Abstract
PURPOSE:
Abusive head trauma (AHT) can have debilitating sequelae for children who survive. A retrospective medical record review was used to describe short-term developmental outcomes of children with AHT and identify predictors of poorer outcomes.
METHOD:
Children with AHT who received follow up by the hospital’s rehabilitation department for 12 to 24 months post-injury were included in this review. Data for 85 children were collected on hearing, vision, gross motor, fine motor, speech and language, cognition, play, adaptive functioning, behaviour and personal-social skills.
RESULTS:
Global assessment found 42% of children had a good recovery, 34% had a moderate disability and 24% had a severe disability. For whom there was data, more than half had abnormal cognition, behaviour and personal-social skills, whilst more than a third had abnormal speech and language, neurological signs on last assessment, vision, play skills, and gross and fine motor skills. Factors that predicted poorer prognosis across all developmental domains included paediatric intensive care unit admission, longer length of hospital stay, breathing difficulty and lower Glasgow Coma Scale on presentation.
CONCLUSION:
This study highlights the substantial number of children who have abnormal development in the short-term post-AHT and assists in identifying those who require extensive long-term follow up.
Keywords
Introduction
Abusive head trauma (AHT) in children is defined as physical abuse that results in brain injury [1]. Incidence rates across the world range between 14 and 29.7 infants per 100,000 children under one year of age [2, 3, 4, 5, 6]. AHT is the leading cause of fatal head injury in children under two years of age with mortality rates reported as high as 25% [7, 8].
AHT most commonly occurs in infants younger than two years old, with a mean age of 6.2 months [9, 10]. This is a time period where children are more susceptible to shaking or impact injuries for anatomical reasons [8], as well as being a critical period of brain development [11]. As a result, for children who survive, AHT can often be a debilitating condition resulting in developmental disability, sensory deficits, seizures, motor impairments, feeding difficulties and later behavioural and educational dysfunction [8]. Multiple studies have identified the global impairment of children with AHT using the King’s Outcome Scale for Childhood Head Injury (KOSCHI) or the Glasgow Outcome Scale. A meta-analysis of 18 retrospective and prospective studies examining global outcomes of children with AHT after a mean follow up period of 39 months (total sample size of 837; mean age of injury
Key areas of child development include fine and gross motor, speech and language, cognition, personal-social skills, adaptive functioning skills, play and behaviour. Studies reporting specific developmental outcomes following AHT are scarce. Those that exist often have variable outcome measures, inconsistent or variable follow up, or examine a subset of the AHT population such as children who have been admitted to ICU and tend to have small sample sizes, often less than 25 children. These studies have found that 18–65% of children with AHT are blind or vision impaired [2, 13, 14, 15, 16], 37–95% have speech and language difficulties [13, 17, 18, 19], 77% have delayed cognitive development [2], 38–53% have behavioural abnormalities [12, 13], and 38% have motor impairment [12]. No studies have examined the impact of AHT on play or adaptive functioning. Further investigation of these outcomes is required in larger sample sizes that are more representative of the breadth of AHT injuries to describe the types of developmental delays and quantify the extent of those delays.
A further source of inquiry relates to the factors that are associated with poorer outcomes following AHT. Factors associated with poorer global outcomes include pre-injury demographics (e.g. low socioeconomic status [SES], quality of child care, pre-existing medical issues) [12, 20, 21], clinical factors indicative of greater severity of brain injury (e.g., respiratory distress or low Glasgow Coma Scale [GCS] on presentation, need for neurosurgical intervention, paediatric intensive care unit [PICU] admission, neuroimaging findings) [22, 23, 24], delay in accessing treatment [20, 25] and post-injury factors relating to environment and ongoing medical complications [8, 22]. The factors influencing specific developmental outcomes rather than global measures have not been investigated.
