Abstract
PURPOSE:
Atypical skeletal development is common in youth with myelomeningocele (MM), though the underlying reasons have not been fully elucidated. This study assessed skeletal maturity in children and adolescents with MM and examined the effects of sex, age, sexual development, ethnicity, anthropometrics and shunt status.
METHODS:
Forty-three males and 35 females with MM, 6–16 years old, underwent hand radiographs for bone age determination. The difference between bone age and chronological age was evaluated using Wilcoxon sign rank tests. Relationships between age discrepancy (skeletal-chronological) and participant characteristics were assessed using multiple linear regression with forward selection.
RESULTS:
Overall, forty percent (31/78) of MM participants had an advanced bone age of 1 year or
greater (median: 2.5 years), while 47% (37/78) were within 1 year above or below their
chronological age (
CONCLUSIONS:
Advanced skeletal maturity is common in children/adolescents with MM over 8 years of age who have reached puberty (65%), particularly those who are overweight (80%). Hormonal effects associated with adiposity and sexual maturity likely influence skeletal maturation. Clinicians may use Tanner stage and weight or BMI to gain insight into skeletal maturity.
Introduction
Myelomeningocele (MM), the most common and severe form of spina bifida, results from failed neural tube formation in utero and affects approximately 3,000 pregnancies in the United States annually [1]. In addition to muscle weakness and paralysis, skeletal development can be altered in those with MM, with decreased bone density and differences in skeletal maturity having been previously documented [2, 3, 4, 5, 6]. The underlying causes of these pathologies have not been fully elucidated, but the effects of mechanical loading, nutrition, bone metabolism and the endocrine system have been investigated as possible contributors [2, 3, 4, 5, 6]. Altered bone development can contribute to increased fracture risk in both the short and long term.
Reduced bone density and incongruent skeletal maturity are likely multifactorial. Previous studies have linked lower bone mineral density in children with MM to wheelchair use and overall limited ambulation [2, 3] while another study correlated increased calcium excretion to lower bone density [4]. Incongruent skeletal maturity, both advanced and delayed, has been documented in the MM population. Previous work suggests that neither gender nor neurosegmental level plays an instrumental role in bone age; however, there are mixed reports on the effects of shunt status [5, 6]. The presence or absence of a shunt has been used to evaluate the effect of hydrocephalus and dysfunctions of the hypothalamic-pituitary axis on skeletal maturation. Feeley et al. found that presence of a shunt was associated with delayed bone age [5], whilst Kalen and Harding found no effect of hydrocephalus or number of shunt revisions on bone age in children with MM [6], possibly due to the relatively low number of participants with shunts in the Kalen study. The Kalen study did find advanced skeletal maturity in 32% of children with MM over 8 years of age. The inconclusive results of previous studies may be, at least in part, due to differences in the methodologies used to assess bone age (Gruelich and Pyle vs. Oxford method) as well as differences in the MM participants studied.
Skeletal maturity and sexual development are inter-related [7], but past work has not examined this relationship in children with MM who often have precocious puberty [8]. Additionally, obesity has been correlated to advanced bone age in youth without disability, possibly due to hormonal effects associated with adipose tissue [9, 10, 11, 12]. However, even though 50–83% of patients with MM are overweight or obese [13, 14], the role of excess weight in incongruent skeletal maturity in MM has not been previously investigated. Finally, fracture rate and bone age have been shown to differ among ethnicities in populations without MM [15, 16]. Given the increased incidence of MM amongst Hispanic populations and the high incidence of obesity in the MM population, ethnicity and body mass index (BMI) are important to consider when assessing differences in bone age.
An understanding of which patients with MM are likely to be advanced or delayed in skeletal development could help improve patient management and treatment decision making, particularly regarding the timing of orthopaedic interventions and treatment of precocious puberty. Fractures, spinal deformity and deformity of the lower limbs are common in children and adolescents with MM, and treatment decision making to address these problems necessitates an understanding of skeletal growth remaining, which depends on bone age [4, 17]. The purpose of this study was to evaluate the relationship of multiple factors including age, sex, sexual maturity, anthropometric measures, ethnicity and shunt status on skeletal maturity in the MM population. It was hypothesized that chronological age, sexual maturity (Tanner Stage), anthropometric measures, ethnicity and shunt status would be related to advanced skeletal maturation in the MM population, while sex would not.
Methods and materials
Following approval by the institutional review board at Children’s Hospital Los Angeles, appropriate written assent and consent were obtained from all participants and their parent(s) or legal guardian(s). This prospective, cross-sectional study included participants who had a diagnosis of myelomeningocele and were between 6–16 years of age. Potential participants were excluded if current use of glucocorticoids or other medications affecting growth and/or development was reported, if they had metal implants in their tibias bilaterally, or if they had other diagnosis excluding hydrocephalus and asthma as determined from guardian and participant self-report. All participants were part of a larger study investigating the relationship between bone mass and ambulation [18].
