Abstract
PURPOSE:
Noxious sensory inputs from the neonatal Intensive Care Unit (NICU) and lack of placental support negatively impact neuronal organization which has implications later in life. Evidence regarding early interventions (EI) on preterm neonates (PN) at high risk for developmental motor disorders is limited and inconclusive. This study focuses on neuromotor changes following Multimodal stimulations (MMS) with sensory and motor interventions among stable hospitalized PNs.
METHODS:
This single-center, non-blinded pre-test post-test control group study will recruit 60 PNs admitted to the Level II and III NICU of a recognized tertiary care teaching hospital by convenience sampling method into two groups by block randomization. Group A (n = 30) will receive MMS trial lasting for 30 minutes per session for five days per week, until discharge of the neonate from the NICU; Group B (n = 30) will receive regular lifesaving care from the NICU. Anthropometric evaluation, physiological status, and Infant Neurological International Battery (INFANIB) will be the outcome measures used to analyze the neuromotor behavioral modifications among the hospitalized PNs. All the outcome measures will be recorded at baseline, after every five days (to compare trajectories of scores between the groups), and at the end of the intervention at the time of discharge of neonate from the NICU.
RESULTS:
Demographic and outcome measures will be assessed for their normality using the Shapiro-Wilk test. Within and between-group comparisons will be analyzed by the repeated measures analysis of variance/Friedman test and independent t-test/Mann-Whitney U test respectively.
CONCLUSION:
MMS, which includes both sensory and motor interventions, will, to the best of the authors’ knowledge, be the first trial for modifying the neuromotor behavior of hospitalized PNs. If successful, the clinical effects of this protocol could be revolutionary in mitigating developmental impairments of PNs.
Introduction
About 15 million premature births occur annually across the globe, and in India alone 3.5 million out of 27 million infants are born prematurely each year [1, 2]. Over 70% of these preterm neonates (PNs) need hospitalization in Neonatal Intensive Care Units (NICUs) for physiological support and developmental maturation [2, 3]. Most of the PNs have atypical maturation of the brain due to extra-uterine sensory experience and atypical sensory stimuli in the NICU [4]. Noxious sensory inputs that PNs experience in the NICU negatively impact neuronal organization, which typically occurs during the last trimester of pregnancy [5–7]. The neonatal musculoskeletal system is fragile and vulnerable to injury [8] as cartilaginous joints have a high risk of developing osteopenia and bone mineral deficiency [6, 8]. Commonly used biochemical markers of bone metabolism such as blood calcium, phosphate levels, and plasma alkaline phosphatase levels can measure osteopenia and bone deficiency [8]. PNs cartilaginous joints are also vulnerable to the effect of gravity due to immature muscle fibers and reduced flexor tone. Tone development in PNs occurs in the caudo-cephalic direction resulting in hyperextended posture. PNs have limited endurance and an immature capacity to control more complex movements against gravitational forces [6].
The NICU is essential for the support and developmental maturation of neonates born preterm [3]. With their improving survival rates of preterm and increasing births of low-birth-weight infants, there is an increase in PNs with neuromotor impairments later in life which range from developmental coordination disorders to cerebral palsy (CP) [9]. Myelination begins during the second trimester and is most rapid in the first year of life; the process continues up to 30 years of age. Therefore, it is important that infants with motor dysfunction are identified early so that appropriate interventions can be implemented [10].
Due to the advancements in NICU equipment, as well as evidence-based diagnostic and treatment techniques, the survival rate of PNs has increased significantly [2, 12], causing an increase in the developmental impairments and motor issues [9, 14]. The disability of PNs has a major impact on family and society [15]. Multi-disciplinary Early Intervention (EI) care in the NICU that includes sensory and kinesthetic stimulations has made an immense contribution toward minimizing the morbidity and hospital stays of PNs. EI has prevented and helped to treat the possible respiratory, neurological, musculoskeletal, and stress-related complications of PNs, including delayed growth, and abnormal bone mineralization [8, 16]. These interventions improve the quality of life and limit disabilities in later part of their life [8, 16].
