Abstract
PURPOSE:
Multiple synostoses syndrome (MSS) is a rare genetic condition. Classical features consist of joint fusions which notably start at the distal phalanx of the hands and feet with symphalangism progressing proximally to carpal, tarsal, radio-ulnar, and radio-humeral joints, as well as the spine. Usually, genetic testing reveals a mutation of the NOG gene with variable expressivity. The goal was to present the anatomical, functional, and radiological presentations of MSS in a series of patients followed since childhood.
METHODS:
Patients with more than 3 synostoses affecting at least one hand joint were included. When possible, genetic screening was offered.
RESULTS:
A retrospective study was performed from 1972 to 2017 and included 14 patients with a mean follow-up of 18.6 years. Mutation of the NOG protein coding gene was seen in 3 patients. All presented with tarsal synostoses including 9 carpal, 7 elbow, and 2 vertebral fusions. Facial dysmorphia was seen in 6 patients and 3 were hearing-impaired. Surgical treatment of tarsal synostosis was performed in 4 patients. Progressing joint fusions were invariably seen on x-rays amongst adults.
CONCLUSION:
Long radiological follow-up allowed the assessment of MSS progression. Feet deformities resulted in a severe impact on quality of life, and neurological complications secondary to spine fusions warranted performing at least one imaging study in childhood. As there is no treatment of ankylosis, physiotherapy is not recommended. However, surgical arthrodesis for the treatment of pain may have reasonable outcomes.
Abbreviations
Age at onset Bone Morphogenetic Protein Computerized Tomography distal interphalangeal Fibroblast Growth Factor Fibroblast Growth Factor receptor 1 Oncogene Partner Growth Differentiation Factor Magnetic Resonance Imaging Multiple Synostoses Syndrome proximal interphalangeal
Introduction
Multiple synostoses syndrome (MSS) was first described by Maroteaux et al. in 1972. Today, the diagnosis remains clinical and is based on three associated signs: symphalangism of hands and feet, deafness due to auditory ossicle ankylosis, and facial dysmorphism [1]. Historically, symphalangism was defined by Cushing in 1916 [2] to describe the failure of interphalangeal joint formation, either proximally or distally. Later, the clinical and molecular heterogeneity of this syndrome was demonstrated, and the definition expanded to include different conditions such as multiple synostosis [3–5].
MSS is a very rare autosomal dominant genetic disease with an estimated prevalence of less than 1 in 1,000,000. The molecular basis for this disorder was first identified in 1998 with the demonstration of a mutation in the NOG gene, located at 17q21-22 [6]. This gene encodes a NOGGIN protein which controls the Bone Morphogenetic Protein (BMP) and is involved in skeletal and neural development throughout embryogenesis. Until now, 4 mutations have been identified corresponding to different clinical presentations with variable expressivity [3–11]. The 4 MSS types corresponding to 4 different clinical presentations are summarized in Table 1.
4 MSS types and clinical descriptions
4 MSS types and clinical descriptions
Recently, it was confirmed that overgrowth to thrive was associated with the NOG spectrum which is often useful in making a diagnosis [12]. Three other genes have been described that are associated with a similar phenotype, including MSS and other features, namely GDF5, GDF6, and FGF9 [4, 8–11]. There is great variability in mutations and associated symptoms, however the three main signs of the MSS remain constant.
The main differential diagnoses include Char syndrome with interphalangeal synostosis, ocular malformation, facial gestalt and hearing loss [13], filaminopathies with carpotarsal coalition [14], cardiospondylocarpotarsal syndrome [15], and atypical cases of fibroblast growth factor receptor 1 oncogene partner (FOP).
The management of MSS currently includes physiotherapy, joint mobilizations, and orthoses. Surgical treatment is also an option but is delicate and often omitted due to young age of patients. However, deformities and stiffness often remain despite these interventions. Pain, decreased range of motion, and lower quality of life require professional intervention. The present article aims to describe radiological findings, clinical symptoms, orthopedic management, and long term outcomes. This is a rare case series, since most other published descriptions are single case reports due to the paucity of this pathology.
This is a retrospective study conducted in a monocenter between 1972 and 2017.
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent was obtained from all individual participants included in the study.
The inclusion criteria included those presenting with clinical MSS defined by hearing impairment, facial dysmorphia, and synostoses in at least one interphalangeal joint [1, 2]. The diagnosis of ankylosis was made in the pediatric orthopedic surgery unit using clinical and radiological data showing bony fusion.
The exclusion criteria included the absence of functional symptoms or lack of data.
The medical history of the patients was analyzed, including clinical symptoms, functional consequences of upper and lower limb stiffness, and the presence of spinal ankylosis. Moreover, the type of treatment they required, surgeries performed, and the type and duration of physiotherapy performed were recorded. Numeric pain intensity and health-related quality of life auto-questionnaires were systematically sent to the patients to assess consequences in daily life.
All x-rays, MRIs, and CT scans were reviewed by radiologists to better describe the skeletal presentation. Finally, for those who had been evaluated in the genetics department, the diagnosis conclusion and the results of molecular screening were recorded.
Results
A total of 14 patients were included in this study (7 males, 7 females). The mean age of patients at the time of inclusion was 25.9 years old (11–48). The mean follow-up was 18.6 years [1972; 2017]. A total of 6 patients were seen in the genetic center of the hospital where genetic confirmation was obtained for only 3 patients with the identification of NOG mutations.
Three families, with 3 or 4 individuals with MSS were included in this series. They had varied clinical presentations. The most frequent synostosis locations were tarsal (100%) and carpal (64%) coalitions.
