Abstract
BACKGROUND:
Hypoxic brain injury results in severe disabilities that require extensive acute inpatient and outpatient rehabilitation to promote maximal functional and cognitive recovery. Brain hypoxemia can result from a multitude of causes, including but not limited to cardiac arrest, drug overdose, and/or shock. While recovery from a hypoxic brain injury alone can be challenging, dealing with concurrent debilitative diagnoses such as Guillain Barré Syndrome (GBS) further complicates the recovery and rehabilitation course.
CASE REPORT:
The current case study highlights the acute inpatient rehabilitation course of a 16 year old male who presented with cerebral hypoxia secondary to strangulation and subsequently developed GBS. Physical examination of the patient upon rehabilitation consult was inconsistent with a purely hypoxic brain injury, including the absence of rectal tone. This prompted further potential spinal cord injury evaluation and work up, with diagnostic testing confirmatory of GBS.
RELEVANCE:
This case is important as, to our knowledge from literature review, the first known documented instance of hypoxic brain injury complicated by GBS. Moreover, it highlights the importance of identifying all potential causes of functional disability, particularly when presented with physical exam findings inconsistent with chief diagnosis, in order to maximize functional recovery and rehabilitative gains during acute inpatient rehabilitation.
Introduction
The sequelae of an anoxic brain injury can result in significant impairment, disability, and handicap that often hinders patients’ recovery [1, 2]. Individuals affected by an anoxic brain injury often require extensive acute inpatient and outpatient rehabilitation to promote and maximize functional and cognitive recovery. By definition, anoxic brain injury is injury to the brain due to lack of oxygen, and while cerebral anoxia is characterized by a brain which has had significant deprivation of oxygen. Common mechanisms resulting in cerebral anoxia include carbon monoxide poisoning, cardiac arrest, drug overdose, shock, and/or in our current case presentation, strangulation. Typically, affected areas include cerebral regions and structures of higher metabolic demand, such as the hippocampus, cerebellum, and basal ganglia, as well as arterial watershed border-zones [3, 4, 5, 6].
While recovering from brain injury alone can be challenging, dealing with concurrent diagnoses such as spinal cord injury complicates the recovery and rehabilitative process. Our current case study outlines the acute inpatient rehabilitation course of a 16-year-old male who initially presented with cerebral anoxia with subsequent development of Guillain-Barré Syndrome (GBS). GBS is an autoimmune related neurological disorder characterized by the body’s immune system mistakenly attacking parts of its peripheral nervous system. The exact mechanism is not known, although it is thought that in some cases the immune attack is triggered when the immune system is fighting an infection or chemicals associated with a bacterial or viral infection that resemble nerve cells, which then subsequently also becomes targets of an immune system attack [7]. Literature review has previously reported of the coinciding diagnosis of GBS in the setting of traumatic brain Injury (closed or open), but to date the development of GBS in setting of an anoxic/hypoxic brain injury has never been documented [8, 9, 10].
Case description
Our patient is a 16-year-old male with no past medical history, who presented to acute care upon being found by family after a suicide attempt by hanging. The patient was noted to have had a Glasgow Coma Scale (GCS) Score of 4 (no eye nor vocal response) with decerebrate posturing. Due to the incompleteness of medical documentation and difficulty in obtaining acute care medical records, details regarding the initial cardiac rhythm and time until return of spontaneous circulation during patient’s initial presentation to the acute care hospital was unknown. However, per patient’s family an approximate three-hour lapse between when the patient was last known well to when he was found by a family member was noted. Attempt at cardiopulmonary resuscitation (CPR) was initiated by the family member who found the patient and continued until approximately 15 minutes later when Emergency Medical Services (EMS) personnel arrived. Furthermore, the patient had a prolonged hospitalization complicated by constant agitation due to autonomic storming.