Evidence is limited and inconclusive regarding the effectiveness of EI in infants at high risk for developmental motor disorders due to the heterogeneity of these interventions [6, 18]. There is a definite need to establish the effectiveness of a standardized protocol comprising Multimodal stimulation (MMS) in PNs to determine whether or not this intervention will have a significant effect on neuromotor outcomes among hospitalized PN. The aim of this study is to identify the effects of a standardized MMS protocol versus standard NICU care among stable hospitalized PN.
Methods
Ethical statement and protocol registration
The MMS trial will be a single-center, non-blinded pre-test post-test control group study, performed on stable hospitalized PNs in the NICU of Maharishi Markandeshwar hospital, Mullana, Haryana, India. The study protocol was approved by the institutional ethics committee of Maharishi Markandeshwar (Deemed to be University) (MMDU/IEC/1566) on 10th December 2019 and is registered in the publicly open access protocol registration and results system, Clinicaltrials.gov, approved by WHO’s International Clinical Trials Registry Platform (ICTRP) and the International Committee of Medical Journal Editors (ICMJE) with unique Universal Trial Number (UTN), U1111-1236-9478 and unique registration no. NCT04247308 on 30th January 2020 (https://clinicaltrials.gov/ct2/show/NCT04247308) [19]. The study will be performed according to the principles laid out by the Declaration of Helsinki (Revised 2013) and the Council for International Organizations of Medical Sciences (CIOMS) guidelines; namely, the International Ethical Guidelines for Health-related Research Involving Humans (2016), and the National Ethical Guidelines for Biomedical Research Involving Children (2017). Before recruiting the preterm neonates in this trial, signed informed consent will be obtained from the parents or legal guardians.
Study population
Sample size and power
Sample size for the study was calculated using G*Power ver. 3.1.9.7 software (Heinrich- Heine-Universität Düsseldorf, Düsseldorf, Germany; https://www.gpower.hhu.de/) [20] for the sample size calculations with Effect size (ES) = 0.978 (Control group = 59.16±3.23; Study group = 62.24±3.07; Effect size = Group difference / Pooled standard deviation; Effect size = 62.24 –59.16 / √(3.07)2+(3.23)2 / 2; Effect size = 3.08 / 3.15) [21]. By substituting ES with the level of significance as 0.05 (αerror prob), 0.90 [Power (1-β err prob)] with 1:1 allocation ratio of a two-tailed test, it was be estimated that the sample size in each group has to be n = 23. In considering a 30% drop-out rate (n = 6.9), the final minimal required sample must be n = 30 (rounded off to the next whole number) in each group. Accordingly, the minimum sample size required for the study to attain 90% power (Type II error = 10%) with 5% level of significance (Type I error = 5%) is n = 60.
Recruitment
The selection criteria for recruited participants are presented in Table 1. Hospitalized PNs (target n = 60) who will meet all the selection criteria will be identified by the convenience sampling technique and a signed informed consent form will be obtained from the parents to ensure appropriate volunteer participation in the study.
Selection criteria for MMS trial
Selection criteria for MMS trial
After demographic information is collected, recruited PNs will be randomly divided into two groups, group A and group B, using block randomization. There will be four blocks with 15 rows each. Each row contains 4 chits (2 chits for each group), totaling 60. By the block randomization method, PNs will be randomly allocated using sequentially numbered, opaque, sealed envelopes (SNOSE). PNs will be allotted to a group based on the randomly chosen chit by their parents. A physiotherapist who is not involved in this research will perform the randomization. Once the first row of all the blocks are filled, the next row block will be opened for recruitment. Thus, an equal number of PNs will be assigned to each group over time.
Intervention description
Group A will receive MMS (Table 2) lasting for 30 minutes per session for five days per week, until discharge of neonate from the NICU [21]; Group B will receive standard NICU care. The study [22] flow chart describing the details of the study is displayed in Fig. 1.
Preemies’ Neuro-motor Facilitation (PNMF) Protocol
Preemies’ Neuro-motor Facilitation (PNMF) Protocol
PNMF Protocol should be administered for 30 min, 5 times per week.
