Abstract
PURPOSE:
The incidence of dystonic cerebral palsy causing significant morbidity is on the rise. There is a paucity of evidence for the management of dystonia in children.
METHODS:
Forty-one children aged 6 months-5 years with predominantly dystonic cerebral palsy were started on a predetermined protocol of trihexyphenidyl (0.25–0.52 mg/kg) and followed up at 3, 6 and 12 weeks. Dystonia severity, motor function and developmental age at baseline and 12 weeks were compared using the Global Dystonia Scale (GDS), the Gross Motor Function Measure (GMFM), and Fine Motor/Perceptual Subscale of the Early Developmental Profile-2. Thirty-four children completed the entire 12 weeks of intervention.
RESULTS:
The mean age of participants was 25±11 months. A significant decrease in median total dystonia scores on the GDS was observed post-intervention (74.5 to 59, p < 0.0001), and 64% of participants gained motor milestones. GMFM scores increased significantly from a median of 19.8% pre-intervention to 26.5% post-intervention (p < 0.0001). There was improvement in the fine motor domain as compared to the baseline (p < 0.0001). The number of children classified at Gross Motor Function Classification System levels 1 and 2 increased to 47.05% from 5.88% in the pre-intervention group.
CONCLUSION:
Trihexyphenidyl significantly improved dystonia, motor function and development in children with dystonic cerebral palsy in this study. Additional studies are needed to clarify its role in larger numbers of children with this condition.
Introduction
Cerebral palsy (CP) is a common neurodevelopmental disorder with a reported pooled global prevalence of 2.11 per 1000 live births [1], and recent systematic analysis from India has shown comparable results [2]. However, because of constraints in both quality and accessibility of health care, as well as the country’s large resource and rural-urban divide, the actual statistics in India may be substantially higher. Though there is limited evidence for the beneficial effects of early intervention on motor development, there is growing interest in diagnosing CP as early as 6 months of age or even earlier [3–5]. A study of 1000 patients with CP in northern India found that 73% had spastic CP and 7% had dyskinetic/athetoid CP [6]. Recent studies suggest that the frequency of dystonic CP is higher, 4–17% of cases, even in children with normal birth weights [6, 9].
At present, spastic CP accounts for the vast majority of cases worldwide [7, 8] but there has been a growing interest in understanding the dystonic component in children previously diagnosed with spastic CP [10, 11]. Dystonic CP is frequently linked with significant movement impairment as well as other impairments such as seizures, intellectual disability, and vision, hearing, communication, swallowing and feeding problems, all of which have a detrimental impact on the individual’s and caregiver’s quality of life [12]. Early focused intervention may aid in reducing morbidity and enhancing treatment quality; however, there are few therapy choices available, particularly for children [13].
Baclofen, benzodiazepines, clonidine, gabapentin, levodopa, trihexyphenidyl, and tetrabenazine are some of the oral drugs recommended for children with CP who have dystonia. Trihexyphenidyl (also known as benzhexol) is a selective muscarinic acetylcholine receptor antagonist that blocks cholinergic activity both centrally and peripherally. According to recent hypotheses, the striatal cholinergic system appears to be retained or uncontrolled following hypoxic-ischemic damage, as evidenced by an increase in cholinergic fibers and cell bodies as well as acetylcholine release [14].
Trihexyphenidyl has been demonstrated to be beneficial in treating dystonia in older children and adults in a few trials; however, the majority of these used subjective modes of assessment [15, 16]. The purpose of this research was to objectively investigate the effect of trihexyphenidyl on dystonia in young children. It is hypothesized that there might be a likelihood of better results in the period of rapid neural development, and hence this study was conducted in young children with dystonic CP.