This study includes 1 case of brachymetacarpia (first metacarpal) and 4 cases of brachymetatarsia (median digits). It also includes Two cases of vertebral fusions, 2 of thoracolumbar scoliosis, 1 of craniosynostosis, 1 of femoropatellar instability, and 1 of neck hemangioma. All patients presented with either bilateral carpal or tarsal fusions and 8 presented with both. Three children had an early age at onset (AAO) but this was not screened in most cases (no x-rays collected before puberty).
The mean score for the health-related quality of life auto-questionnaire was 3.5/5. The median was 4/5.
The main results are presented in Table 2.
Main Results
Main Results
In this study 1 patient had a cervical fusion, discovered at 7 years old (Fig. 1).
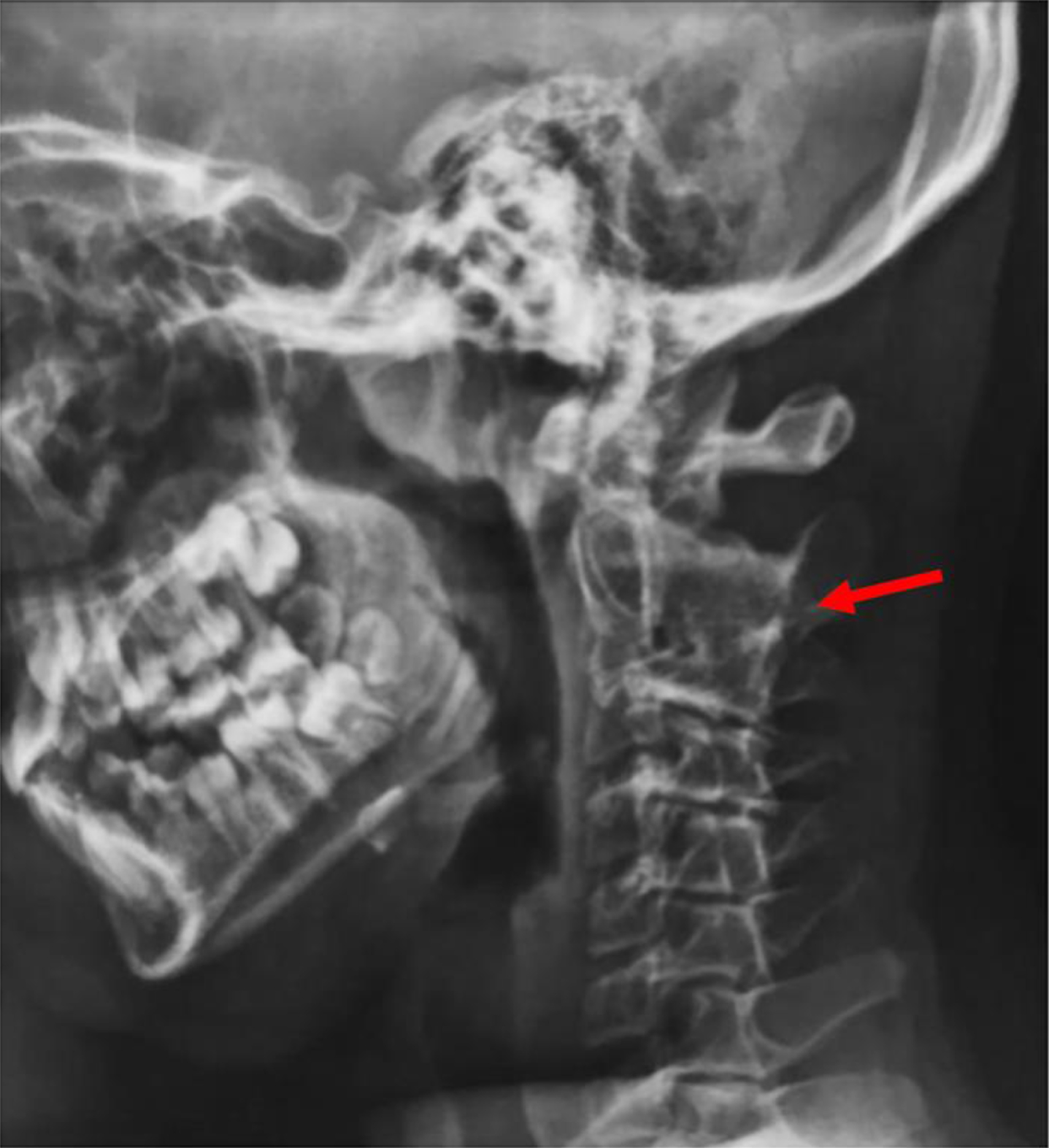
7-year-old girl with lateral X-Rays showing cervical C2-C3 vertebrae fusion.
Two patients included in this study also had cases of congenital scoliosis with large curves due to spinal fusion discovered during childhood.
These deformities were rigid and not reducible on traction x-ray. One case of congenital scoliosis was treated with a with C7-L5 posterior instrumented fusion (Fig. 2).
