Abstract
PURPOSE:
Most patients with spina bifida require ventriculoperitoneal (VP) shunt placement. Some also require bladder augmentation, which may increase the risk of VP shunt malfunction and/or failure. The aim of this study was to assess whether bladder augmentation affects the rate of VP shunt failure in this population.
METHODS:
Using the Pediatric Health Information System, we studied patients with spina bifida born between 1992 and 2014 who underwent VP shunt placement. Using conditional logistic regression, we compared age- and hospital-matched patients who did and did not undergo a bladder augmentation to determine their difference in rates of VP shunt failure.
RESULTS:
There were 4192 patients with spina bifida who underwent both surgical closure and VP
shunt placement. Of these, 203 patients with bladder augmentation could be matched to
593 patients without bladder augmentation. VP shunt failure occurred within 2 years in
7.7% of patients, the majority of whom were in the group who underwent bladder
augmentation (87%). After adjusting for confounders, undergoing bladder augmentation was
independently associated with VP shunt failure (HR: 33.5, 95% CI: 13.15–85.44,
CONCLUSION:
Bladder augmentation appears to be associated with VP shunt failure. Additional studies are necessary to better define this relationship and identify risk-reduction techniques.
Introduction
Spina bifida without anencephaly is estimated to occur at a prevalence of 3.5 per 10,000 live births in the United States, with an estimated number of new cases per year approximating 1460 [1]. It is well established that children with spina bifida are at an increased risk for renal injury [2]. Although most children born with spina bifida have normal renal function, a systematic review of adult patients with myelomeningocele reported that 25.7% (range 3–81.8%) had some degree of renal damage, with 1.3% having end stage renal disease [3]. The neurologic defect associated with spina bifida often leads to varying degrees of bladder dysfunction. It is this potential dysfunction with the child’s ability to store and eliminate urine that leads to renal morbidity. The ultimate goal of every urologist evaluating a newborn with spina bifida is to prevent or at least delay the development of renal injury. In some cases, bladder augmentation is necessary in order to increase the capacity and compliance of the bladder.
Many patients with spina bifida also require ventriculoperitoneal (VP) shunt placement for hydrocephalus. VP shunt placement is associated with a substantial burden on patients and healthcare systems due to the high rates of complications and/or necessary revisions [4, 5, 6, 7]. Studies of patients with bladder augmentation and VP shunts report that bladder augmentation is not associated with an increased risk of infection in the prostheses [8, 9]. These series, however, were single institutional studies. We do not yet know whether bladder augmentation affects the rate of VP shunt failure in this patient population. The aim of this study was to assess whether bladder augmentation is associated with the rate of VP shunt failure in children with spina bifida.
Methods
Data source and cohort development
We performed a multi-institutional retrospective cohort study using data
from the Pediatric Health Information System (PHIS), a large administrative database that
includes discharge data for inpatient, ambulatory surgery, emergency department, and
observation unit visits at 49 tertiary children’s hospitals in the United States. These
hospitals are affiliated with the Children’s Hospital Association (Overland Park, KS).
Data quality and reliability are assured through a joint effort between the Children’s
Hospital Association and participating hospitals. Portions of the data submission and data
quality processes for the PHIS database are managed by Truven Health Analytics (Ann Arbor,
MI). For the purposes of external benchmarking, participating hospitals provide
discharge/encounter data including demographics, diagnoses, and procedures. Nearly all of
these hospitals also submit resource utilization data (e.g. pharmaceuticals, imaging, and
laboratory) into PHIS. Data are de-identified at the time of data submission, and data are
subjected to a number of reliability and validity checks before being included in the
database. For this study, we used International Classification of Diseases,
9
Patient and hospital-level characteristics
We compared age and hospital-matched patients who did and did not undergo bladder augmentation (ICD-9-CM procedure code 57.87). The group of patients who did not undergo a bladder augmentation included only eligible age and hospital matches to the patients who had a bladder augmentation; eligible meant that the potential match had a birth date within 6 months of the patient who received bladder augmentation, was treated at the same hospital as the patient who received bladder augmentation, and did not have a bladder augmentation procedure either prior to or within two years after the date of the start of follow-up. The patient characteristics used to describe the study population included age at surgical closure of spina bifida defect, age at first VP shunt placement, birth year, number of VP shunt replacement procedures after initial placement (prior to the start of follow-up), gender, race, gestational age at birth, birth weight, primary payer at time of surgical closure of spina bifida defect, number of congenital anomalies (distinct ICD-9-CM diagnosis codes in 740–759, other than spina bifida), diagnosis of congenital renal agenesis/dysgenesis (ICD-9-CM diagnosis code 753.0), cystic kidney disease (ICD-9-CM diagnosis codes 753.1x), congenital anomalies of the heart (using the list of diagnosis codes used for risk adjustment for congenital heart surgery (RACHS) method [10]), and congenital anomalies of the respiratory system (ICD-9-CM diagnosis codes 748.x). These comorbidities were considered to be present if they were listed at the admission at which the spina bifida defect was closed or at any other hospital admission prior to the start of study follow-up.
