Abstract
PURPOSE:
Pediatric outpatient procedures can be traumatic experiences for patients. This retrospective study, evaluates intramuscular midazolam as a safe option for anxiolysis during spasticity management injections.
METHODS:
We performed a retrospective chart review of 72 patients from a Tertiary Pediatric Hospital Outpatient Clinic. One hundred and twenty injections were administered over two years by a single practitioner. Comorbidities included asthma, sleep apnea, chronic obstructive disease, and epilepsy. Duration of sedation, safety of midazolam as determined through the use of the REACT (Respiration, Energy, Alertness, Circulation, Temperature) score, and frequency of side effects (prolonged sedation, breakthrough crying, medication reversal, and emergent evaluation) were recorded. A student’s
RESULTS:
The average duration of sedation was 29 minutes (95% CI 26.51–31.35) with an average dose of midazolam of 0.20 mg/kg (95% CI 0.9–0.21). None of the subjects required medication reversal or emergent evaluation. 39% of the patients had prolonged sedation (
CONCLUSION:
Intramuscular midazolam is a possible effective anxiolytic medication strategy for outpatient pediatric injections. Additional studies are needed to ensure its safety and efficacy.
Keywords
Introduction
Spasticity is a velocity-dependent increase in tone of muscles, generally found in upper motor neuron disorders. While it can be functionally helpful for some patients, it is functionally and hygienically limiting for others. There are several available options for treatment of spasticity and one effective temporary treatment is botulinum toxin injections in the affected muscles. Children with neurological disorders, specifically cerebral palsy, tend to have spasticity that requires treatment with botulinum toxin, and generally requires multiple and repeat injections [1].
Undergoing these injections can be a traumatic experience for the child, causing significant distress on the patient and caregivers. Options to address the distress range from providing distraction techniques to sedation in the operating room. Several studies looked at providing in-clinic intravenous medication for anxiolysis; the agents included intravenous forms of ketamine, propofol, fentanyl, and midazolam [2, 3]. Many of these studies are limited to the context of gastroenterology procedures or premedication to general anesthesia, and they are administered through oral, rectal, intranasal, or intravenous routes. Few studies look at the administration of the medication as an anxiolysis for intramuscular botulinum toxin injections. Zier et al., looked at inhaled nitrous oxide compared to enteral midazolam as anxiolysis agents for botulinum toxin injections, and found inhaled nitrous oxide to provide a quicker onset sedation as well as some analgesia when compared to enteral midazolam [4]. Additionally, they had a higher incidence of nausea and emesis in the nitrous oxide group compared to the enteral midazolam, and while no documented aspiration pneumonias resulted, it is still a potential risk. Enteral midazolam provided a longer sedation period but had a slower onset, and did not provide an analgesic effect per their study [4].
In the author’s literature review, we found limited studies looking at intramuscular administration of intramuscular midazolam; specifically, they were limited to intranasal, oral, rectal or intravenous administration. Anisha et al., compared the use of intranasal midazolam vs. placebo for sedation during imaging. They found intranasal midazolam at a dose of 0.5 mg/kg to be safe, reduced the need for additional IV medications, and eased separation from the parents [5]. A Cochrane review looking at effective anxiolytic and retrograde amnesia agents (including various administration routes of midazolam compared to placebo), revealed inconclusive results with low quality of evidence [6]. In this retrospective case series, we look at the effectiveness and safety of using intramuscular midazolam administration as anxiolysis for botulinum toxin and phenol injections in children. Additionally, we evaluate comorbidities and their influence on duration of sedation with midazolam administration. We hope to add to the current literature of providing alternatives for anxiolysis treatment other than general anesthesia or IV sedation requiring an intensive care unit setting.
Methods
This retrospective study was approved by the Institutional Review Board from Tufts Medical Center. We performed a retrospective chart review of 72 patients from a Tertiary Pediatric Hospital Outpatient Clinic. Seventy-one of the seventy-two charts were analyzed as one record had insufficient data. A total of 120 injections were administered over the course of two years with 31 patients undergoing more than one injection procedure at separate time points (Fig. 1). Out of the 71 patients, 61% were male and 39% were female with a median age of 12 years (Fig. 2).