Participant characteristics including height, weight, and BMI were measured by
an experienced pediatric physical therapist. Height was measured in standing for
participants who could stand upright and supine for those who could not. Laboratory-specific
intra- and inter-rater reliability for height and weight measurements was within 0.4 cm and
0.9 kg, respectively. Height, weight, and BMI percentiles for age were determined using
growth charts from the Centers for Disease Control and Prevention, and participants were
classified into groups based on cutoffs for height (
Demographic and anthropometric characteristics of the study participants
Demographic and anthropometric characteristics of the study participants
Continuous variables (age, age difference, and anthropometric percentiles) are
presented as median (interquartile range) [range]. Categorical variables (Hispanic,
shunt, Tanner) are presented as
Statistical analyses were performed using Stata14 (Stata Corp, Texas).
Descriptive statistics are presented using median and interquartile range (IQR).
Participant characteristics were compared between males and females using Mann Whitney
rank sum tests for continuous variables (age, height, weight, BMI percentiles) and
Fisher’s exact tests for categorical variables (sex, Hispanic ethnicity, Tanner stage, and
shunt status). Differences between bone age and chronological age were assessed using
Wilcoxon sign rank tests and are described by the median difference, IQR, and 95%
confidence interval (CI) of the median difference. Age discrepancy was defined as
age
Results
Age, sex, and sexual development
Seventy-eight participants with MM (35 females) were included in the
analysis; 14 were classified as sacral level, 12 as low lumbar, 41 as mid lumbar and 11 as
high lumbar and above (5 high lumbar, 6 thoracic). The overall median chronological age
was 9.8 (IQR 7.7 to 12.2) years, and median bone age was 10.5 (IQR 7.5 to 14.0) years. The