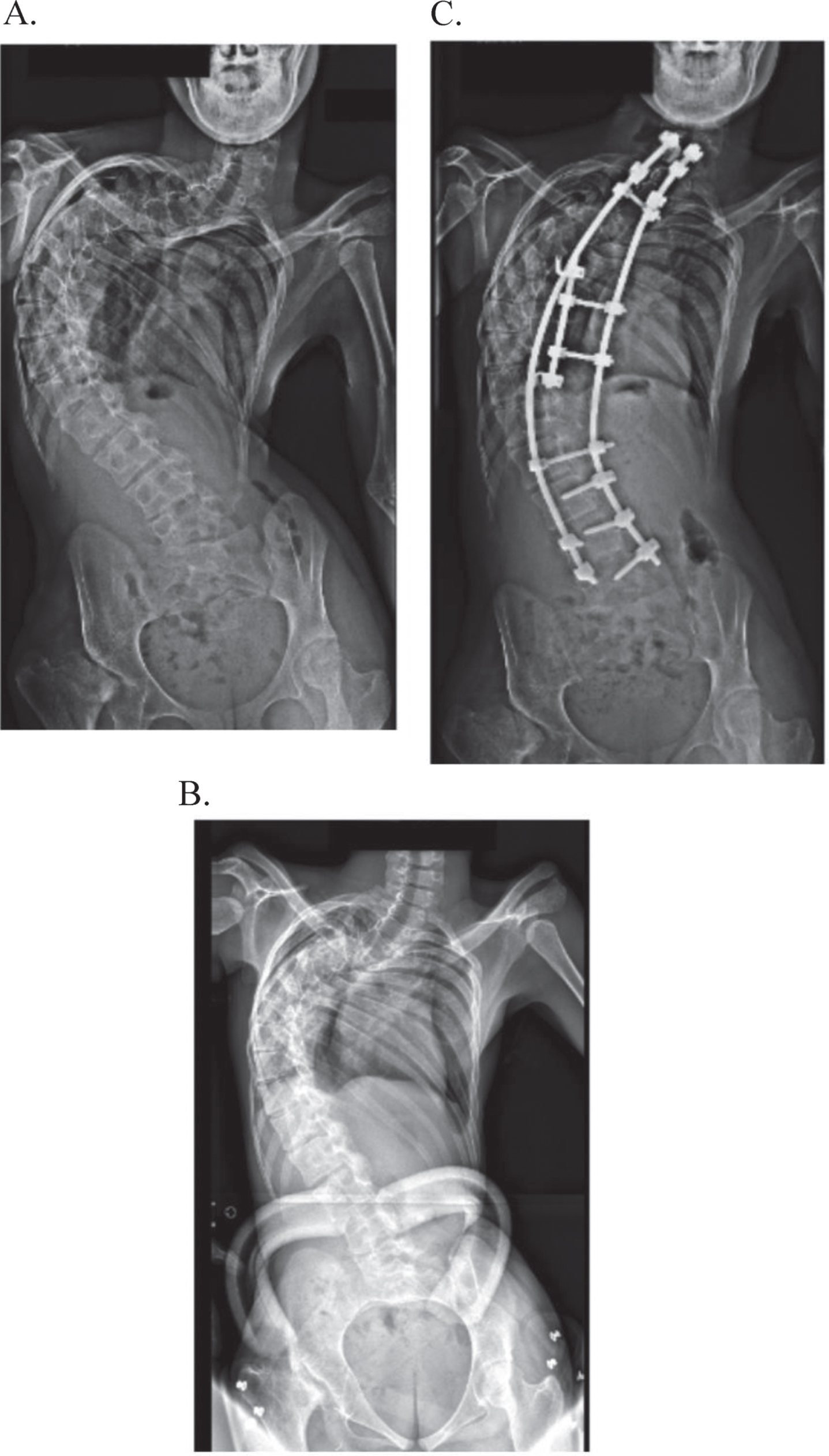
14 year old girl suffering from severe kyphoscoliosis. Frontal x-rays showing a 160° deformity (A) and no reducibility on bending image (B). Treatment was with a posterior vertebral arthrodesis (C).
All patients presented with proximal interphalangeal symphalangism with fusion, which was one of the principal inclusion criteria in the study (Fig. 3).
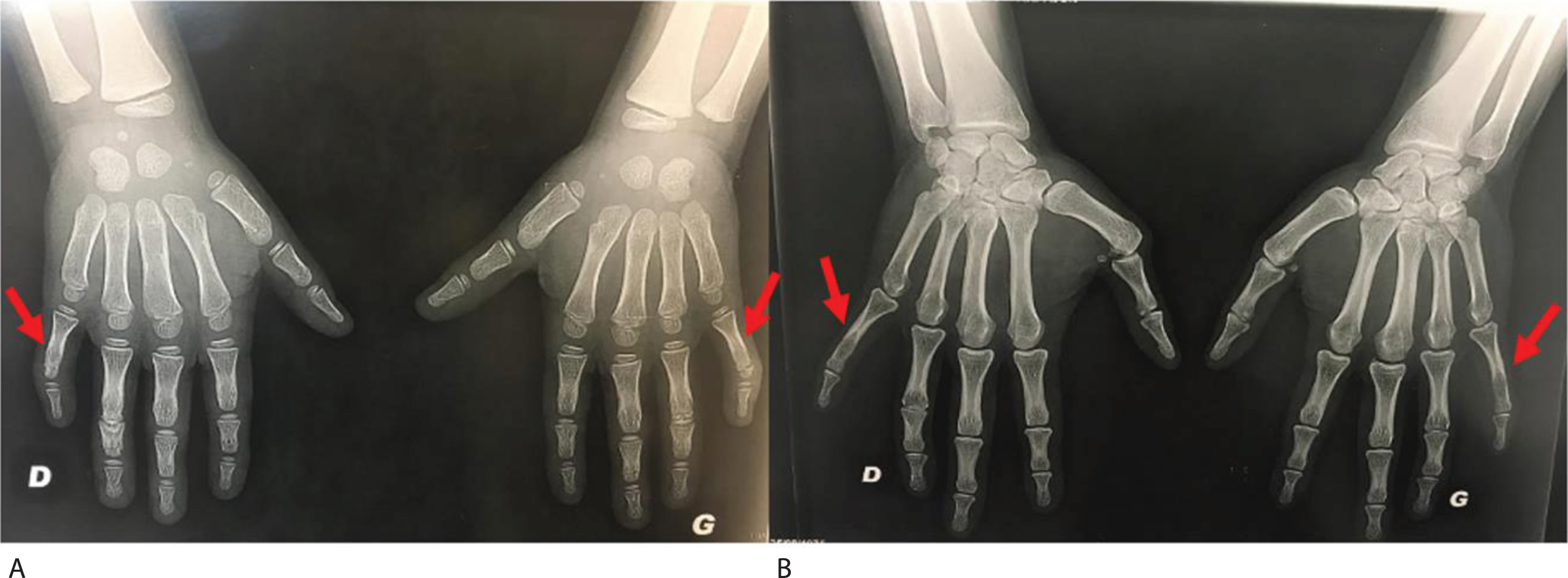
Hand x-rays of son (A) and mother (B) showing fourth and fifth digit PIP symphalangism.
