Abstract
OBJECTIVE:
To evaluate the safety of combining salivary gland onabotulinumtoxinA (BTX-A) injections for sialorrhea with intramuscular BTX-A injections for spasticity in the same procedure.
DESIGN:
A retrospective cohort study in a tertiary hospital center. Patients selected were younger than 20 years, diagnosed with cerebral palsy, and received their first salivary gland BTX-A injection between March 2011 and July 2015. Chart review and telephone interviews were performed. The primary outcome measure was the rate of adverse events after combined BTX-A injections into salivary glands and muscles. The secondary outcome measure was the efficacy of injections, as reported by patients.
RESULTS:
Twenty-five of the 52 selected patients received salivary gland BTX-A injections only, and 27 received concurrent salivary gland and multi-level intramuscular chemodenervation with BTX-A with or without alcohol. The rate of adverse events was
CONCLUSIONS:
Combining BTX-A injections for sialorrhea with multilevel intramuscular BTX-A injections (with or without alcohol) appears to be safe and effective and allows treatment of patients for both conditions simultaneously.
Introduction
Poorly controlled drooling (sialorrhea) is a common complication associated with diseases of motor development and is estimated to occur in approximately 10–58% of children with cerebral palsy (CP) [1]. It can be caused by motor function difficulties, such as severe dysphagia due to spasticity or weakness of the oropharyngeal and esophageal musculature, as well as oral sensory dysfunction and poor head control [2]. Sialorrhea causes serious medical problems such as choking, aspiration, and pulmonary infections. Additionally, sialorrhea causes skin irritation, leads to social embarrassment, and increases the burden of care, including frequent suctioning, bib/clothing changes, and cleaning of household items. Thus, sialorrhea adversely affects a child’s health, social integration, and quality of life [2, 3].
While sialorrhea can be addressed by a variety of modalities, direct injection of onabotulinumtoxinA (BTX-A) into the salivary glands is known as a safe, minimally invasive, and effective treatment for drooling in children and adults. Injection of BTX-A has become a common tool in the treatment of sialorrhea [3], and it benefits patients by avoiding the systemic adverse effects caused by oral medications, which are reportedly as high as 44% with glycopyrrolate, one of the most commonly used oral medications [4]. This treatment also precludes the need for surgical intervention and can be helpful if surgery fails.
Besides sialorrhea, one of the most common problems in patients with CP and similar motor developmental conditions is spasticity. Spasticity can result in a decreased range of motion and even cause contractures, leading to challenges with ambulation, transfers, positioning, ease of care, skin breakdown, and pain. Intramuscular BTX-A injections are a common therapeutic modality for treating localized spasticity and pain in individuals with CP. Due to diffuse spasticity, multiple intra-muscular injections are often done throughout the body in a single session, termed a single event multi-level chemoneurolysis or SEMLC. Injections can be used alone or in combination with oral anti-spasticity medications or surgical interventions, such as soft tissue lengthening, intrathecal baclofen pump placement, or selective dorsal rhizotomy. Thus, patients with CP who may benefit from BTX-A injections for sialorrhea also frequently benefit from intramuscular BTX-A injections for spasticity. Based on clinical experience, this is common for patients with quadriplegic CP.
To avoid antibody formation, many healthcare pro- viders wait at least 3–4 months [16] between injections, by which time the effects of BTX-A have often unfortunately waned. If a patient cannot receive BTX-A injections into the salivary glands and muscles concurrently, one must be chosen over the other for that 3–4 month cycle, thus denying the patient optimal therapy. An additional benefit of combining injections is that the number of procedures can be minimized, which is especially important when general anesthesia is required, given the risks and adverse events associated with it. Therefore, although it is not a universal practice, administering both injections concurrently may be the best option for patients who require both spasticity and sialorrhea management.
Currently, the information on the safety and efficacy of a combined procedure with BTX-A injections into the salivary glands and muscles is limited. The only available literature is a previous case report describing two patients with severe spastic quadriplegic CP who received BTX-A injections into both salivary glands and multiple intramuscular sites during the same procedure [5]. Both patients showed improvements in drooling and spasticity without any adverse events. With such limited data, there is a need for further research to validate the safety and efficacy of combining BTX-A injections for spasticity and sialorrhea in the same procedure.