Methods
This study was an open-label single arm interventional trial conducted at a tertiary hospital. Children aged 6 months to 5 years, diagnosed with CP according to American Academy of Neurology criteria, and having significant motor delay and abnormal neuro-motor presentation in the form of variable hypertonia and/or abnormal twisting postures were eligible to participate in the study. The trial excluded children with poorly controlled epilepsy, glaucoma, significant cortical visual impairment, congenital heart block, or a history of anti-cholinergic medication allergy or adverse response. Children who received trihexyphenidyl/ levodopa or titration of baclofen doses in the previous 3 months and those who received botulinum toxin or underwent orthopedic surgery in the previous 6 months were also excluded.
The children were assessed for inclusion in the study by the principal investigator (a paediatric postgraduate resident). When clinically appropriate, screening for inborn errors of metabolism was performed as a part of routine care to rule out progressive disease. Genetic mutations, on the other hand, could not be tested in individuals without clear aetiology due to lack of resources. The study procedure was described to the parents and, after obtaining a written informed consent, children who met the study requirements were recruited.
This study was given due ethical clearance from the institutional ethical committee.
Study flow
As no published studies were found that examined the effect of trihexyphenidyl using the Global Dystonia Scale (GDS), Gross Motor Function Measure (GMFM), or Early Developmental Profile-2 (EDP-2) scores as the primary end measure in children with dystonic CP, the sample size for this study was calculated based on the difference in Melbourne Assessment scores between baseline and 15 weeks [25]. To detect a mean change of 2.97 with a standard deviation of 6.69, a sample size of 57 with a threshold of significance of 5% (single tailed) and power of 95% was required. A convenience sample of at least 30 children was chosen after estimating the number of children with predominantly dystonic CP who met the study criteria and could be followed at the centre at specified intervals during the study period.
Baseline evaluation
A detailed pro forma listing the patient’s medical history, developmental status, presence of co-morbidities, anthropometry as per the World Health Organization and Indian Academy of Pediatrics charts [17, 18], and neuroimaging findings was filled out. The children’s functional status was assessed using the Gross Motor Function Classification System (GMFCS) scale [5, 19].
Clinical assessment including comprehensive prenatal/perinatal/neonatal history, physical examination, neuro-imaging reports and other relevant tests were used to ascertain the aetiology of CP. A designated radiologist at the centre analysed and reported the neuroimaging of all the study participants.
Assessment measures
The GDS, the GMFM, and the Fine Motor/Perceptual Subscale of the EDP-2 were used to assess the severity of dystonia and motor function of the children at the beginning of the study [21]. The assessment was repeated at 12 weeks. GDS and GMFM assessments were performed by an occupational therapist or physiotherapist; the EDP was scored by a psychologist.
After the initial evaluation, all children included in the trial were started on oral trihexyphenidyl based on a predetermined age-based protocol. At the initial visit, the drugs for the first three weeks were provided, and parents were asked to return for follow-up visits at 3 and 6 weeks, during which drug dispensing, titrating of dosages and side effects monitoring were done. The 6-week ceiling dosage was maintained for another 6-week period. Besides the study medication, all the enrolled children continued to undergo occupational therapy and/or physiotherapy and received additional drugs (anti-epileptic medications, baclofen at stable doses and/or nutritional supplements) as decided by the treating unit during the study period. The doses of baclofen were not changed during the trial. Periodic telephone reminders were offered to assure compliance and minimize follow-up loss.
Post intervention assessment
After 12 weeks of treatment, the severity of dystonia and motor function were assessed using the measures stated above. Functional status was examined using the GMFCS.
Assessment of measures
The three main outcome variables were change in GDS score, change in GMFM score, and change in EDP-2 Fine Motor/Perceptual Subscale score.
The GDS, a scale for assessing the severity of dystonia, is used to score severity based on a Likert scale ranging from 0–10 in ten different body regions: 1) eyes and upper face, 2) lower face, 3) jaw and tongue, 4) larynx, 5) neck, 6) shoulder and proximal arm, 7) distal arm and hand including elbow, 8) pelvis and upper leg, 9) distal leg and foot, and 10) trunk. The total score is the sum of the individual scores in all the body areas. Both sides are assessed individually to give a maximal total score of 140. A higher score indicates more severe dystonia. The pre- and post-intervention scores were calculated and compared.