This retrospective study describes the developmental outcomes of children with AHT one to two years post-injury and identifies factors predictive of poorer outcomes. This follow up period was selected because the most accelerated recovery post brain injury occurs in the first six months post injury, with patients displaying persisting impairments from 18 months onwards [26, 27]. We hypothesise that factors indicative of severe brain injury will be associated with poorer developmental outcomes.
Methods
Sample
The study included children who presented with AHT to a large tertiary, level 1 trauma children’s hospital in Sydney, New South Wales (NSW), Australia (The Children’s Hospital at Westmead [CHW]) prior to their fourth birthday between 2000 and 2015. S.M, a physician from the Child Protection Unit (CPU), M.-C.W and J.H reviewed the clinical presentation of all children as outlined in the medical records, and identified those with a diagnosis of AHT based on criteria from the Committee on Child Abuse and Neglect [28] as articulated by Lind et al. [13]. Children who were referred to CHW following initial treatment at other hospitals were also included. Children who presented to CHW with AHT were assessed acutely by the rehabilitation department and follow up was always attempted regardless of injury severity on presentation due to the possibility of a long “sign-free interval”. All children included in this study received follow up by the hospital’s rehabilitation department and their outcomes between one and two years post-injury were examined. To prevent the presence of pre-existing conditions from confounding the data, several children were excluded from the study. These pre-existing conditions included traumatic and non-traumatic brain injury, significant developmental delays, or neurological disorder. Furthermore, any child who sustained an additional brain injury during the study period, that was not AHT, was also excluded.
Thirty-two children were lost to follow up between one and two years post-injury (Fig. 1). Of these, data were accessible for 24 children. Demographics and initial injury characteristics for these children are outlined in Table 1.
Child characteristics on presentation to CHW
Child characteristics on presentation to CHW
Notes.
Children included in the study.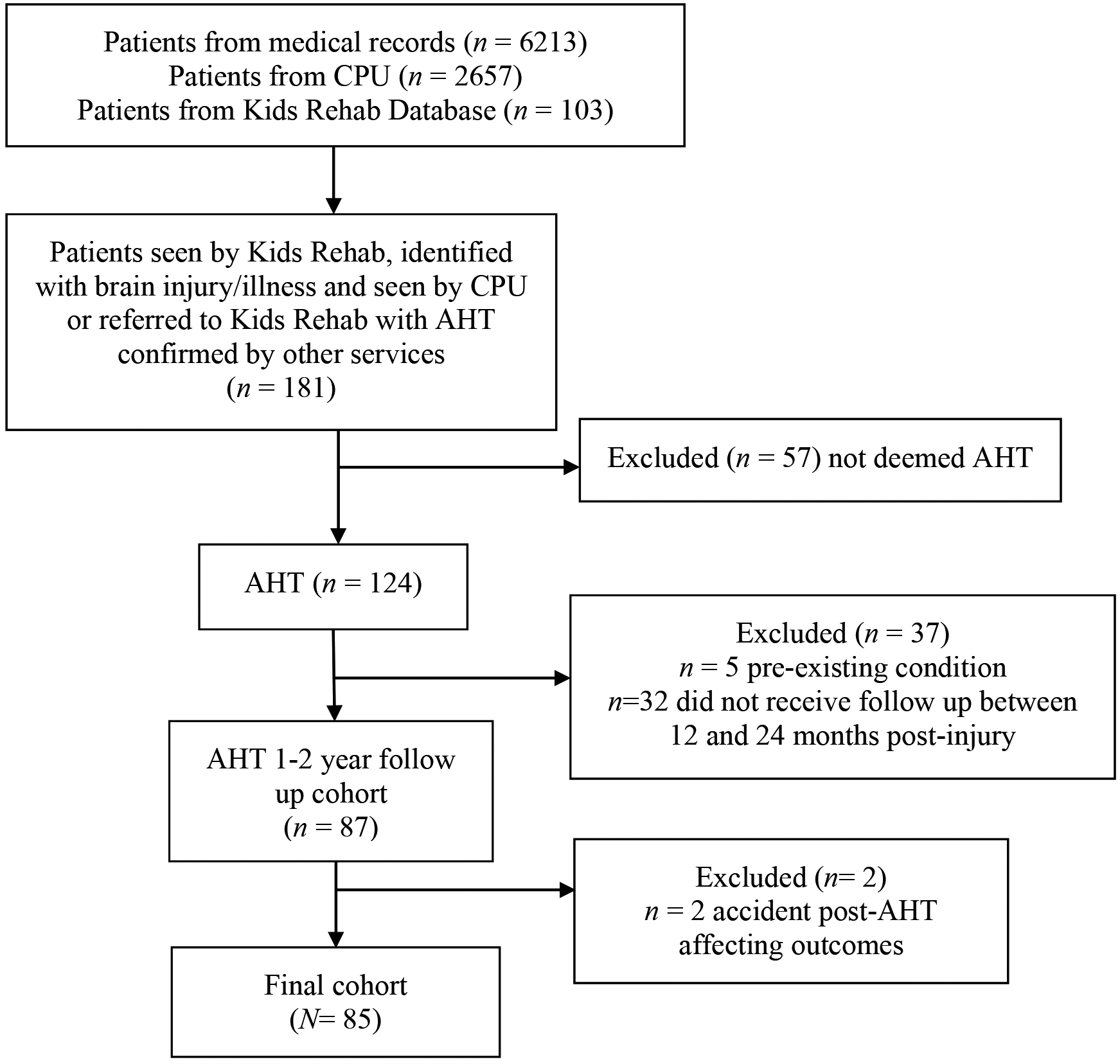
Ethics approval was granted by the Sydney Children’s Hospitals Network Human Research Ethics Committee (LNR/16/SCHN/236) and The University of Notre Dame Australia Human Research Ethics Committee (017064S).
