Abstract
PURPOSE:
To examine the results of repeated episodes of 21-day pediatric constraint induced movement therapy (PCIMT) paired with gross motor training (GMT).
METHODS:
Nineteen children, age 14 months – 6 years with unilateral upper extremity impairment enrolled in this cohort study to receive repeated episodes of 21 day PCIMT-GMT. Outcome measures included the Peabody Developmental Motor Scales-2 (PDMS-2), the Assisting Hand Assessment (AHA) and the Canadian Occupational Performance Measure (COPM).
RESULTS:
All children demonstrated improvement in raw scores following each episode of PCIMT-GMT with a statistically significant change in the least squares estimated mean for all measures except the PDMS-2 total motor raw score and gross motor quotient for the preferred hand in the fourth episode (
CONCLUSION:
Children with unilateral upper extremity impairment demonstrate improvements in fine motor, gross motor, and bimanual skills, along with functional changes in participation in daily life, following PCIMT-GMT. Participation in repeated episodes can lead to further improvements.
Keywords
Introduction
The development of hand function and motor skills in young children with unilateral upper extremity impairment is an important issue with implications for activity level and participation in daily life. This study examined activity level and participation outcomes in children with unilateral upper extremity impairment after repeated episodes of pediatric constraint induced movement therapy (PCIMT) paired with gross motor training (GMT). While seemingly promising, this repeated approach to rehabilitation has not been studied in depth thus far.
Pediatric constraint induced movement therapy
PCIMT is an intervention strategy targeting impaired upper extremity function in children with hemiparesis, by constraining the preferred hand while providing intensive training. Research indicates it to be effective in the improvement of unimanual and bimanual skills in these children [1, 2, 3]. PCIMT demonstrates modest to strong treatment effects for improving movement quality and efficiency of the more impaired upper extremity when compared with usual care [3]. Improved upper extremity activity and participation is also observed when PCIMT is compared to observation or sham intervention [1]. Despite a wide range of PCIMT treatment protocols, all randomized controlled clinical trials of PCIMT have achieved improvement in upper extremity skills for children with unilateral cerebral palsy [4]. At this time, there is limited information on the impact of repeated episodes of PCIMT, though this has been identified as a high priority for future study [4].
Ramey, DeLuca and Coker-Bolt have identified five essential components that should be present for a treatment program to qualify as being an evidence-based form of PCIMT [5]. These include constraint of the preferred arm, high dosage in a concentrated time frame, use of shaping techniques and repetitive task practice, learning functional skills in a natural and diverse setting and having a post-treatment transition plan. They further classify treatment as signature, modified or alternative based on intensity of treatment and adherence to the five essential components [5]. Signature PCIMT includes all five essential PCIMT components and uses a high dosage of therapy in a concentrated period of time (minimum of three hours per day for five days/week for at least two weeks). Modified PCIMT includes all five essential PCIMT components but the dosage is less. Alternative PCIMT does not include all five essential components.
Eliasson et al. also have classified PCIMT into three categories, signature, modified, and hybrid [4]. Signature constraint induced movement therapy (CIMT) consists of restraint of the preferred arm for 90% of waking hours while providing intensive training to the more involved arm for three or more hours each day for a minimum of two weeks. Modified constraint induced movement therapy (mCIMT) requires a lessened treatment intensity than signature CIMT, with several variations in terms of the amount and type of constraint and the type, setting and intensity of treatment. Hybrid CIMT incorporates variations of CIMT and bimanual therapy (BiT), and protocols for the latter provide high intensity training of upper extremity activities without constraining the preferred arm.
The effects of repeated episodes of PCIMT have been described in two research studies and two case reports. Both single subject case reports found improvements in fine and gross motor skills and spontaneous use of the assisting hand [6, 7]. Improved quality of movement, amount of arm use, and emergence of new motor behaviors were noted for 28 children after receiving repeated episodes of signature PCIMT [8]. Improvements in movement efficiency, speed and dexterity, and caregiver perception of amount of use and quality of movement of the involved limb were attained for eight children who completed modified PCIMT [9]. The children assessed in the aforementioned studies all demonstrated improvement with repeated episodes of PCIMT, though type of the utilized PCIMT and outcome measures varied.
