Abstract
PURPOSE:
Feeding problems are common in premature infants (PIs) and may lead to negative consequences such as malnutrition, dehydration, excessive weight loss, as well as developmental and psychological deficits. Moreover, they are associated with increased length of hospital stay/cost. There is not enough evidence on how feeding problems should be treated in PIs. The goal of this study was to investigate the effects of non-nutritive sucking combined with oral motor stimulation and oral support on feeding performance in PIs.
METHODS:
A single-blind randomized clinical trial was performed on 44 PIs with feeding problems. Patients were randomly categorized into two groups: (1) combined intervention (CI) and (2) non-nutritive sucking (NNS). The CI group received NNS, oral motor stimulation and oral support simultaneously. Infants in both groups received 14 treatment sessions for 14 consecutive days. The Preterm Oral Feeding Readiness Assessment Scale (POFRAS) was used as the primary outcome measure. Weight, volume of milk intake, time to achieve full oral feeding, and length of hospital stay were secondary outcome measures. All measures were assessed before treatment, after the 7th session, after the 14th session, and after 7 days after the end of treatment.
RESULTS:
Both groups improved in all outcome measures across time (P < 0.001). The improvements in the POFRAS, volume of milk intake, and time to achieve full oral feeding were significantly greater in the CI group than the NNS group (P < 0.001). The improvements attained in weight and length of hospital stay were not significantly different between the CI and NNS groups (P > 0.05). Large effect sizes were found for POFRAS score in both CI (d = 3.98) and NNS (d = 2.19) groups.
CONCLUSION:
The current study showed that the combined intervention including NNS, oral motor stimulation, and oral support significantly improved the feeding performance in PIs.
Introduction
Premature infant (PI) refers to the neonate who’s born at less than 37 weeks gestation [1]. Premature birth is the second main cause of death in newborn babies [2]. As reported by the World Health Organization in 2016, 15 million PIs are born annually and approximately one million of them die due to complications of preterm labor [3]. Premature birth may cause many health complications such as physical disabilities, pulmonary diseases, difficulties in feeding, and developmental neurological concerns [3–9]. These problems then result in prolonged and costly hospital stays.
Feeding problems, defined as any difficulties in the safe and effective feeding performance, are the primary issues in the PI [10]. This is a dangerous condition because of its potential complications such as malnutrition, dehydration, excessive weight loss, and developmental and psychological deficits [10]. Moreover, it can lead to adverse family, economic, and social consequences [9, 10] due partially to very expensive increased lengths of hospital stays. Early diagnosis and intervention for feeding problems is necessary to improve feeding performance in PIs [9–11].
There have been various interventions using oral motor stimulation, non-nutritive sucking (NNS), and oral support that are well documented to improve feeding performance in PIs [12–24]. Oral motor stimulation (OMS) strengthens oral muscles in the lips, tongue, cheeks, soft palate, and face. Such exercises are passive and improve strength and range of motion in muscles that are engaged during oral feeding [13]. NNS enhances the coordination of jaw and tongue movements, and consequently promotes the functional patterns of nutritive sucking [13, 14]. Oral support facilitates sucking competence through bottle feeding and improves feeding performance by providing stability and pressure to the mandible [12].
There is not sufficient evidence to show that NNS combined with OMS and oral support is more effective than NNS alone. Therefore, this study aimed to investigate the effects of NNS combined with OMS and oral support on feeding performance in PIs.
Methods
Study design
This study is a single-blind randomized clinical trial. Each assessor was blinded to treatment allocation.
The research council of rehabilitation faculty and the ethical committee of Iran University of Medical Sciences (IUMS) approved the protocol of this study (ethics code: IR.IUMS.FMD.REC1396.9411360007). This trial was registered with the code IRCT20171023036956N2 in Iran registry of clinical trials.
Patients
PIs with feeding problems were recruited from the neonatal intensive care unit (NICU) of the Firoozgar, Rasule-e Akram and Aliasghar hospitals of the IUMS in Tehran, Iran, between August 2017 and October 2017. The inclusion criteria were: 1) born at 30 to≤36 weeks gestational age (GA), 2) Apgar score≥3 at 5 minutes, 3) birth weight appropriate for GA, 4) presence of feeding problems based on physician diagnosis, and 5) feeding via nasogastric or orogastric tube. The exclusion criteria were: 1) presence of conditions including facial abnormalities, intraventricular hemorrhage, congenital syndromes, or respiratory, cardiovascular, neurological, or digestive disorders as determined by physician diagnosis, or 2) reluctance of the infant’s parents to participate in this study. All the parents of eligible infants signed a consent form before participating in the study.