A retrospective review of the medical records of identified children was undertaken. Data was sourced from clinic visits through Kids Rehab, a multidisciplinary team including paediatricians who specialise in rehabilitation and senior paediatric therapists (occupational therapists, physiotherapists, speech pathologists and psychologists). Data extracted included details regarding: demographics (age, gender, presenting hospital, gestational age, postcode of residence at the time of injury to inform index of relative socioeconomic advantage and disadvantage [ISRAD] as a marker of SES, indigenous status, discharge location); clinical presentation and injury details (mechanism [determined by S.M and M.-C.W based on neuroradiology and injury details], neuroimaging [from radiology reports], retinal haemorrhages, neurological signs of tone, power and reflexes, GCS, cardiopulmonary resuscitation [CPR], admission to PICU, neurosurgical intervention, length of hospital stay); and developmental outcomes at follow up (fine and gross motor skills, speech and language, cognition, adaptive functioning skills, behaviour, play and personal-social skills). Data relating to outcomes as close as available to two years post-injury was extracted by S.B.
The extracted information was used to assign a global outcome score using the KOSCHI, with scoring undertaken by S.B. The KOSCHI is a five-point scale used to assess level of disability following traumatic brain injury in children [29]. “Good recovery” (5a or 5b) is defined as when a child has made a complete recovery with no detectable sequelae or has a disability that does not interfere with the child’s function. “Moderate disability” (4a or 4b) refers to a child who is mostly independent but needs supervision or help for physical or behavioural problems, or has residual problems with learning, behaviour or neurological sequelae affecting function. “Severe disability” (3a or 3b) refers to the child being able to move part of their body or eyes to command, make purposeful spontaneous movements, or has a high level of dependency but is able to assist in daily activities. “Minimally responsive” and “death” are self-explanatory categories, with no participants fitting these categories in this study. The KOSCHI has satisfactory inter-rater reliability between clinicians with different experience [30].