median difference between bone age and chronological age for all participants was
0.50 years (IQR –0.5 to 1.9; 95% CI 0.20 to 1.05;
Difference between skeletal and chronological age in participant subgroups
Difference between skeletal and chronological age in participant subgroups
Values are presented as median (IQR) [range]. P-values are from Wilcoxon Sign Rank tests.
Difference between skeletal age and chronological age as a function of
chronological age in boys and girls.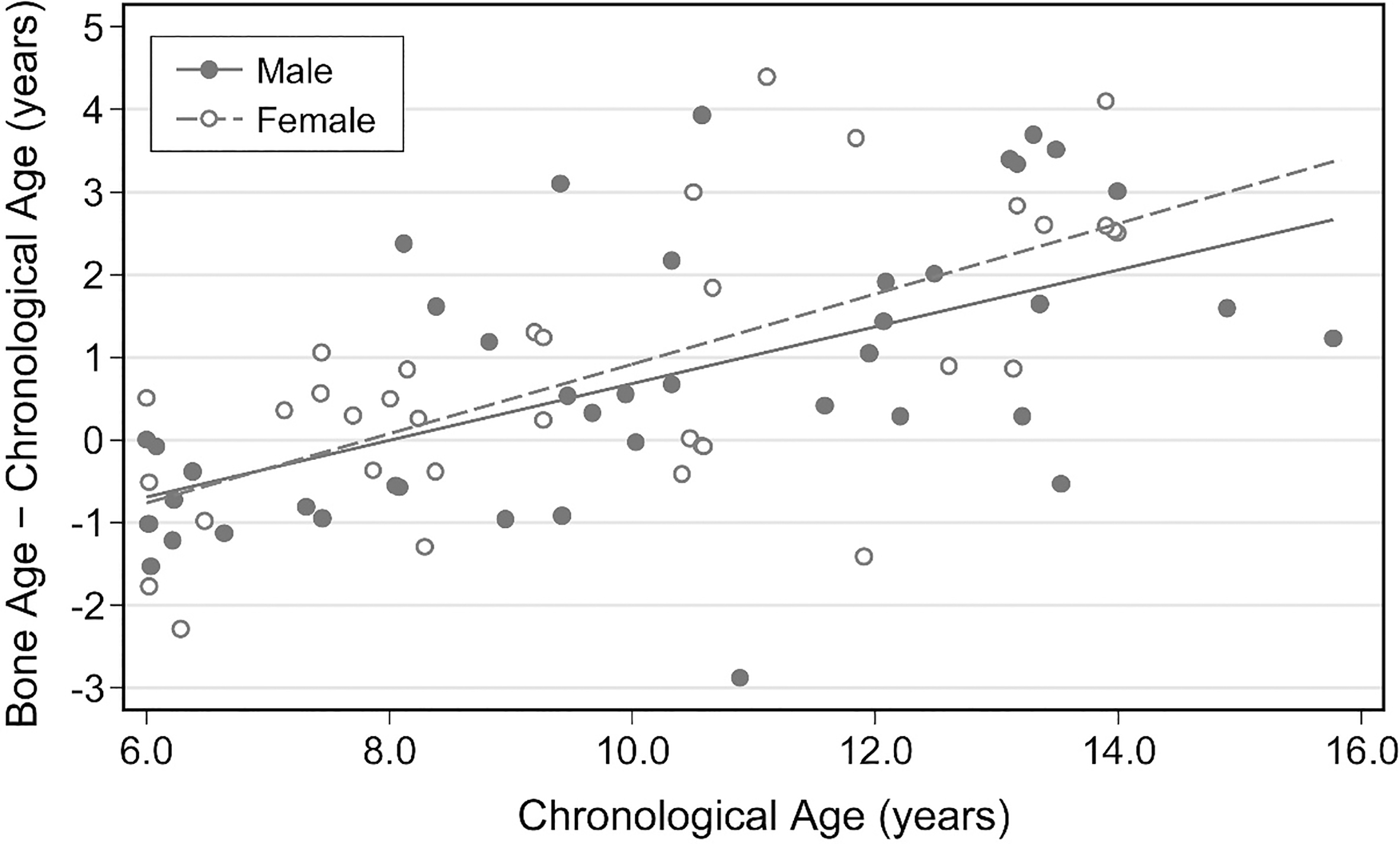
The difference between bone age and chronological age increased with age
in both males and females, progressing from slightly delayed bone age in younger children
to increasingly advanced bone age after a chronological age of approximately 8 years
(
Results of simple linear regression predicting the difference between skeletal and chronological age
Coef.
Difference between skeletal age and chronological age as a function of Tanner stage
of sexual maturity.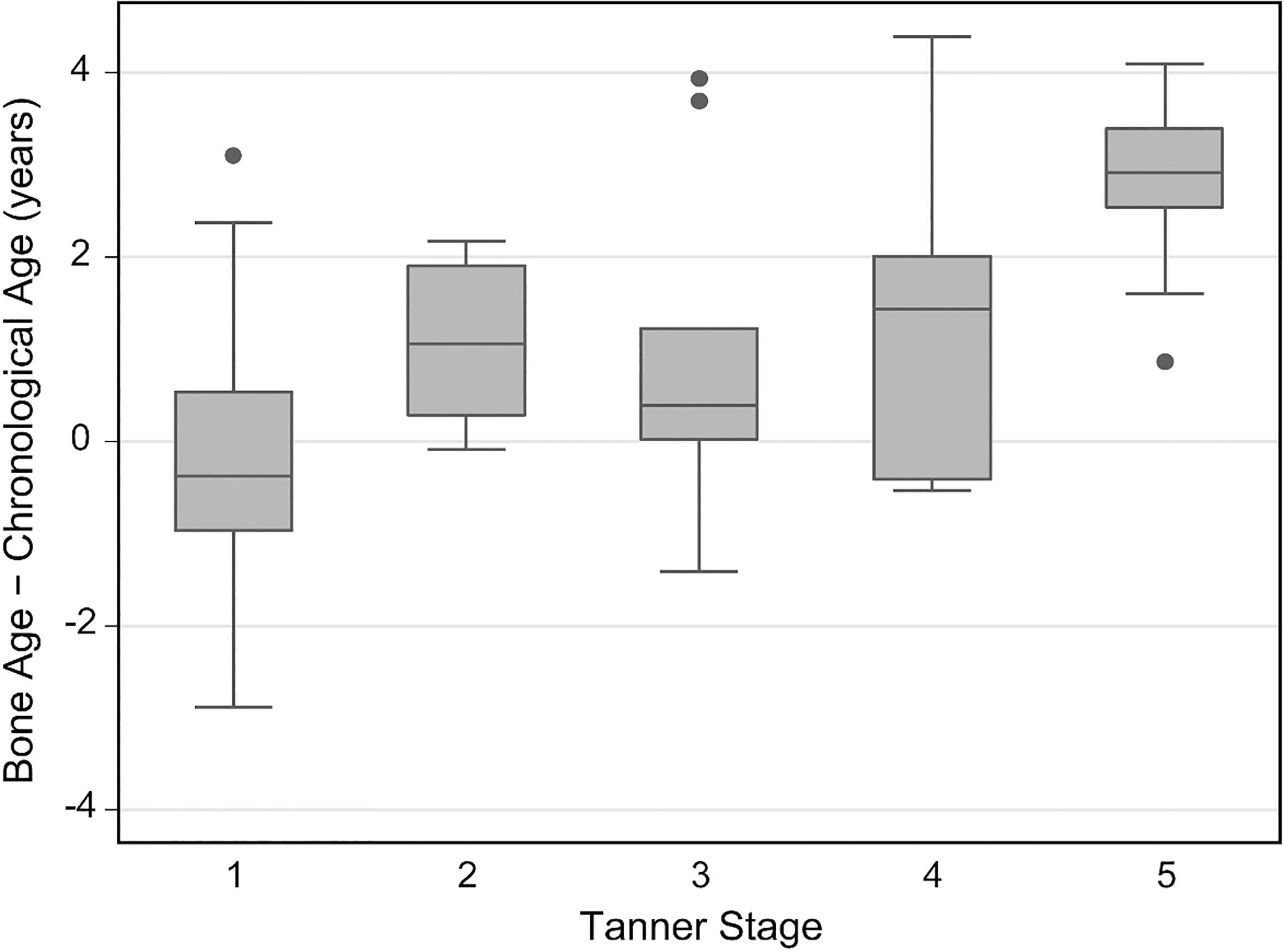