Gross motor activities have been included in several PCIMT studies [6, 7, 10, 11, 12]; however, the specific time spent training gross motor skills was not clearly defined and/or the training failed to include the lower extremities and trunk. Gross motor improvements as noted on the Gross Motor Function Measure (GMFM) sections for standing, walking-running-jumping [11], and gait changes measured on the six-minute walk test [12] and GAITRite system [10, 12] have been reported by studies using mCIMT, PCIMT, and hybrid PCIMT. These changes were in addition to changes in upper extremity function. One study using BiT with activities that also targeted the trunk and lower extremity found improvement in bimanual skills, manual ability in daily life, gait, and social participation [13]. These results suggest that PCIMT paired with gross motor training has the potential to result in global motor changes. Currently, there is limited information on the impact of repeated episodes of intensive gross motor training on the development of gross motor function over time [14].
This report examines outcomes of repeated episodes of PCIMT-GMT provided in a clinic setting. This protocol distinctly pairs three weeks of signature PCIMT with one week of bimanual therapy, and includes gross motor skill training to address the global motor learning needs of the child. In this prospective cohort study, we sought to measure the effect of PCIMT-GMT on activity level and participation in daily life for children with unilateral upper extremity impairment.
Methods
Participants
This prospective cohort study included children between the ages of fourteen months and six years who received multiple episodes of PCIMT-GMT at a pediatric hospital-based outpatient clinic from October 2006 to December 2015. Children were screened prior to entering the study to verify they were appropriate candidates for the treatment protocol. Inclusion criteria were as follows: weakness of one upper extremity; medical stability; grade 2/5 strength as measured by manual muscle testing of the fingers, elbow, and shoulder of the more impaired upper extremity; ability to participate in six hours of play per day; and parental/guardian commitment to support the high intensity treatment schedule.
Children with significant gross motor and cognitive delays were not excluded unless they could not meet the inclusion criteria. Medical diagnoses represented include cerebral palsy-hemiplegia, cerebral palsy-triplegia, traumatic brain injury, brain tumor, schizencephaly, dysplasia, cerebral vascular accident, chiari malformation, and cerebral palsy – spastic quadriplegia. The one child with a medical diagnosis of cerebral palsy – spastic quadriplegia had a clinical presentation of triplegia based on movement assessment and standardized testing; they were therefore included in our sample.
Repeated episodes of PCIMT-GMT were completed if the child continued to meet the inclusion criteria and the parents/guardians chose further treatment. Ten children completed two episodes, seven completed three episodes, and two completed four episodes of PCIMT-GMT. The repeated episodes were completed no earlier than nine months following the conclusion of the previous episode, in order to allow the child to practice and integrate their new skills in addition to the need for prudent use of resources. Each episode was 21 days over the course of four weeks, Monday through Saturday the first week, Monday through Friday for each week two through four. If a child was ill or a session was missed the time was made up during the four week episode, so all children received 126 hours of therapy. Institutional review board approval was obtained for the study along with parental consent for data collection of standardized tests, video, photographic documentation, and standard clinic documentation.
Procedure
The children participated in six hours of treatment daily for 21 days over the course of the four-week protocol. Treatment consisted of developmentally appropriate fine motor, gross motor and daily living activities utilizing principles of shaping, repetition and motor learning. The entire treatment and assessment were conducted by two experienced pediatric physical therapists who completed formal training in the ACQUIREc protocol, a form of signature PCIMT [15, 16, 17].
Constraint was provided by use of a long arm cast that was worn 24 hours daily for three weeks. During the fourth week, the cast was removed during the six-hour treatment day to focus on bimanual skill training, and then placed back on the child prior to returning home for the day. Daily treatment was provided primarily by one therapist, while the second therapist fabricated the cast and conducted weekly cast care. Cast care consisted of skin checks, washing and applying lotion to the constrained arm, weight bearing activities and active range of motion.
Treatment was conducted in a hospital-based outpatient clinic. A typical treatment day for a younger child consisted of activities of daily living (eating, dressing, brushing teeth), fine motor activities (grasp, release and placement, hand opposition, and finger isolation), gross motor activities (transitions, gait, balance, agility, stairs, mobility on ride toys, ball skills, and coordination), upper and lower extremity weight bearing/strengthening, core strengthening, and stretching as needed. Gross motor activities were specifically trained for two of the six hours each day. Therapeutic activities were selected and graded with the goal of keeping the child motivated and engaged throughout the day.