Sample size
The primary outcome measure of this study was feeding readiness measured using the Preterm Oral Feeding Readiness Assessment Scale (POFRAS). Assuming an effect size of 0.88 for POFRAS with alpha set at 0.05 and a power of 0.8, as well as accounting for a 10% rate, the calculated necessary sample size was 22 patients in each group.
Randomization
Eligible infants were randomized using opaque sealed envelopes with a computer- generated random list of numbers. They were randomly allocated to the CI group (n = 22) or NNS group (n = 22).
Intervention
The treatment sessions were carried out at the NICU of the Firoozgar, Rasule-e Akram and Aliasghar hospitals of the IUMS. All infants in both groups received treatment for 14 consecutive days 30 minutes before the initiation of scheduled tube feeding. Treatment was employed at the beginning of 30 weeks postmenstrual age (PMA) after the pediatrician allowed for gavage feeding and the infant was medically stable. All infants were routinely monitored for vital signs (such as changes in respiratory rate, heart rate, or SpO2), behavioral cues, and stress symptoms (such as changes in color, state of alertness, and breathing). In order to prevent the above symptoms, treatment was delayed until clinical stability was established. Infants did not receive any undesirable stimuli 30 minutes before the treatment sessions. Moreover, treatments were provided when they were in a quiet waking state according to Holditch et al. [15]. Treatments were provided by the principal researcher who is an expert in the treatment of feeding problems in PIs. It is necessary to mention that the reliability of the treatments used in this study has never been formally tested or published. The only oral motor program with published intervention fidelity is Lessen’s (2011) Premature Infant Oral Motor Intervention [16].
All infants received their mother’s breast milk for feeding, and when this was unavailable pasteurized human donor breast milk was used. However, the results of this study were not analyzed according to the type of milk. The infants were fed via nasogastric/orogastric tube before treatment. The timing when they could orally receive milk from the breast or bottle, the duration, volume of milk, and frequency of oral feeding were predetermined according to physician’s orders. The physician was blinded to group assignment. Before treatment, infants’ families were asked not to participate in other feeding treatment programs.
Non-nutritive sucking
In the NNS method, the pacifier was placed in the infant’s mouth and the infant was allowed to suck on it. If the infants made no effort to suck, the pacifier was manipulated by the therapist to stimulate sucking. The NNS was provided for 5 minutes, 7 to 8 times per day [14] for 14 consecutive days.
Combined intervention
The infants in the CI group received NNS (5 minutes, 7-8 times per day), OMS (12 minutes, once a day), and oral support (10 minutes, twice a day) for 14 consecutive days. OMS included gentle stroking of oral structures (cheeks, lips, gums, and tongue) according to Fucile et al. (2002). OMS was provided for 12 minutes, once a day [14, 24]. Oral support was given to support the chin and cheek during feeding and to improve deglutition. In oral support, the cheeks are compressed towards the lips by the thumb and ring fingers on both sides to reduce fluid loss. The little finger supports the chin to stabilize the jaw. Oral support was provided for 10 minutes, twice a day [12].
Outcome measures
Primary outcome measures
The POFRAS was used as the primary outcome measure. Feeding readiness was measured using the POFRAS before treatment (T0), after the end of the 7th session (T1), after the end of the 14th session (T2), and 7 days after the end of treatment (T3).
The POFRAS is a clinical tool for examination of the readiness of infants for full oral feeding [25, 26]. The POFRAS contains 18 items on the corrected gestational age, behavioral conditions, oral reflexes, oral posture, NNS, and other significant signs such as the accumulation of saliva in the mouth, skin color changes, apnea, tonus variations, changes in positions of body, tremor of the tongue or jaw, hiccups, and crying. This is a quantitative scale and each item is scored 0–2. The maximum possible score is 36, which is the sum of all sub-tests. A score below 30 indicates that the infant is not ready to feed. The POFRAS is valid and reliable with a sensitivity of 60% and a specificity of 75% [26, 27].
Secondary outcome measures
The secondary outcome measures were volume of milk intake, infant weight, time required to achieve full oral feeding, and length of hospital stay.
