barely detectable mirror movement), 2 (mild mirror movements), 3 (sustained, stronger mirror movements) and 4 (mirror movements sufficient to be comparable to movement on the volitional side) [34].
The present study described lower extremity strength, selective motor control, and mirror movements by functional motor level in the spina bifida population. Regarding lower extremity strength, the data showed a trend of more cephalad motor levels being associated with decreased muscle strength. Despite this trend, many individuals with spina bifida had functional strength that would not have been anticipated based on previous data reporting neurosegmental innervation of the lower extremities and the patients’ assigned motor level [13, 36]. Specifically, individuals with more cephalad motor levels tended to show better than expected strength, particularly in muscle groups located in the proximal lower extremity that were not anticipated to be innervated given their motor level. For example, most individuals with high- and mid-lumbar levels showed strength in the knee extensors, hip abductors, and knee flexors, which are all muscle groups with substantial innervation at or below L3 [13, 36]. Interestingly, this pattern of strength is similar to results in a study by McDonald et al., which found medial hamstring, quadricep, and gluteal strength showed stronger correlation with cephalad innervated muscles as compared to more caudal innervated muscles with which they should share similar innervation [13, 36]. Strength in these muscle groups may be particularly clinically relevant, as previous studies have reported that strength of the iliopsoas, quadriceps, and glutei are important determinants of an individual’s ambulatory status [38]. However, it should also be noted that, even among the individuals with low-lumbar motor levels, less than half showed strength in the plantarflexors, which play an important role in propelling the leg into swing phase and stabilizing the limb during the gait cycle [39, 40].
A number of individuals in the present study alternatively showed fewer muscles with at least 2/5 strength than would have been anticipated based on their functional motor level. This weakness was particularly seen in the dorsiflexors and ankle inverters of those with lower-lumbar and sacral motor levels. However, compared to patients with proximal weakness, isolated distal lower extremity weakness may not functionally impact patients as significantly given that distal weakness can be compensated for, to a certain extent, by ankle-foot orthoses [41]. Additionally, individuals in the present study with myelomeningocele appeared to have more lower extremity weakness compared to those with fibrolipomas of the filum terminale or spinal cord lipomas.
This variability in motor outcomes has been observed by a number of other authors [16, 42–44] and is likely due to several factors. First, if the spina bifida lesion results in not only central nervous system but also peripheral nerve injury, there may be improved reinnervation of proximal versus distal musculature as has been documented for other types of peripheral nerve injuries [45]. This reinnervation pattern may explain the unexpected strength in many proximal lower extremity muscles of those with more cephalad functional motor levels and unexpected weakness in many distal lower extremity muscles of those with more caudal functional motor levels. Additionally, although individuals with spina bifida have a neural tube defect, the severity of the lesion can differ such that certain individuals may have more or less motor function preservation caudal to the lesion [46]. Furthermore, some authors suggest that motor function can, to a certain extent, be improved in individuals with higher activity levels [47]. This variation is also likely due to interindividual variation in lower extremity neurosegmental innervation, with previous authors suggesting that 27.9% of lower limbs show innervation that differs from classically understood segmental innervation patterns [36]. In addition, there can also be intraindividual variation in innervation with the same individual having significant side to side differences in innervation and even differential innervation within the same muscle group [48, 49]. Lastly, comorbidities can result in additional neural injury, such as tethered cord and perinatal spinal hemorrhage, which may be culpable for poor motor outcomes in certain individuals with similar motor levels [10, 50–53].
Although others have previously examined patterns of strength in spina bifida, to the authors’ knowledge, the present study represents the first systematic examination of selective motor control in this patient population [5–8, 10]. Similar to the trends seen in lower extremity strength, there was a trend of better selective motor control with more caudal motor levels and in those muscle groups showing good strength. Interestingly, a similar association has been seen between selective motor control and strength in specific types of cerebral palsy that result in injury to the corticospinal tract, implying the possible role of corticospinal tract injury in causing similar motor impairments in the spina bifida population as well [54, 55]. Better understanding of impaired selective motor control is important as it may offer an additional target for physical therapy intervention to improve lower extremity motor function, particularly as partial recovery of selective motor control can occur through compensation by the rubrospinal tract [55].