Baseline characteristics of patients with spina bifida who underwent VP shunt
placement, with or without bladder augmentation
Baseline characteristics of patients with spina bifida who underwent VP shunt placement, with or without bladder augmentation
Data are shown as median (IQR) or N (%).
The outcome investigated was the occurrence of VP shunt replacement or revision procedures or VP shunt malfunctions (ICD-9-CM 02.4x, 02.22-02.39). The rate of these occurrences through June 2015 was examined, and the proportion of patients who had such an occurrence within two years of the start of their follow-up was also examined.
Statistical analyses
After deriving the study cohort using m:1 age-matching of bladder
augmentation patients to non-bladder augmentation patients treated at the same hospital,
characteristics of the two groups at the time of the start of follow-up were compared
using chi square tests or Fisher exact tests for categorical variables and Mann Whitney U
tests for continuous variables. The primary outcomes of time to first VP shunt
replacement, revision, or malfunction and the occurrence of any VP shunt replacement,
revision, or malfunction within two years were compared between groups, and across other
baseline characteristics of interest, using stratified Cox proportional hazards and
conditional (stratified) logistic regression models respectively, in order to account for
the matching of patients by age and hospital. In order to adjust for all measured
confounders, whether weak or strong, any baseline characteristic that was associated with
both the treatment and the outcome at
Predictors of VP shunt malfunction, revision, or replacement in patients with spina
bifida
Predictors of VP shunt malfunction, revision, or replacement in patients with spina bifida
Cohort Development. Of 7523 patients born with spina bifida, 1946 were excluded
because they did not undergo a surgical closure of their defect by day of life 4, 40
were excluded because of data quality issues (including duplicate medical record
numbers), and 88 were excluded because they were managed at a hospital that stopped
participating in PHIS. An additional 1257 patients were excluded because they did
not undergo a VP shunt procedure and 3396 were excluded because they either did not
