Abstract
PURPOSE:
The purpose of this study is to describe the efficacy and adverse events of neuraxial anesthesia for post-operative pain control in non-ambulatory children with cerebral palsy with pre-existing intrathecal baclofen (ITB) pumps undergoing hip reconstructive or palliative surgery.
METHODS:
Twelve children (mean age 11.25 years) were included in the study with the following neuraxial anesthesia methods: indwelling epidural catheter (8 patients), neuraxial opioids administered through the side port of the ITB pump (3 patients), and single injection spinal anesthetic (1 patient). Observational pain scores and opioid requirements were quantified for all patients.
RESULTS:
There were no ITB pump or surgical complications at a mean follow-up of 2.2 years. The average length of stay was 6 days. Patients had good post-operative pain control with a mean observational pain score of 0.7 and mean morphine equivalent use of 0.26mg/kg/day. Four patients required anti-emetics to control nausea and three patients had urinary retention requiring repeat catheterization, but all medical complications resolved prior to discharge.
CONCLUSION:
Neuraxial anesthesia can effectively control post-operative pain in children with a pre-existing ITB pump. Utilizing the side port of the ITB pump for administration of neuraxial opioids is an option when epidural or spinal anesthesia is not possible.
Introduction
Up to one in three children with cerebral palsy (CP) are at risk for progressive hip displacement, with risk increasing to 70 –90% in those that are non-ambulatory (Gross Motor Function Classification System Levels IV and V) [1]. When hip displacement progresses, hip reconstructive surgery can improve health-related quality of life outcomes and reduce pain [2]. For hip dislocations in which reconstruction is not possible, palliative surgeries have been shown to improve pain and outcome [3]. Muscle spasticity, decreased pulmonary function, developmental delay, and impaired communication can make post-operative pain control after reconstructive and palliative surgeries challenging [4–6]. A multi-modal pain program can be effective in controlling post-operative pain after hip surgery in children with CP [7], with supplemental epidural anesthesia demonstrating improved pain management over catheter-based local infiltration analgesia [8].
An intrathecal baclofen (ITB) pump is an effective way to control severe spasticity in children with CP [9], and some of these children may require major hip surgery following their ITB pump implantation. Anesthesiologists treating a child with CP with an ITB pump may be reluctant to place epidural catheters due to the risk of ITB catheter damage or dislodgement, infection, or iatrogenic dural puncture [10–12]. Moreover, these children have been specifically excluded from randomized control trials for epidural anesthesia [8]. There have been case reports of successful epidural analgesia administration in the presence of an ITB catheter in young adults with CP [10, 11]. Recently, there has been an increased interest in epidural catheter placement in children with CP and an indwelling ITB pump utilizing regional pain techniques [13]. Indwelling pain pump tubing has been used to administer spinal anesthesia in adult patients [12], but the ITB pump has not been used in a child with CP for neuraxial opioid administration.
The goal of this study is to report the efficacy and side effects of neuraxial anesthesia for post-operative pain control in children with CP and a pre-existing ITB pump undergoing hip reconstructive or palliative surgery.
Methods
This retrospective study was approved by the institutional review board (IRB) and informed consent requirements were waived due to the retrospective study design. Children with CP and a previously implanted ITB pump were included if they underwent either a bony reconstructive (femoral osteotomy and pelvic osteotomy) or palliative hip surgery (proximal femoral resection or proximal femoral valgus osteotomy) and received neuraxial anesthesia for post-operative pain control between January 2012 and October 2018.