Chart review process.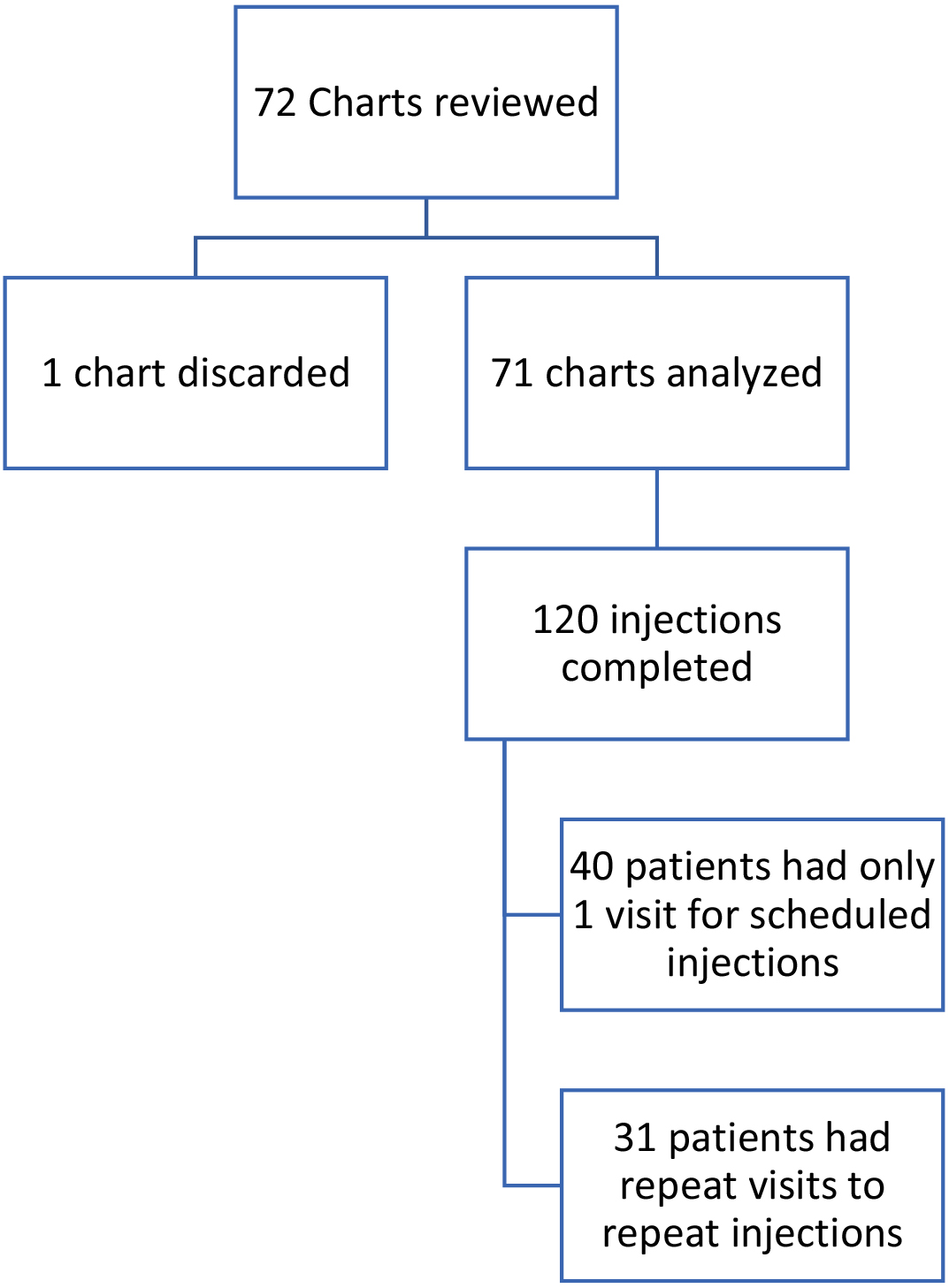
Demographics.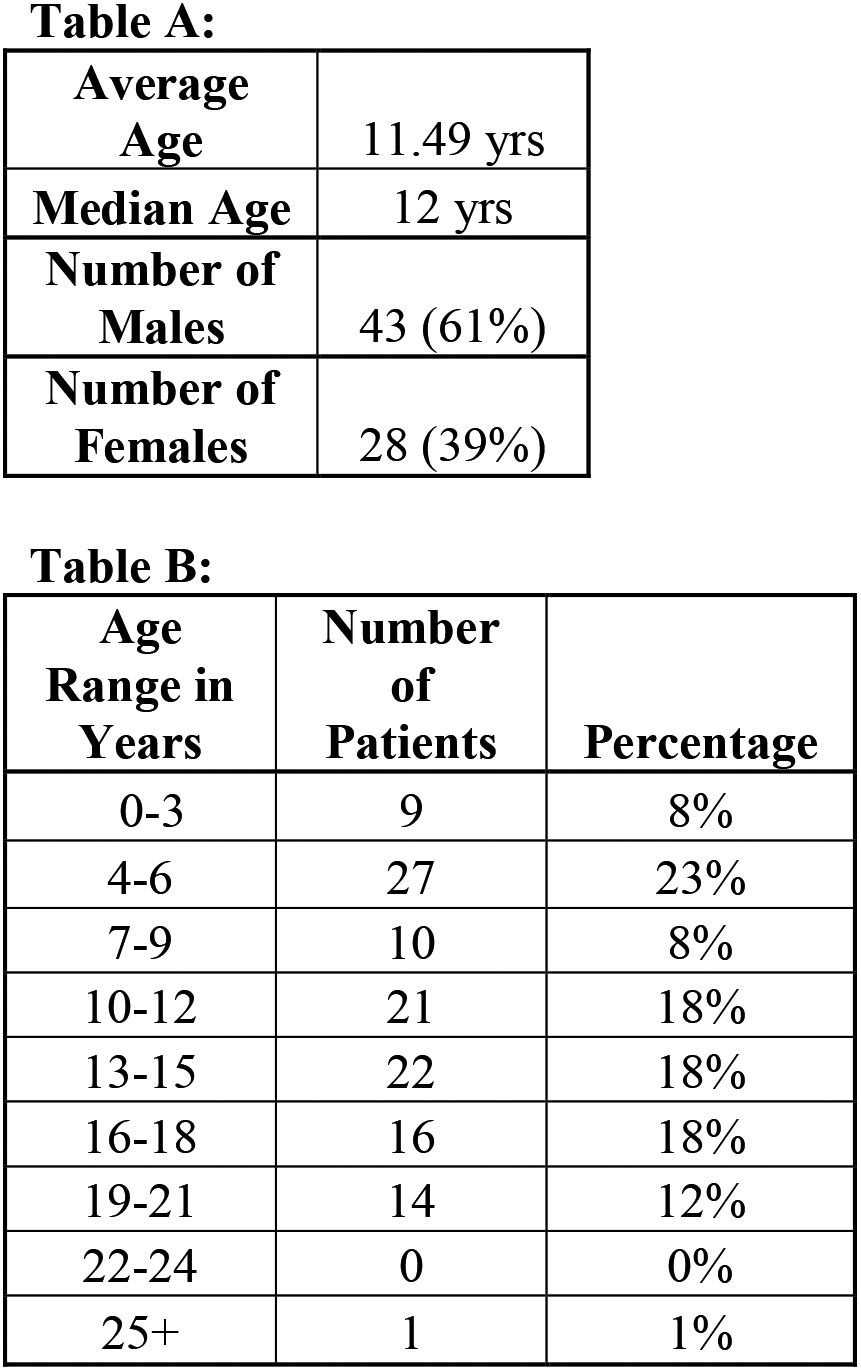
Diagnosis and co-morbidities.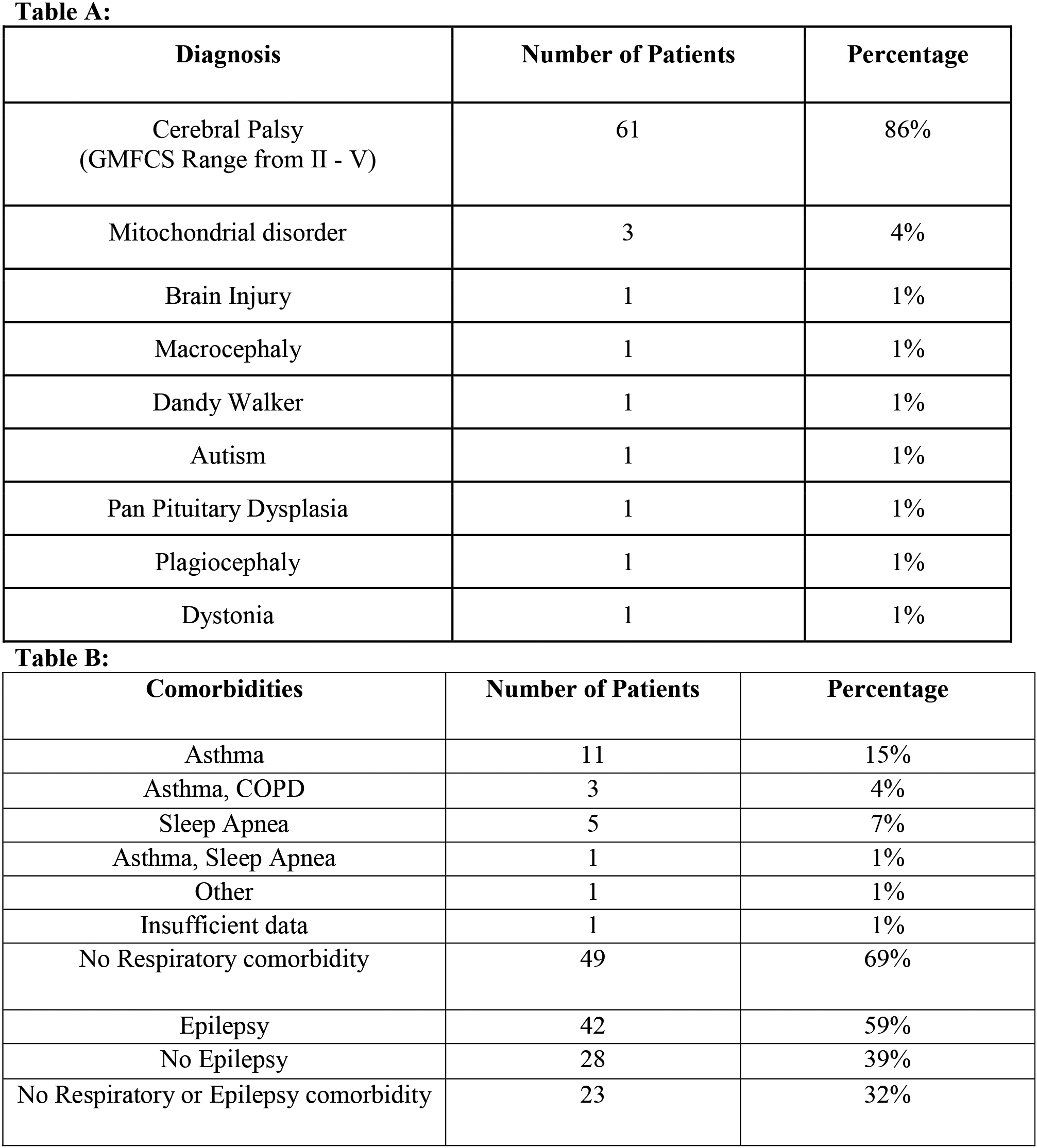
The children had known neurological diagnoses, predominately spastic cerebral palsy (86%) with Gross Motor Function Classification System (GMFCS) range of II–V. Other diagnoses included mitochondrial disorders (4%), traumatic brain injury (1%), and autism (1%; Fig. 3, Table A). In addition to their neurological diagnoses, 30% of the patients had a respiratory diagnosis (asthma, chronic obstructive pulmonary disease, sleep apnea) and 59% had a known seizure disorder (Fig. 3, Table B). Other comorbidities of dysphagia, dysarthria, cardiac, endocrine disorders, or skeletal abnormalities were not recorded.