The volume of milk intake per feeding was measured using a syringe. The mean (±SD) of volume intake per feeding was recorded at T0, T1, T2, and T3. The infant’s weight was recorded using digital balance at T0, T1, T2, and T3. The time required to achieve full oral feeding from the breast or bottle was considered as the number of days from the beginning of intervention until the infant obtained independent oral feeding. All infants received gavage feeding before treatment. Oral feeding means that infant could orally feed at least once a day with no signs of oxygen desaturation, apnea, or bradycardia. The criterion for full independent oral feeding was reaching eight oral feedings per day [23]. The length of hospital stay was defined as the number of days between the NICU admission and discharge date.
Statistical analyses
SPSS Statistics V22, SPSS Inc., Chicago, IL, USA was used to analyze all data. The Kolmogorov-Smirnov test revealed normal distribution of data. Independent t-test and chi-square tests were used to compare data between the groups at baseline. Repeated measures ANOVA was used to determine the main and interaction effects of time and group on the outcome measures. The Bonferroni test was used for post hoc analysis. The independent t-test was used to analyze the between groups comparison of the hospitalization period and the time required to achieve full oral feeding. Finally, the effect sizes (Cohen’s d) of the changed scores were calculated to determine the treatment effects. The effect sizes were defined as: < 0.20 (negligible);≥0.20 but < 0.50 (small);≥0.50 but < 0.80 (moderate); and≥0.80 (large) [28]. Results were reported with a 95% confidence interval and the significance was considered at p < 0.05.
Results
The flow chart of participants is shown in Fig. 1. From the 96 premature infants that were admitted to the NICU of the hospitals of the IUMS, a total of 44 PIs (mean gestational age at birth: 32.5, SD = 2.01) were included in this study. All subjects completed the study protocol. There were no significant differences in demographic characteristics between the groups (Table 1) (p > 0.05). Moreover, there were no significant differences between two groups in regard to POFRAS, infant’s weight and volume of milk intake at T0 (Table 2) (p > 0.05).
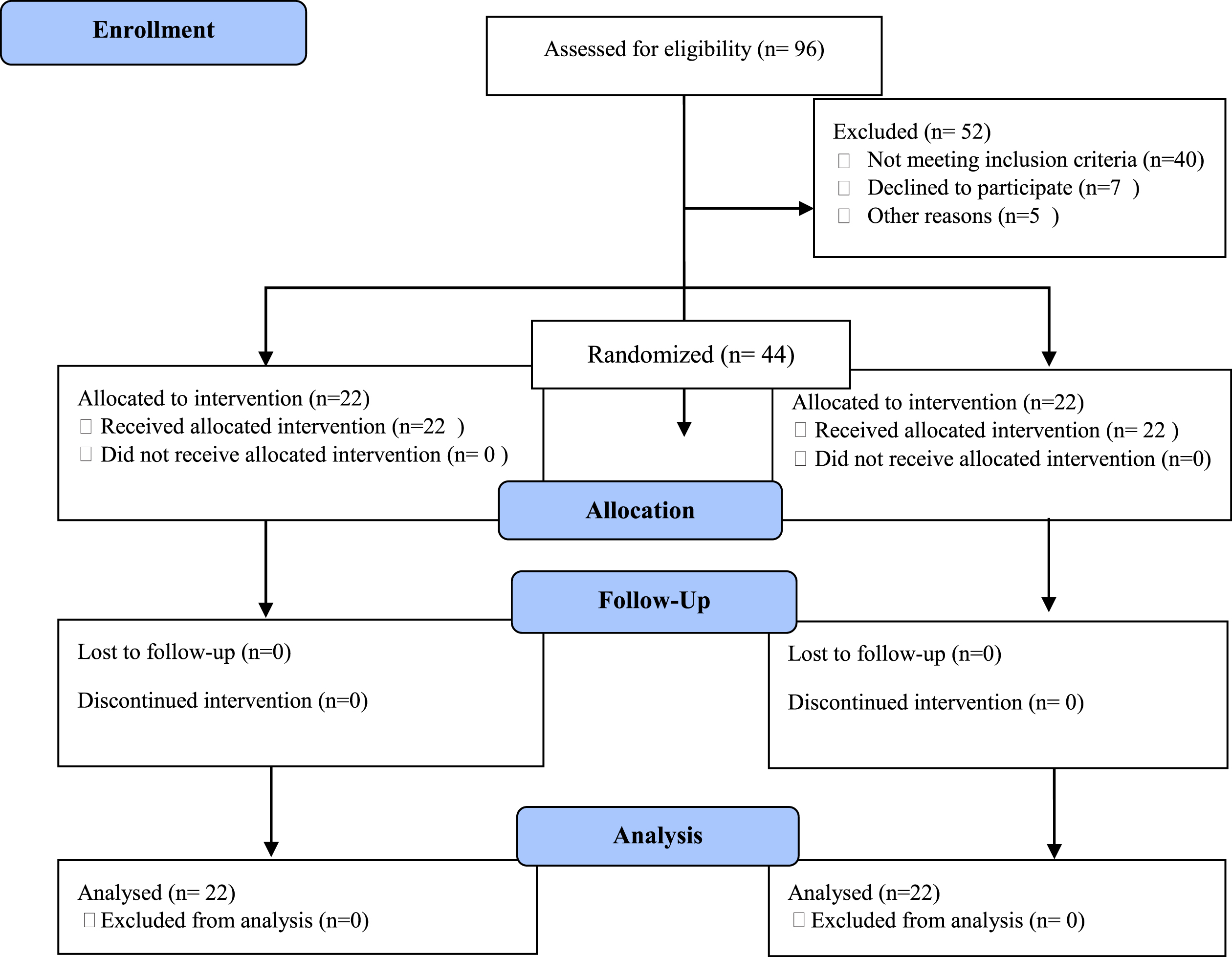
Flow chart of participants.