Prior to the use of botulinum toxin, alcoholic compounds such as phenol or ethanol were used for chemodenervation in patients with spasticity. While BTX-A effects spasticity by targeting the presynaptic membrane of the neuromuscular junction, alcohol blockade works by denaturing proteins within the motor nerve axon. Treating spasticity with simultaneous intramuscular injections of BTX-A and alcohol is a common clinical technique to maximize the number of muscles covered while minimizing using excessive doses of either medication [15]. Single event multi-level chemoneurolysis, or SEMLC, can now be done with BTX-A alone, with BTX-A with ethanol, or less commonly with ethanol alone.
The objective of this study was to evaluate the safety and efficacy of treating sialorrhea with BTX-A alone or concomitantly with SEMLC (intramuscular injections with BTX-A alone or in conjunction with alcohol injections). SEMLC has been used for individuals with CP to manage both diffuse and multilevel muscle spasticity. However, little data exists on simultaneous use with intraglandular injections.
The primary hypothesis of this study was that the concomitant use of multi-level intramuscular BTX-A for spasticity and intraglandular BTX-A for sialorrhea would not increase their adverse events or decrease their efficacy. Because both sialorrhea and spasticity are often present in patients with CP, the potential significance of this retrospective study is to provide information that will empower clinicians to treat both conditions simultaneously.
Methods
Study design, participants, and data sources
This was a retrospective cohort study, approved by the committee on research ethics at the institution in which the research was conducted in accordance with the Declaration of the World Medical Association, performed in a tertiary-care hospital center. Parental consent was obtained for all subjects, and patient assent was obtained when appropriate.
The independent variable assessed was the inclusion of intramuscular spasticity management during BTX-A injection for sialorrhea. The dependent variables were the occurrence of adverse events and the reported improvements in drooling and spasticity reported by the patients.
The inclusion criteria were all patients with CP seen at the study institution who were younger than 20 years old and had received their first BTX-A injection into salivary glands between March 2011 and July 2015 consecutively. Patients who received SEMLC with either BTX-A alone or with BTX-A and alcohol were included for power. Further analysis was performed to evaluate if there was a difference between these two SEMLC groups (BTX-A only or BTX-A
The start date was the first salivary gland injection at the study institution. The end date was chosen after consultation with the biostatistics department to allow for an adequate interval of at least 2 years (average
There was a total of 57 patients, of which five (9%) were lost to follow-up, leaving 52 patients. Of these, 25 patients received BTX-A injections into salivary glands only, and 27 patients received BTX-A injections into both salivary glands and SEMLC during the same procedure (with or without concomitant alcohol injections).
After consultation with a statistician, the decision was made to limit the analysis to the patient’s first BTX-A injection and not to include repeat injections in order to eliminate variables that were likely to skew the outcome measures. These potential confounders included the number of BTX-A injections received previously, self-selection for subsequent injections, and timing between injections.
Efficacy outcome measures for salivary gland injections and intramuscular injections
Efficacy outcome measures for salivary gland injections and intramuscular injections
Patient characteristics at initial BTX-A injection
Abbreviations: GMFCS, gross motor function classification system; SEMLC, single-event multilevel chemoneurolysis; BTX-A, OnabotulinumtoxinA.
Medical records were reviewed for past medical and surgical histories significantly related to salivation (tracheostomy, gastroesophageal reflux, tube feeds, history of fundoplication, and constipation), medications (glycopyrrolate, a scopolamine patch, benzodiazepines, antihistamines, gastrointestinal medications for gastroesophageal reflux, and anti-seizure medications), the number of incidents and dates of aspiration pneumonia, and other treatments for salivation such as surgery. The records were also reviewed for medications and other treatments for spasticity. The goals of injections, whether the goals were met, and any adverse events observed were reviewed. Additionally, the patients (or parents/legal guardians/caregivers, as appropriate) were called to confirm, clarify, and add details to the information above.
All injections were performed under the guidance of one senior pediatric physiatrist (H.K.) at a tertiary medical center. All intraglandular and intramuscular injections were performed using ultrasound and electrical stimulation guidance. For salivary glands, BTX-A was injected into the parotid and/or submandibular salivary glands at a dose of 1 U/kg, with a maximum of 25 U per gland and a total of 100 U per session. Intramuscular dosing was performed based on clinical history, examination findings, and safety recommendations of the manufacturer. No patient received greater than the weight-based recommended dose of BTX-A to avoid systemic toxicity and adverse events.