Clinically, both mother and son had PIP and DIP joint stiffness with absent palmar and dorsal creases.
Although 4th and 5th metacarpals and phalanges were more frequently involved, fusions of all rays were seen in 5 patients (Fig. 4).
X-rays (A) and clinical image (B) of a 37 year old women showing symphalangism affecting all digits.
One patient presented with 1st brachymetacarpia and carpal fusions such as trapezo-metacarpal, trapezo-trapezoidal, or midcarpal joint fusions. Membranous syndactylia was seen in 2 patients. A humero-ulnar fusion led to elbow ankylosis in 2 patients (Fig. 5).
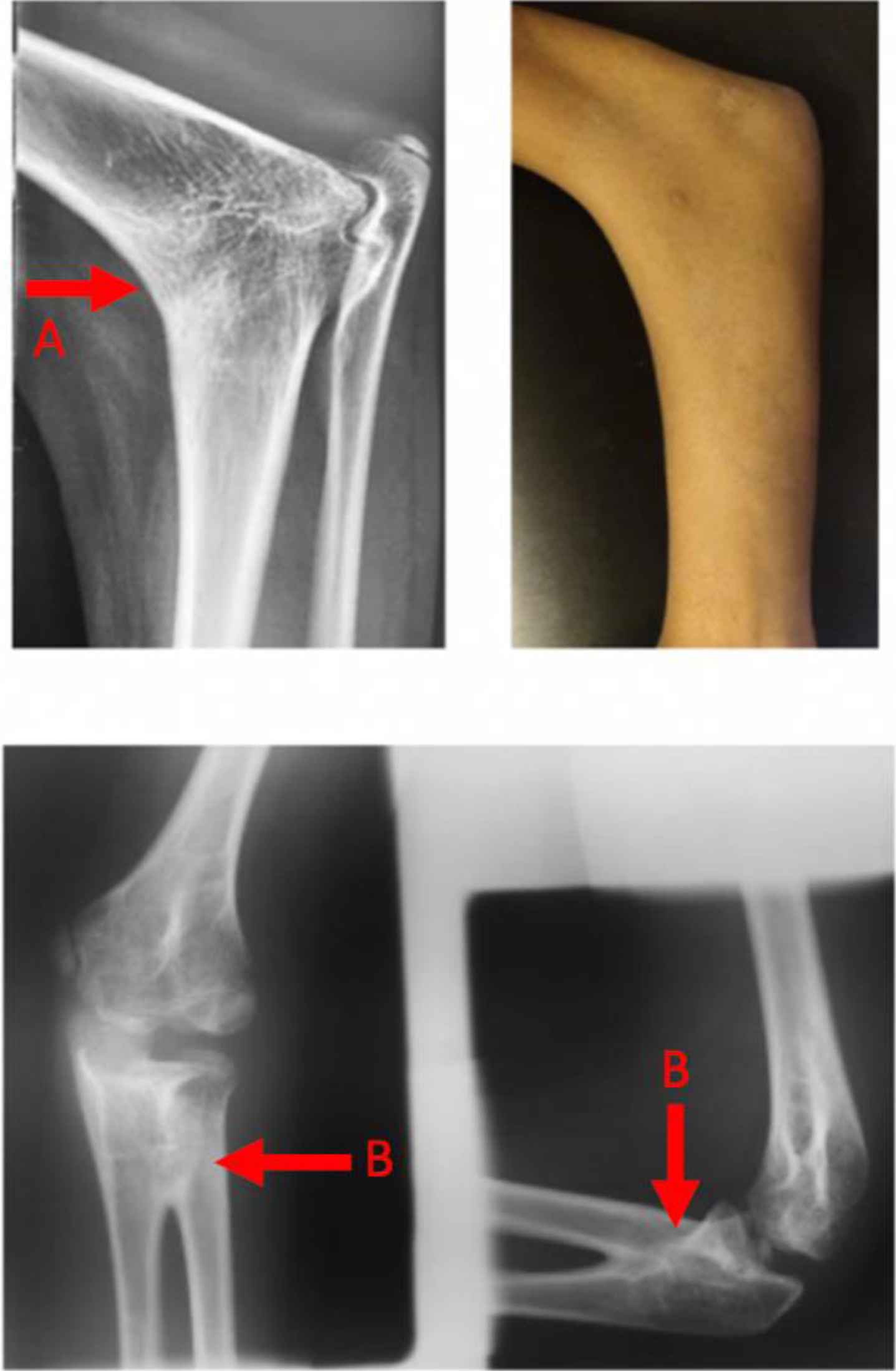
Frontal and lateral x-rays and clinical image showing an elbow ankylosis humero radial fusion (A) and radio ulnar fusions (B) in an 8 year old boy.