The pediatric anesthesiology team followed two protocols for post-operative pain control in this group of patients with an ITB pump (SynchroMed II, Medtronic, Minneapolis, MN): Standard neuraxial anesthesia protocol –For epidural catheter placement, the anesthesiologist used standard sterile technique and placed it distal to the scar for the ITB pump (Figs. 1, 2). Local anesthetic with preservative-free morphine was delivered by epidural catheter pump for up to post-operative day 3 based on the preference of the surgical team. All patients receiving an indwelling epidural catheter followed a standardized post-operative multi-modal pain management pathway with co-management with pediatric intensivists and pediatric anesthesiologists (Appendix 1). For single injection spinal anesthetic, the anesthesiologist injected 5 mcg/kg of preservative-free morphine (Duramorph-West-Ward Pharmaceuticals, Eatontown, NJ), and 0.5 % preservative-free isobaric bupivacaine was injected through the L5-S1 interspace using standard sterile technique. A physical medicine and rehabilitation (PM&R) physician confirmed proper functioning of the ITB pump pre- and post-operatively. Baclofen pump administered neuraxial opioids –A single dose of intrathecal opioids was administered through the side port of the ITB pump in consultation with a PM&R physician familiar with ITB pump programming and management. After induction of anesthesia, the ITB pump was paused temporarily by the PM&R physician using the external controller. The ITB pump has a main reservoir