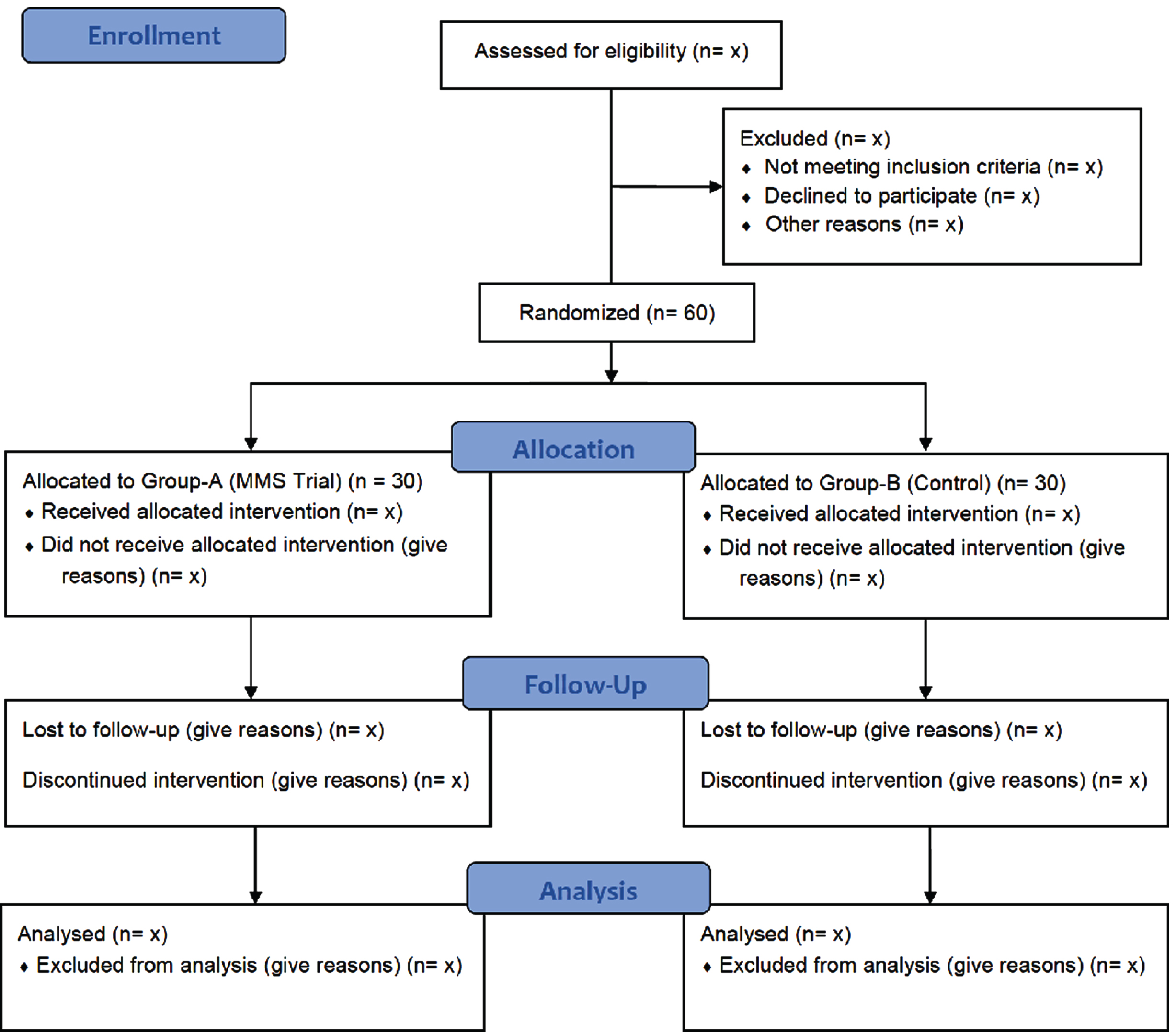
Study flow diagram for the randomized controlled trial protocol.