Data analysis
Data for gross and fine motor skills, along with speech and language, were extracted from qualitative clinical documentation written by experts in screening for developmental delays including paediatric rehabilitation specialists and senior paediatric therapists. Where available, this included standardised assessments. The information gathered depicted the milestones the child achieved, and was assessed as normal or abnormal for their age based on a framework founded on the best available level of evidence for development [31]. If abnormal, meaning the child was not meeting age appropriate developmental milestones, the data collected was then used to appraise each child’s level of development in months in gross and fine motor skills, and speech and language. Data was coded by S.B, with 15% of the data cross-coded independently by an experienced paediatric rehabilitation physiotherapist (J.H). Inter-rater reliability was 83% as calculated by Cohen’s kappa statistic.
The level of development in months for each child was subtracted from their actual age to give a percentage of function based on age ((age
For example, a child aged 18 months who was standing but not yet walking was assessed as having a delay of 4 months for gross motor function. Their percentage of function based on age was calculated as
Children were grouped as having a severe to very severe delay (0–50% function based on age), mild to moderate delay (51–99% function based on age) and normal (100% function based on age).
Outcomes relating to vision and hearing were assessed as normal or abnormal based on formal vision and hearing assessments by trained optometrists and audiologists at CHW. Adaptive functioning, behaviour (e.g. agitation, aggression, temper tantrums, self-injurious behaviour, impulsivity, and inattention), and play were assessed as normal or abnormal for each child based on clinical judgement by their physician or therapist at the time of the child’s outpatient visit. Cognition and personal-social skills were assessed as normal or abnormal based on standardised developmental assessments only (e.g. Gessell Developmental Schedules-Revised, Griffiths Mental Development Scales and Bayley Scales of Infant and Toddler Development). Cognition was evaluated through a combination of sensory-perception, knowledge, memory, problem solving and early language [32]. Personal-social skills were assessed on how a child understands themselves and engages with others. When insufficient information was available in medical records, the data was considered missing. Data were coded by S.B.
Descriptive statistics (median, range) were calculated for outcomes measured on a continuous basis (e.g. SES) and the percentage of children falling into each category was calculated for outcomes assessed categorically. Only descriptive statistics were performed on the outcomes for cognition and personal-social skills as there were less than ten participants with data for these outcomes. Univariable binomial logistical regression was performed to identify predictors of all other outcomes. Predictors of global outcomes using the KOSCHI score were examined comparing those with severe disability (3a and 3b) with those with moderate disability and good outcome (4a, 4b, 5a and 5b). For gross motor, fine motor and speech and language outcomes, comparisons were made between those with mild-moderate and severe delays. Odds ratios with 95% confidence intervals were calculated. Analyses were conducted using SPSS v25 software [33]. The level for statistical significance was set at
Results
A total of 124 children who had AHT received follow up by the rehabilitation department within the study period. Thirty-nine [39] were excluded from current analyses, with a final cohort of 85 children with AHT (see Fig. 1). As such, we had data on 75% of children who presented to CHW during the study time period who were not excluded for other reasons. Demographic details of both participants included in the study and a subsection of those lost to follow up are presented in Table 1. There was no statistical difference between the two groups.
As data collection relied on the information found in clinical notes, it was missing for many patients. Table 2 highlights the missing data by providing sample sizes for each outcome. Gross motor, fine motor and speech and language were the main focus of this study as they are key milestones in early years of development. Assessment of missing data for additional outcomes (adaptive skills, play, behaviour, cognition, personal-social skills) based on the KOSCHI was performed to investigate correlation with injury severity. Of the 36 children with a good recovery (KOSCHI 5a and 5b), seven (19%) children did not have any data for any additional outcomes. Similarly, of the 29 children with moderate disability (KOSCHI 4a and 5b) six (20%) children had missing data. Notably, this increases to eight out of the 20 children with a severe disability (KOSCHI 3a and 3b).
Outcomes of children with AHT
Outcomes of children with AHT
Notes.