undergo a bladder augmentation or they could not be age- and hospital-matched to a
patient who did undergo bladder augmentation. PHIS: Pediatric Health Information
System. VP: ventriculoperitoneal.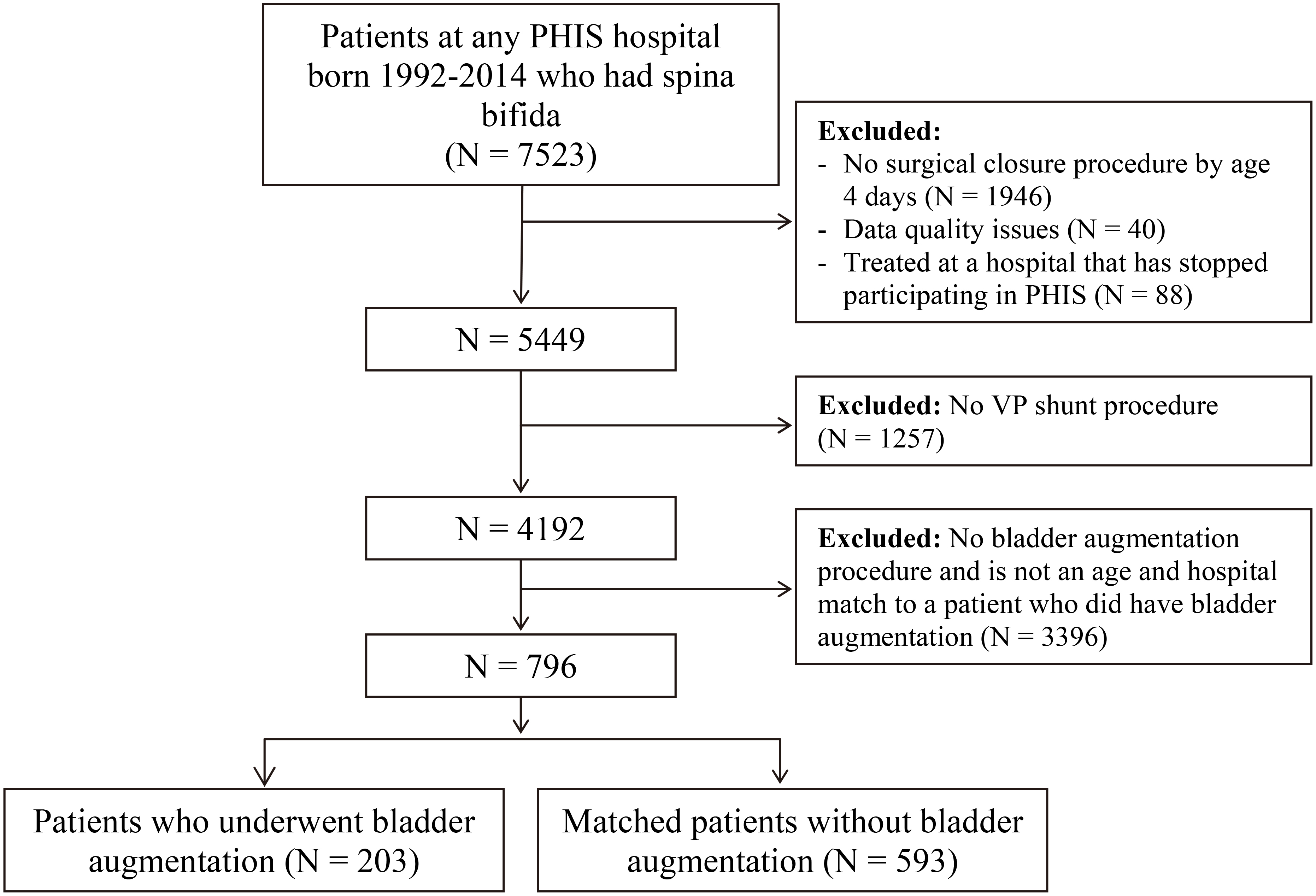
We identified 7523 patients born between 1992 and 2014 with spina bifida
(Fig. 1). Of those patients, 1946
were excluded because they did not undergo a surgical closure of their myelomeningocele by
day of life 4, 40 were excluded because of data quality issues (including duplicate medical
record numbers), and 88 were excluded because they were managed at a hospital that stopped
participating in PHIS. An additional 1257 patients were excluded because they did not
undergo a VP shunt procedure and 3396 were excluded because they either did not undergo a
bladder augmentation or they could not be age- and hospital-matched to a patient who did
undergo bladder augmentation. The final analyses included 796 patients who either underwent
bladder augmentation (
There were no differences in demographics or baseline characteristics between age- and hospital-matched patients who did and did not undergo bladder augmentation (Table 1). In our cohort, the median ages at closure of the myelomeningocele, VP shunt placement, and bladder augmentation were 1 day, 7 days, and 7 years, respectively. The age range of patients at the time of bladder augmentations was 1 to 21 years. The majority of patients were White, and approximately half were male. Most patients had at least 1 other congenital anomaly.
Predictors of VP shunt malfunction, revision, or replacement in patients with spina
bifida
Predictors of VP shunt malfunction, revision, or replacement in patients with spina bifida
Kaplan-Meier Curves for Time to VP Shunt Malfunction, Revision, or Replacement. A
univariable stratified Cox proportional hazard model suggested that bladder
augmentation patients had a much higher rate of VP shunt issues (Hazard Ratio
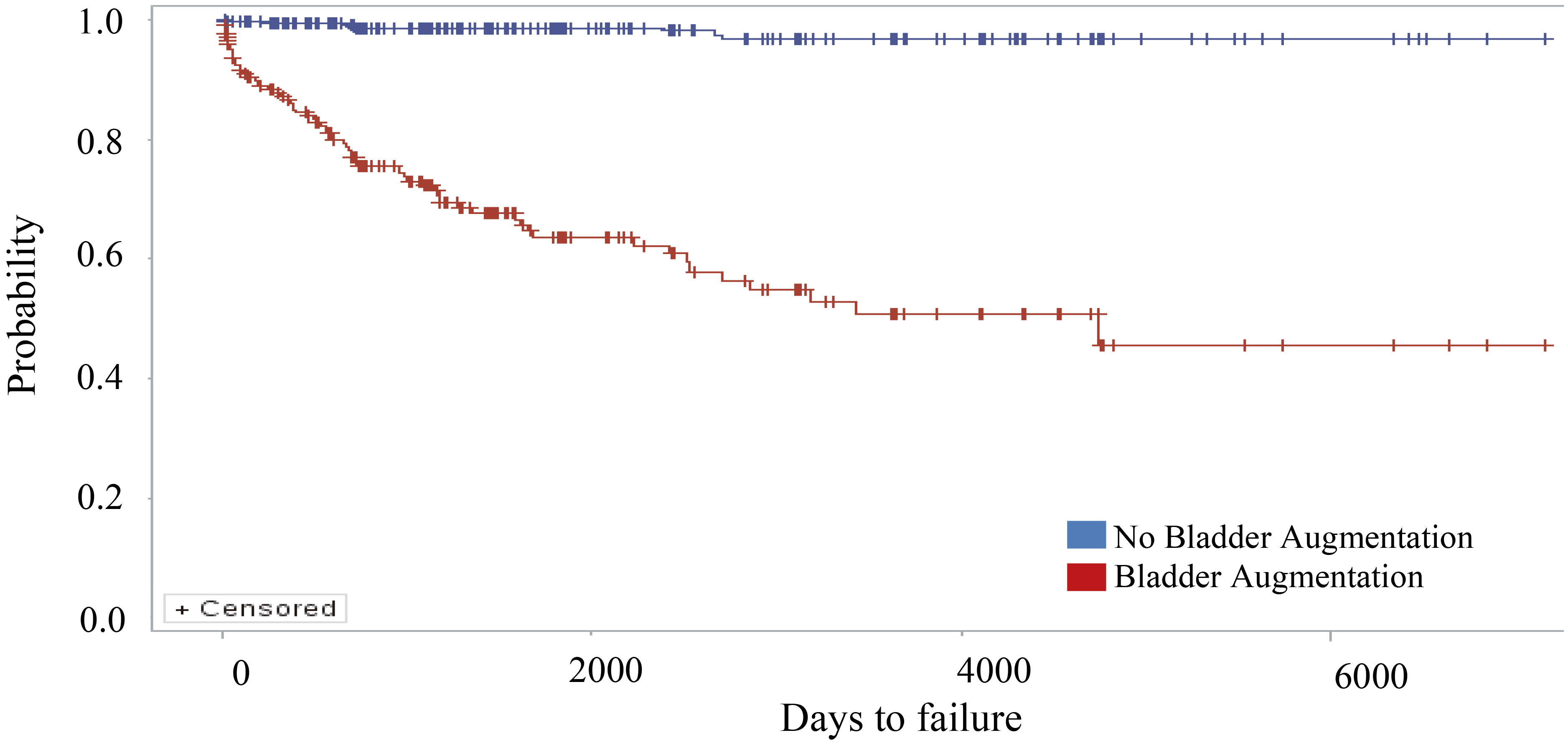
There were 597 patients who had at least 2 years of follow-up data. Of the 597
patients, 438 were in the non-augmentation group and 159 were in the augmentation group.
Among those patients, VP shunt malfunction, revision, or replacement occurred in 46 (7.7%)
patients. The majority of those patients (87%) had undergone bladder augmentation; VP shunt