Demographic Characteristic of Preterm Infants
a: Postmenstrual age (gestational age plus chronological age); b: before treatment; c: Nasogastric; d: Orogastric; e: between groups comparisons at T0.
Mean and SD of primary and secondary outcome measures for nonnutritive sucking stimulation (NNS) and Combined Intervention (CI) groups
T0 = before treatment; T1 = after 7th session; T2 = after 14th session; T3 = 7 days follow-up. a: Preterm Oral Feeding Readiness Assessment Scale; b: between groups comparisons at T0.
The mean (SD) of the POFRAS score, weight and volume of milk intake of both groups at T0, T1, T2, and T3, as well as p-values for baseline differences are presented in Table 2. All three outcome measures showed improvement in both groups.
A significant effect of time (p < 0.001) was seen on POFRAS. Bonferroni post-hoc test revealed that POFRAS scores increased across time in both groups (p < 0.001). The improvement of POFRAS scores was maintained at T3 (p = 0.13). The main effect of Group on POFRAS was significant (p = 0.03) but the Time×Group interaction was not significant (p = 0.45) (Table 3). Large effect sizes were found for POFRAS score in both the CI group (d = 3.98) and the NNS group (d = 2.19).
Main effect of Time and Group and their interaction effect on primary and secondary outcome measures (CI = 95 %)a
Main effect of Time and Group and their interaction effect on primary and secondary outcome measures (CI = 95 %)a
a: The effect of NNS versus CI on outcome measures was analyzed using a group (NNS and CI)×Time (T0, T1, T2 and T3) mixed model ANOVA at significant level≤0.05; b: Preterm Oral Feeding Readiness Assessment Scale.
A significant effect of Time was seen on volume of milk intake (p < 0.001). Post-hoc analysis showed that this increased significantly from T0 to T2 (p < 0.001) in both groups. Moreover, the improvement of volume of milk intake was maintained at T3 (p = 0.21). The main effect of Group on volume of milk intake was significant (p < 0.001). There was not a significant Time×Group interaction (p = 0.61) for the volume of milk intake (Table 3). Large effect sizes were found for in both the CI group (d = 2.63) and the NNS group (d = 2.62).
There was a significant effect of Time on weight (p < 0.001). Post-hoc analysis showed that weight increased significantly across the time (p < 0.001) and continued to increase until T3 (p = 0.04) in both groups. The mixed model ANOVA comparing weight did not demonstrate a statistically significant Group effect (p = 0.67). The Time×Group interaction was not significant (p = 0.07) (Table 3). Small effect sizes were found for weight in both the CI group (d = 0.26) and the NNS group (d = 0.07).
The duration of hospitalization and the time required for achieving full oral feeding are presented in Table 4. There was no significant difference between the two groups for hospitalization length (p = 0.72). The time required to achieve full oral feeding was significantly different between groups in favor of the CI group (p = 0.007).
Mean (SD) days of hospitalization period and time to achieve oral feeding in nonnutritive sucking stimulation (NNS) and Combined Intervention (CI) Groups
Mean (SD) days of hospitalization period and time to achieve oral feeding in nonnutritive sucking stimulation (NNS) and Combined Intervention (CI) Groups
a: between groups comparisons.