that holds baclofen, but it also has a side port that allows for direct access to the external catheter that terminates in the intrathecal space. The ITB pump side port was accessed using the Catheter Access Port Kit (Medtronic, Minneapolis, MN) using sterile technique (Figs. 3–5). Approximately 2 ml of fluid was aspirated via the side port to ensure that the baclofen was cleared from the external catheter and to ensure free return of cerebrospinal fluid. Under direction of the anesthesiologist, 5 mcg/kg of preservative-free morphine (Duramorph-West-Ward Pharmaceuticals, Eatontown, NJ) was injected into the external catheter via the ITB side port, followed by 1 ml flush of preservative-free normal saline. To prevent respiratory depression due to phrenic nerve blockade, local anesthetic was not used because the ITB catheter usually terminates at the high thoracic level. The PM&R physician then used a priming bolus to fill the external catheter with baclofen and resumed infusion of baclofen through the ITB pump at the previously set rate. Proper functioning of the ITB pump was confirmed by the PM&R service post-operatively, and the patient was closely monitored for signs of baclofen withdrawal. At the discretion of the anesthesiology and surgical team, children also received a regional fascia iliaca block prior to initiation of surgery. Use of the SynchroMed II pump side port to administer neuraxial opioid is off-label, while the administration of intrathecal Duramorph is consistent with approved indication of this drug.