The difference between bone age and chronological age was positively
correlated with height, weight, and BMI percentiles (Table 3). Most notably, advanced skeletal maturity manifested at the highest BMI
percentiles. Bone age was advanced more than 1 year in 49% (22/45) of children who were
overweight or obese and in 80% (20/25) of these who were also over 8 years of age had
entered puberty (Tanner
Difference between skeletal age and chronological age for normal/healthy weight
(BMI 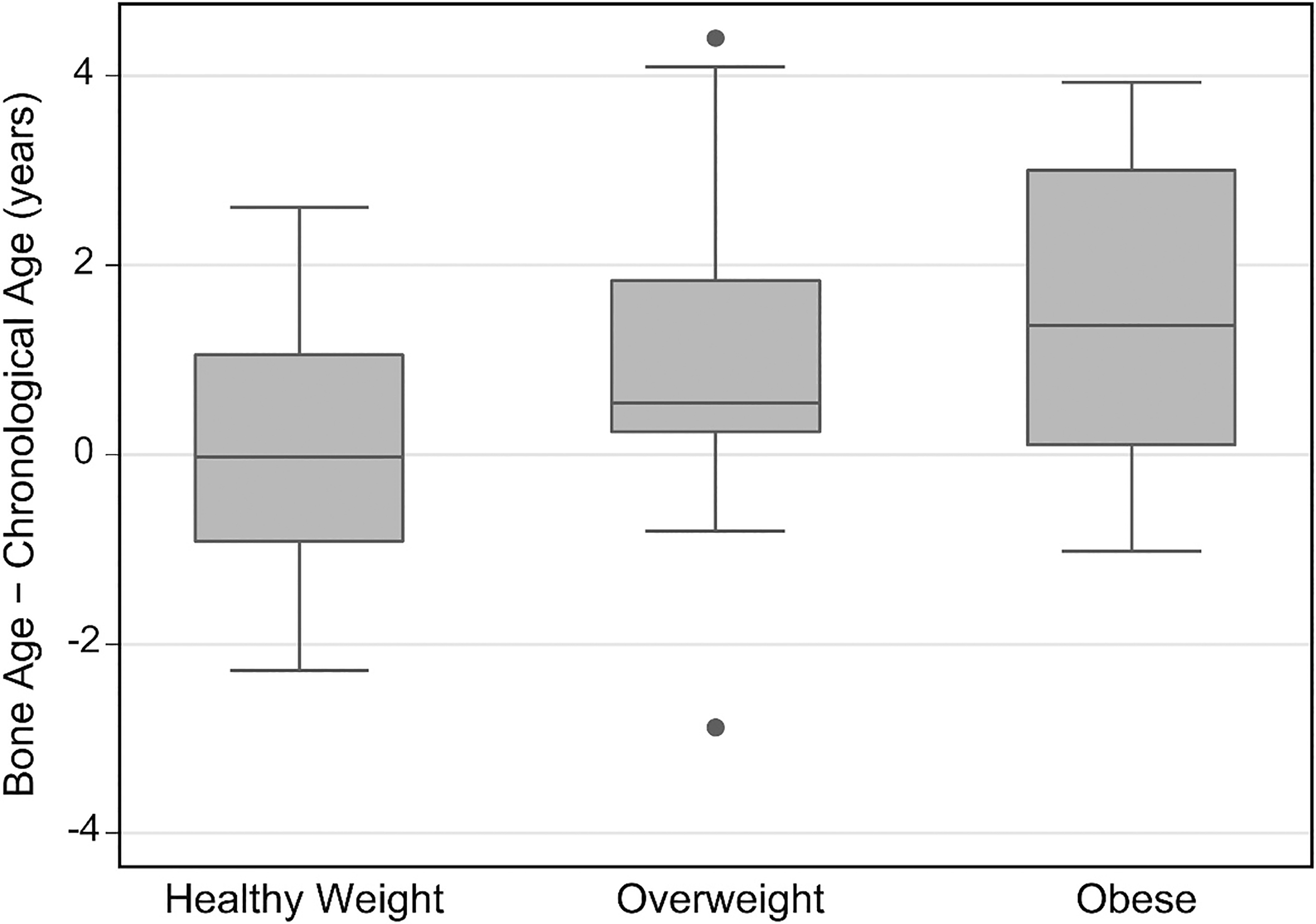
Difference between skeletal age and chronological age for participants with height
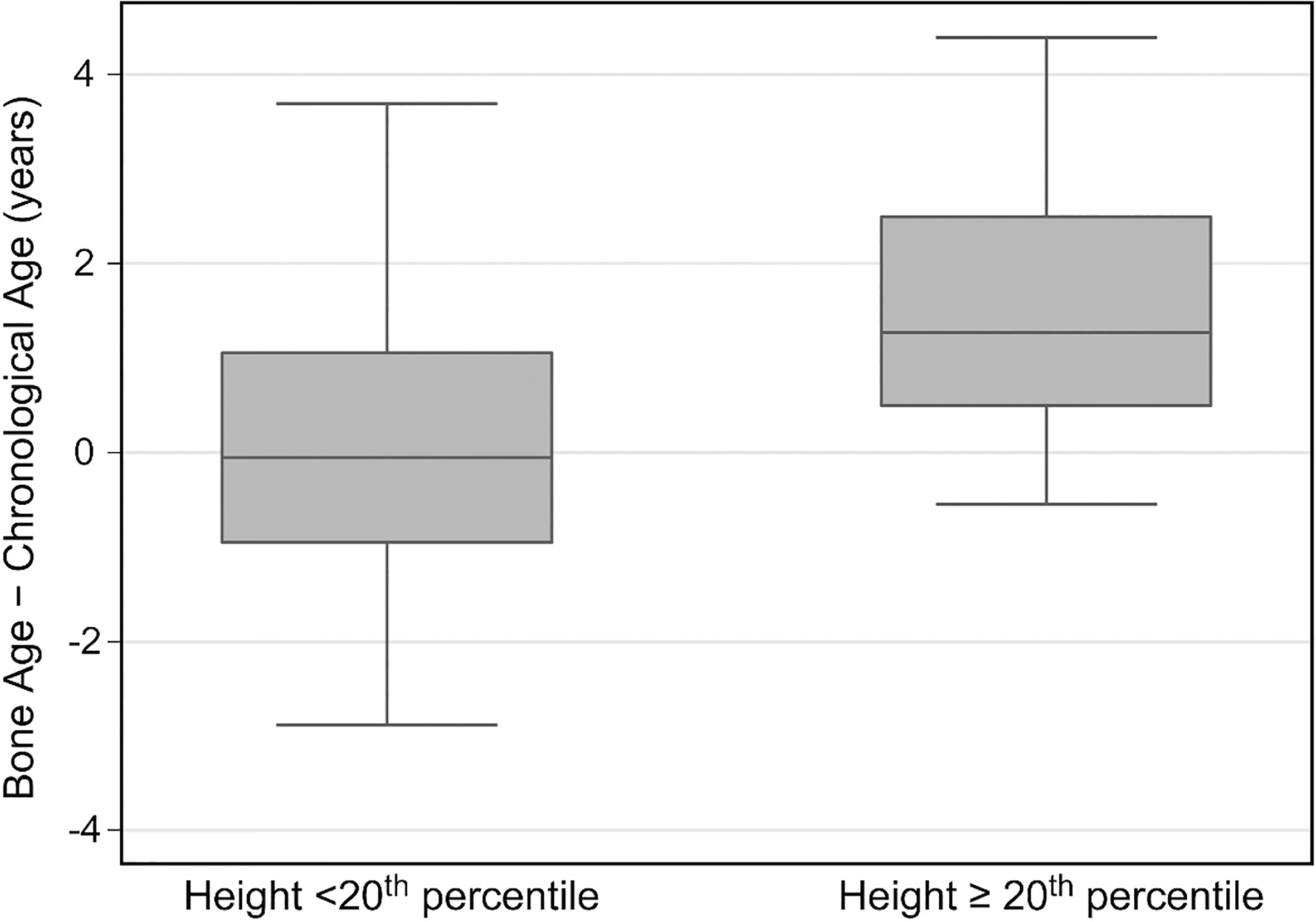
Bone age was advanced in Hispanic participants, but did not differ significantly from chronological age in non-Hispanic participants despite their having a larger median difference (Table 2, Fig. 5). Bone age was significantly advanced in the participants with a shunt, but not in those without a shunt (Table 2, Fig. 6).