The primary outcome measure was the number of adverse events after injections. Adverse events were defined inclusively as any adverse event reported by the patient/caregiver, or identified via chart review. The latter included incidents such as those observed by the physician during the procedure on physical exam, or on post-procedure monitoring of heart rate, blood pressure, and resumption of pre-op cognitive status and feeding. Patients were seen in clinic approximately 4–6 weeks, post-injection for follow up. At that time a physical exam was performed and adverse effects from the BTX-A procedure were obtained via history.
Efficacy scale vs. number of patients for first BTX-A injection for the 3 groups. 0 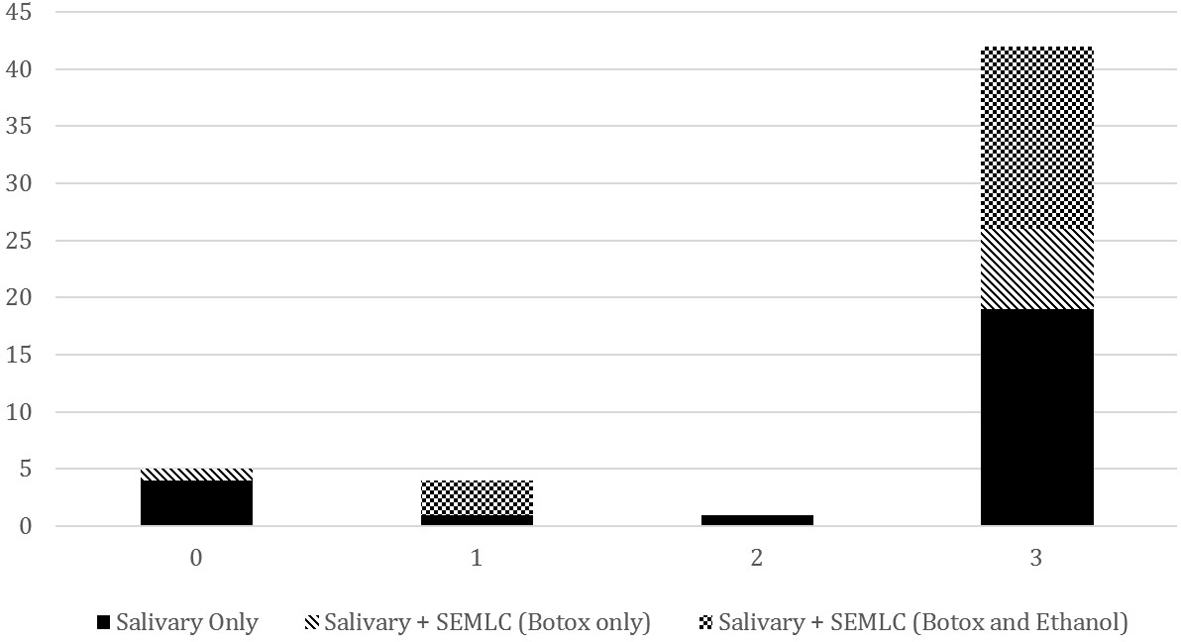
The secondary outcome measures were the efficacy of injections to salivary glands and muscles. Efficacy was defined as at least 50% improvement in a predetermined injection goal (see Table 1), which lasted for at least 2 months. For objective should be goals (i.e., frequency of suctioning or number of bibs used), the patients were asked about goals of injection before the procedure, and the degree of goal accomplishment was discussed on follow up. For subjective goals (e.g., quality of gait, embarrassment from drooling), a percentage was determined by asking the patient on follow up what percentage their symptoms improved from baseline, or determined by the change in Likert scale from 1–10. This information was available, as the line of questioning is standard practice in our clinic. Before each injection, each child and or caregiver identified the goals of injection. Unfortunately, standardized assessment tools were not consistently performed in clinic to better qualify efficacy in this retrospective study.
Statistical analysis was performed using the IBM SPSS Statistics for Windows, Version 24.0. (IBM Corp., Armonk, NY, USA). The Fisher’s exact test was used to compare categorical data. Spearman correlations were performed to examine the relationships between variables. For all analyses, statistical significance was set at
Results
Patients
Of the 52 patients who carried through for this study, 25 received BTX-A injections into their salivary glands only (Group 1, “Salivary Only”) and 27 received BTX-A injections into their salivary glands and muscles during the same procedure (Group 2, “Salivary
Injection information
In patients who received intramuscular injections with only BTX-A in addition to salivary gland injections, an average of eight muscles were injected. In patients who received intramuscular ethanol injections in addition to BTX-A, an average of nine muscles were injected. The three most common muscles injected with BTX-A, in order of decreasing frequency, were the iliopsoas, hamstring, and hip adductor longus. The three most common muscles injected with alcohol, in order of decreasing frequency, were the gastrocnemius, brachialis, and hip adductor longus.