Treatment goals were created for each episode in collaboration with each child’s parents/guardians based on initial assessment results. Information on the child’s progress was shared daily with each family and feedback on the child’s functional status at home was received in kind. The parents/guardians were instructed in a formal individualized home program on the final day of treatment to assist with transfer of newly acquired skills into daily life. Whenever possible, the treatment outcome was discussed with the child’s primary outpatient occupational and/or physical therapists located in a variety of healthcare systems to assist with carryover of the new skills once the episode of PCIMT-GMT was completed. The children resumed standard occupational therapy and/or physical therapy as needed after each episode (1-2x/week), except for one child who did not receive services between the first episode and second episode of PCIMT-GMT due to family circumstances. No children had surgery on an upper or lower extremity during the timeframe in the study. One child received Botox to the assisting hand one month prior to participation in a second episode of PCIMT-GMT.
Outcomes and measures
Outcomes included activity level for fine motor, gross motor, and bimanual skills, as assessed by the PDMS-2 and AHA; participation in daily activities, measured by the Canadian Occupational Performance Measure (COPM); retention, defined by participants’ scores on standardized measures from day one of a subsequent episode compared with day one of the previous episode; and severity, using the Gross Motor Classification Scale-Expanded and Revised (GMFCS-E&R) and mini-Manual Ability Classification System (MACS) for children 1–4 years of age or MACS (children 4–12 years of age).
Outcomes were evaluated by comparing outcome measures administered by a single physical therapist for each child on day one (pre) and day 21 (post) of each episode of PCIMT-GMT. Children with two episodes of care had four data collection points over time (pre episode one, post episode one, pre episode two, post episode two), and those with three and four episodes had six and eight points over time, respectively. Each child was assessed using the PDMS-2 (
The PDMS-2 is a criterion and norm reference activity level outcome that was utilized as a measure of fine and gross motor skill capacity. Inter-rater reliability and test-retest reliability is well-established for children with disabilities, specifically for children with cerebral palsy [18]. As utilized previously in CIMT research, the left and right hands were tested and scored separately for unimanual skills in the object manipulation, grasping and visual motor integration subtests of the PDMS-2 [19, 20, 21]. Two children exceeded the upper age limit on the PDMS-2 during their second episode of PCIMT-GMT; therefore total motor raw scores were obtained but quotient scores were not used. These children had motor and cognitive impairment levels such that they could not complete age level motor tests such as the Bruininks-Oseretsky Test of Motor Proficiency, Second Edition (BOT-2), and their scores on the PDMS-2 did not approach ceiling levels, so the PDMS-2 was deemed the most appropriate measurement tool to assess their skill level.
The AHA is a standardized, criterion-referenced test for children 18 months to 12 years who have unilateral upper limb impairment, designed to assess a child’s typical performance when performing tasks rather than their best effort or capacity [22]. It was selected as an activity level measure of how effectively the child used their more affected hand (assisting hand) in bimanual activities [22]. The AHA has demonstrated inter-rater reliability of 0.98, intra-rater reliability of 0.99 [23], and test-retest reliability of 0.98–0.99 [24]. Administration was standardized as both evaluating physical therapists completed AHA certification to ensure appropriate use of the measure. The smallest detectable difference is 5 or more AHA scaled score units [25]. One child was tested at 14 months with scoring considered representative as they could actively engage with all the items presented. The Mini-AHA had not yet been created at the time of this study. As mentioned in the Participants section, one participant who had a diagnosis of cerebral palsy – spastic quadriplegia actually presented with unilateral impairment with fine motor skills in the average range in their dominant hand. Therefore, the AHA was appropriate for all children measured, from a clinical standpoint.
The COPM is a client-centered measure designed to detect change in a client’s self-perception over time that was used as a participation level measure of the functional status of the child. It is validated for use with a parent or caregiver when the child is not able to identify or verbalize problems on their own [26]. Test-retest reliability is 0.84 to 0.92 [27]. The COPM demonstrates internal consistency, content and construct validity, and responsiveness when adapted for use with parents of children with spastic hemiplegic cerebral palsy [28]. A change score of 2 or more points is deemed clinically meaningful [27].