The GMFM is a clinical tool designed to evaluate change in gross motor function in children with cerebral palsy. Items on the GMFM span from activities in lying and rolling to walking, running and jumping skills. The GMFM requires the child to demonstrate various motor skills as outlined in the GMFM administration and scoring guidelines. There is a four-point scoring system for each item on the GMFM. Specific descriptors for scoring items are detailed in the administration and scoring guidelines. The GMFM item scores can be summed to calculate raw and percent scores for each of the five GMFM dimensions, selected goals areas and a total GMFM score.
The EDP-2 is a lesser-known developmental assessment scale in which different domains are assessed such as gross motor, fine motor, cognition, language, socio-emotion, feeding, toilet, and dressing in children aged 0–36 months. The EDP-2 was scored and interpreted as per the manual.
Statistical analysis
The data was entered in a Microsoft Excel spreadsheet. Children who were lost to follow-up were excluded and per protocol analysis of the remaining children was done using Statistical Package for Social Sciences (SPSS) version 21.0. A p-value of < 0.05 was considered statistically significant. Categorical variables such as sex, presenting complaints, socioeconomic status, comorbidities, GMFCS levels, and Developmental Quotient (DQ) were presented in number and percentage (%). Continuous variables such as age, developmental age, and scores were presented as mean±SD/median. GDS and GMFM scores were presented in the median and interquartile range (IQR). The normality of data was tested by the Kolmogorov-Smirnov test. A Wilcoxon signed-rank test was used to compare the pre- and post-intervention GDS, GMFM and EDP-2 scores. Univariate linear regression was used to determine significant factors affecting improvement in GDS and GMFM scores. The independent variables were parameters such as age, anthropometry, and baseline GDS, EDP-2, DQ, and GMFM scores.
Results