Difference between skeletal age and chronological age for non-Hispanic and
Hispanic participants.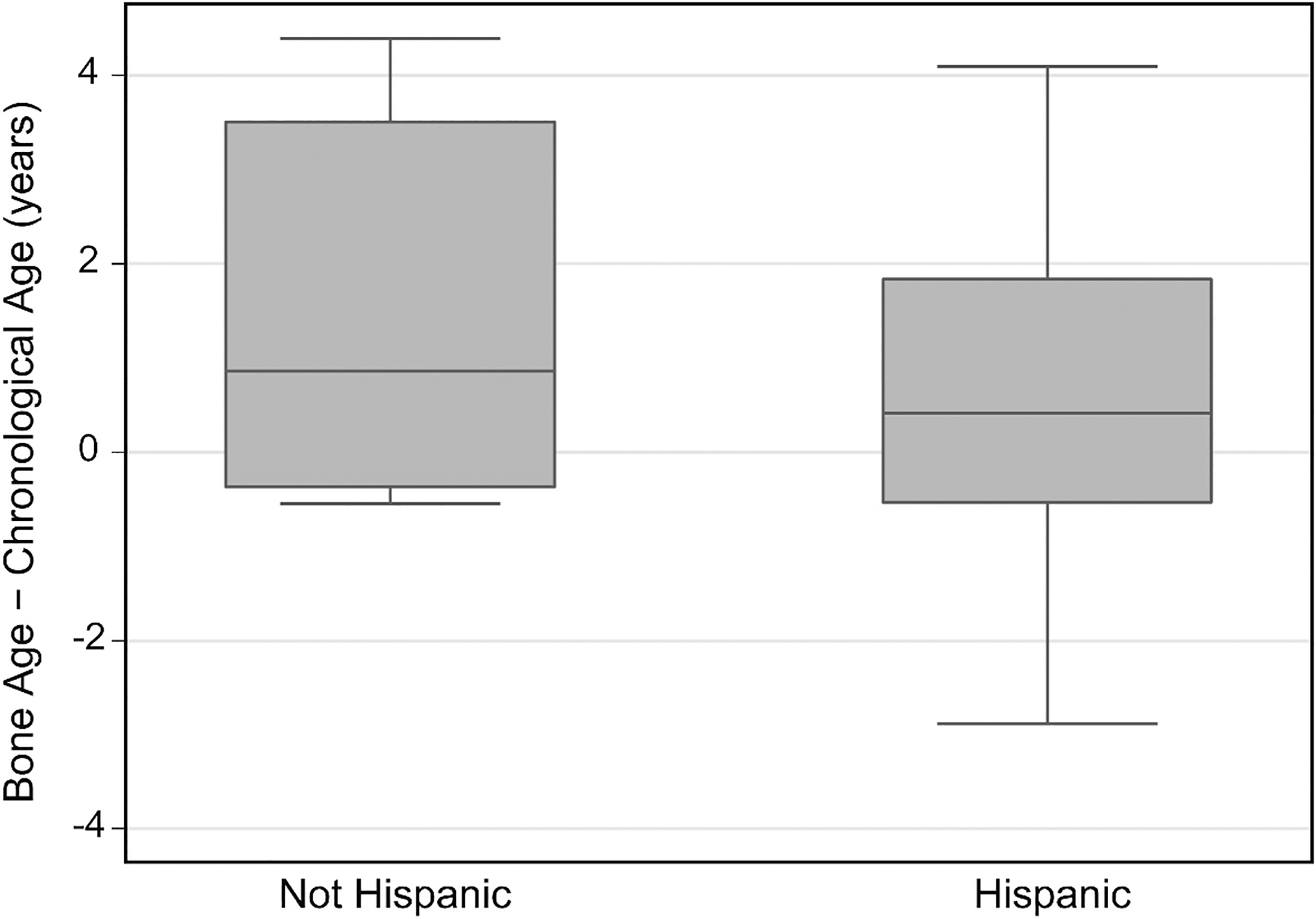
Difference between skeletal age and chronological age for participants without
and with a shunt.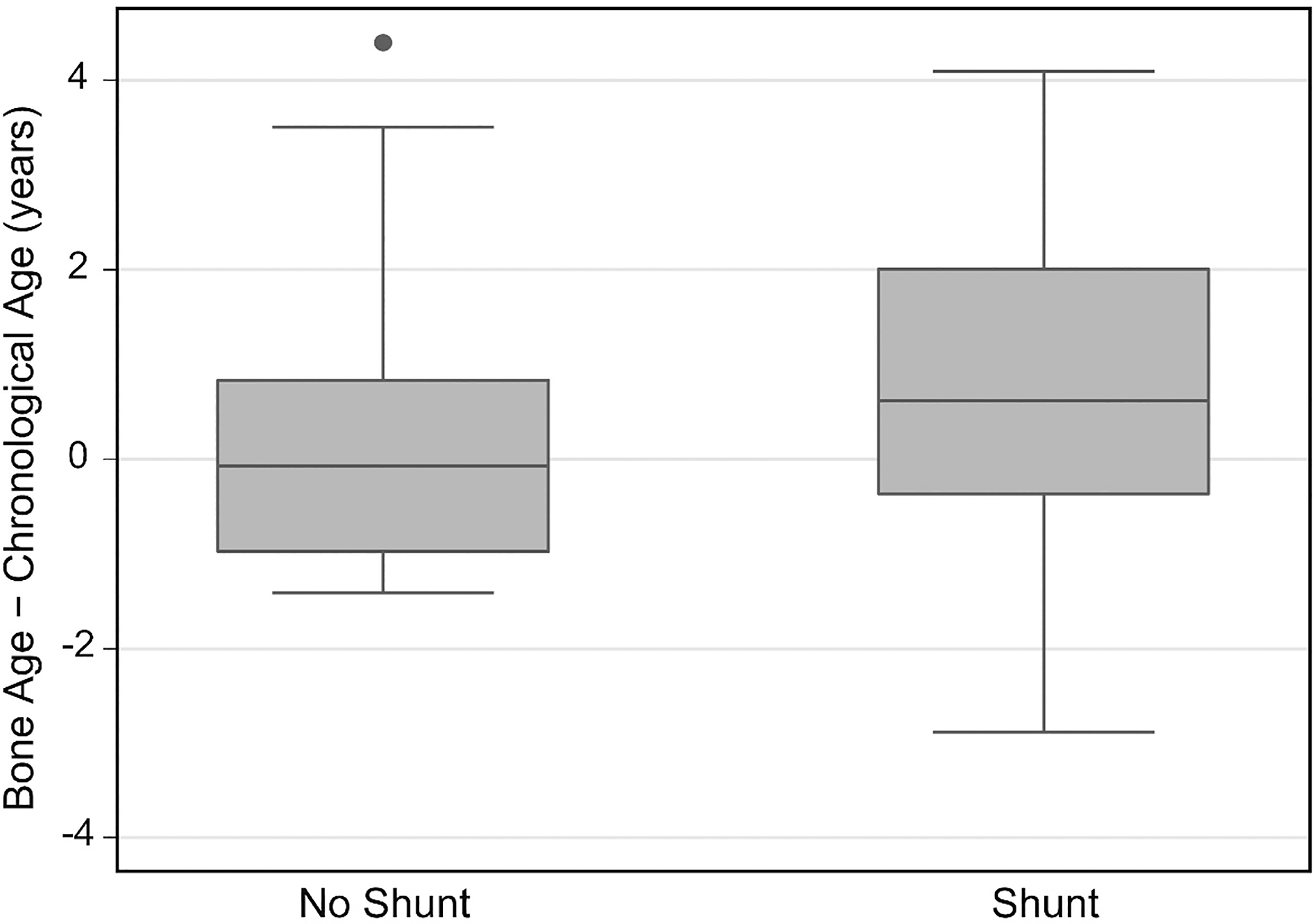
In multiple regression analysis, the difference between skeletal
and chronological age was most strongly associated with higher Tanner stage of sexual
development and higher weight, height or BMI percentile. Chronological age had a
significant effect only when Tanner stage was removed from the model. Of the
anthropometric measures, weight percentile produced the strongest model although
statistical significance was also obtained using height or BMI percentile instead
(
Results of multiple linear regression predicting the difference between skeletal
and chronological age
Results of multiple linear regression predicting the difference between skeletal and chronological age
Coef.
For the pediatric orthopedist, knowing a patient’s skeletal maturity and time to growth cessation is important in the timing of many orthopedic interventions such as epiphysiodesis, hemi-epiphysiodesis, limb lengthening, angular and rotational osteotomies and spinal fusion [25, 26]. In fracture management, knowing the amount of skeletal growth remaining aides decision making as to an acceptable amount of angulation in fracture alignment that will remodel [27]. Bone age is critical to decide on the timing of growth modulation in the axial and appendicular skeleton to correct angular deformity or determine how much scoliosis is likely to progress [28, 29]. Additionally, information regarding skeletal maturity would help pediatricians in monitoring growth and development as short stature and precocious puberty are common in the MM population [30]. Patients with delayed bone age and short stature but normal pubertal development might be referred to endocrinology for evaluation of possible growth hormone deficiency. Patients with advanced bone age and advanced pubertal development might be referred to endocrinology for evaluation of precocious puberty and possible GnRH analogue therapy.
Consistent with prior studies, the current study found a significant difference between chronologic age and bone age in children with MM. It was observed that bone age changed from being delayed to advanced at about 8 years of age for both girls and boys, consistent with prior work [6, 8]. While the median discrepancy between skeletal age and chronological age was approximately 6 months, discrepancies as great as 4.4 years were observed. The underlying reason for the acceleration of skeletal maturation around 8 years of age cannot be fully ascertained from this study; however, the timing is synchronous with the typical onset of sexual development in the MM population and suggests a premature reactivation of the hypothalamic-pituitary-gonadal axis [31, 32]. For clinicians caring for the children with MM, this would be an appropriate age to evaluate skeletal maturity and contemplate the timing of interventions such as growth modulation or scoliosis correction that may be reliant on time to growth cessation.