Available imaging and impressions provided by the acute care hospital came in the form of a Magnetic Resonance Imaging (MRI) of the head and brain without contrast, which showed restricted diffusion in both thalami with lesser extent in the basal ganglia and medial temporal lobes bilaterally, as well as decreased perfusion in the right parietal region correlating with anoxic brain injury. Per limited documentation from acute care, the first 29 days of acute care management was significant for continual sedation and intubation, as well the development of severe paroxysmal autonomic hyperactivity. Medication management administered by acute care included, but was not limited to, the use of benzodiazepines, Haloperidol, Quetiapine, Clonidine, and Propranolol. Furthermore, his acute care stay was also complicated by the development of sepsis secondary to a urinary tract infection. During the latter stages of his acute care hospitalization, the patient began to develop severe spasticity and tonicity along with multiple uncontrolled episodes of paroxysmal autonomic hyperactivity manifesting as fever, tachycardia, and increased muscle rigidity/spasticity.
On post-injury day 29, a request for patient transfer from the acute care hospital to the acute inpatient rehabilitation unit was initiated in order to assist with uncontrolled management of severe spasticity/tonicity. On post-injury day 31, the patient was transferred to the acute rehabilitation unit and was notably sedated and intubated during initial evaluation by the rehabilitation team. Significant findings on initial physical examination included tachycardia with a heart rate of 112, increased tonicity of bilateral upper and lower extremities, and clonus of the left wrist. The patient was slowly initiated on an antispasmodic regimen which incorporated the administration of Baclofen and Dantrolene, as well as Propranolol and Clonidine to assist with management of notable paroxysmal sympathetic hyperactivity. On day 53 post-injury, the patient was noted to be flaccid in bilateral lower extremities, which was a change from baseline. Given concern for a possible new spinal cord injury, MRI of the cervical and thoracic spine was completed and Neurology was consulted. MRI findings were suggestive of GBS vs meningitis for which lumbar puncture was obtained and ganglioside antibody panel was sent out to Mayo Clinic. Electromyography (EMG) was performed with corresponding nerve conduction study (NCS) and electromyogram (EMG) studies to help confirm the clinical diagnosis of GBS. NCS/EMG obtained (seen in Table 1) demonstrated sensorimotor axonal polyneuropathy consistent with differential diagnoses of GBS versus critical illness myopathy. GM1b and GD1a IgG Antibodies (seen in acute motor and sensory axonal neuropathy [AMSAN] variant of GBS) were then found to be positive (titer results unknown) due to test processing as a send out, with the reporting laboratory only providing information regarding positive vs negative findings [11, 12, 13, 14]. Per neurology consult, initially a five day course of Intravenous Immunoglobulin (IVIG) treatment was administered on Day 57 post-injury. However, after completion of the IVIG treatment, the patient demonstrated poor response per Neurology and on Day 78 post-injury, Neurology recommended for the use of plasmapheresis for treatment of AMSAN variant GBS. On Day 92 post-injury, after completion of his second plasmapheresis, the patient began to exhibit improved lower extremities motor function (increased spontaneous movement of bilateral lower extremities as well as some active abduction and adduction) despite continuing to demonstrate abnormal signal enhancement in the spinal nerves and the nerve roots of the cauda equina. The patient subsequently completed the course of five sessions of plasmapheresis on Day 99 post-injury. On day 112 post-injury, the patient was transferred to a brain injury-specific rehabilitation center for continued care and rehabilitation. At follow up, his functional status had improved dramatically from discharge as shown in Appendix 1.
Electrodiagnostic exam results and findings
Electrodiagnostic exam results and findings
Muscles tested: R – Right, L – Left, Stim – Stimulation Site, Dist – Measured Distance, mm – millimeter, mV – millivolts,
Given the mechanism injury, initial concern for his neurological presentation included a potential cervical spinal cord injury superimposed on his anoxic brain injury. In particular, the lack of anal sphincter tone was worrisome for a spinal cord injury. Other differential considerations included critical illness myopathy and metabolic myopathy. However, due to the state of medical instability he initially presented with, neurologic imaging needed for confirmation was not performed until Day 27 post-anoxic injury during his inpatient rehabilitation stay.