Regarding mirror movements, some degree of mirrored movements of the lower extremities in spina bifida patients was common, but they were not strongly correlated with motor level or patient age, at least in this small sample. Although a few case reports and case series have reported mirror movements in the upper extremities, this is the first systematic study demonstrating that lower extremity mirror movements in the spina bifida population are a more common motor phenomenon than previously appreciated [28–30]. The etiology of mirror movements in spina bifida remains incompletely elucidated, but other authors have speculated that they may be due to injury of the lower medulla resulting in atypical development and perhaps insufficient decussation of the corticospinal tract, or alternatively they may be due to the inadequate pruning of the corticospinal tracts due to aberrant neuronal input after neuronal injury [55, 56]. Mirror movements may also be another therapeutic target for interventions (e.g., physical therapy) to improve lower extremity motor function in those with spina bifida.
Several limitations affect the generalizability and conclusions of the present study. First, the study was performed at a single pediatric hospital in the United States where the majority of the patients had lumbar level spina bifida, did not have prenatal closure of their spina bifida lesion, and had a diagnosis of myelomeningocele. Therefore, results may not be generalizable to the wider spina bifida patient population. In addition, a relatively small sample size was available for each motor level group, especially when evaluating mirror movements. Also, selection bias may be present as patients qualified for the study only if they completed a gait lab analysis, with gait data collection occurring between 22 years before to five years after NSBPR data collection. Furthermore, some of the variability, particularly in strength and selective motor control, could have been due to different therapists grading similar patients differently. It may also reflect a change in clinical status (e.g., tethered cord) occurring between gait laboratory and NSBPR data collections; however, tethered cord release only occurred for 13% of the sample between these data collection episodes. Additionally, the use of at least 2/5 as the cutoff to define functional motor level, which was used to ensure that the study was consistent with diagnostic criteria established by the NSBPR, would lead to differential rates of unexpected versus expected strength as compared to using at least 3/5 strength. Lastly, due to the retrospective nature of the study, different activity levels among participants could not be documented or controlled for, which may explain some of the variation in motor outcomes among individuals.
Future studies in this area are needed to clarify several remaining questions. These include exploring the implication of and possible therapeutic interventions to address impaired selective motor control and mirror movements, which in turn could improve function and quality of life. Next, further research is needed to determine if timing of spina bifida lesion repair affects strength, selective motor control, and mirror movements. Finally, if the unexpected motor findings in this study are found to be clinically meaningful, this may emphasize the need for more consistent and functionally based classification of spina bifida patients in addition to motor level. Such a classification system could improve patient and family counseling, better identify a worsening superimposed pathologic process (e.g., tethered cord), and provide a meaningful covariate for research. This system could be a new classification scheme or an adaptation of an existing framework such as the Dias Functional Classification of Myelomeningocele, which has been shown to have good correlation with real-world lower extremity function including walking [57, 58].
In conclusion, results of the present study indicate that muscle strength and selective motor control tend to be more intact among spina bifida patients who have more caudal spinal motor levels. However, approximately 90% of individuals with spina bifida show either preservation or loss of strength that would not be anticipated based on functional motor level classification [13, 36]. Additionally, lower extremity mirror movements may be a more common motor phenomenon than previously appreciated, based on a small sample of spina bifida patients. Additional research will be required to further determine the clinical and functional impacts of these unexpected motor findings. If clinically meaningful variability in lower extremity motor function among individuals with similar motor levels exists, there may be a need for the routine use of an improved, more functionally meaningful spina bifida classification system that does not rely solely on motor level to describe an individual’s functional abilities.
Footnotes
Acknowledgments
We are grateful to the patients and families who participated in the study. We also appreciate the efforts of Maykala Owens, Aleksys Patterson, Victoria Blake and Rhonda Cady who help to maintain and assisted with data extraction from the Gillette based NSBPR. The authors acknowledge the Endowed Fund for Research in Cerebral Palsy Treatment of Gillette Children’s as the funding source for author ERB. This project is also supported in part by the generous donors to Gillette Children’s and by the CDC under the Cooperative Agreement for Award Number 5U01DD001268. The content is solely the responsibility of the authors and does not necessarily represent the official views of the CDC.
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical considerations
This project was approved by the Institutional Review Board at the University of Minnesota with approval numbers 1410M54583 (approved 2/28/2019) and STUDY00005641 (approved 1/15/2015). Informed consent was obtained from participants or their caregivers during the course of the study.