Sensory stimulations
Sensory stimulations will consist of a soft lullaby using a miniature speaker, gentle stroking massage, visual stimulation with a black and white card, gentle rocking, and oral stimulation including stroking the cheeks, lips, jaw, and tongue as well as rubbing the gums [22]. Each stimulation will be given for three minutes [21].
Movement therapy
Movement therapy will be administered for three minutes each per the following interventions: guided range of motion (bicycle riding pattern), antigravity movements in prone position (neck and spinal extension), antigravity movements in sitting position (supported), and upright positioning. A multi-parameter NICU monitor will be used to record physiological stress during the intervention. Recruited PNs will be considered to be in physiological stress if heart rate (HR) >200 or < 100 bpm, respiratory rate (RR) >20 over baseline, or oxygen saturation (SpO2) <86% for more than 15 seconds [15]. Similarly, signs of behavioral stress will be monitored; if a recruited PN startles, tremors, yawns, finger splays, averts gaze, cries, hiccups, protrudes their tongue, or displays tone or state-related behavior changes during treatment session, the MMS intervention will be modified. The particular stimulation will be paused for 15 seconds and only resumed if parameters recover. The infant’s physiological and behavioral stress responses will be monitored to prevent overstimulation. This standardized MMS protocol was named as Preemies’ Neuro-motor Facilitation (PNMF) Protocol, tabulated in Table 2. The PNMF Protocol by the first author was copyrighted under the Copyright Office of the Government of India with unique registration no. L-89359/2020 dated 11th February, 2020 (copyright filed with diary no., 19789/2019-CO/L dated 10th December, 2019).
Group B - Control group
The control group will receive standard life-saving NICU care from the medical team and daily maternal care, such as being held in the mother’s arms [23].
Though both the groups may not be identical, they differ only by MMS intervention and thereby pre-post intervention changes due to the full attribute of the MMS intervention could be verified. Hence, this design could be considered as the acceptable scientific standard for establishing a cause-and-effect relationship in clinical research [24].
Study measures
Demographic and clinical information
Date of birth, prematurity, type of delivery, APGAR scores [25], and the New Ballard score will be obtained from hospital records, whereas length (cm), head circumference (cm), and weight (grams) will be measured before the interventions.
Outcomes
Primary outcome
Infant Neurological International Battery: The Infant Neurological International Battery (INFANIB), which is easy and efficient to administer [25–28], will be used as the primary outcome measure to assess the neuromotor development of the preterm neonate using corrected gestational age [21]. INFANIB has excellent reliability of ICC = 0.9 with 90% sensitivity and 83% specificity and acceptable predictive validity in detecting developmental delay [28, 29]. It can be successfully used among preterm infants to detect the short-term effect of multisensory interventions on the neuromotor development of preterm neonates [21]. INFANIB consists of a total of 20 items to be evaluated in five positions: supine, prone, sitting, standing, and suspended [25]. Among them, only 14 items are able to be administered at birth. The 14 included items are: 10 items in supine position - hands closed/open, scarf sign, heel to ear, popliteal angle, leg abduction, dorsiflexion of the foot, foot grasp, tonic labyrinthine, asymmetric tonic neck reflex and pull to sitting; two items in prone position –all fours and tonic labyrinthine; one item in sitting position –sitting; and one item in standing position –weight-bearing. Thus, a total score of 70 will be used instead of 100 [30]. The neurological cut-off scores proposed by the developer, Patricia H. Ellison (1986), are as follows: Abnormal:≤48, Transient: 49–65 and Normal:≥66 [30]. Following interpretation, these scores will be used to describe the characteristic of recruited PNs. Pre-post MMS intervention changes of INFANIB will be recorded at baseline and considered for the data analysis, after every five days (to compare trajectories of scores between the groups,) and at the end of the intervention at the time of discharge of neonate from the NICU.
Secondary outcomes
Bone minerals (serum calcium, serum phosphate, and bone specific alkaline phosphate) will be measured at baseline, after every five days, and at the end of the intervention (discharge of the neonate from the NICU) [26, 28] to compare the trajectories of scores between the groups.
Parathyroid hormone (serum level) will be measured at the onset and the end of the intervention (discharge of neonate from the NICU) [23].