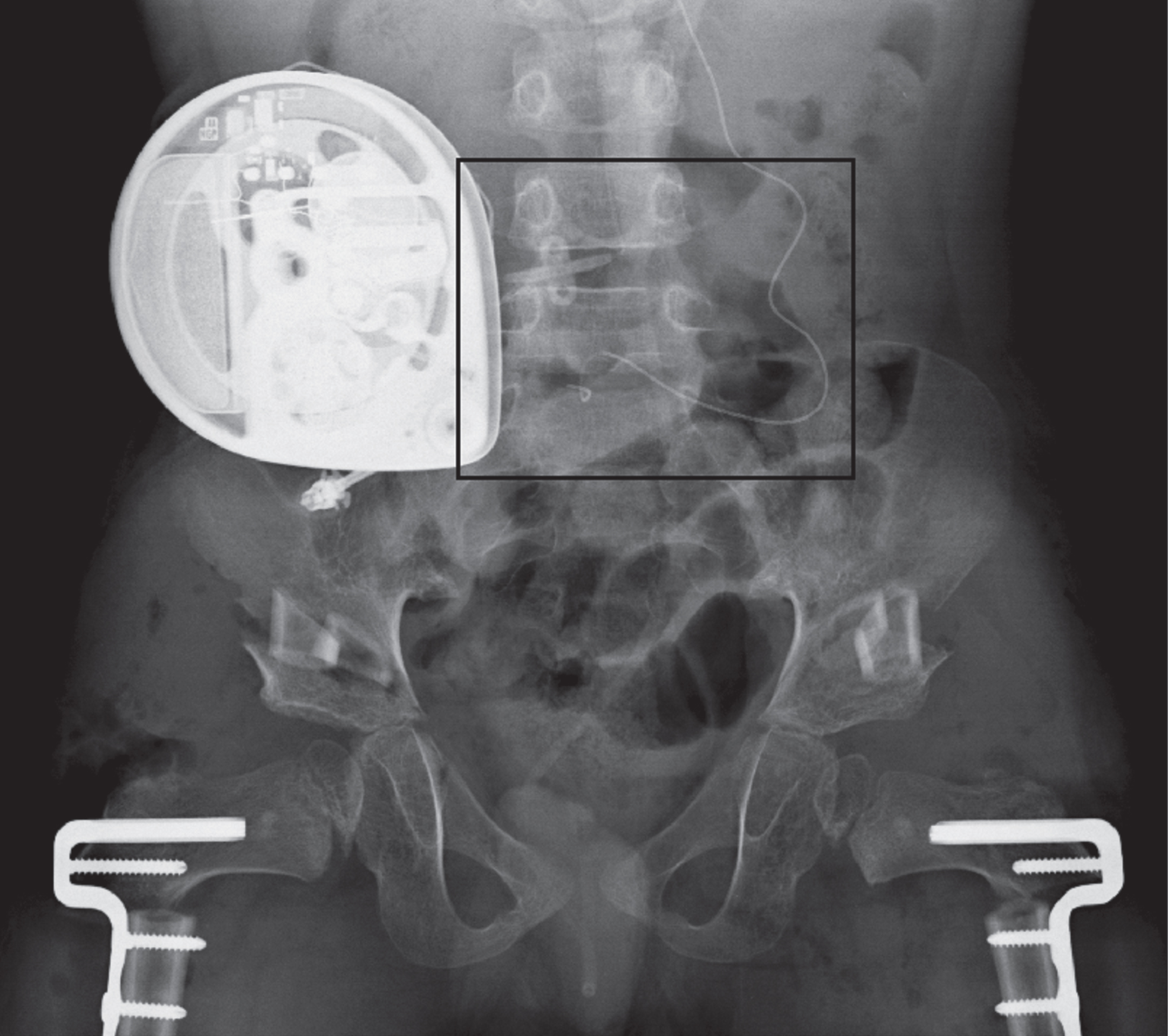
Subject 8 had a lumbar epidural catheter placement following bilateral femoral varus derotation osteotomies with Dega pelvic osteotomies, with a black box around the area of interest shown in greater detail in Fig. 2.
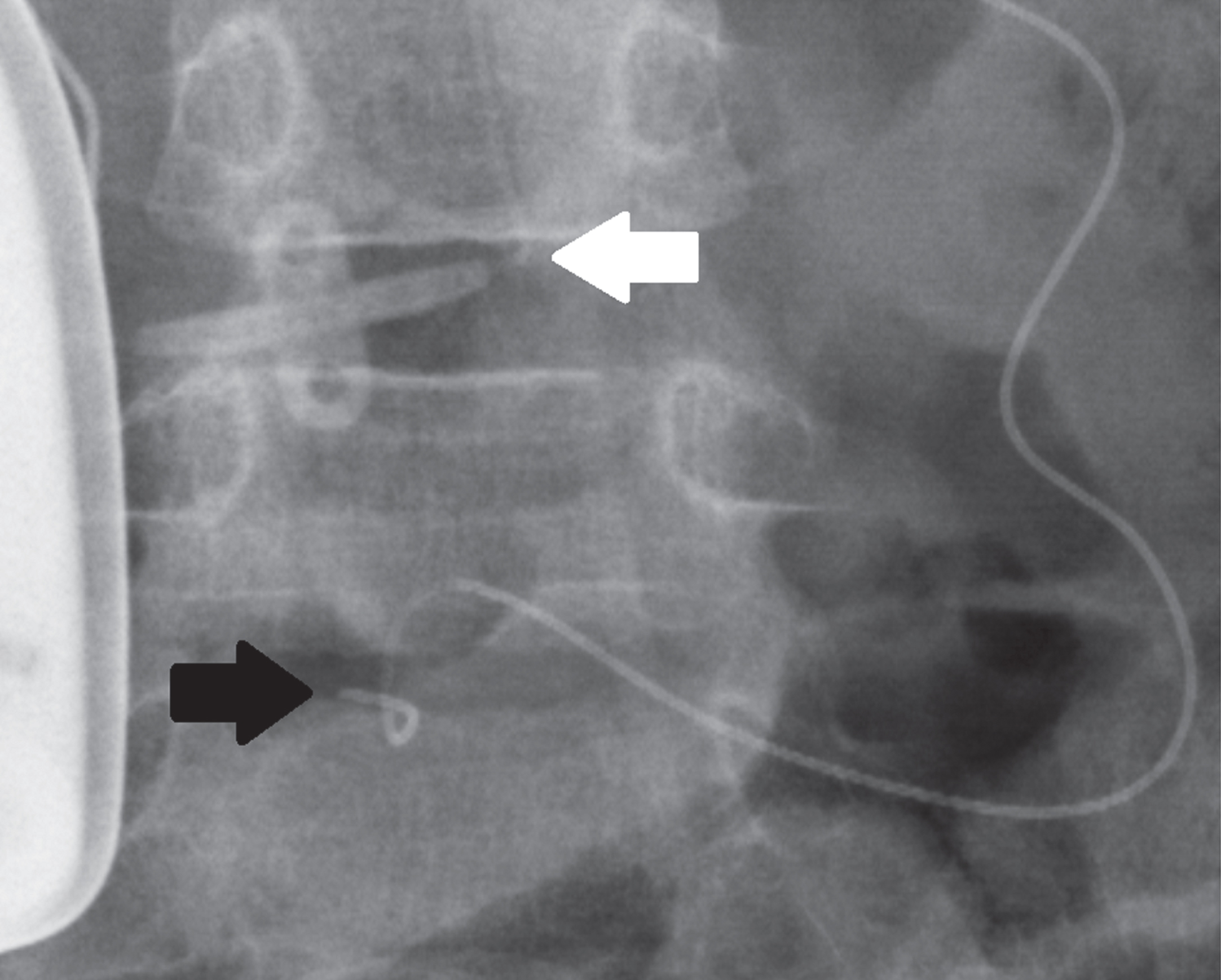
Magnification of the area of interest from Figure 1 shows the intrathecal baclofen pump catheter entering the thecal sac at the L4-5 interval (white arrow) and the epidural catheter entering at the L5-S1 interval (black arrow).
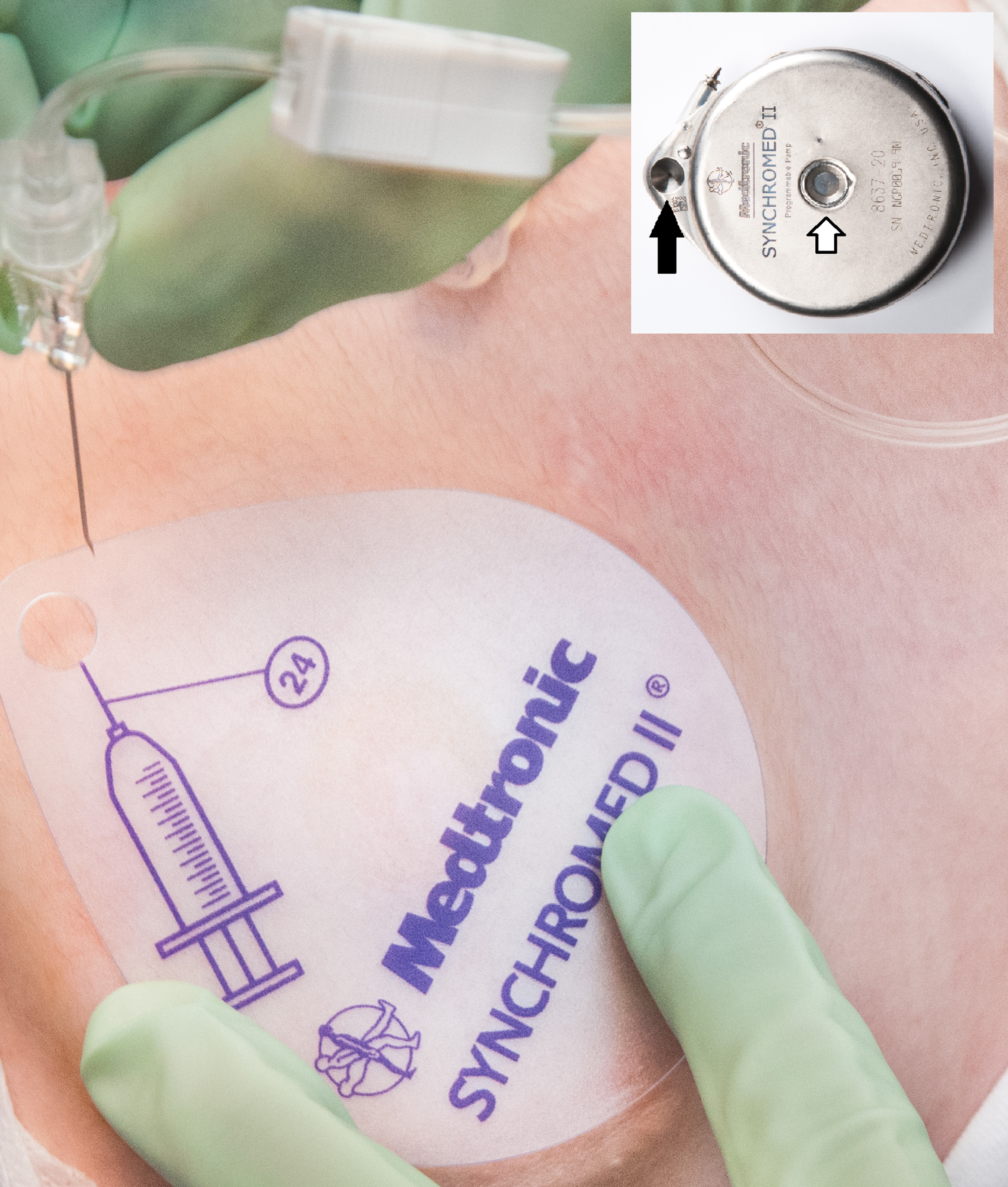
Utilizing the side port (inset, black arrow), a single injection of neuraxial opioids can be delivered through intrathecal pump (SynchroMed II, Medtronic, Minneapolis, MN). The main reservoir for baclofen refill (white arrow) is not used for this technique.
The following details were extracted from the medical record: demographic information, diagnosis, surgery, type of anesthetic technique, intra- and post-operative medication administration, post-operative complications, and observational pain assessment using the Observational Pain Assessment Score (OPAS) [14]. The OPAS rates restlessness, muscle tension, facial expression, vocalization, and wound guarding with scores between 0–2 for a total between 0 –10, with lower values indicating less pain. OPAS has been showed to strongly correlate (
Patient characteristics