Results
Electromyography findings
EMG findings of the upper extremities were notable for low amplitude with normal distal latency and velocity on bilateral median nerve studies without conduction block or temporal dispersion on compound muscle action potentials (CMAP). Sensory nerve action potentials (SNAPs) of bilateral upper extremities were of low amplitude with normal distal latencies and conduction velocities.
In regards to the lower extremities, CMAPs were non-responsive/unobtainable for most tested nerves including right tibial nerve, left peroneal nerve, and left tibial nerve (see Table 1). CMAP of the right peroneal nerve showed very low amplitude with prolonged distal latency, but normal conduction velocity. No conduction block or temporal dispersion was noted. SNAP of the right sural nerve was of normal amplitude, distal latency, and conduction velocity while the left sural nerve was unresponsive.
F-wave latencies of the upper extremities were borderline normal.
EMG findings of both the upper and lower extremities were consistent with sensorimotor polyneuropathy, likely the axonal type. The lower extremities were affected more than the upper extremities, and the left lower extremity worse than the right lower extremity. The differential diagnosis based on these results included axonal type AMSAN variant GBS and critical illness polyneuropathy.
Imaging findings
MRI of the cervical and thoracic spine obtained on Day 56 post-injury were significant for diffuse abnormal enhancement of the spinal nerves in the distal thecal sac, an appearance that can be seen with Guillain-Barré Syndrome, with possibility of underlying meningitis or neoplastic origin along with mild spinal cord atrophy in the lower cervical and upper thoracic cord without cord signal abnormality (Fig. 1).
MRI of the Cervical and Thoracic Spine (Left – T2 Weighted Sagittal Thoracic Spine MRI, Right – T2 Weighted Axial Low Thoracic/Upper Lumbar Spine MRI), demonstrating abnormal signal enhancements of the spine nerves in the distal thecal sac (arrows). Per discussion with neuroradiologist, the abnormal signal changes observed are seen as “fraying” of nerve roots and adhesion-like presentation nerve roots in axial view as opposed to normal clearly circumscribed nerve roots.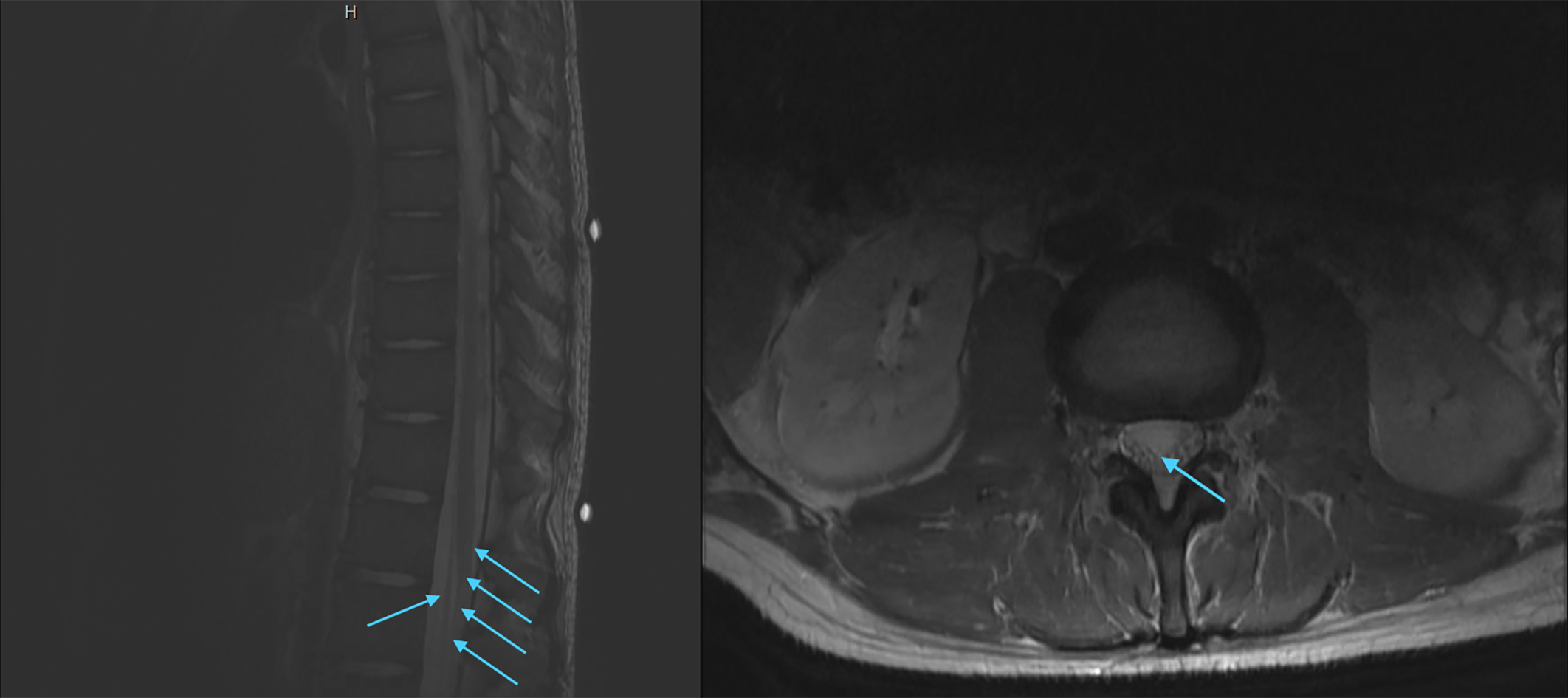
Repeat MRI obtained on Day 91 post-injury redemonstrated enhancement of the anterior and posterior spinal nerves and the nerve roots of the cauda equina similar to the previous examination. No obvious intraspinal fluid collection was identified.