Data analysis
To examine change within episodes we conducted a linear random effects model with patient random effects for the change between pre and post AHA, COPM and PDMS-2 variables.
To examine change between episodes we computed linear and quadratic growth curves. These were run using SAS Version 7.12 Proc Mixed (SAS Institute, Inc., Cary, NC, USA), using a
Descriptive data of participants (
19)
Descriptive data of participants (
GMFCS-E&R
Activity level for fine motor, gross motor, and bimanual skills
Nineteen children successfully completed the full protocol of PCIMT-GMT (
Episode least squares means: AHA, COPM, PDMS-2
Episode least squares means: AHA, COPM, PDMS-2
AHA
Mean scores by episode of PCIMT-GMT across time (by group with 2, 3 or 4 episodes of PCIMT-GMT)
Note: Data collection point by episode over time 1
Mean PDMS-2 Total Motor Raw Score-Assisting Hand (1a) and Preferred Hand (1b) across episodes of PCIMT-GMT (by group with 2, 3 or 4 episodes of PCIMT-GMT). Note: Data collection point by episode 1 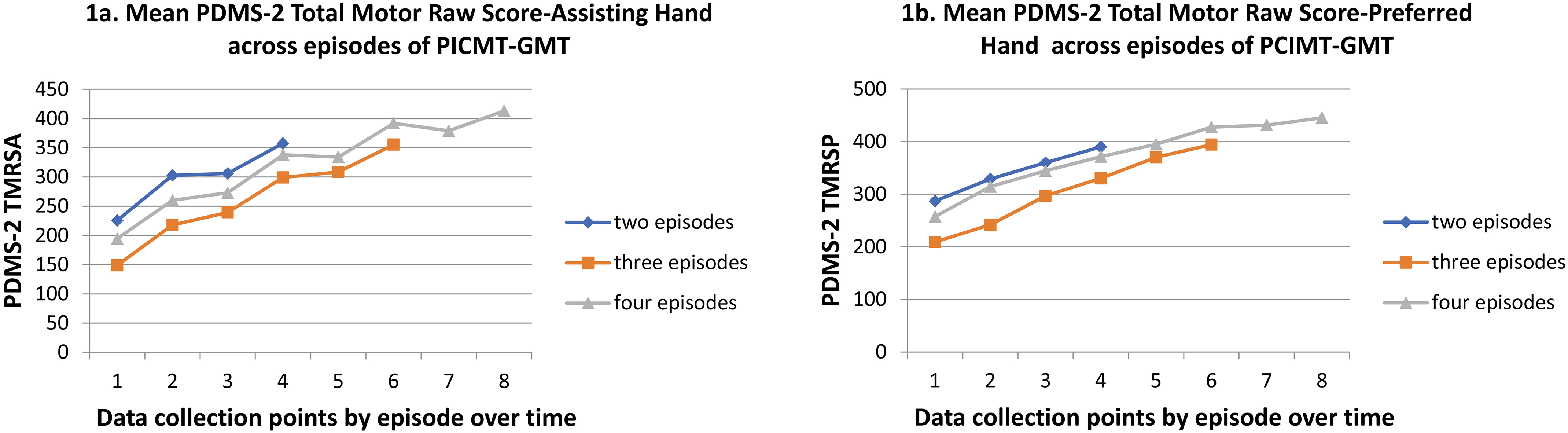
