Abstract
PURPOSE:
The Gross Motor Functional Classification System (GMFCS) provides a standardized classification of motor disability in children with cerebral palsy (CP) that is typically considered longitudinally stable. Here, changes in GMFCS levels of children with CP who received repeated botulinum toxin A (BoNT-A) injections within an intensive rehabilitation approach were evaluated.
METHODS:
This retrospective, observational study included 503 children with CP (GMFCS I-V) treated in a single university rehabilitation clinic. Individualized treatment plans and a variety of rehabilitation techniques were incorporated within an integrated model of care. Each child received≥2 repeated BoNT-A injections as well as an intensive rehabilitation program.
RESULTS:
GMFCS level following repeat treatment improved in 42.1% of the patients and worsened in 3 patients; 14.9% of children showed improvement after 1 treatment cycle, 12.3% after 2 cycles, 7.6% after 3 cycles, 3.6% after 4 cycles, and 3.8% after≥5 cycles. Type of involvement (
CONCLUSION:
Children with CP may change GMFCS level when intensive rehabilitation programs are combined with repeated BoNT-A injections.
Introduction
One of the most commonly used methods of classification of cerebral palsy (CP) is the Gross Motor Function Classification System (GMFCS), which categorizes CP severity on the basis of self-initiated movement abilities [1]. It is generally agreed that children with CP reach their gross motor potential by the age of 3.5–5 years. After this time, the GMFCS is classically considered to be longitudinally stable, even following surgical intervention [2–4].
Currently there is a movement within paediatric rehabilitation towards the provision of periods of intensive rehabilitation [5]. A wide range of intensive physical and occupational therapies, often following targeted injections of botulinum toxin A (BoNT-A), are provided at the Kocaeli University Department of Physical Medicine and Rehabilitation (PMR) and Izmit Rehabilitation Centre. The philosophy of the centre is to provide goal-oriented treatment at an intensity and duration that will impact neuroplasticity and is tailored to the child’s needs and preferences [6, 7]. BoNT-A therapy is used when required to reduce focal spasticity and ‘enable’ the child to participate in the chosen targeted therapies [8–10].
Striking changes in GMFCS have been observed in the centre’s patient population following repeat BoNT-A injections. These observations prompted a more formal retrospective review to evaluate the potential for change in GMFCS levels of children with CP who received intensive rehabilitation following repeated BoNT-A injections at the centre between 2007 and 2017.
Methods
Setting
This was a retrospective, observational study conducted at the Kocaeli University Department of Physical Medicine and Rehabilitation and Izmit Rehabilitation Centre. In this setting, all children are managed by a multidisciplinary team consisting of PMR physicians, physical and occupational therapists, special education and recreational sports specialists, and orthotists. BoNT-A injection parameters are individualized according to the pre-defined goals of treatment, clinical presentation and previous BoNT-A experience.
Inclusion criteria and treatment
All children with a diagnosis of CP and lower- limb hypertonia treated with≥2 BoNT-A treatment cycles within an integrated rehabilitation program between 2007 to 2017 and who had com-plete GMFCS level data for analysis were included in this review. Children were re-treated as required to meet their personal treatment goals, with≥12 weeks between injections. Following each BoNT-A injection, the children were entered into a 3-week intensive rehabilitation program (half or full day) starting 7 to 10 days after BoNT-A injection.
Although there was no single standardized rehabilitation approach, all children participated in a goal-directed post-BoNT-A therapy program consisting of a combined approach of relevant stretching and strength training, as well as function, daily living, play and school-based activities (including dance therapy and hippotherapy). Muscle stretching exercises were usually directed to agonist BoNT-A injected muscles using passive and/or active, non-weight bearing and/or weight-bearing techniques. Muscle activation training, selectivity and strength training were usually applied to the antagonist but also to the agonist muscles.
Outcomes assessment
Changes in GMFCS levels of children who received intensive therapy following repeat BoNT-A injections were documented. Comprehensive data on clinical assessments are regularly collected by the centre as part of its involvement in several clinical trials. Muscle tone and spasticity were assessed by Modified Ashworth Scale (MAS) and Modified Tardieu Scale (MTS) [11], and gait function by Observational Gait Scale (OGS) [12, 13]. Assessments were documented at the baseline of each treatment cycle with a follow-up at week 4-6 post injection (peak effect), and students’
In order to evaluate predictors of change (vs. no change) in GMFCS level, a logistic regression analysis (Hosmer Lemeshow) with backward stepwise method was performed. This analysis included age, gender, baseline GMFCS level, type of involvement, presence of severe dystonia, number of treatment cycles, presence of speech/communication problems, and cognitive dysfunction as categorical covariates. Children with GMFCS level I were excluded from this analysis as there was no possibility of improvement (and none worsened).