The following results describe the findings for those included in the study only. Mechanism of injury was able to be determined in 50 cases, acceleration/deceleration injury being the most common (64%), followed by combination of acceleration/deceleration and impact (31%) and impact only (5%). The average length of follow up for included participants was 20 months (range 12–24 months). The median age at injury was 4.5 months, with a male to female ratio of 3:2. The majority of children (79%) first presented to their general practitioner or local hospital before being referred to CHW, with the remainder first presenting to a hospital in the SCHN. The median ISRAD was 38.5 compared to 50 in NSW, indicating the majority of children were from a low socioeconomic background. The majority were discharged into out of home care. There was a higher rate of children who identify as Aboriginal, 11.7% in our study compared with 3.3% of the general population [34] and higher rates of prematurity 35% in our study compared with 10% of the population [35].
Of the 63 children for whom chronicity was reported on neuroimaging, it was revealed that 17 children (27%) had only an acute brain injury (injury less than three days old) on presentation to hospital, with the remainder acquiring their injury greater than three days prior or sustaining multiple injuries over a period of time. Almost all children (93%) had a subdural haemorrhage with the majority having additional findings indicative of brain injury on neuroimaging. The majority of children also had retinal haemorrhages (64%) and extracranial injuries (85%). On admission, 18 (22%) children required CPR, and 41 (51%) children were admitted to PICU with the majority of these children requiring intubation. The median length of hospital stay was 17 days, however, there was a wide range from 1 to 166 days, with three outliers of 121, 127 and 166 days due to a combination of injury severity and social issues.
Outcomes
Global outcomes
As categorised by the KOSCHI, 36 children (42%) had a good recovery (5a and 5b), 29 (34%) had a moderate disability (4a and 4b), and 20 (24%) had a severe disability (3a and 3b). No children died or were in a minimally responsive state during the study period.
Univariable logistic regression revealed that those with a severe disability as per the KOSCHI compared to those with good outcome or moderate disability were more likely to have clinical signs indicative of a more severe brain injury. This included significantly greater likelihood of: admission to PICU (OR
Developmental outcomes for gross motor, fine motor and speech and language as grouped by severity (% function based on age).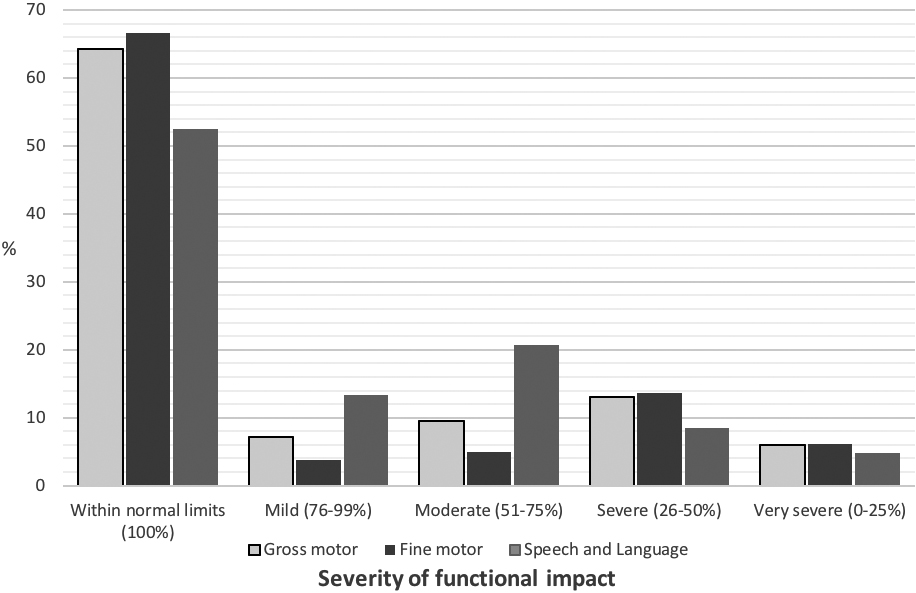
Predictors of global outcomes
Notes.