During the study period, 238 children met the inclusion criteria. Parents of 69 children denied consent because they did not live in the study area and would be unable to attend planned follow-ups for the duration of the study. Ninety-one children who already received trihexyphenidyl for more than 3 months, 9 children with poorly controlled seizures, 17 children on increasing dosages of anticholinergic medications (glycopyrrolate) and 11 children with significant cerebral visual impairment were excluded. A total of 41 children were enrolled, of which five were lost to follow-up (four families relocated and one child lost a parent). Two children were unable to complete the study owing to intolerable side effects. 34 children completed the entire 12-week intervention period.
The flowchart depicting the process of inclusion and exclusion of participants is illustrated in Fig. 1.
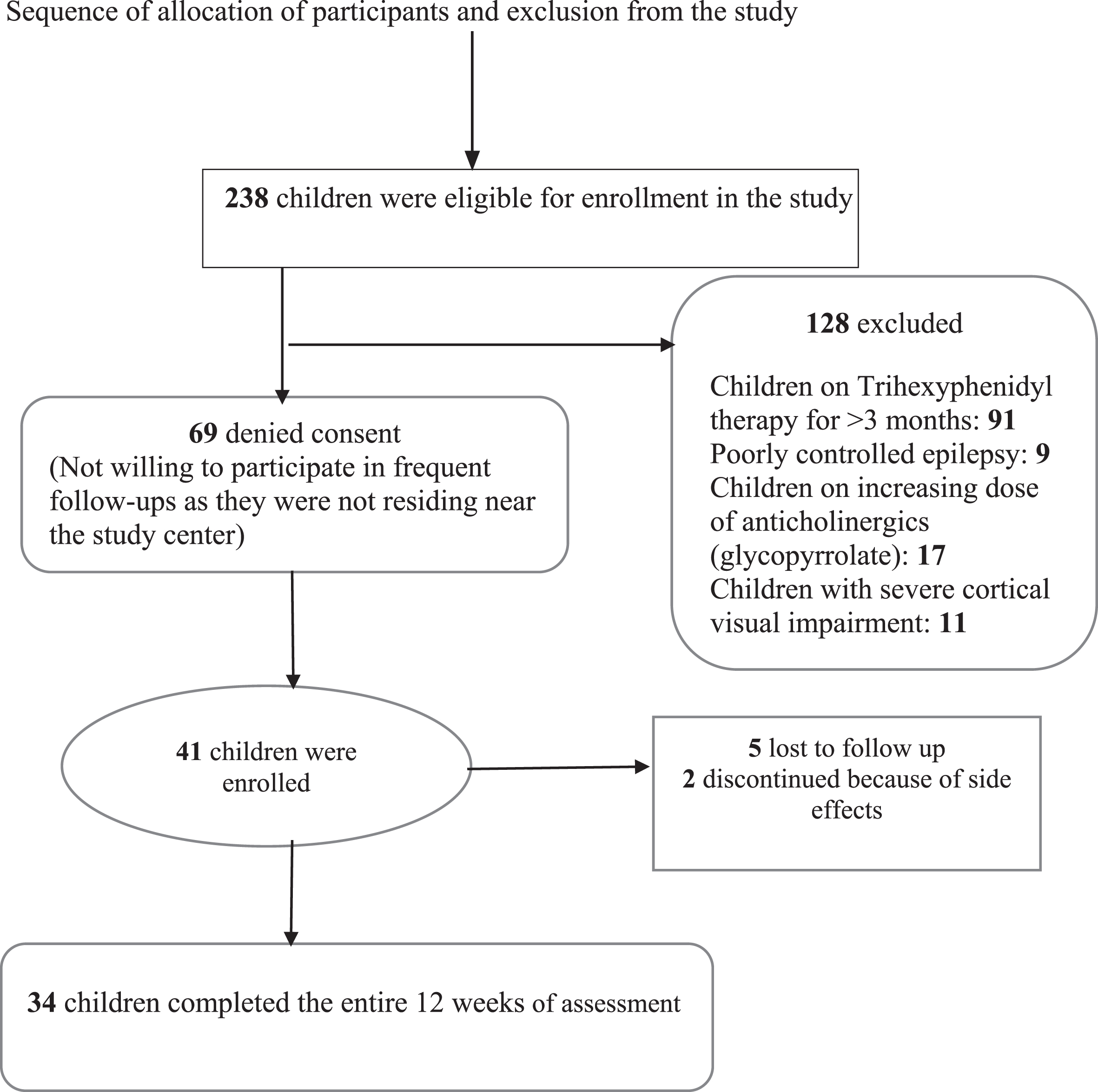
Flow chart depicting sequence of allocation of participants.
Demographic profile
The mean age of the participants was 25.09±11.89 months. The majority (18, 52.94%) of them were in the 13–24 month age group (Table 2). Eighteen (52.94%) were male. Developmental delay was the most common presenting complaint, seen in 31 (91%) children, and 11 (32%) presented with intermittent twisting of limbs and abnormal postures (Table 2).
Dose of trihexyphenidyl by age
Dose of trihexyphenidyl by age
Table depicting weekly Trihexyphenidyl dosage schedule. Tid: thrice daily dosing.
Baseline evaluation
All children had at least one comorbidity, the most frequent of which were speech problems such as delayed or unclear speech in 33 (97.05%) children, followed by feeding problems in 31 (91.17%) and constipation in 16 (47.05%) children (Table 2). Behavioural problems were seen in 24 children, of whom 23 had temper tantrums and one was diagnosed with autism spectrum disorder.
Aetiology
Perinatal asphyxia was found in 25 (73.52%) children and was the most common etiological diagnosis. When neonatal admission records were unavailable, a probable diagnosis of birth asphyxia was made based on history of delayed cry at birth, resuscitation requirement, poor feeding, irritability/lethargy, and/or seizures within 3 days of birth, supplemented by neuroimaging findings. Bilirubin Induced Neurological Dysfunction was documented in only 2 children (5.88%) [Table 2].