Results
Retrospective analysis of case records identified 503 children with CP who fulfilled the inclusion criteria (Fig. 1); baseline characteristics are given in Table 1. Across the study, patients received 2–18 BoNT-A treatment cycles (doses per muscle are given in Table e1). No child received intrathecal baclofen during the follow-up. Less than 5% of children at GMFCS levels I–III used oral medications for tone reduction at some point during the follow up. Of the 55% of children at GMFCS levels IV and V who used oral pharmacotherapies, only 24% remained on oral treatment at their last visit.
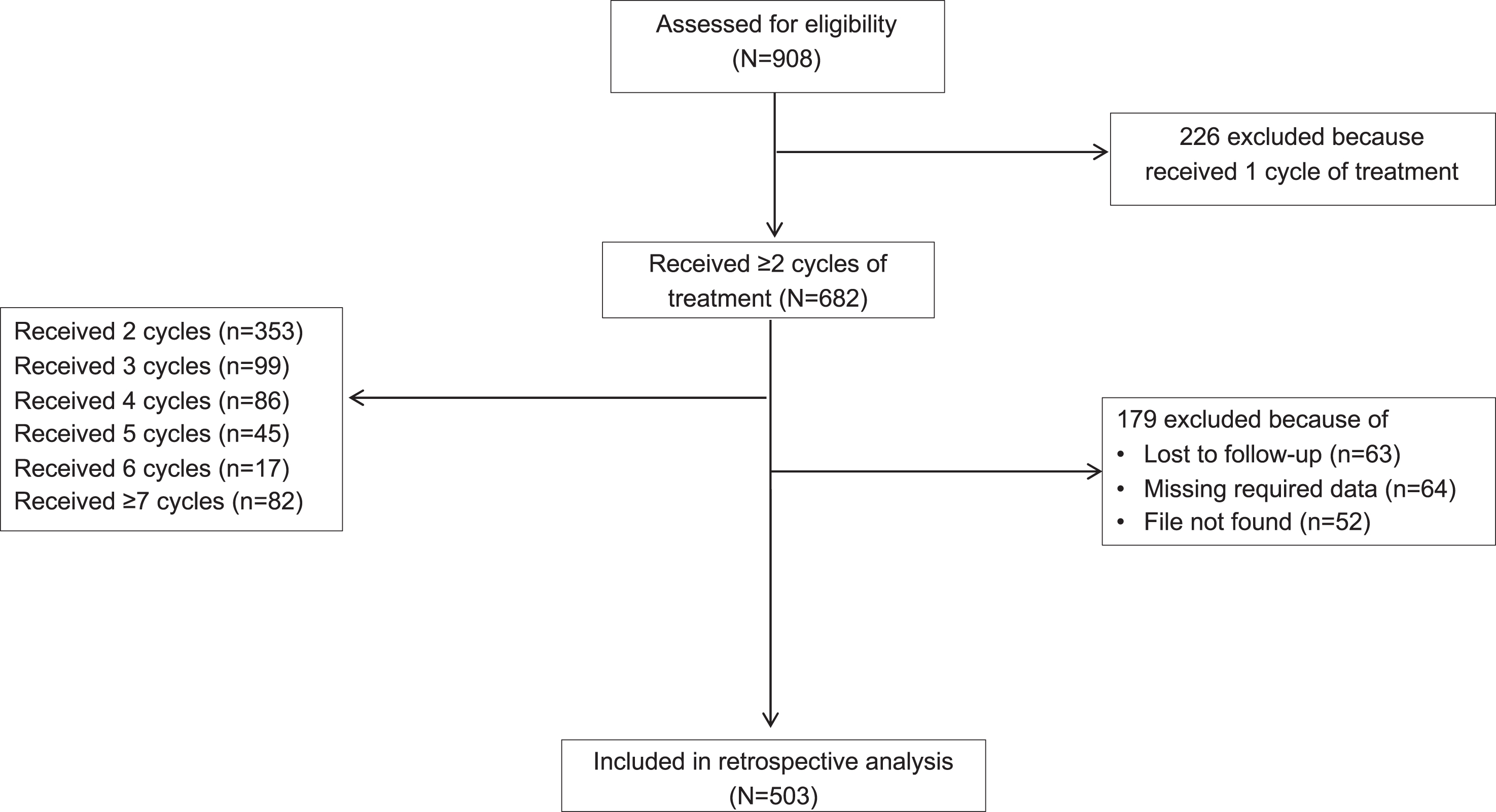
Number of children included in the retrospective analysis.
Baseline characteristics and associated conditions
Orthotics were used by most children (91.8%); 70.8% received functional ambulatory training, 58.6% Biodex balance training, 58.6% whole body vibration, 45.3% intermittent or weekly progressive serial casting (typically with functional ambulatory training as part of the casting protocol), 44.9% virtual reality training, 40.2% hippotherapy, 12.7% robotic rehabilitation, 10% dance therapy and 8.5% intensive advanced ambulatory training.