The NNS and the OMS are among the most common treatments for oral feeding problems in PIs [29]. This single-blind randomized clinical trial investigated the effect of NNS combined with OMS and oral support on feeding performance in PIs. The results revealed that the POFRAS score, volume intake of milk, weight, time to achieve full oral feeding, and hospitalization duration improved in both the CI and NNS groups. However, the improvements in the POFRAS score, volume of milk intake, and time to achieve full oral feeding were significantly greater in the CI group.
Primary outcome measures
The results of this study showed that the POFRAS score improved in both groups, but the magnitude of improvement differed between groups. Scores improved significantly more in the CI group. There is a dearth of similar studies with which to compare the findings of this study. The results of this study indicate that the combination of NNS, OMS, and oral support have beneficial effects on oral motor skills and feeding performance in PIs. As a result, one can conclude that combined intervention is more effective than NNS alone for acquiring oral feeding readiness in infants. Zhang et al. demonstrated oral motor stimulation alongside NNS can be efficient on feeding performance in PIs [14].
This study revealed the large effect sizes of the POFRAS score after the end of intervention in both CI and NNS groups. These findings suggest that both interventions were greatly effective in improving the infants’ feeding performance. However, the effect size achieved for POFRAS score in the CI group was remarkably larger than that in the NNS group (3.98 and 2.19 respectively) which demonstrates that more significant improvement occurred in the CI group.
Secondary outcome measures
The results of this study demonstrate that volume of milk intake was improved in both groups and the improvement was significantly greater in CI group. Moreover, this study revealed the large effect sizes of volume of milk intake after the end of treatment in both CI and NNS groups. These findings imply that although feeding performance improves in both groups, CI techniques were more effective in increasing the volume of milk intake than NNS. The improvements in the CI group may have been achieved by improving oral sensory motor skills, and by supporting the chin and cheeks of premature infants [24, 30].
This study revealed increases in the infants’ weight in both the CI and NNS groups. There is not a similar study with which to compare this one, but there is some evidence that NNS is an effective method to increase an infant’s weight [31–33]. However, Sehgal et al. (1990) showed that NNS was less effective in improving the outcomes of oral feeding such as weight gain in PIs [34]. This discrepancy in the effectiveness of NNS may be due to the fact that the ability to perform NNS does not provide information on the oral feeding performance or the suck-swallow-breathing coordination, as respiratory patterns do not change during NNS in the same way that they would during nutritive sucking [35]. Moreover, other studies showed that OMS alone or in combination with NNS had differing results, including negative effects on weight gain in PIs [14, 36].
Furthermore, the findings demonstrated small effect sizes of weight in both CI and NNS groups. These findings suggest that both interventions were not remarkably effective in increasing infants’ weight. However, the effect size achieved for weight in the CI group was larger than that in the NNS group (0.26 and 0.07 respectively) which demonstrates that more improvement occurred in the CI group.
There is no meaningful difference in hospitalization duration between the two groups. However, the hospitalization period in the CI group was somewhat shorter than in the NNS group. This finding indicates that the combination of three interventions can be slightly more effective than NNS in reducing hospital stay length.
On average, infants in the CI group achieved full oral feeding only after 8 days of intervention, which is considerably shorter than in the NNS group. There is not a similar study to compare the results of this study. However, there is published evidence that shows various interventions such as oral motor stimulation, oral support, and NNS can reduce the time to achieve oral feeding [12–24]. Moreover, there are some studies that show that NNS plus OMS can lead to full oral feeding in a shorter period of time [14].
Although the CI group reached full oral feeding earlier than the NNS group, hospitalization length did not significantly differ between the groups. This finding is in accordance with Zhang et al. [14] and is in contrast to findings of Rocha et al. [13]. This may be because the hospitals involved in this study do not have specific discharge strategies and access to full oral feeding was not an important factor for infant discharge in hospitals.
Conclusion
This single-blind randomized clinical trial revealed that combining NNS with oral motor stimulation and oral support was more effective than NNS alone in improving the feeding performance of PIs.
The main limitation of this study is that the oral motor stimulation and oral support were not provided separately, and the effects of the CI was compared only with NNS. Therefore, future clinical trials are needed to compare this combined intervention with other feeding interventions to achieve the most effective treatment in infants suffering from feeding problems. Moreover, in this study the treatments were provided by one principal researcher without any involvement of infants’ parents. Since parental involvement is a key component in the neuroprotective care models [37] and parent-infant interactions have critical effects on infants’ physical and psychological development and normal growth [38], future studies are needed to investigate the parental participation in feeding practices.
Conflict of interest
The authors have no conflict of interest to report.