Adverse events of initial BTX-A injection
Adverse events of initial BTX-A injection
Abbreviations: BTX-A, OnabotulinumtoxinA.
In patients who received intramuscular injections with BTX-A only, the average dose was 12 U/kg, with an average total dosage of 283 U per session. In patients who received alcohol and BTX-A, the average dose of BTX-A was 13 U/kg, with an average total dosage of 287 U per session. For alcohol injections, 45% ethanol was used at 0.14 mL/kg, with an average total dosage of 2.8 mL per session. Range of dosages injected into muscles can be seen in Table 4.
One patient in Group 1 (4%) and two in Group 2 (7%) experienced adverse events. All three were related to salivary gland injections. The patient from Group 1 reported difficulty in swallowing after the first injection. Because of the concern for dysphagia associated with the spread of BTX-A, a modified barium swallow examination was performed, but was normal. The symptoms lasted for 2 months, at which point there was complete resolution. The patient did not have aspiration pneumonia or other respiratory illnesses during this time. In Group 2, the only two patients who reported adverse events were part of Group 2b, who received ethanol and BTX-A. One patient reported erythema at the injection site (face), which lasted for one day, and the other reported dry mouth which lasted for 2 weeks (see Table 3). In summary, adverse events occurred in both groups at a rate of
Efficacy
Both groups showed good efficacy, and they were not statistically different from each other (chi-square test,
In Group 2, all patients met their goals for IM injections and 23/27 patients (85%) met their goal for salivary gland injection (at least 50% improvement for over two months). Three patients (11%) showed some efficacy, but did not meet their salivary gland injection goal. One patient (4%) showed no efficacy of the salivary gland injections. Subgroups 2a (BTX-A only) and 2b (BTX-A
Muscles injected with range of units of BTX-A used
Muscles injected with range of units of BTX-A used
Primary outcome: Adverse events
The rate of occurrence of adverse events in Group 2 was less than
Previous studies on intraglandular BTX-A injections for sialorrhea alone showed adverse events in 10–45% of cases [7, 8]. Most of these studies had similar dosing protocols to ours. Moller et al. reported adverse events in 11% of patients who received BTX-A to salivary glands, dosed at 100–120 U to all four glands [8]. Bothwell et al. observed adverse events at a frequency of 11% when injecting a total of 20 U into two glands [9]. Reid et al. [7] injected 100 U into four glands and reported a 23% frequency of adverse events (56% of non-ambulatory patients, which could be viewed as GMFCS level III and above). Scheffer et al. [10] reported 30% of patients experienced adverse events with dosing at 60–100 U divided into four glands (50% of patients with GMFCS levels IV–V), and Ong et al. [11] injected 60–80 U into four glands and reported an adverse event frequency at 43% (53% of patients with GMFCS levels IV–V). Previous studies using intramuscular injections of BTX-A showed adverse events at a rate of 2–28% [12], and the rates of adverse events for intramuscular injections of ethanol and BTX-A ranged from 7% [13] to 21% [14].
In this study, it appears that injections for spasticity combined with injections for sialorrhea caused relatively few adverse events, even in severely affected CP patients. Besides having an experienced practitioner perform injections and using electrical stimulation and ultrasound guidance for the confirmation of correct placement, the selection of muscles and careful consideration when selecting ethanol versus BTX-A is critical for preventing adverse effects. There are certain muscles, such as the gastrocnemius, where anatomical localization of motor end plate zone have been established, where it is therefore preferable to use ethanol injections compared to using BTX-A. We believe that this combination is what accounted for the low rate of adverse events in this study. Additionally, this finding is important, as the ability to perform injections for both problems limits the need to perform separate procedures spaced over time to avoid antibody formation.