Mean AHA Scaled Score across episodes of PCIMT-GMT (by group with 2, 3 or 4 episodes of PCIMT-GMT). Note: Data collection point by episode over time 1 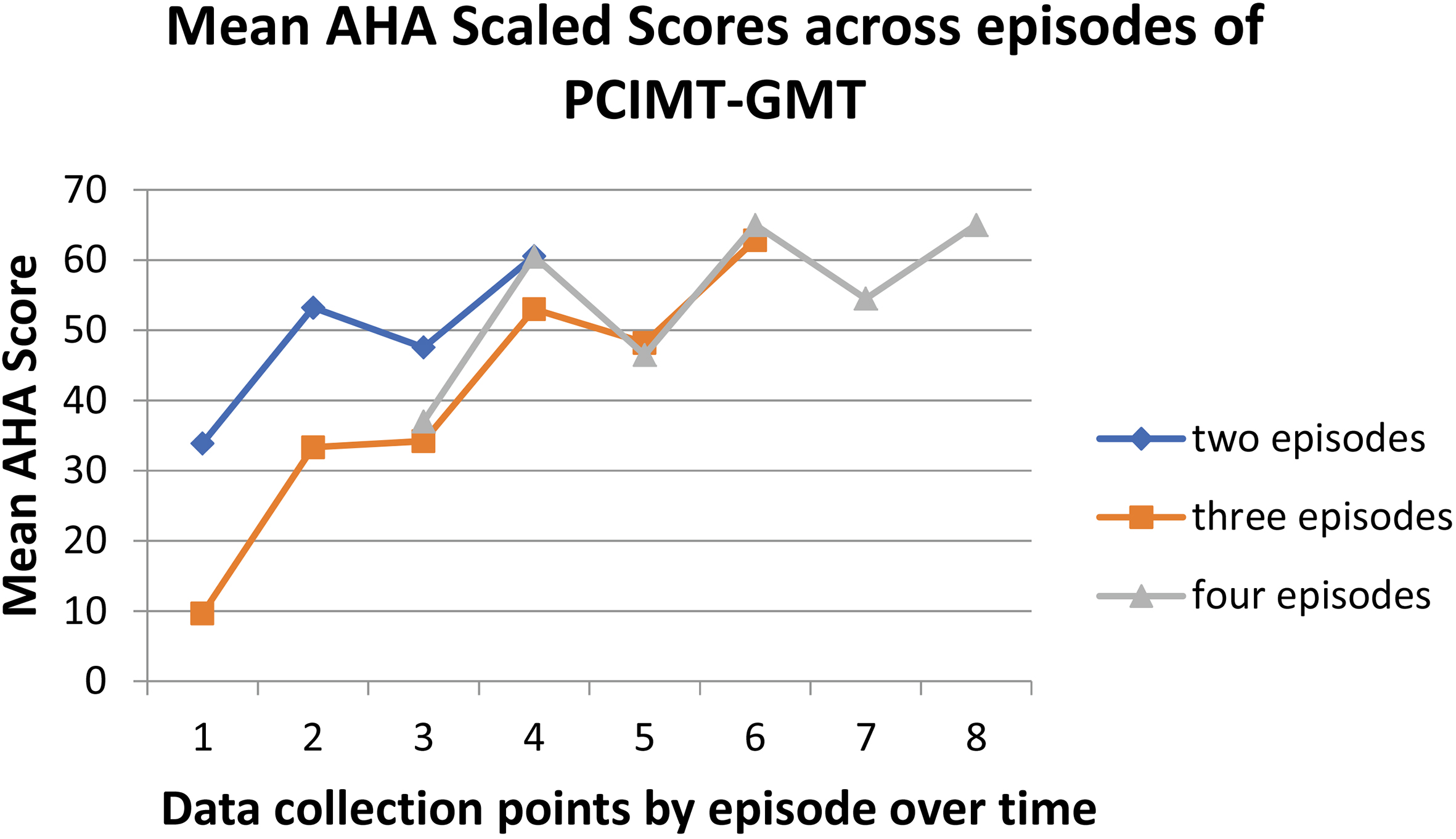
Linear growth curves across four episodes
AHA
The raw mean change in PDMS-2 TMRSA, PDMS-2 TMRSP, and AHA scaled score are shown by group in Figs 1 and 2 and in Table 3. The groups of children who had two, three and four episodes of care show a similar response to treatment. Rates of change on the scaled score for the AHA and PDMS-2 TMRSA show notable change from day one to day 21 of PCIMT-GMT with a leveling off of change or decline between episodes of PCIMT-GMT.
The linear change between episodes for both pre scores and post scores on the AHA and PDMS-2 are shown in Table 4. Both the pre and post scores on the AHA sum and scaled scores are significant as are the pre and post PDMS-2 TMRSP and PDMS-2 TMRSA scores. Not shown are the results of the quadratic growth curves which were, in all cases, insignificant at the 0.05 level.