Of those with details pertaining to gross motor function (
Univariable logistic regression comparing children functioning between 51 and 99% expected for their age with children functioning at a level between 0 and 50% expected for their age was conducted separately for the three developmental outcome areas. Predictors of poorer function for each of the three outcomes were abnormal neurological signs on presentation, breathing difficulty, PICU admission, longer length of hospital stay and lowest recorded GCS (Table 4). Significant predictors specific to each developmental outcome including odds ratios are shown in Table 4.
Predictors of individual developmental outcomes
Predictors of individual developmental outcomes
Notes.
Of those with details about hearing outcomes (
Vision
Thirty-one children (38%;
Cognition, play, adaptive functioning, behaviour and personal-social skills
Outcomes related to cognition, play, adaptive functioning, behaviour and personal-social skills are presented in Table 2. Cognition and personal-social skills had the smallest sample sizes as only standardised assessments were reviewed and due to the age of children, formal neuropsychology assessment had not been performed. Three of 6 children (50%) had abnormal cognition for their age on standardised assessment, whilst 5 children (50%) out of a total 10 who were assessed had impaired personal-social skills. Due to the small number of children with assessments of cognition or personal-social skills, logistic regression analyses were not conducted.
Of the 34 children with details of play skills, 13 (38%) had comments indicative of abnormal play skills relative to their developmental age. Abnormal adaptive functioning was reported for 7 (18%) of the 39 children. Behavioural difficulties were reported in 25 of 42 children (59%). Univariable logistical regression revealed PICU admission, length of hospital stay and lowest recorded GCS to be indicative of poorer outcomes for play, adaptive functioning and behaviour (Table 4). Significant predictors specific to each developmental outcome including odds ratios are shown in Table 4.
Neurological signs at follow up
Of the 74 children with details regarding neurological findings, 30 (43%) had abnormal neurological signs on last assessment (Table 2), including 17 with hypertonia, 3 with hypotonia, 10 with hemiparesis, 7 with hemiplegia, 4 with abnormal Babinski reflex, and 2 with clonus. Univariable logistic regression revealed those with abnormal neurological signs on follow up were more likely to have a PICU admission (OR
Discussion
This study describes the impact of AHT on global and individual developmental outcomes and identifies predictors of poorer outcomes. It is the largest known study of individual outcomes of children with AHT with a consistent follow up of between one and two years post-injury. Fifty-eight percent of children with AHT in this study had a moderate to severe disability based on their KOSCHI score, although 62% had at least one abnormal developmental milestone. This demonstrates the importance of looking not only at global outcomes, but developmental milestones individually to gain a greater understanding of the function of a child with AHT. The most prevalent impairments at two years post-AHT were in more complex skills such as behaviour, and speech and language. Cognition and personal-social skills also appear to have significant impairments despite the low sample size. Factors that predicted poorer developmental outcomes were clinical factors indicating a more severe brain injury.
Missing data for the secondary outcomes was prevalent in this study and it challenges our ability to make comments on these outcomes. Assessment of this data indicates that it was more likely to be missing if a child had a more severe disability. This is likely due to it not being possible to screen for these developmental milestones in such severely disabled children. However, these outcomes supplement our results regarding the key developmental milestones of gross motor, fine motor and speech and language, and highlight the need for formal timely assessment and rehabilitation. Data was not able to be collected on maternal education and its impact on follow up and outcomes, however this would be beneficial in future studies.