Efficacy
In terms of efficacy, 76–85% of patients (depending on whether injections were performed with BTX-A only or BTX-A and ethanol) met both salivary and intramuscular injection goals completely. The success rate was consistent with the results obtained in isolated intraglandular BTX-A injection studies conducted by other groups [7, 8, 9, 10]. The majority of patients in this study who received intramuscular injections with BTX-A also received injections with ethanol. This spared some BTX-A for injections into glands, which was especially critical in this patient population with the majority having spastic quadriple- gic CP at a GMFCS level of V. Additionally, careful selection of BTX-A vs. ethanol is critical for efficacy. For example, BTX-A is preferred when direct pain relief is a goal, as BTX-A acts on calcitonin gene-related peptide and substance P which affect pain transmission. On the other hand, an advantage of alcohol is that it has a longer duration of efficacy [17].
It is also worth noting that in all three groups, the majority of patients reported “Very Good” improvement in drooling for greater than two months (the best response possible; see Fig. 1). 42/52 (about 81%) subjects reported “Very Good” results for drooling. Interestingly, of the ten subjects who reported a score of 2 or less (any score other than the highest score), six of the subjects received only salivary gland injections, and 4/6 of those subjects reported the lowest score. But simply, it appears that patients who only received salivary gland injections were more likely to report a low efficacy. Further study with a larger patient population would be useful to illustrate if this is a statistically powerful finding.
Incidentally, two of the subjects who showed no efficacy after the first salivary gland injection decided to undergo repeat injection. Both met or exceeded their goal for over 2 months after the second injection. There was no clear reason for the failure of the first injection and success of the second injection since there were no significant changes in medical conditions. Regardless, this suggests that patients who fail the initial treatment may benefit from subsequent injections.
It should also be noted, as per Table 2, that the average GMFCS level for patients in this study was indicative of relatively severe symptoms at level IV or above. This indicates that intramuscular and glandular injections can be successful in controlling spasticity and drooling, even in individuals with severe motor function impairment.
Confounding factors
No subjects had surgery during the time of this study, and thus the improvements observed after injections could not be attributed to surgical interventions. The improvements also could not be explained by changes in medications (e.g., a scopolamine patch or glycopyrrolate). In fact, the few patients who were previously on such medications to decrease drooling were able to decrease or discontinue these medications after successful BTX-A injections. Although there were some differences in medical history, there were no significant associations between injection efficacy and gastroesophageal reflux, feeding tube use, and seizure history. Notwithstanding, all patients with gastoesophageal reflux and/or seizures were treated for their conditions prior to BTX-A injections, according to clinical practice.
Limitations and future research
The study was a retrospective cohort study with a limited number of subjects and injections. Applicability of the study’s results may be narrow, as study participation was limited to patients with a diagnosis of CP who were younger than 20 years and had not received prior injections. This also resulted in decreased power of the study. Notably, the population in this study had severe CP, with
Another limitation is that it is possible that the case number was too small to demonstrate the difference between the groups. While this may be the case, it is nevertheless clinically relevant that the side effect profile was low for both groups.
Additionally, dosage in this study should be noted with an average total dosage of 283–287 U per person per session and 12–13 U/kg depending on whether BTX-A or ethanol were given. Generalizing the safety profile in this study to higher doses of BTX-A is not advisable. While goals created with patients were based on the most clinically relevant portion of the Drooling Impact Scale to them (number of bibs, presence of rash, embarrassment etc.), due to the retrospective nature of the study, there was insufficient data to use standardized outcome measures (such as the Drooling Impact Scale). Therefore, it would be inaccurate to try to draw major conclusions regarding efficacy of SEMLC with either BTX-A alone or in conjunction with ethanol combined with salivary gland injections.
However, the data available do indicate that there was no major loss of efficacy when the two procedures are combined. A prospective study in which standardized drooling and spasticity assessment tools were utilized in both pre- and post-injection evaluations would be more powerful in proving efficacy.
Conclusion
This study suggests that combining the procedures for BTX-A injections into the salivary glands and multi-level intramuscular injections with BTX-A injection with or without ethanol for diffuse spasticity in a single session is a safe procedure in children with CP, allowing both conditions to be treated simultaneously. Our data indicated that efficacy of both salivary gland and intramuscular injections is likely maintained. A future, large-scale prospective trial would be beneficial to support these findings.
Footnotes
Conflict of interest
Heakyung Kim has received research grant from Allergan and Ipsen, has received honorarium from Catalyst Medical Education LLC, American Academy of Physical Medicine & Rehabilitation (AAPM&R), and is a Allergan and Ipsen Pediatric Spasticity advisory member. All other authors have no conflict of interest to report. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