Anthropometric parameters
Weight (g) of the neonate will be further stratified into normal birth weight (NBW) from 2500 g to 3999 g, low birth weight (LBW) which is considered less than 2500 g [31], appropriate for gestational age (AGA), small for gestational age (SGA), length (cm), cephalic perimeter (cm), and type of delivery (normal vaginal delivery [NVD] or Lower segment Caesarean section [LSCS]) [26].
Physiological parameters including HR, RR, body temperature (BT), and BP will be obtained from a multi-parameter NICU monitor before and after every session. In addition to this, the length of hospital stay will be noted. Both the primary and secondary outcomes with anthropometric parameters will be recorded at baseline, after every five days, and at the end of the intervention at the time of discharge of the neonate from the NICU to compare the trajectories of scores between the groups. The recommended content for the schedule of enrolment, interventions, and assessments of the MMS trial is displayed in Fig. 2.
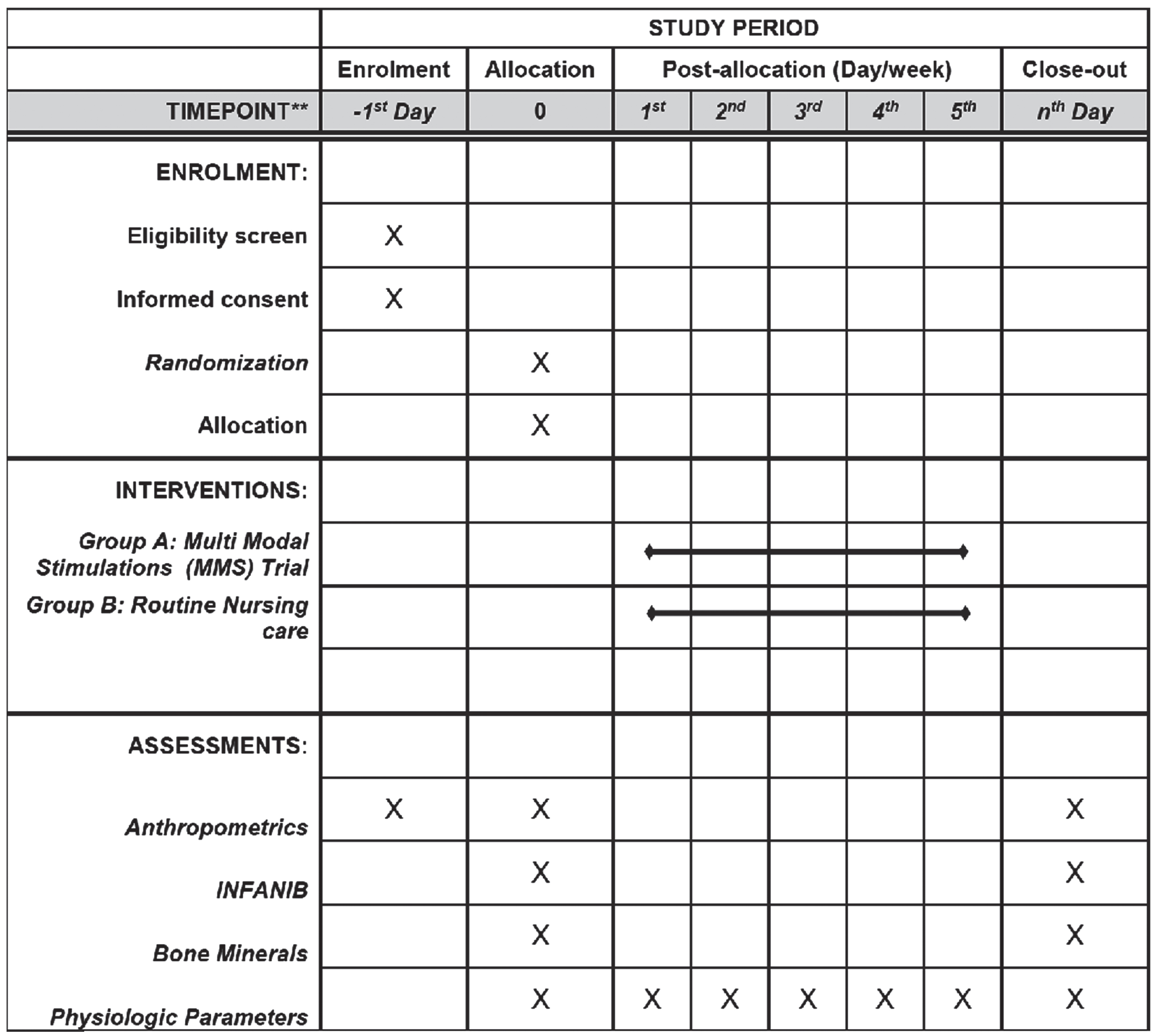
Recommended content for the schedule of enrolment, interventions, and assessments of MMS trial.