One patient presented with a brachial plexus birth palsy. That could be secondary to an elbow ankylosis complicating childbirth procedures. Six others had proximal radio-ulnar synostoses.
Management included regular physiotherapy and surgical treatment.
The efficacy of physiotherapy on IP fusion was difficult to demonstrate. Some patients seemed to feel better with routine exercises and stayed on a regular program. For others, the absence of a noticeable results led them to end their participation in these programs. According to the patients, symphalangism was not disabling.
Various surgical treatments were proposed. Membranous syndactylia were treated surgically without complication. A patient with brachymetacarpia of the first ray underwent successful metacarpal lengthening with an external fixator to improve gripping.
Radio-ulnar synostoses were treated surgically to correct the rotational deformity of the forearm and to place the hand in a functional position. This technique consisted of an osteotomy through the synostosis and immobilization by cast or pinning. The aim was to position the dominant hand in light pronation and the other hand in slight supination. Functional results after this surgery were excellent.
Lower limb locations
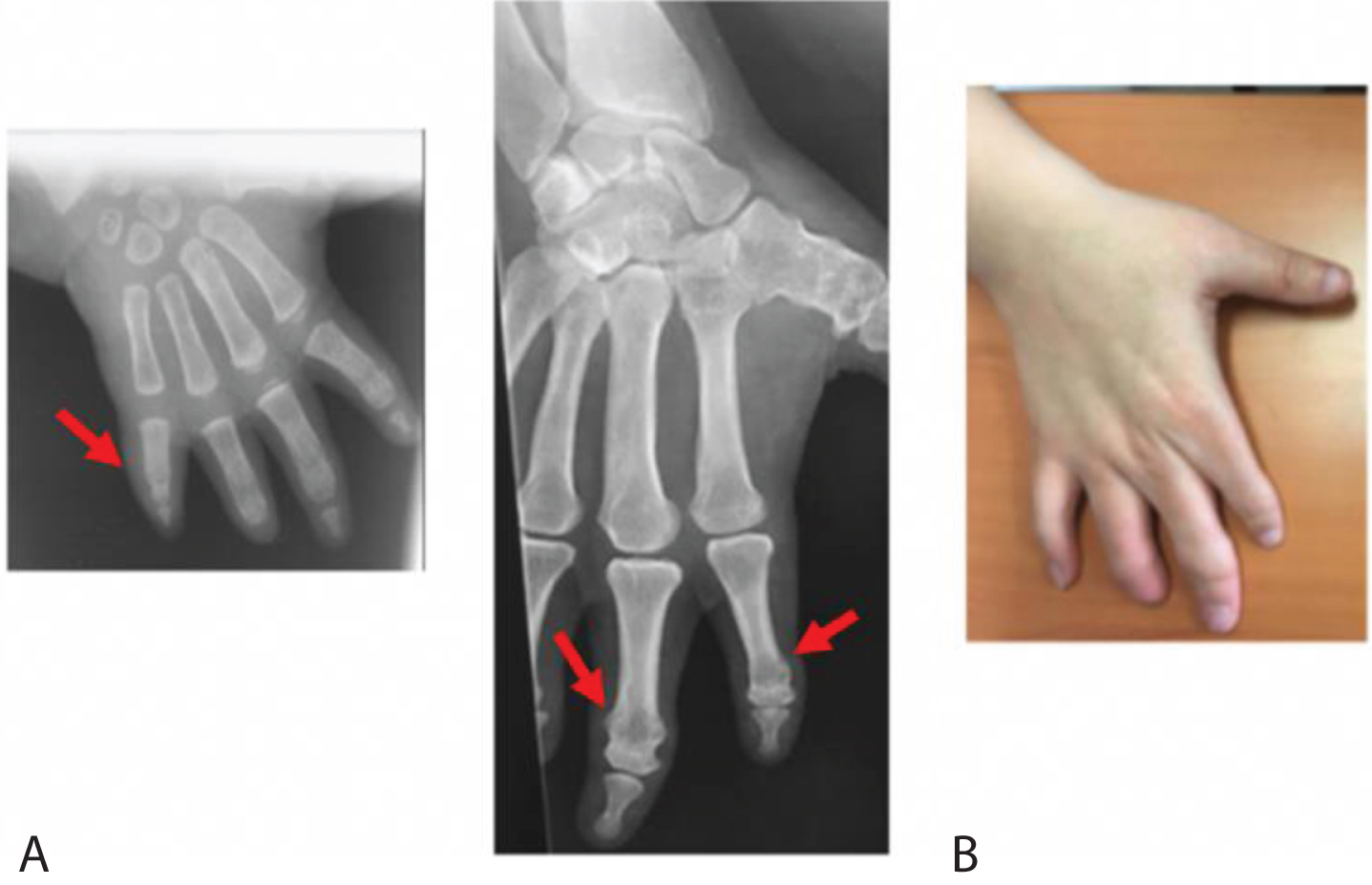
The toes were the most commonly affected by symphalangism, in particular at the 5th ray. Brachymetatarsia of the 2nd, 3rd, or 4th ray was seen in 4 patients (Fig. 6).