Clinically meaningful change on the COPM was reported by a child’s parent/guardian in all 17 episodes where this outcome measure was utilized. From day one to 21, the raw mean change in COPM Performance score was 4.16 (
Retention
Progression in raw mean scores was analyzed for the PDMS-2 TMRSA, PDMS-2 TMRSP, and AHA scaled score over time for children receiving two, three and four episodes of care (Figs 1 and 2, Table 3). The mean time interval between episodes of PCIMT-GMT was 18 months between episodes 1 and 2, 16 months between episodes 2 and 3, and 13 months between episodes 3 and 4. The PDMS-2 TMRS and AHA scores on day one of each repeated episode were higher than the scores from day one of the previous episode for all children.
Severity
Children were classified retrospectively using the GMFCS-E&R and mini-MACS or MACS (Table 1). The raw mean change on the AHA scaled score and PDMS-2 TMRS for the preferred hand and assisting hand demonstrated no significant difference between classification levels on the GMFCS-E&R and mini-MACS through ANOVA analysis (Table 5).
Comparison of mean differences in amount of change each episode across levels of GMFSC-E&R and MACS/mini-MACS
Comparison of mean differences in amount of change each episode across levels of GMFSC-E&R and MACS/mini-MACS
GMFCS-E&R
In this study of repeated episodes of PCIMT-GMT, participants demonstrated statistically significant gains in fine motor and gross motor skills after each episode of PCIMT-GMT, as measured by the PDMS-2, and bimanual skills as measured by the AHA. Additionally, parents reported clinically meaningful gains in their children’s ability to participate in daily activities as measured by the COPM. Children with a range of functional levels benefited from this intervention. Our eligibility criteria were intentionally inclusive of children with more severe limitations. While we included a more heterogeneous pool of conditions than other studies of similar therapies, all participants presented with unilateral upper extremity impairment.
These findings contribute to a small body of evidence on repeated episodes of PCIMT, although previous studies have paired different intensities and treatment components than in our study. The children in the study by Charles [9] received 60 hours of treatment compared to 120–126 hours for those assessed by DeLuca [8] and this study. Different measures were used by Charles and DeLuca; therefore it is not possible to determine if intensity of treatment had an influence on the amount of change. All children in this study made additional gains with each repeat episode of PCIMT-GMT, which is consistent with the previous literature [6, 7, 8, 9]. The gains noted here in both fine motor and gross motor skills as observed by changes in PDMS-2 scores are supported by two case reports discussing repeated episodes of PCIMT [6, 7], where positive change was also noted. Direct comparison is not possible, however, because those case studies utilized the PDMS-2 as an outcome measure for fine motor skills, but used separate outcome measures for gross motor skills. The PDMS-2 raw score gains observed in this study were larger than those reported in previous PCIMT studies utilizing single episodes of care; however, those studies only assessed fine motor skills versus global motor skills [19, 21, 29]. These results on PCIMT-GMT suggest that it is possible to treat gross and fine motor skills and to achieve gains in global motor skills without inhibiting improvements in other domains. This is consistent with findings from intensive BiT paired with lower extremity training [13]. The stimulation of multiple areas of the brain with a wide variety of meaningful tasks may in fact enhance global changes in function.
The PDMS-2 TMRSA is primarily a reflection of unimanual gains by the affected hand. The children studied demonstrated very little change in this score between episodes of PCIMT-GMT (Fig. 1). This is consistent with previous evidence that suggests rates of change during PCIMT are much larger than gains measured with standard care delivery models [3]. Although further study is required, standard care may prevent significant loss of skill between episodes of PCIMT-GMT.
All children assessed by the AHA demonstrated change greater than the smallest detectable difference, with every child making further gains with each repeated episode of PCIMT-GMT. These results provide evidence that the rate of change can be positively impacted by repeated episodes of PCIMT-GMT over the course of several years (Fig. 2) in children less than 6 years of age. The rate of change observed in the PDMS-2 TMRSA, PDMS-2 TMRSP, and AHA during PCIMT-GMT is notably larger than the rate of change recorded between episodes of PCIMT-GMT when children typically received standard care (Figs 1 and 2). The ability to make large improvements over four weeks has the potential to provide a highly beneficial developmental advance for children as they strive to keep up with peers. It is not possible to fully rule out the effect of maturation on the functional gains observed during PCIMT-GMT; however, the significant difference between the rates of change observed during and between episodes of PCIMT-GMT suggests it is highly unlikely for maturation to be a major influence. It appears that repeated episodes of PCIMT-GMT were able to change the course of functional hand development for the individual children in this study, though there is not a control to allow more definitive comparison. Klingels et al. found that children with unilateral cerebral palsy show a decline in AHA scores after the age of nine [30]. Further data on the long-term outcomes into late childhood and adolescence would be beneficial and warrants further investigation.