The injections were performed by a single practitioner who made the clinical decision whether the patient was a candidate for an in-office injection versus general anesthesia. By family request or if the patient had unsuccessful sedation with a prior midazolam dose, the patient was given the injections under general anesthesia. The data collected represents only the patients who received midazolam sedation in the clinic, and not under general anesthesia. Topical lidocaine-prilocaine cream was applied to the injection sites for analgesia, and an intramuscular dose of midazolam based off of the patient’s weight was administered by a certified pediatric nurse practitioner. The dose of midazolam was given with a goal to provide minimal to moderate sedation, and given only once prior to the procedure. Repeat administration of midazolam during the procedure did not occur.
Pre-sedation protocol followed American Academy of Pediatrics Sedation Guidelines, under the guidance of a board certified pediatric physiatrist and board certified pediatric nurse practitioner. The protocol included equipment checks, full history and physical exam (especially cardiorespiratory and airway), consent, fasting after midnight the night before, and a full set of vitals including weight on the day of procedure. During the procedure, physiological monitoring was done with a continuous pulse oximeter and noninvasive intermittent blood pressure monitoring.
Frequency of injections and average sedation and midazolam dose.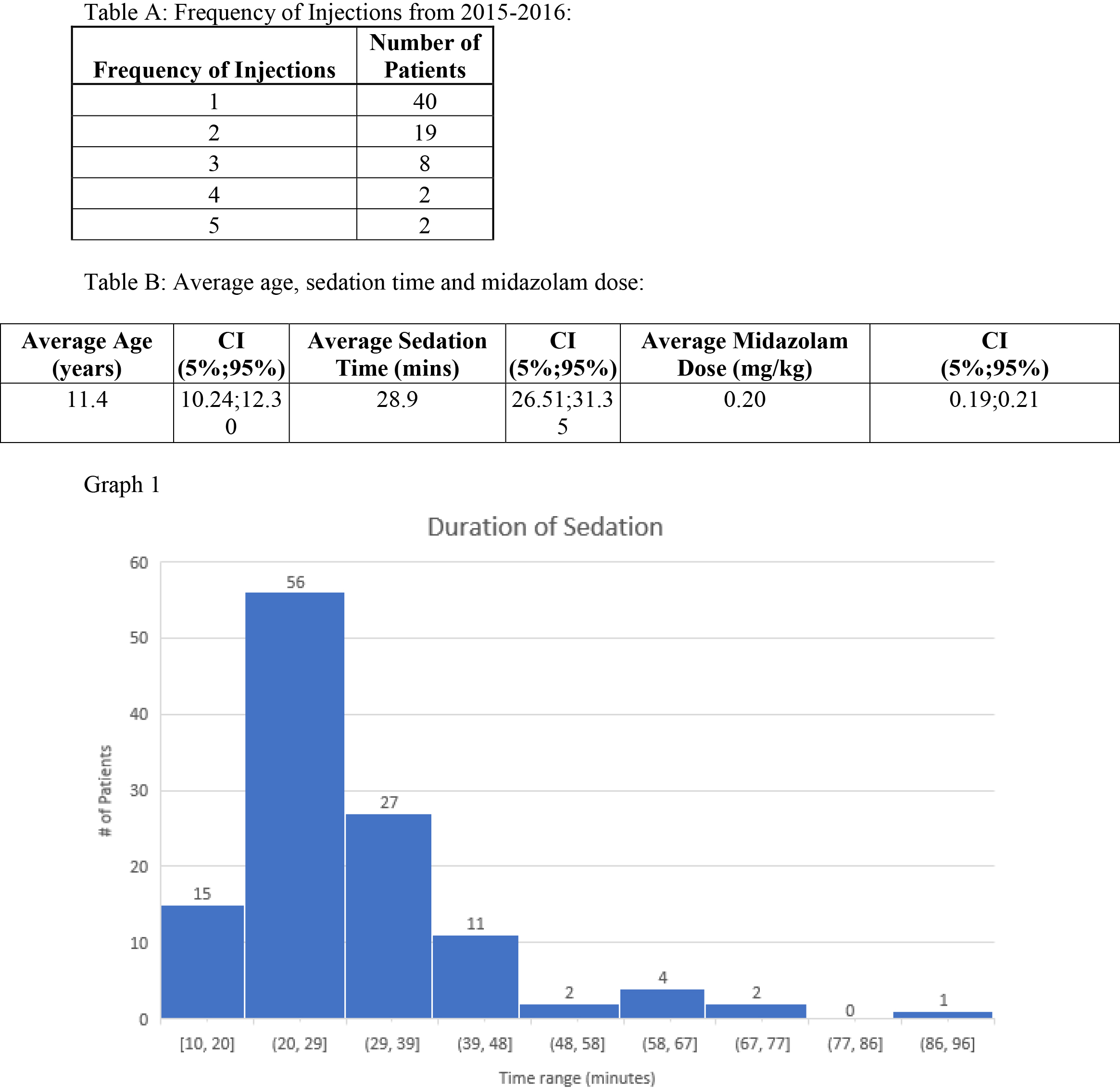
Frequency of side effects.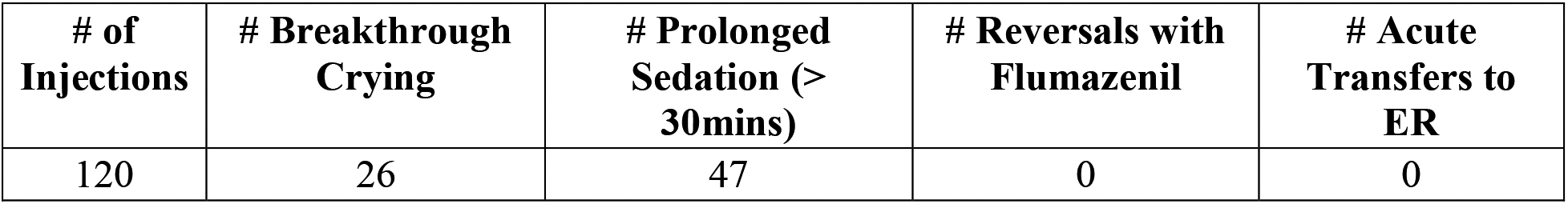
Duration of sedation vs prior respiratory disorder vs prior seizure disorder vs midazolam dose.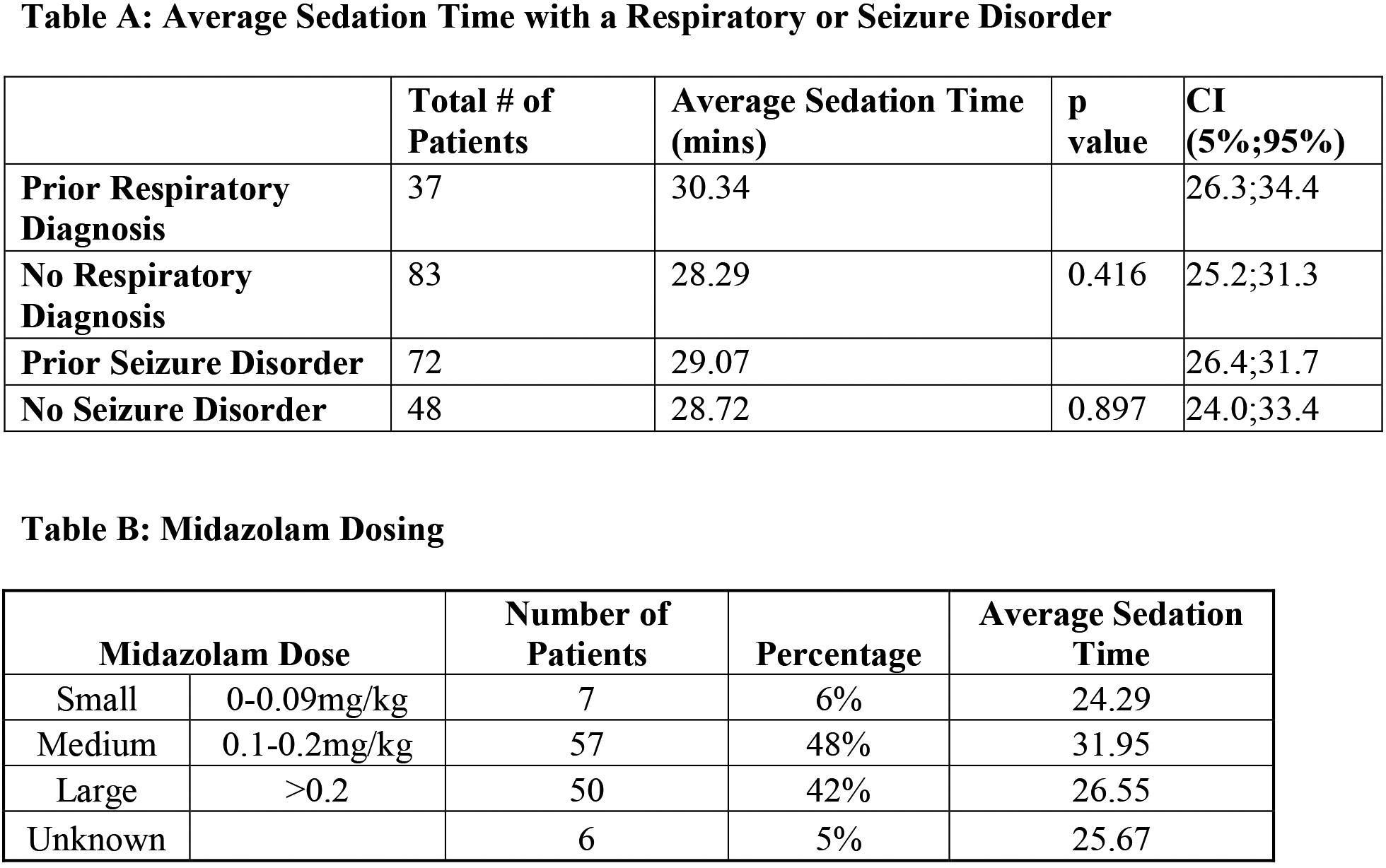
We measured the safety of midazolam administration based on the post-recovery anesthesia scale REACT (Respiration, Energy, Alertness, Circulation, Temperature), scaled from 1–10. We looked at the frequency of negative side effects including prolonged sedation, breakthrough crying, and respiratory compromise. Sedation time was defined as the length of time from intramuscular administration of midazolam to the patient achieving a REACT score of ten. The REACT score was evaluated every 15 minutes during and after the procedure. Prolonged sedation was defined as the patient taking more than 30 minutes to achieve a REACT score of ten. Breakthrough crying was defined as any signs of crying or irritability during the procedure witnessed by the physician or nurse practitioner. Respiratory compromise was evaluated by continuous pulse oximetry with saturations
As a secondary measure of our chart review, we used Microsoft Excel software to perform a student’s
Out of the one hundred twenty injections performed with 31 patients receiving more than one injection in the two-year period, the average time of sedation was 29 minutes (95% CI 26.51–31.35) with an average dose of midazolam of 0.20 mg/kg (95% CI 0.9–0.21). None of the subjects required reversal of midazolam or a transfer to the emergency room. Additionally, we found 39% of the patients with prolonged sedation (
Looking at the duration of sedation in patients with a prior respiratory diagnosis (
Duration of sedation can also be influenced by a prior seizure diagnosis, making a patient more susceptible to sedation with a benzodiazepine. We found an average of 29.07 minutes (95% CI 26.4–31.7) of sedation in patients with a prior seizure disorder compared to 28.72 minutes (24.0–33.4) for patients without a documented disorder. Again, statistical analysis did not identify this as significant (
Lastly, it is expected that a higher dose of midazolam would lead to a longer sedation period.