A total of 108 children with CP underwent hip reconstructive or palliative surgery at a single institution between 2012 and 2018, with fourteen children having pre-existing ITB pumps at the time of surgery. Two of the patients were managed without neuraxial anesthesia prior to development of the multi-modal pain protocol, and so the twelve that had neuraxial anesthesia following development of the protocol (mean age of 11.25 years, range 6.3 to 16.4 years) were included. Eight patients had an indwelling epidural catheter, three patients had neuraxial opioids administered through the side port of the ITB pump, and one patient had a single injection spinal anesthetic. The epidural catheter was used for continuous infusion for an average of 2.5 days (range 2 –4 days). The estimated blood loss of the cohort was 338 ml (range 100 –600 ml). The average length of stay of the cohort was 6 days (range 3.2 –10.3 days). Details of patient demographics and anesthetic utilization are highlighted in Table 1.
Neuraxial Anesthesia Group Characteristics
Epidural catheter (E); intrathecal baclofen pump spinal (BPS); fascia iliaca regional block (FI); single injection spinal anesthetic (SS); femoral varus derotation osteotomy (VDRO); Dega pelvic osteotomy (Dega); femoral valgus osteotomy (FVO); proximal femoral resection (PFR); femoral valgus osteotomy with femoral head resection (FVOH); Gross Motor Function Classification System (GMFCS); gastrostomy tube (G); ventriculo-peritoneal shunt (V); epilepsy (Y); spinal fusion (S); acetaminophen (A); ketorolac (T); non-steroidal anti-inflammatory medication (I); ketamine (K); dexmedetomidine (D); estimated blood loss (EBL); length of stay (LOS).