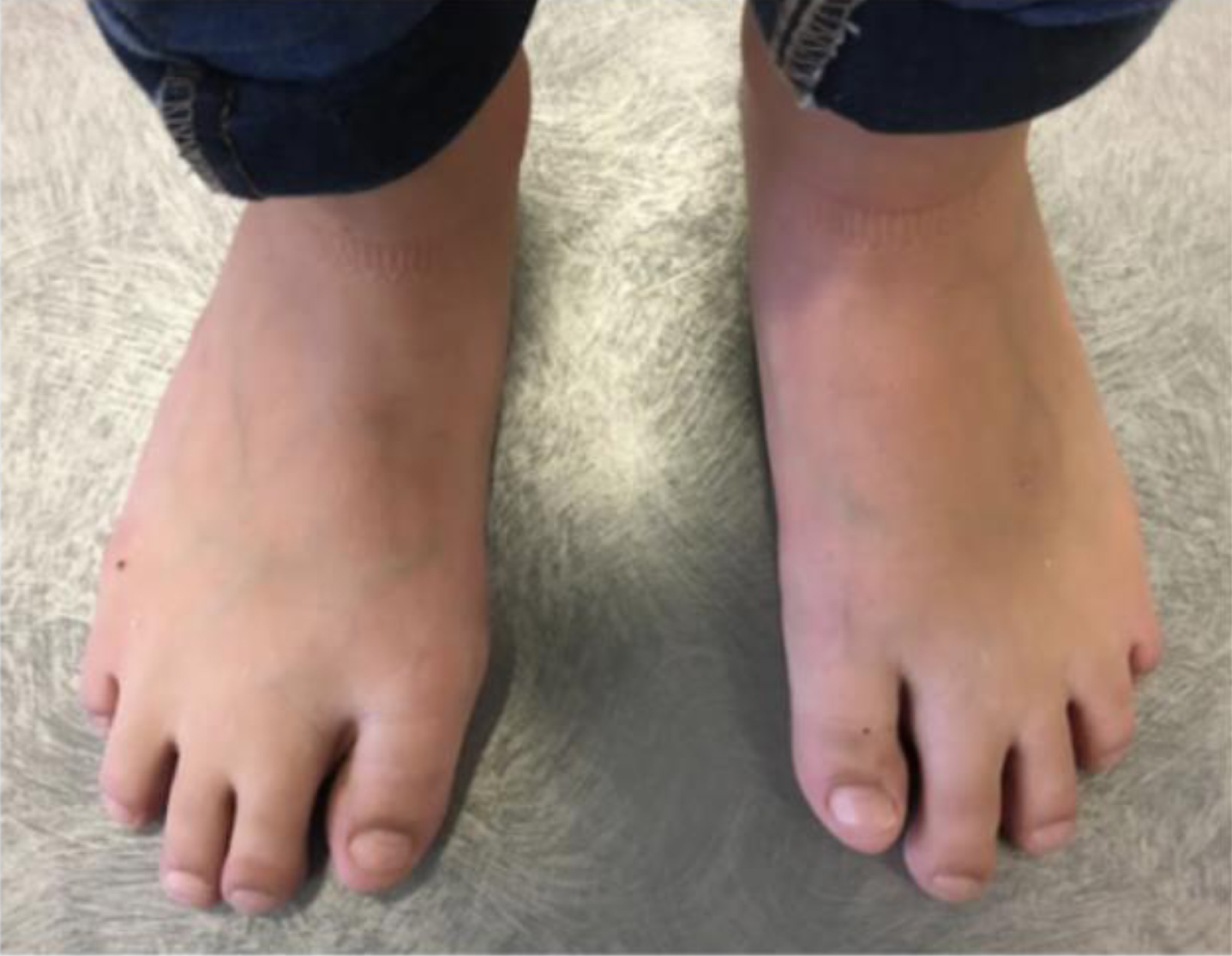
Clinical image showing first digit brachymetatarsia in a 11 year old girl.
All patients had hindfoot synostoses, most commonly in the subtalar joints which created rigid varus or valgus foot deformities. One patient had an entire tarsal fusion (Fig. 7).
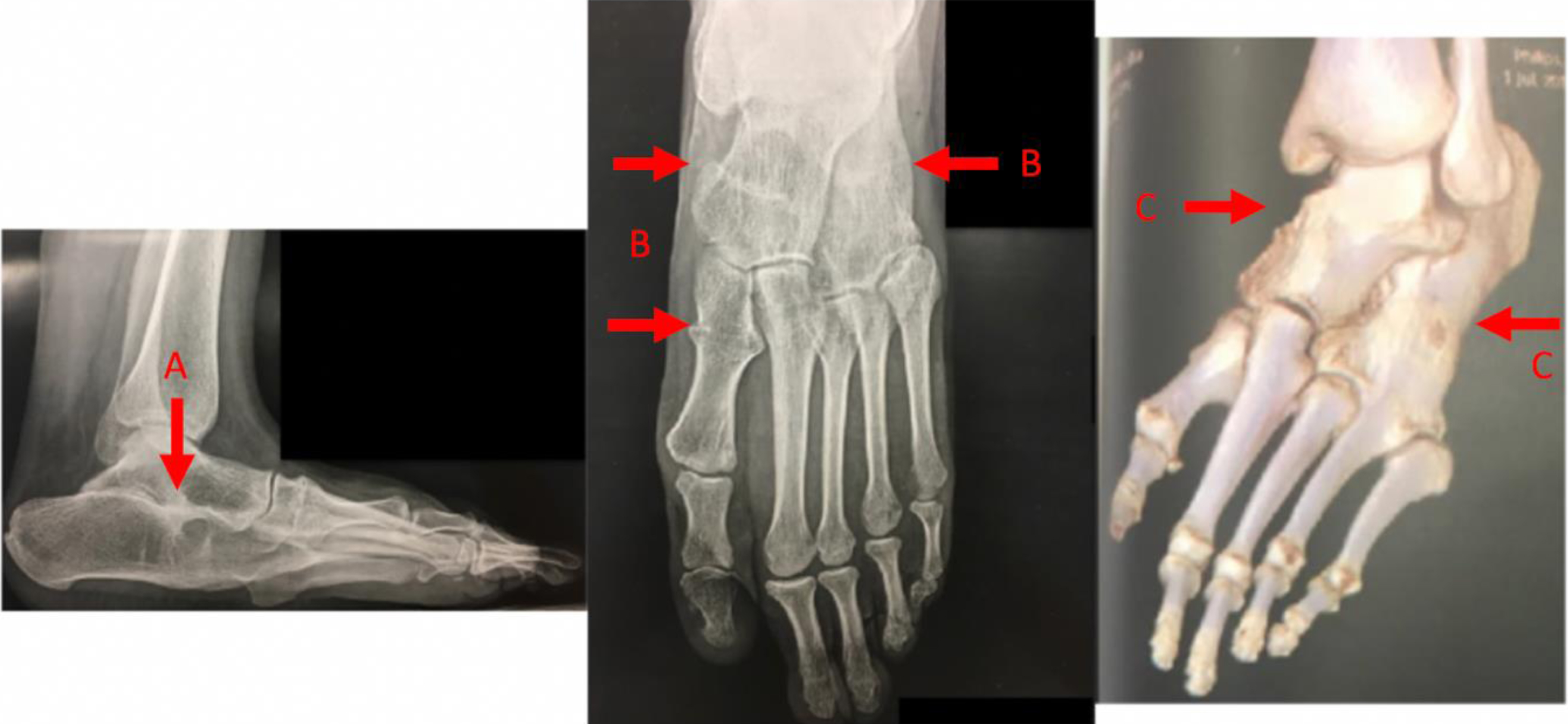
Lateral x-rays showing subtalar fusions (A) in a 15-year-old girl. Anteroposterior x-rays showing talonavicular, calcaneocuboid and tarsometatarsal fusions (B). 3D CT scan confirming hindfoot fusions (C).