Functional improvements in participation for individual goals set in collaboration with the parents/guardians utilizing the COPM demonstrated clinically meaningful change in all cases. Typical gains observed included improved ability to grasp everyday objects, dress, eat, and move safely and efficiently about the child’s environment. Though participation was not assessed on the COPM for all children, feedback families provided on the program was positive for all children in this cohort. This satisfaction with the outcomes of treatment is also reflected in the number of families who elected to have their child complete repeated episodes of the program. There is some concern that higher intensity treatments could have a negative social impact on the child and their family [31]. This experience suggests that the children and families managed the higher intensity of treatment quite well and found it to be a positive experience.
All children, regardless of their range of initial bimanual, gross motor, and fine motor skill abilities, were able to complete the PCIMT-GMT program and demonstrate significant positive change. The changes in PDMS-2, TMRSA, and AHA scaled score, though diminishing over time, were still quite large. No participants dropped out of the study. Children with more severe limitations in fine and gross motor function often are excluded from other CIMT treatment and studies [4]. Though the numbers were small in this study, this model of PCIMT-GMT with a 1:1 treatment ratio was beneficial for children regardless of impairment level. Group based PCIMT with less direct support and training might make this less feasible for children with more severe impairments. The high level of direct individualized treatment in this protocol may make it possible for children with more severe impairments to learn new skills and improve participation without excessive frustration or stress.
Limitations
The full interpretation of the results of this study is limited by the absence of a control group and small sample size. The assessments were not blinded, which has the potential to introduce bias into the results. However, the strong reliability and test-retest reliability of the selected outcome measures help to control for some of this potential variability. The families who elected to enroll their children in an intensive treatment program had adequate support systems to manage this commitment in their daily schedules and may not be representative of the general community, a limitation also noted by another recent clinical cohort [16]. The addition of the AHA and COPM during the study period limited the number of measurements we could capture using these tools; ideally these measures would have been collected throughout the study. However, there is still value to reporting these findings, and future research should include them from the beginning. The use of the PDMS-2 for two children who exceeded the upper age limit in their second PCIMT-GMT episode, and AHA for one child who did not meet the lower age limit, while not consistent with the validated age range for that tool, followed from the realities of clinical practice, as both tests were the most feasible options for measuring motor skills given the impairment levels of those participants. The intervals between PCIMT-GMT treatment episodes were not standardized, making the analysis of change open to more external influences. Currently, there are no set standards for type, frequency, or amount of PCIMT, leaving therapists in clinical situations to make professional judgments based on individual patient needs, circumstances, and outcomes. As such, this study reflects the current clinical practice reality of providing this therapy. As the body of evidence for repeated PCIMT grows, standards and best practices may be established.
Conclusions
The purpose of this study was to investigate the effects of repeated episodes of a 21-day PCIMT-GMT on activity and participation levels for children with unilateral upper extremity impairment. The results indicate that children with a range of impairment levels may demonstrate improvements in fine motor, gross motor, and bimanual skills, along with functional changes in participation in daily life following PCIMT-GMT. Participation in repeated episodes of PCIMT-GMT can lead to additional improvements. Further research is needed to assess the impact of repeated episodes of PCIMT-GMT treatment into adolescence and adulthood. While an optimal number of episodes of PCIMT-GMT is not known, this study provides evidence of substantial progress in outcomes through four episodes.
Footnotes
Acknowledgments
The authors thank Kristen Griffin, MA, MPH and Jennifer Henrickson, PT, DPT, PCS for their assistance with reviewing and editing the manuscript. There are no funding sources to disclose.
Conflict of interest
The authors have no conflicts of interest to disclose.