Neuraxial Anesthesia Group Characteristics
Epidural catheter (E); intrathecal baclofen pump spinal (BPS); fascia iliaca regional block (FI); single injection spinal anesthetic (SS); femoral varus derotation osteotomy (VDRO); Dega pelvic osteotomy (Dega); femoral valgus osteotomy (FVO); proximal femoral resection (PFR); femoral valgus osteotomy with femoral head resection (FVOH); Gross Motor Function Classification System (GMFCS); gastrostomy tube (G); ventriculo-peritoneal shunt (V); epilepsy (Y); spinal fusion (S); acetaminophen (A); ketorolac (T); non-steroidal anti-inflammatory medication (I); ketamine (K); dexmedetomidine (D); estimated blood loss (EBL); length of stay (LOS).
All patients received multi-modal pain medication management, with nearly all patients (11 of 12) receiving two or more synergistic agents during their hospital stay. The mean pain score for the cohort was 0.7 for the entire admission, and peaked on post-operative day 1 at 1.2 (Table 2), indicating excellent pain control equivalent to or better than published results [7, 8]. Total opioid use following neuraxial anesthesia in the first three post-operative days was between 0.1 and 0.37 MME/kg/day (Table 3), a lower rate of opioid administration than published results for patients using patient-controlled anesthetic (PCA) [7] and similar to epidural anesthesia without an ITB pump [8].
Observational Pain Assessment Scores
Epidural catheter (E); intrathecal baclofen pump spinal (BPS); fascia iliaca regional block (FI); single injection spinal anesthetic (SS); post-operative day (POD).
Observational Pain Assessment Scores
Epidural catheter (E); intrathecal baclofen pump spinal (BPS); fascia iliaca regional block (FI); single injection spinal anesthetic (SS); post-operative day (POD).
Opioid Use and Adverse Events
Epidural catheter (E); intrathecal baclofen pump spinal (BPS); fascia iliaca regional block (FI); single injection spinal anesthetic (SS); mean morphine equivalent (MME); emesis/vomiting (V); urinary retention (U); decubitus ulcer (D); pruritus (P); ileus (I); Standard Deviation (SD).
Across the entire cohort, there were no ITB pump or surgical complications at a mean follow-up of 2.2 years (range 6 months to 4.5 years). Four patients (33%) experienced post-operative nausea or emesis that was controlled with medication and resolved by the time of discharge. Three patients (25%) had post-operative urinary retention that resolved by discharge with catheter re-insertion and bladder rest. One patient experienced pruritus that was controlled with medication and resolved by discharge. One patient had a post-operative ileus that resolved with bowel rest but extended the hospital stay to 10 days, and one patient developed a Stage 2 sacral decubitus ulcer after discharge from the hospital that resolved with outpatient dressings.
Discussion
Comfort following major hip surgery in children with CP is a high priority for the patient and caregiver. Multi-modal medication pathways can improve pain control in children with CP, and the addition of neuraxial anesthesia improves patient comfort while minimizing the side effects of large post-operative doses of opioid [5]. The authors’ institution began an ITB pump program in 2000, and early experience with sub-optimal pain control utilizing patient-controlled analgesia pumps for patients undergoing major hip surgery mirrored the published experience [7]. To improve comfort after surgery, the pediatric anesthesiology, PM&R, pediatric intensive care, and pediatric orthopaedic surgery teams developed a comprehensive protocol in 2014 to administer neuraxial anesthesia to this small and medically complex cohort of children. Based on this protocol, the results of this study show that pain control was clinically comparable or superior to published standards. The patients in this study experienced average pain scores of less than 2 on all post-operative days, which is comparable to the published pain scores (mean rFLACC = 1.8) in children without ITB pumps who were managed post-operatively with epidural analgesia [8]. The average length of stay in this cohort was 6 days, which is comparable to the length of stay of children reported in a recent publication of children with CP without pre-existing ITB pumps undergoing hip reconstructive surgery (median length of stay 6 days, 70% greater than 5 days) [4]. Utilization of neuraxial anesthesia in the current study’s cohort also reduced opioid requirements (MME/kg/day = 0.26) compared to published data on a comparable cohort managed with patient-controlled analgesia pumps alone (MME/kg/day = 0.49) [7]. Importantly, no major ITB pump or surgical complications were noted during the study period.