Following repeat treatment, 42.1% of children showed at least one level of GMFCS improvement, with worsening seen in only 3 patients (Fig. 2). In addition, 8.5% of the GMFCS III and IV patients had two levels of GMFCS improvement. Improvements in GMFCS level were observed in 14.9% of children after 1 treatment cycle, 12.5% after 2 cycles, 7.8% after 3 cycles, 3.6% after 4 cycles, and 4.0% after≥5 cycles. Worsening was observed in one child after the 2nd treatment cycle, one after the 3rd and one after the 5th. Once achieved, improvements were sustained through the follow-up period. Logistic regression analysis (Table e2) showed that baseline GMFCS level (
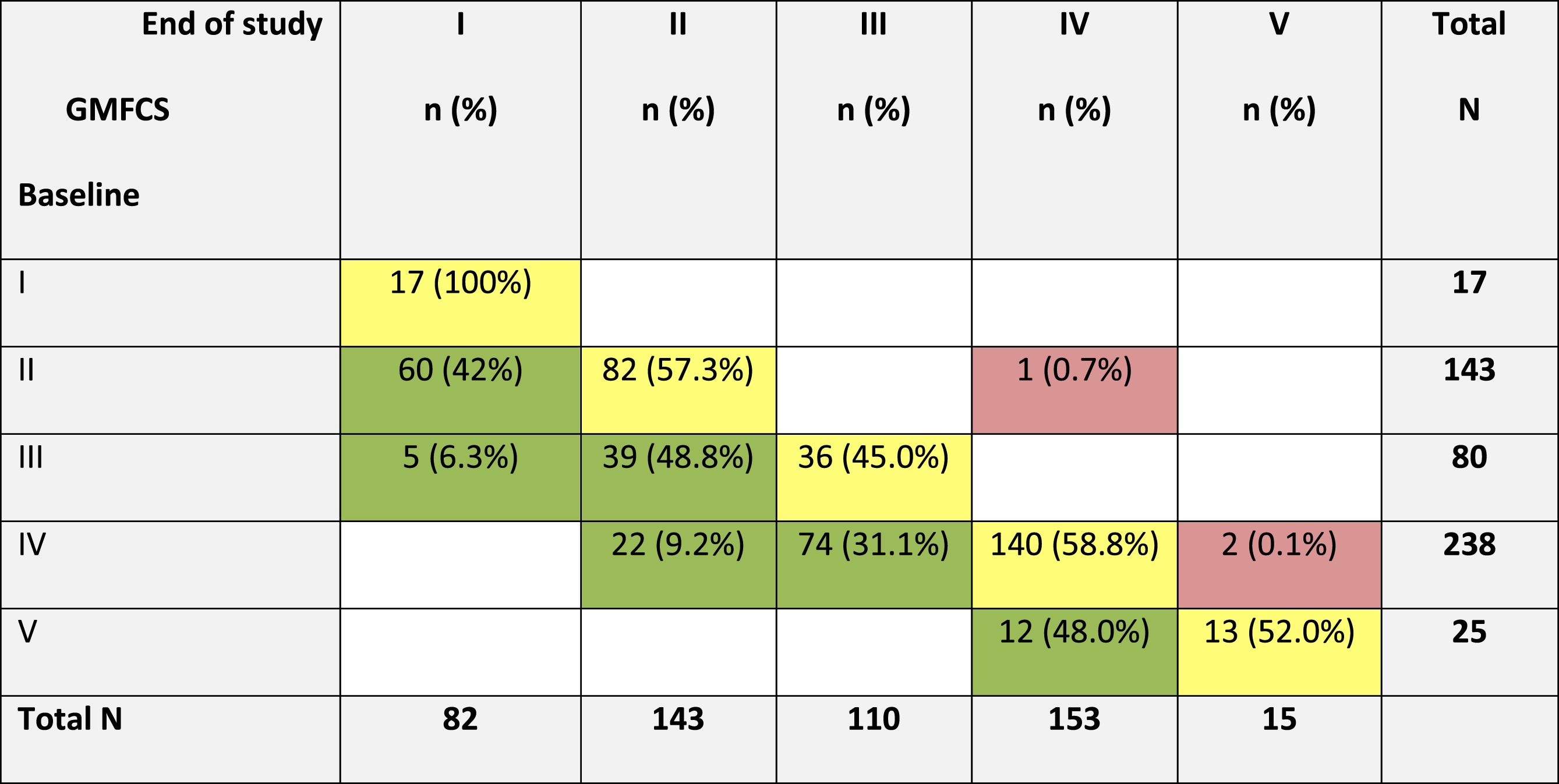
Shift diagram showing changes in Gross Motor Function Classification System (GMFCS) scores, from baseline to end of study.