Neuroimaging
All 34 children underwent non-contrast computed tomography (NCCT) or magnetic resonance imaging (MRI), 13 and 21 children respectively. Twenty children (58.8%) had abnormal findings, out of which hypoxic ischemic encephalopathy (HIE) sequelae was the most common finding, seen in 12 children. The other findings were periventricular leukomalacia, periventricular bleed, and infarcts (Table 2). Neuroimaging was normal in 14 children (11 with NCCT and 3 with MRI); however, the neonatal discharge papers for 11 of them had a diagnosis of HIE. In the remaining three children, the aetiology was unknown. Among six children who had a non-contributing perinatal history, three children had neuroimaging suggestive of HIE; metabolic workup at the time of presentation to the study centre turned out to be normal (Figs. 23).
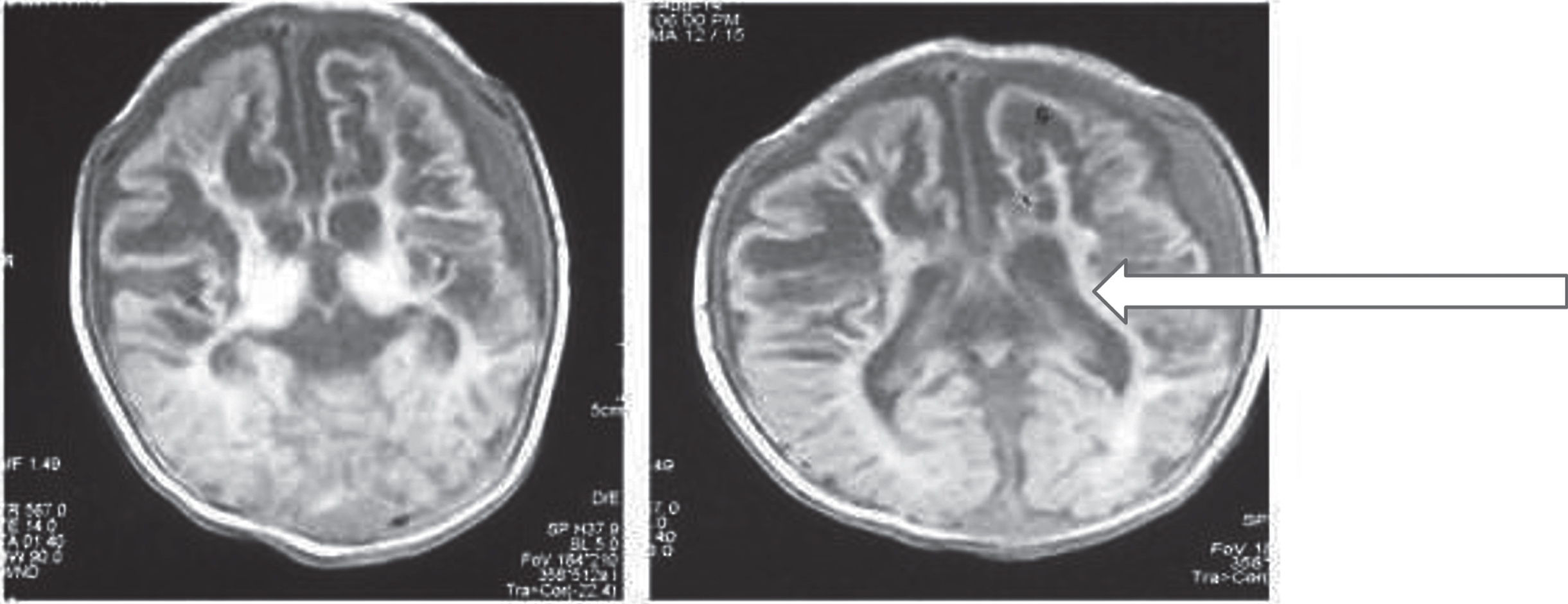
Brain MRI showing lamina necrosis in bilateral basal ganglia.