Fidelity of therapist
The application of the MMS trial and outcomes will be assessed by a pediatric physiotherapist practicing in NICU with five years’ experience in handling the neonates. This therapist will be known as the Investigating Therapist (IT).
Fidelity of data management
The gathered data will be then overseen by the Data and Safety Monitoring Committee (DSMC) of the University. The DSMC operates independently from the IT and has a key role in surveilling the outcome of the trial, monitoring adverse effects, and supervising IT. After at least 50% of neonates have been randomized, an interim analysis will be done by the DSMC to check the expected or other unintended effects of the MMS trial. Twice per month, the DSMC will conduct a surprise audit of the MMS trial.
Confidentiality
Each recruited PN will be given a serial code. (Example: C1 for the first PN of the control group and M1 for the first PI of the MMS trial group). Only the IT will hold the key to the code matching demographic details of the PNs. An encrypted user password will be used to secure the collected data in the Microsoft® Office Excel 2019 and stored in a personal desktop of the IT to prevent unauthorized data access and data theft. All the encrypted user passwords will be shared only between the IT and the data analyst. Data will be archived for future reference without the names of PNs or any other features which can reveal the identities of PNs.
Data analysis
Qualitative variables (nominal and ordinal scales) of the demographic dimensions collected will be reported in frequency rounded off to one decimal with percentages. An example of a qualitative variable is type of delivery. For quantitative variables (interval and ratio scales) such as, demographic, physiological parameters including HR, RR, Body Temperature (BT), and BP and outcome measures (Primary: INFANIB and secondary: Bone minerals and parathyroid hormone), “goodness of fit” will be determined by Shapiro-Wilk (SW) test [32]. The SW test will be applied to assess for their normality, as the sample size of an individual group is less than 50 (n < 50) [33–35]. The normality test can be sub-classified into descriptive statistics and theory-driven methods which use both graphical and numerical methods [34, 36]. The normality will be verified by numerical methods, skewness, and kurtosis for descriptive statistics and SW test for the theory-driven method. If skewness approaches zero (0) and kurtosis approaches three (3), then according to the descriptive statistics method, the data will be considered normal [36]. Similarly, if SW test is not significant (p > 0.05), then the distribution will be considered normal [32]. The descriptive statistics of the normally distributed data will be expressed in mean±standard deviation. A paired t-test will be adopted to find out the differences within Group-A and group-B for pre-post intervention changes. If PNs continue to stay longer than five days duration, repeated measures ANOVA/Friedman test will be used to compare the intervention changes at regular intervals till the time of discharge of neonate from the NICU within the groups. Independent t-test will be used to compare the changes in mean values of the outcome measures between Group A and Group B at baseline, after every five days, and at the end of the intervention at the time of discharge of neonate from the NICU, thereby allowing for comparison of the trajectories of scores between the groups. If the data does not follow a normal distribution, then the descriptive statistics will be reported in medians with interquartile ranges (IQR). Wilcoxon signed-rank test will be adopted to find out the differences within Group A and Group B for pre-post intervention changes and to compare the intervention changes at regular intervals till the time of discharge of neonate from the NICU, Friedman test will be used. A Mann-Whitney U test will be used to compare the changes in median values of the outcome measures between Group A and Group B at baseline, after every five days (to record trajectories of scores between the groups), and at the end of the intervention at the time of discharge of neonate from the NICU in order to compare the trajectories of scores between the groups. In this design, type of intervention (MMS and control) is the independent variable, neuromotor outcome measured by INFANIB and bone minerals are the dependent variable, and categories of preterm [extremely preterm (less than 28 weeks), very preterm (28 to 32 weeks), and moderate to late preterm (32 to 37 weeks)] are covariates, the potential confounding variables for which to control. Hence, analysis of covariance (ANCOVA)/Quade’s test for non-parametric Analysis Of Covariance (ANCOVA) will be used to make adjustments in the dependent variable. In addition to the above objective measurements, subjective visual observation method will be used to describe the status of the PNs. Both the primary (INFANIB) and secondary (bone minerals) outcomes with anthropometric parameters will be recorded at baseline, after every five days, and at the end of the intervention at the time of discharge of neonate from the NICU. All the data will be analyzed using, the statistical package for the social sciences (SPSS), IBM SPSS version 20.0 (Armonk, NY: IBM Corp.). For all the analyses, p < 0.05 will be considered as statistically significant.