This study describes a broader application of the previously published technique of delivering a spinal anesthetic utilizing an indwelling pump [12]. The ITB pump has two access ports, a primary reservoir that holds the infusion drug, and a side port that allows for access to the external catheter which terminates in the intrathecal space (Fig. 3). The technique described in this study utilizes the side port to deliver a dose of opioids directly through the external catheter into the intrathecal space. This technique requires a PM&R physician to temporarily pause the continuous baclofen infusion from the pump’s primary reservoir while the side port is being accessed for the intrathecal opioid administration, allowing for minimal disruption in the administration of the patient’s ITB and limiting the risk of baclofen withdrawal. The most important indication for this anesthetic technique is for a child who presents for hip reconstructive surgery with both a prior implant of an ITB pump and a history of spinal fusion surgery. This technique also allows for the theoretical administration of a second dose of neuraxial opioids on the surgical floor, although this was not needed in this study’s group of patients. For an effective ITB pump-delivered neuraxial opioid administration, the intra-operative and post-operative presence of a PM&R team that is familiar with programming and accessing the ITB pump is critical. Access to the ITB pump may be challenging in children with obesity, suboptimal pump placement, pump inversion or rotation, and scar formation over the side port [19], and further studies may clarify whether the use of additional imaging such as ultrasound may assist in accessing the side port in these specialized situations. As was done in this study, all children with ITB pumps should have confirmation that there is no break in the administration of ITB to minimize the chance of baclofen withdrawal.
Children with CP undergoing major hip reconstructive or palliative surgery have multiple medical co-morbidities and are at a high risk of perioperative complications, with one study reporting 91% of patients having at least 1 minor medical adverse event that resolved by the time of discharge or with outpatient treatment [4]. In the current study, 9 of the 12 patients (75%) experienced at least 1 minor medical complication that resolved by discharge. The two most common medical adverse events, nausea and urinary retention, are well-documented minor adverse events in children with CP undergoing anesthesia [5, 20]. While it is not certain that an alternative pain strategy that avoids neuraxial anesthesia would provide a superior result in this medically complex group of patients, strategies to avoid and manage these common post-operative challenges would be better addressed by a prospective study.
This study has several limitations. The first and most important is the small number of patients enrolled, which reflects the small number of children with CP who undergo major hip surgery and have a previously implanted ITB pump, mirroring the experience of a recently published study from another high-volume center (16 subjects over 10 years) [13]. The small sample sizes from single-center studies highlight the need for a multi-center prospective study to make more sound conclusions on comparative effectiveness of post-operative pain approaches. Second, as this was a retrospective study, there was significant variability in adherence to the multi-modal pain protocol and this could explain the variability in pain control. While the pain management protocol was created with broad consensus across treating physicians and teams at the institution, children with CP are not a homogenous group and individual providers were encouraged to use their professional expertise to tailor the post-operative pain regimen to the patient. It is believed that this represents a real-world application of a multi-modal pain protocol and that the results would be reproducible at other centers.
Conclusion
Neuraxial anesthesia for post-operative pain control is an option after major hip surgery in this unique population of children. The side port of the ITB pump for administration of intrathecal opioids neuraxial can be used when epidural catheter placement or single injection spinal anesthetic is not possible or feasible. A multi-disciplinary approach, with coordination between the pediatric orthopaedic surgeon, pediatric anesthesiologist, and PM&R physician, is essential for the safety of children with CP and pre-existing ITB pumps undergoing neuraxial anesthesia.
Footnotes
Acknowledgments
No funding was used to support this study.
Conflicts of interest
Jon R. Davids is a paid consultant for Orthopediatrics Inc and Board Member of the Foundation for the Advancement of Pediatric Orthopaedics. Sundeep Tumber is a paid consultant for Pacira Biosciences. The remaining authors have no conflicts of interest to report.
Ethical considerations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional review board and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was approved by the host institutional review board. The host institutional review board waived informed consent requirements for this retrospective study.