Improvements in GMFCS level were supported by improvements in gait (OGS scale) and measures of hypertonia (MAS) and spasticity (MTS) across repeated treatment cycles (Tables e3-e8). At peak effect, an improvement of 4-5 points was seen in OGS-Total scores. MAS scores in the hip adductors, hamstrings and plantar flexors reduced (improved) by an average of 2 points during the first two treatment cycles. Improvements in the angle of catch (XV3), spasticity angle (X) and spasticity grade (Y) were also revealed by MTS scores.
The results of this single-centre retrospective review demonstrate that intensive rehabilitation programs facilitated by repeat BoNT-A injections have the potential for improvement in GMFCS level of a proportion of children with CP. In this retrospective study, improvement was shown in 42% of children treated in this way. When any improvement in GMFCS level occurs, it is, by definition, clinically relevant. For example, an improvement from GMFCS level IV to level II might mean a reduced need for mobility devices or an improved ability to climb stairs independently.
The proportion of children showing improvement in this study was much larger than reported elsewhere [2, 3]; however, the local situation at the time meant many children attending the centre had previously been inadequately treated. For this reason, it is not possible to compare this treatment approach with early intervention, which also maximises the potential for motor improvement [14, 15]. In addition, the magnitude of change seen probably reflects the intensity of treatment available at the specialist centre, which surpasses that available in many community-based practices. While there was no standard approach to the rehabilitation, all children had at least 3 weeks of intensive goal-directed therapy amounting to at least 60–120 hours of therapy input.
Although other studies have shown that various task-specific intensive rehabilitation approaches can significantly improve gross motor function within just a few weeks of intensive therapy [16], most have used the Gross Motor Function Measure and only a few have looked at change in GMFCS levels [16–18]. A large Swedish registry study of 736 showed that children initially classified as GMFCS levels II and III have a greater propensity for change (upward and downward) than those classified as level I or IV [4]. This finding is supported by the current study as children initially classified at GMFCS level III or IV were able to improve by 2 levels (to GMFCS I or II, respectively) and that GMFCS level at baseline was a significant predictor for improvement. The study by Alriksson-Schmidt also showed that children at GMFCS I were the least likely to worsen, probably due to less spastic paresis and soft tissue deformities [4].
Number of repeat treatments (i.e., repeat injections followed by intensive therapy) was another prognostic factor for GMFCS improvement. BoNT-A was often used as an enabler of intensive therapy (e.g., permitting casting [19]) and, in this retrospective review, the effects of this chemodenervation cannot be easily separated from the effects of intensive therapy. While some studies such as the Space Bop study found no evidence for the added value of BoNT-A injections for lower-limb therapy interventions [20], other recent studies have reported significant benefits of repeat BoNT-A treatment on gross motor function [21, 22].
Strengths of the study include the consistency and experience of the assessing clinicians throughout this study. However, analyses are limited to the data that had been systematically collected over the years, so, while functionality was the focus of the rehabilitation, there was no systematic data for other functional outcomes available. Although the relationship between changes at body function level and activity level can be debated [20], recent systematic reviews have found a high level of evidence for improvement of gait with posture therapy after BoNT-A injection [9] and some evidence for improvements in gross motor walking for BoNT-A coupled with physiotherapy [23].
The study is also limited by its retrospective design and lack of a control group. There was no standardization of therapy –the children had varied treatment goals and were treated based on their clinical needs, motivation and preferences. This includes oral antispasticity medications, which were mainly used in children at GMFCS levels IV and V. While any influence of these oral treatments on gross motor findings cannot be ruled out, they only provide mild tone reduction and, in clinical experience, are not well adhered to because of their associated systemic side-effects. In addition, the median age of children was already 5 years old at the start of treatment, and this relatively late start might explain why age did not have a predictive effect. The type and duration of therapy as well as injection parameters (including dosing) all contribute to the efficacy of treatment. Over the 10-year follow-up period, some therapies were discontinued in favour of newer technologies, for example. Therefore, prospective clinical trials and observational studies are needed to verify these findings across different settings and treatment backgrounds. It is also important to evaluate whether improvements in GMFCS level are sustained over longer durations and into adulthood.
Footnotes
Acknowledgments
Funding for this retrospective analysis was provided by Ipsen Pharma as part of an unrestricted educational grant. The authors wish to thank all children and their parents who contributed to the study. We thank Anita Chadha Patel for providing medical writing support, which was funded by Ipsen (Paris, France) in accordance with Good Publication Practice guidelines.
Conflict of interest
ND reports research support from Ipsen, Allergan, and Merz. MA, TG, MA, EY, CK, ED have no conflict of interest to report.
Ethical considerations
The study was approved by the ethics committee of Kocaeli University.