One patient had recurrent patellar instability that resolved at the end of growth without radiographical skeletal abnormalities related to her syndrome.
One patient had a significant discrepancy between digits of his foot that caused difficulty to put shoes on and underwent epiphysiodesis of the base of the 3 median metatarsals. Hindfoot arthrodesis with deformity correction osteotomies was performed in 4 patients due to pain before 12 years of age (Fig. 8). All patients noted improvement following surgery.
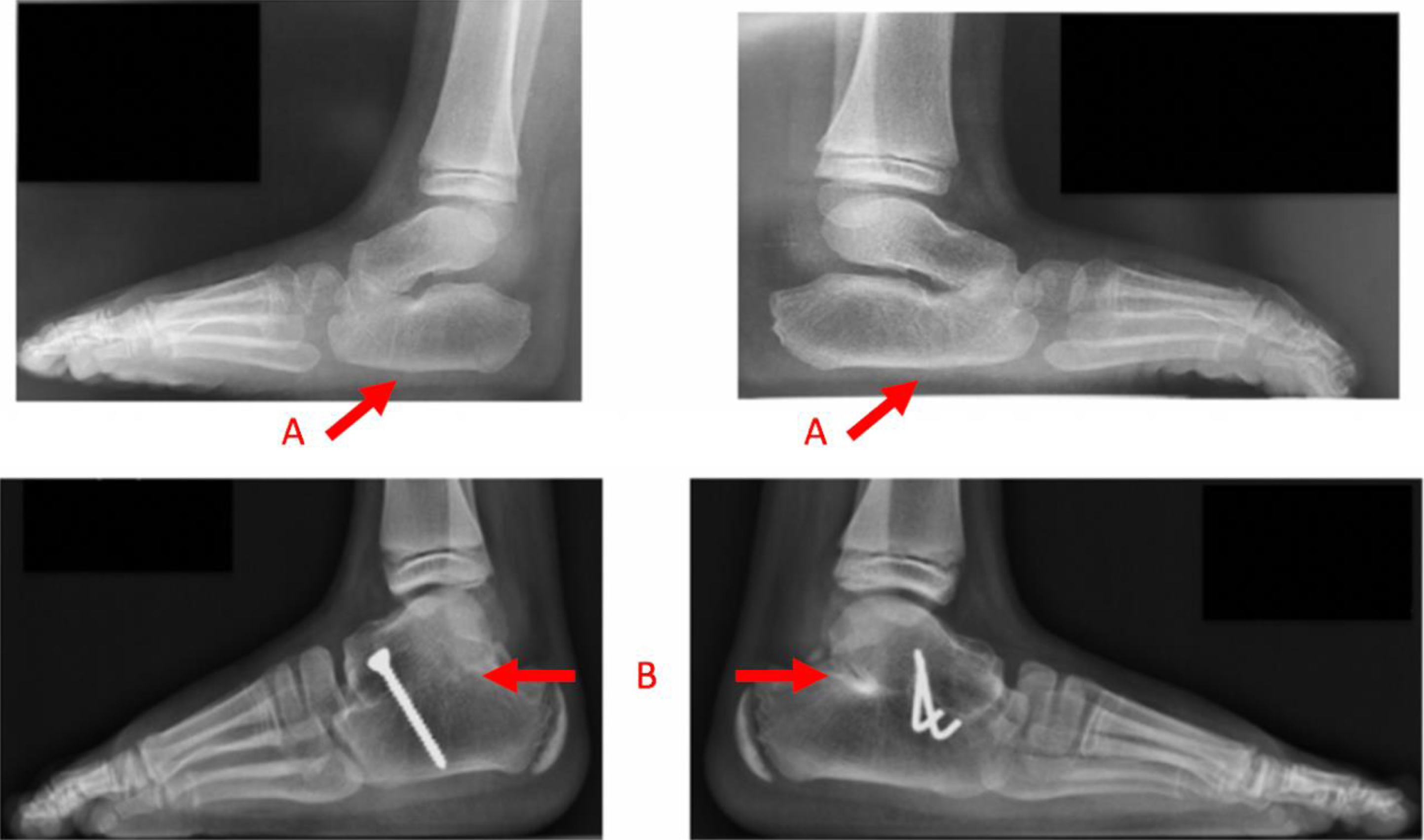
Lateral x-rays showing bilateral flat foot secondary to tarsal fusions (A) and subtalar arthrodesis (B) in a 6 year old boy.