Respiratory culture was significant for pseudomonas aeruginosa, with antibiotic sensitivity significant for Ciprofloxacin.
Microbiology testing was also performed and was negative for key viral antigens. Lumbar puncture specimen was also negative for herpes simplex virus (HSV), varicella zoster virus (VZV), and cytomegalovirus (CMV) without growth from culture.
A ganglioside antibody panel (Asialo-GM1, GM1, GM2, GD1a, GD1b, and GQ1b) was also obtained and was significant for +GM1b and +GD1a antibodies, both of which are known antibodies associated with Guillain-Barré Syndrome [11, 12, 13].
The coinciding radiologic and serologic evidence, along with the EMG findings, strongly suggested the diagnosis of GBS.
Treatment and prognosis
Even after treatment, approximately 3 percent of patients with GBS die from the condition, and 60 percent of pediatric patients have been known to become non-ambulatory during the illness [1, 2, 15, 16]. As noted in current literature, treatment of GBS involves the application of IVIG and plasmapheresis/exchange with variable results [17]. IVIG has generally been more preferable due to lower complication rates and with higher rates of treatment completion and tolerance [18, 19]. In most cases, independent ambulation and shortened hospital stays were documented. Comparison of therapeutic regimens has been limited by differences between treatment groups, but a recent meta-analysis of those trials conducted to date suggests that IVIG is as effective as plasma exchange in accelerating recovery from GBS [19, 20, 21]. Alternatively, plasmapheresis has been well tolerated among the pediatric age group [22]. Furthermore, two independent randomized controlled trials have demonstrated marked benefit regarding the use of plasmapheresis in the treatment of GBS [23, 24]. Similarly, however, two other randomized controlled trials have demonstrated equal efficacy of IVIG when compared to plasmapheresis in treatment of GBS [18, 19]. Regardless, IVIG ad plasmapheresis have both been shown to be efficacious in the treatment of GBS [25]. In the case of our patient, there was minimal motor function improvement after completion of IVIG. However, after the initiation of plasmapheresis, notable motor function recovery was appreciated after two sessions, prompting completion of the five session course of plasmapheresis. Historically, pediatric patients diagnosed with acute inflammatory demyelinating polyneuropathy (AIDP) typically recover more quickly than patients afflicted with acute motor axonal neuropathy (AMAN) or acute motor and sensory axonal neuropathy (AMSAN) variants of GBS [26].