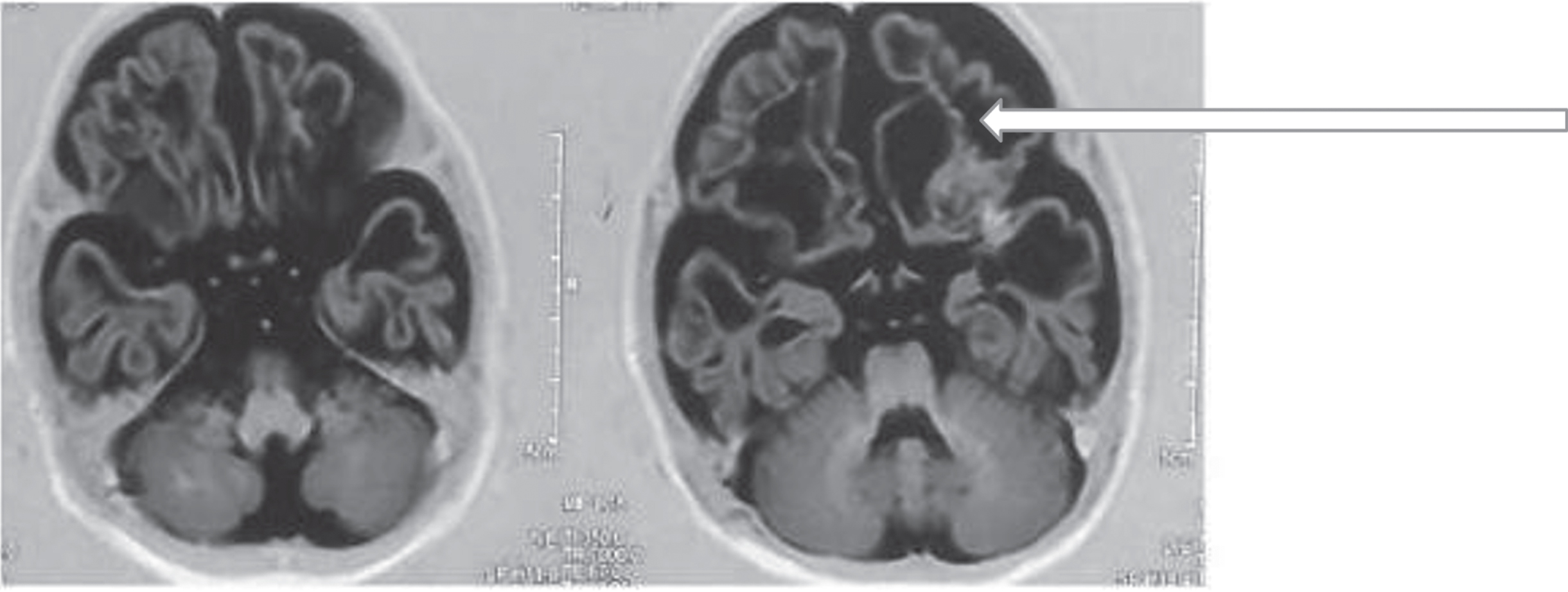
Brain MRI showing bilateral cystic encephalomalacia.
Initial developmental assessment revealed a DQ > 70 in 3 children (8.8%) and DQ 50–70 in 11 (32.3%), while DQ < 50 was seen in the majority: 20 (58.82%) of the participants. Median baseline developmental age (in months) in the domains of gross motor (7.12 IQR [3.3–11.5]) and fine motor (8.9 IQR [4.7–16]) were lower than that of cognition (8.6 IQR [6.5–20]), language (10.4 IQR [5–18.2]), socio-emotional (10.25 IQR [7–25.6]), and feeding (10.8 IQR [7–17.6]). The median baseline total GMFM score was 19.8% (IQR [10.1–24.600]) and total GDS score was 74.5 (IQR [64–92]). Gross motor functional ability measured using the GMFCS was assessed for all children at baseline (Table 6). The baseline data of those lost to follow-up were similar to the rest of the study population.
GMFCS levels pre- and post-intervention
GMFCS levels pre- and post-intervention
Table depicting change in GMFCS scores after 12 weeks of trihexyphenidyl.