MSS is a rare and heterogeneous condition for which there is a paucity of literature but which results in significant functional impairments for patients. This diagnosis is made clinically after identifying several findings.
One of these clinical signs is hearing loss for which a proposed treatment is palliative surgery [16]. Another symptom is facial dysmorphia which could include narrow facies, hypoplastic alae nasi, thin vermilion border of the upper lip, or a short philtrum [17].
Though MSS is very rare, isolated or associated carpal and tarsal fusions are regularly encountered in this condition.
In this series and at last follow up, all patients reported a negative impact of their hand or foot ankylosis on their quality of life. Among 8 patients who were employed professionally, 90% needed accommodations. All adult patients except for one required occupational rehabilitation and accommodation as handicapped workers.
According to Gaal et al. [16], the results of this study demonstrated that patients adapted to upper limb disease through occupational therapy. There is no curative treatment currently available for upper limbs fusions. Two patients with humero-ulnar fusions were not operated on since they had fixed 70-80° flexion and 15° pronation which allowed daily life movements. At their final follow-up, those patients had jobs that required computer work for which pronation facilitated typing. In the 1980s, Palmieri [18] proposed to make fixed joints mobile by interposing silicone caps or implants but the results were poor. Further, that technique is not appropriate for growing children. This study included several cases of ungual hypoplasia or agenesis and abnormalities of dermatophytes such as an absent palmar crease or digital prints. These symptoms were also described by Da Silva who found, in addition to classical synostoses, absence of phalanges and nails [19]. In accordance with the results of this study, Hachimi found brachymetacarpia and clinodactyly in a 39 year old woman as well [20].
Conversely, lower limb synostoses required intervention in several of the patients in this study. Flat foot deformity or rigid cavo-varus foot caused pain and instability with walking, justifying orthopedic surgical management. The 4 patients included in this study that underwent hindfoot surgical arthrodesis were improved their pain and proprioception in gait.
Other lower limb variations were found including a case of bilateral hip dysplasia and proximal radio-ulnar synostosis in a patient with symphalangism but without a genetic diagnosis confirmation. Hip dysplasia was treated non-surgically. A case of patellar instability resolved at skeletal maturity with physiotherapy and without evidence for a link with MSS. This is an illustration of phenotypic and genetic variations.
Some rare cases of spinal cord compressions were reported in the literature in patients with congenital scoliosis [21]. Hence, a neurological exam must be regularly performed throughout follow-up. Vertebral fusions may lead to spinal stenosis and in such cases laminectomy may be necessary, although there was no example of this in this study.
In conclusion, surgical treatment for feet deformities should be offered even in young children as it can result in a painless and stable gait. Vertebral fusions and hip dysplasia should be detected on x-rays in early childhood.
Rudnik reported a case of failure to thrive [22]. In this series, there were 2 similar cases which were managed by an endocrinologist.
While those with MSS may have a large variety of mutations, this series revealed some shortcomings. More than 30 mutations are today described in the literature [23] but indeed, very few genetic confirmations were available for the patients in this study. For older patients, molecular screening was not available and some of them have been lost to follow up. Among the younger patients, one family refused genetic testing and one analysis is still in progress at the time of publication. This study reported at least one de novo mutation.
Most authors hypothesize that the disease progresses with time, notably at the hand, proximally to distally and ulnar to radial. In fact, MSS may probably be a late diagnosis on x-ray owing to the endochondral ossification and the difficulty to assess PIP and DIP joints in newborns. It is possible that reduced mobility exists at birth but is difficult to objectively diagnose. This could explain the brachial palsy observed in this study.
Finally, diagnosis of MSS must be considered when seeing a newborn presenting with stiff joints and family history, even though it is necessary to keep in mind that some mutations can appear de novo. Genetic confirmation and a familial genetic investigation should be offered to every patient. Radiological exams should be obtained during the growth of the child to prevent complications, particularly of the spine. Growth, puberty, and functional consequences should be monitored in children to prevent complications in adulthood.
Conclusion
MSS is clinically diagnosed in the presence of joint fusions, deafness, facial dysmorphia, and family history. This diagnosis can be confirmed with genetic testing. It appears that, except for forearm synostosis, upper limbs fusions only require functional rather than surgical intervention. In contrast, foot pain and instability can be improved by surgical stabilization. In all cases, pain and stiffness should be managed by rheumatologists, rehabilitation physicians, and occupational therapists as there is no curative treatment for ankylosis. Although rare, the severity of the damage to the spine and the hips in the case of synostosis in these locations necessitates screening radiography before children with MSS reach their pubertal growth spurt. All children should have deafness screening and growth curve monitoring, which should prompt endocrine consultation and repeated neurological examinations.
Footnotes
Acknowledgments
The authors wish to acknowledge Dr. G. Baujat, Geneteic Department, Institut Imagine, Paris, France, for her help in preparing this manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