Discussion
This case demonstrates the importance of identifying all attributing pathology for symptoms that a patient presents with, particularly in the setting of a physical examination that does not correlate with a patient’s initial diagnosis. As noted in literature, the functional outcome of individuals afflicted with an anoxic brain injury often result in greater detriments to cognition while reaching mobility goals earlier when compared to individuals who sustained a traumatic brain injury [27]. In our current case study, however, the presenting bilateral lower extremity weakness simply did not correlate with his mechanism of injury and thus prompted further investigation. In doing so, we were able to identify the coinciding diagnosis of GBS which necessitated the timely treatment of the condition before further detriment was imposed on his recovery. More specifically, the predominant motor deficit noted among individuals afflicted with GBS is lower extremity weakness [28].
Had the patient’s GBS been unrecognized and untreated, his overall functional outcome could have taken a drastically different trajectory with potentially fatal consequences [1, 16, 29]. Specifically, without plasmapheresis treatment, his lower extremity functionality would have been significantly hindered and may have blunted his progress to regain functional ambulation during his inpatient rehabilitation stay. This could have not only altered his overall functional goals during inpatient rehabilitation, but could have also drastically changed family education and training for potential needs of the patient on discharge. For instance, family training and education would have been necessary for bed mobility, transfers, bowel and bladder management, as well as community ambulation. However, by recognizing and identifying the discrepancies with his presentation and physical exam findings, we were able to alter this rehabilitation course to maximize his potential for a safe community discharge with appropriate family training.
Footnotes
Conflict of interest
The submitted manuscript has not been published previously and is not under consideration for publication elsewhere. All authors take responsibility for the contents of the manuscript, including review and approval of this version, and satisfy the requirements for authorship. There are no relevant financial disclosures nor conflicts of interest.
Appendix I – Functional Measures following Brain Injury Specific Rehabilitation post-acute rehabilitation discharge
Min A – Minimal Assistance; Mod A – Moderate Assistance; Max A – Maximal Assistance; CGA – Contact Guard Assist; UE – Upper Extremity; LE – Lower Extremity
Physical Therapy
Bed Mobility:
Rolling Left and Right – Min A to Supervision
Supine to Prone – Max A to Supervision
Squat Pivot – Max A to CGA
Gait:
Arjo Walker, 80 feet to 100 feet with 2-Person
Assist
Standing:
5 seconds without Assistance in Arjo Walker
Sitting:
Edge of mat with Stand By Assist
Occupational Therapy
Bathing:
Bathing – Mod A
Dressing, UE – Mod A
Dressing, LE – Max A (due to incoordina-
tion, motor planning, command following
deficits)
Speech Therapy:
Increased vocal production, “when he needs to
sit, when he wants to talk, stand, etc.”
More appropriate with verbal responses, com-
pleting tasks with less time for processing
Increased accuracy with 2 Step Directions
Dietary upgrade to Regular Textures, Cut Up.
Physical Therapy
Strength:
3/5 in Hip Flexion and Knee Extension (Left
Lower Extremity)
2/5 in Hip Flexion and Knee Extension (Right
Lower Extremity)
Mobility:
Sit to Stand – Mod A to Min A
Bed Mobility – Min A
Car Transfer – Min A (3 people)
Occupational Therapy
ADL Max A to Min A
No more agitation noted per Therapy Staff
Speech Therapy:
Follow 1 Step Directions to 90% accuracy, 2
Step Directions to 50% accuracy, increasing to
70% with max visual cues
Talking at short sentence level, can be 100%
intelligible