In the current sample, 58% of children with AHT were assessed as having a moderate or severe disability as per the KOSCHI. This is similar to the meta-analysis findings of Minns et al. [12], although they found an additional 5% to be minimally responsive. However, Lind et al. [13] found 81% of children to have a moderate to severe disability and 4% to be minimally responsive. Differences in study inclusion criteria are likely to explain this difference, with our study including any child with AHT who received rehabilitation follow up, whereas Lind et al. [13] only included those admitted to PICU, which arguably would represent a sample with more severe brain injuries. Although frequently used, global outcome measures lack the ability to identify specific developmental impairments. For example, when using the KOSCHI, children assessed as having a good outcome may still have minor headaches, abnormalities on brain scan, require anticonvulsants, or mild neurological asymmetry that does not interfere with the child’s wellbeing or functioning. Thus, global outcome rating scales may not account for more subtle developmental impairments and may underestimate the impact of AHT on a child’s function as it is still possible for a good outcome to include children with permanent impairment and behavioural disorders [36]. Although such impairments may not have functional implications for a young child, the ongoing functional implications may be unknown and hence are informative for the focus of rehabilitation.
This is the first study to compare specific developmental outcomes with global outcomes. Our finding that 62% of children had at least one developmental abnormality highlights the importance of examining more than just global outcomes. The number of children who experience delays in their ability to perform at age expected milestones even at a young age is significant. However, as demands placed on the child become increasingly more complex with age, and additional developmental milestones are expected, this study may underestimate the number of children with long-term impairment. Multiple studies have shown that deficits may not emerge for many years after the initial injury, with motor deficits being the first to be recognised followed by behavioural and cognitive impairments [18, 22, 37]. Literature in accidental brain injury suggests that cognition is often the most severely affected developmental outcome in childhood brain injury [37], but this is often not apparent until around four years of age when standardised norms of intelligence and cognition are available [18].
Previous studies have examined gross and fine motor skills together [12, 13, 14]. These skills, however, seem to present in equivalent levels across children with AHT with 36% and 33% of children having impaired skills respectively. Despite the young age of our sample and small sample sizes in some developmental domains, our data indicates that more complex skills including cognition, behaviour, and speech and language are the most affected. As few children received standardised assessments of cognition, and with the increasing expectations of development in the areas of behaviour and speech and language, examination of the longer-term outcomes for this sample are required to assess the true developmental impact of AHT. Future research into the five and ten-year outcomes of the children in this study is planned.
Our results confirm the negative association of multiple clinical factors on global outcomes as reported in a number of studies (e.g. [20, 22, 23, 24]). Our study expands on the knowledge base by identifying predictors of specific developmental outcomes after AHT. The clinical factors identified as significant predictors of poorer outcomes including PICU admission and lower GCS on presentation are indicative of a more severe initial brain injury. Whilst longer length of hospital stay may also reflect the severity of the injury, we suggest that given the nature of AHT psychosocial issues, such as finding appropriate out of home care, may contribute to length of hospital stay. Further factors, such as breathing difficulties on presentation and need for CPR, may lead to a diffuse hypoxic brain injury, a type of severe global brain injury, which would explain their predictive relationship to individual outcomes.
However, other measures of severity of brain injury were not consistently predictive of developmental outcomes. Although CPR, neurosurgical intervention, cerebral oedema and subarachnoid haemorrhage (SAH) were predictors of poorer prognosis for some specific developmental outcomes, they did not predict global developmental outcomes in the current study. This is contrary to previous studies [8, 20, 22, 23]. Rhine et al. [22] found children with cerebral oedema to be 27 times more likely to have a moderate or poor recovery at follow up compared to those with a good recovery. Keenan et al. [38] found CPR to be the strongest predictor of poor outcome at one year. Differences in findings is unclear but may be related to differences in inclusion criteria and follow up period. Further research of the long-term outcomes of the current cohort may highlight if predictors of outcomes change over time.
Age at injury has been found to be a significant predictor of outcome in children with accidental brain injury [39]. However, it was not a predictor of poorer global outcomes in the current study, which is in line with recent studies [13, 14]. Older age at injury was related to poorer outcomes in vision, play and adaptive functioning. This has been identified previously by Greiner et al. [15] who found, of children who required intubation at admission, older age at injury was associated with better developmental outcomes. Children with AHT sustain their injuries at very similar and young ages limiting the likelihood of finding a significant difference in outcome relating to age. As a result, comparison cannot be made to the accidental brain injury literature.