Post intervention outcomes
Comparison of primary outcome variables
A significant decrease in median dystonia scores measured using the GDS pre- (74.5 [64–92]) and post-treatment (59 [47–92]) with trihexyphenidyl was observed in the total (p < 0.0001) as well as individual body regions after 12 weeks (Table 3).
GDS scores pre- and post-intervention
Table depicting change in GDS scores after 12 weeks of trihexyphenidyl in different body areas. *p-values were tested on the change in GDS scores using a Wilcoxon. Signed rank test.
There was a significant increase in GMFM scores in the dimensions of lying, rolling, sitting, crawling, and standing as compared to initial values, indicating improvement in motor function (Table 4).
GMFM scores pre- and post-intervention
Table depicting change in GMFM scores after 12 weeks of trihexyphenidyl; therapy in different motor skills. *p-values were tested on the change in GMFM scores using a Wilcoxon; signed-rank test;
Post-intervention fine motor median developmental age measured using the EDP-2 changed on the right side from 8.9 to 11.15 months and on the left side from 8.5 to 9.5 months (p < 0.0001). Other domains of gross motor, cognition, language, and socio-emotional also showed a significant gain (p < 0.0001) [Table 5]. Twenty-two children documented a gain in motor milestones over 12 weeks. Six children achieved standing, five children went to ‘sitting’ from ‘no sitting,’ and four achieved supported sitting. However, there was no significant change in the DQ post-intervention.
EDP-2 pre- and post-intervention
Table depicting change in the developmental age in various domains after; 12 weeks of trihexyphenidyl therapy. *p-values were tested on the change in the scores of EDP-2 using a Wilcoxon; signed-rank test;
Post-intervention GFMCS assessment revealed that 47.05% of children were classified as GMFCS level 1 or 2 in contrast to only 5.88% in the pre-intervention group. At the end of 12 weeks, 35.28% were still at level 4 or 5 (Table 6).
Stratified analysis showed no significant differences in the change of GDS, GMFM, or EDP-2 scores or GMFCS levels among those who had dystonic CP due to perinatal asphyxia versus other aetiologies.
Univariate analysis was also done to study factors such as age, anthropometry, baseline individual scale scores, and aetiology (perinatal asphyxia versus other causes) affecting the effectiveness of trihexyphenidyl on dystonic CP. However, none were found to be significant.
Side effects
Amongst the patients who completed the study, three had constipation (managed with dietary modification and oral laxatives) and one child reported mild irritability. Two children discontinued medication due to intolerable side effects (increased irritability in one and severe constipation in the other). They were not included in the final analysis.
Dystonia has been identified as the second most common cause of moderate to severe pain in patients with CP; hence measures to control dystonia are essential to improve their quality of life [22]. Prevalence of birth asphyxia in developing countries is quite high, with around 6% of term new-borns being asphyxiated, so dystonic CP is likely to be much more prevalent in India, albeit commonly unrecognized [23].
After publication of the international guidelines for early diagnosis of CP in 2017, there has been an increasing emphasis on early accurate diagnosis of CP so as to utilize neuroplasticity optimally and start early intervention [4, 5]. Keeping abreast of the latest information and guidelines, the study centre has also made early diagnosis and intervention of CP a priority. In view of the benefits of early intervention, this study attempted to assess objectively the effect of trihexyphenidyl on dystonia and functional status of young children with CP.
The results of the present study are encouraging and showed a positive quantifiable effect of the drug on the severity of dystonia, functional status, and motor ability as well as gain in milestones. After 12 weeks of receiving trihexyphenidyl, significant decreases in the total and individual median dystonia scores were observed in most body regions (Table 3). Seventy-five percent of the participants showed improvement in dystonia in upper limbs and 61% in lower limbs as per the GDS. Reflecting improvement in oromotor dystonia, there was also a significant reduction in GDS scores in the lower face, jaws and tongue, and larynx in 55.8%, 82.3%, and 76% of children respectively. Surprisingly, GMFCS levels also improved in the study population, which could be because of the relative instability of GMFCS scores in younger children [5, 24]. None of the participants had complete resolution of symptoms, thus ruling out any transient dystonia.