Discussion
The long-term developmental sequelae associated with prematurity includes developmental delay in the acquisition of age-appropriate motor skills; this may be precipitated by noxious stimuli within the NICU [4–6, 37]. The maturing PN brain is highly sensitive and rapidly-changing and thus EI and a pleasant environment of NICU is required for optimal brain development [5]. Although EI is an umbrella term describing a variety of multi-disciplinary care delivered to children up to five years of age [38, 39], in the current study, it refers to the specific EI MMS protocol.
Various physiotherapy interventions, namely, positioning [40], hydrotherapy [41, 42], motor physical therapies, tactile or massage, vestibular stimulation, and sensory interventions, have been tested independently for their effects on neuromotor outcomes of PNs. The effects of multisensory and tactile-kinesthetic stimulations EI have previously been evaluated in combinations [43, 44].
Many of the reported intervention protocols were started at least a week after birth [5]. In contrast, the MMS trial will be implemented a day after the birth. Also, the majority of studies assessed only cognitive outcomes, with fewer studies assessing motor and behavioral long-term outcomes, and thus providing inconclusive evidence about the effect of early intervention across multiple developmental domains [5, 45]. Hence, neuromotor development of PNs (INFANIB) along with anthropometrics (weight, length, and head circumference) and bone minerals (serum calcium, serum phosphate, and bone-specific alkaline phosphate) will be used to determine the efficacy of the MMS trial. The current study describes the MMS trial which is a uniquely customized early intervention trial to promote sensory and motor maturation among the PNs.
The effects of MMS will be studied systematically by using a standardized MMS protocol with randomized controlled study design where the control group has no intervention except standard medical and maternal care [5].
Like other reported trials this study also might minimize parental stress, improve quality of life, lead to early discharge from the NICU, and provide other hidden benefits to PNs and their families [46]. Furthermore, the study could be extended to document the neurological outcomes of preterm infants at 3, 7, and 10 months as recommended by Liao et al. [28].
Footnotes
Acknowledgments
The authors express their sincere thanks to Dr. Asir John Samuel, PhD (Physiotherapy), Professor, Department of Pediatric and Neonatal Physiotherapy, Maharishi Markandeshwar Institute of Physiotherapy and Rehabilitation, Maharishi Markandeshwar (Deemed to be University), Mullana, Haryana, India, for his assistance in writing sample size and data analysis section. This study protocol forms the partial fulfillment for the completion of structured Doctorate of Philosophy / Philosophiae Doctor (PhD) programme by the first author, Mrs. Vencita Priyanka Aranha (Roll No. 2019001 / Regn No. 14-PCM-076).
Conflict of interest
The authors have no conflict of interest to report
Funding
Mrs. Vencita Priyanka Aranha, first author, was supported by a three year PhD Research Fellowship (MMDU/Ph.D/19/2108 dated 5th Febrauary 2019) from Maharishi Markandeshwar (Deemed to be University), Mullana, Ambala District, Haryana, India during the study period.