In univariate analysis, advanced bone age was found in the children with a shunt, but not in those without a shunt. This finding is contrary to past research reported by Kalen and Harding [6] and Feeley et al. [5]. Kalen and Harding found no relationship between shunt presence and bone age while Feeley et al. found that the presence of a shunt correlated with a younger bone age. In the present study, most participants (62 of 78) had shunts; additionally, the current study assessed the difference between bone age and chronological age within individuals, while the previous studies assessed group differences in bone age. Moreover, the relationship between shunt presence and age discrepancy in the current study was modulated by other variables, as it was no longer observed once Tanner stage and anthropometrics were considered. As prior studies have shown that participants with shunted hydrocephalus have lower levels of growth hormones (e.g., insulin-like growth factor-1, growth hormone, binding protein 3) [33], measuring hormone levels in this population might reveal if their hypothalamic-pituitary axis were dysfunctional and a possible source of abnormalities of skeletal maturation [32].
In the spina bifida population, short stature is common, and excess weight is
more prevalent than in the typically developing population [9, 34]. The current study
found a relationship between higher weight or BMI and advanced bone age. This is consistent
with prior studies in the typically developing population, which have shown advanced bone
age with increased BMI [12]. Knowing that
increased BMI is associated with advanced skeletal maturity in MM is valuable because
obesity is modifiable through physical activity [3]. Decreased adiposity could affect maturation since production of hormones,
including estradiol and leptin, by adipose tissue may stimulate early maturation in people
with increased BMI [12]. Incongruent bone age was
not observed in participants below the 20
Tanner stage of sexual development was positively related to advanced bone age consistent with prior studies in typically developing children that have shown a correlation between pubertal development and skeletal maturation [35, 36]. In the cerebral palsy population, which also has discrepancies between chronologic age and skeletal maturation, Henderson et al. showed a relationship between Tanner stage and skeletal maturation [37]. To our knowledge, the current study is the first to show a similar relationship in MM. The increase in discrepancy between chronologic age and skeletal age as the participants progressed through puberty may suggest an uninhibited hormonal positive feedback loop controlling skeletal maturation in the MM population [38]. Studies in typically developing children have reported that increases in gonadotropin hormones and pubertal progression correlate with advances in bone age [36].
No effect of Hispanic ethnicity was observed in the multiple linear regression analysis. Previous studies in typically developing children have reported inconsistent results regarding the relationship between Hispanic ethnicity and bone age. Zhang et al. and Ontell et al. found advanced bone age in Hispanics around the time of adolescence and puberty, while Mansourvar et al. found normal in bone age in Hispanics [15, 39, 40]. The relationship between skeletal maturity and Hispanic ethnicity may be of particular importance for those with MM as the Hispanic population has a higher prevalence of MM [41]. Additional investigations are needed to fully understand if a relationship between ethnicity and bone age exists and to what extent.
Limitations
This study has several limitations. First, the study sample was skewed with the majority of participants having lesions at the mid-lumbar level and below, and is therefore not representative of the general MM population. Additionally, most participants in this study were ambulatory, with only nine being non-ambulatory. Qualitatively, similar results between ambulatory and non-ambulatory participants were found, but the study was not powered to examine statistical differences between ambulatory and non-ambulatory participants. However, it should be noted that previous studies have found no relationship between neurosegmental level and bone age in children with spina bifida [5, 6] although lack of ambulation is strongly related to low bone density [18, 42]. Additionally, the standard height measure used and the resulting BMI may not be ideal for all children with MM. Standing or supine height may underestimate true height in the presence of joint contracture or scoliosis. Unfortunately, in this study alternative measures of height such as segmental measurement or arm span were not used. Due to this study’s high proportion of Hispanic participants the findings may not be generalizable to non-Hispanic youth with MM although there is a higher prevalence of spina bifida in the Hispanic population [41, 43], which is reflected in the current study’s sample. A larger multicenter study could also enable a larger sample size along with confirming generalizability of the study results.
Conclusions
In conclusion, in the MM population advanced skeletal maturation is common in children and adolescents over 8 years of age who have reached puberty, particularly those who are overweight or obese. Being cognizant that skeletal age can be advanced by as much as 4 years or more when compared to chronologic age may lead the orthopedic surgeon to investigate a patient’s bone age sooner when planning for growth modulation or spinal arthrodesis. The combination of physical examination, bone age and BMI data would greatly assist the pediatrician in clinical decision making, specifically regarding possible use of hormone therapies to treat precocious puberty and to obtain pediatric endocrinology consultation where most beneficial.
Footnotes
Acknowledgments
Support provided by NIH-NICHD Grant # 5R01HD 059826 from the National Institutes of Health – Eunice Kennedy Shriver National Institute of Child Health and Human Development.
Conflict of interest
The authors have no conflicts of interest to disclose.