The recommended dose of IM midazolam for procedural sedation for patients 6 months and older is 0.1–0.15 mg/kg, and can be safely administered up to 0.5 mg/kg with a max dose of 10 mg. No patient received the max dose of 0.5 mg/kg.
In looking at the 120 injections performed, the average dose of midazolam administered was 0.2 mg/kg, 48% of the patients received a dose between 0.1–0.2 mg/kg (medium dose) and 42% received a dose greater than 0.2 mg/kg (large dose). The patients receiving a medium dose had the longest recorded sedation time (95% CI 0.19–0.21) compared to patients receiving small (
Discussion
The intention of this retrospective chart review was to evaluate whether administering intramuscular midazolam is a feasible practice for in-clinic procedures to reduce the anxiety related to the injection. We looked at chemodenervation procedures specifically, and defined feasible by the duration of sedation, the number of patients with signs of discomfort (breakthrough crying), the frequency of requiring chemical reversal of sedation, and the number of patients requiring emergency evaluation.
We found no statistical relationship between a prior respiratory diagnosis (Obstructive Sleep Apnea or Asthma) and the duration of sedation, or a statistical difference with a prior seizure diagnosis and duration of sedation. Having either diagnosis did not interfere clinically with the administration of midazolam or the injection itself. Furthermore, out of the 120 injections performed with intramuscular midazolam for the procedure, none required reversals with flumazenil or transfer to the emergency department for stabilization. In other studies, looking at pediatric sedation prior to procedures, it is not uncommon to have breakthrough crying after administration in sedation. Sajedhi and Habibi found breakthrough crying varying from tearful and on the verge of crying to crying and fearful prior to administration of midazolam and even afterwards in similar numbers. They compared other sedation agents in reducing anxiety prior to general anesthesia and found ketamine to provide less breakthrough crying compared to midazolam, and the combo of ketamine and midazolam as the most effective [2].
Given these findings, we found intramuscular administration of midazolam for in office procedures of spasticity management injections effective to provide adequate sedation without adverse effects.
We are interested in midazolam more than the other agents such as ketamine or propofol, and specifically in its intramuscular form for a few reasons. One, midazolam may provide an amnesic effect which is important in the pediatric population, as remembrance of a distressing event may limit future treatments. Two, the intramuscular form provides a quicker onset and faster drug clearance than the enteral form [7]. Three, Midazolam’s known safety profile allows it to be safely administered in a less intensive setting. In Zier et al.’s study, they concluded nitrous oxide was a better alternative than midazolam. However, they reported several cases of emesis putting children with known dysphagia at risk for developing aspiration pneumonia. While nitrous oxide is also an available option to provide adequate anxiolysis, our retrospective review also shows intramuscular midazolam as a safe option for the clinic setting.
Some limitations in our study include the small sample size, data collection, and retrospective review design. In the procedure record, there was no differentiation between breakthrough crying versus agitation, and no measure of the severity of the breakthrough crying. If a patient cried briefly for a few minutes or throughout the entire procedure, that difference was not recorded. There were also differences in the frequency of data collection between the staff members. The retrospective review design limited our ability to compare patients who did not receive any sedation and monitor their outcomes.
To help with these concerns, we suggest a future prospective study to be performed with a more standardized protocol of data collection which also includes a cohort that only receives topical lidocaine. Additionally, more qualifying information could be obtained with a follow-up questionnaire given to parents regarding their experience with the in-clinic no sedation versus in-clinic mild-moderate sedation versus general anesthesia in the operating room.
Conclusion
Overall, this retrospective review shows administering intramuscular midazolam is a possible approach to reduce anxiety for the pediatric population receiving injections. However, additional studies are needed to ensure its safety and efficacy.
Footnotes
Acknowledgments
The authors have no acknowledgments.
Conflict of interest
The authors have no conflict of interest to report.