Only one previous study has investigated predictors of specific developmental outcomes. Duhaime et al. [40] found loss of grey white differentiation to be associated with children who remained blind, non-verbal and unable to mobilise independently at nine years post-injury. Our finding of cerebral oedema being a predictor of both vision impairment and poorer gross motor skills in this cohort is in line with these findings. However, we did not find an association with poorer speech and language skills. Interestingly, the predictors of gross and fine motor are similar to each other, however speech and language differs in that CPR, midline shift and neurosurgery are also predictors of poorer outcomes. This may be due to these factors being indicative of a more global brain injury or increasing severity of the brain injury. Knowing predictors of specific developmental outcomes helps clinicians in predicting prognosis and focusing rehabilitation.
Although not the purpose of this study, examination of the demographics of the sample within the context of the broader Australian population assists in identifying risk factors for AHT. Aboriginal background, prematurity and lower SES were more greatly represented in this study than the general population in Australia which may indicate demographic pre-injury risk factors or outline a bias in diagnosis.
Limitations
A number of limitations are intrinsic in this retrospective analysis of children with AHT and are predominantly secondary to missing data. This population is inherently difficult to follow up due to the nature of abuse, out of home care arrangements, and children presenting to CHW from interstate and overseas. The nature of retrospective medical review also brings with it some difficulties as there is variation in the recording of achievement. This has meant that clinical history and assessment have been primarily used to inform the data, which can be subjective. Subjective data was correlated by senior clinicians with best available level of evidence regarding developmental milestones. Information regarding developmental milestones may also not have been included in the documentation if the child had normal development or if their disability was so severe that it could not be assessed. Domains that were evaluated using standardised assessment only, such as cognition and personal-social skills, may have inherent bias as the reason for conducting standardised assessments is unknown. Prospective studies that specify the data to be collected at each follow up will be informative.
Data analysis introduces potential error in terms of bias and type I error. Multivariate logistical regression was unable to be performed due to small sample sizes secondary to missing data in some cases. Type I error is possible secondary to the large number of statistical tests undertaken for each outcome. This could be avoided by using Bonferroni correction which would set the statistical significance at
Confounders may be present that make it difficult to determine if the developmental impairment is as a result of the child’s brain injury or due to the environment pre- or post-injury. For example, behavioural impairment could be multifactorial from a direct result of a frontal lobe brain injury or secondary to speech and language abnormalities, genetic and environmental factors. Despite these limitations, this study gives insight into the development of children with AHT up to two years post-injury and reinforces predictors of poor outcomes globally and for specific developmental domains.
Conclusion
This retrospective review of 85 children with AHT describes their developmental progress up to two years post-injury. This paper highlights the substantial number of children who present with abnormal development, even at a young age and relatively early post-injury. This provides evidence for the importance of early intervention as well as long term follow up to track children’s development as they grow into their disability. In addition, it not only confirms predictors of global outcomes found in previous studies, but also highlights predictors of poorer specific developmental outcomes. This understanding of early prognostic factors will assist clinicians to identify children with AHT who require closer monitoring and to focus early intervention. Due to the age of the children at follow up in this study and the “sign free interval” it is likely that abnormalities have been underestimated and predictors of long-term outcomes not elucidated. Hence, there is a need for further long-term outcome studies. Further research into outcomes at five and 10 years in the same cohort is being conducted by this group to examine the trajectory of development post-AHT and to understand longer term outcomes and implications.
Footnotes
Acknowledgments
This work was supported by Slater and Gordon, Health Projects and Research Fund through funding a research assistant. They had no role in the study design, collection, analysis or interpretation of data, or in writing the report.
Conflict of interest
The authors declare that they have no financial or other conflicts of interest in relation to this research and its publication.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/PRM-190624.