Sanger et al. [25] conducted a similar prospective open-labelled study in an older population of 23 children (median age of 8 years vs. 2 years, one month in the present study). That study demonstrated objective motor improvement in essential functional upper extremity movement such as reach, grasp and release using the Melbourne Assessment of Unilateral Upper Limb Function at 15 weeks of treatment. However, they documented no change in GMFM scores and severity of dystonia. They used the Burke-Fahn-Marsden dystonia scale [26] and Barry-Albright scale [27] for documenting change in dystonia. Unlike the GDS, both of these scales grade dystonia based on the percentage of time dystonia is present in a particular region, which may not change significantly in children with dystonic CP wherein dystonia is present during most awake periods. Another reason for better results in the present study could be that younger children have more neuroplasticity. Also, even though maximum doses of trihexyphenidyl were similar (0.7 vs. 0.75 mg/kg/day) in both the studies, it was continued for only 4 weeks in study by Sanger et al. while it was continued for 6 weeks in the present study.
In fact, in a randomized, double-blinded, placebo-controlled, crossover trial with 16 participants aged between 2 and 18 years with predominant dystonic CP, Rice et al. [28] documented a worsening in the overall severity of dystonia assessed at baseline, 12 weeks and 28 weeks by the Barry-Albright dystonia scale with the drug/placebo being administered for 12 weeks each. They documented some side effects, including agitation, constipation, dry mouth or poor sleep, in all the patients. The rapid increase in the dose of trihexyphenidyl as well as a higher mean maximum dose compared to the present study (1.5 vs. 0.7 mg/kg/day) could possibly explain this high incidence of side effects. These side effects also tend to worsen dystonia and may explain the worsening scores in the treatment group.
Most other studies [24, 27–29] have reported a higher frequency of side effects ranging from 45–100%. In comparison, only four children who completed the present study had adverse effects like constipation and mild irritability. An additional two children who discontinued medication due to side effects in the form of severe constipation and irritability were excluded from the final analysis. The lower prevalence of side effects in the present study could be attributed to the younger age of the children as well as a more gradual increase in dose. Also, constipation, a commonly reported side effect, was already present during baseline evaluation and managed as per unit protocol and thus likely did not manifest as a side effect. The low incidence rate could also be attributed to inherent difference in the study population.
Apart from Sanger et al. [25], most other studies [28–30] were retrospective and used varying doses of trihexyphenidyl. Also, their primary outcome measures were qualitative and therefore biased by subjective assessment of caregivers. Objective tools to quantify changes were used in the current study to overcome these limitations. This study is also unique because the GDS was used for the assessment of dystonia in children with CP for the first time in published English literature.
Although stratified analysis did not show any significant difference between children with dystonic CP due to perinatal asphyxia and those with other aetiologies, trihexyphenidyl may have a special potency for reducing dystonia in children who have suffered HIE brain injury [14].
The limitations of this study include its short duration, especially for documenting side effects. Long-term follow-up to ascertain dose-effect relations was also lacking. Another limitation was that genetic evaluation in those without clear aetiology was not done; however, especially without specific treatment, none of the subjects with genetic aetiologies could have improved markedly in a way that would affect the results of this trial.
This was the first study to document effects of trihexyphenidyl in young children with dystonia. In conclusion, this trial documented a statistically significant change, not only in dystonia, but also in the functional status of the children as measured using the GMFM, GMFCS, and EDP-2. There was also gain in gross motor milestones in 65% of children. Though a recent systematic review [31] concluded that trihexyphenidyl is possibly ineffective in reducing dystonia and improving motor function in CP, the results of the current study suggest trihexyphenidyl significantly improved dystonia, motor function and development in children with dystonic CP. Additional studies with larger numbers of children with this condition are required to further clarify its role.
Footnotes
Acknowledgments
The authors are thankful to all patients, their parents, and nursing staff for their cooperation.
Conflict of interest
The authors have no conflicts of interest to disclose.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