issues were much more likely among patients who had undergone bladder augmentation (Odds
Ratio: 22.3, 95% Confidence Interval: 8.7–57.1,
A univariable Cox proportional hazard regression model for time to first VP
shunt problem during follow-up suggested that bladder augmentation patients had a much
higher rate of VP shunt malfunction, revision, and replacement (Fig. 2). In multivariable modeling (Table 3), bladder augmentation was still independently associated with
an increased rate of VP shunt issues (Hazard Ratio (HR): 33.5, HR Confidence Limits:
13.1–85.4,
In this retrospective review, we determined that bladder augmentation was associated with VP shunt issues, including malfunction, revision, or replacement. In our cohort, only a small number of patients developed issues with their VP shunts (7.7%), but the majority of patients who did had undergone bladder augmentation.
VP shunt placement is associated with a high complication rate, which often necessitates multiple revision procedures. In the US, the estimated cost of shunt procedures is $100 million per year. Approximately 41% of all shunt procedures are performed for malfunction and 7–13% of malfunctions are related to shunt infection [4, 5, 6]. Therefore, VP shunt failure places a substantial burden on patients and healthcare systems, as excessive costs are driven from high complication and revision rates. In patients with a VP shunt who are undergoing abdominal surgery, there is a concern that it may increase the risk of shunt failure. Specifically, after bladder augmentation, which includes intra-peritoneal dissection, bowel harvest, and a contaminated field as a result of stool spillage, the concern for a shunt failure is high.
Previous reports suggest that intra-abdominal surgery, especially bladder augmentation surgery, is associated with a low risk for VP shunt failure or infection [8, 11, 12]. This study, however, is the first to determine that bladder augmentation is predictive of shunt malfunction in a matched cohort. The potential reasons for the difference with our findings are the sample size and the duration of follow-up. Some of the series are single institutional reviews with limited numbers of patients and are underpowered. In addition, the short duration of post-operative follow-up is also a limitation. The causative organisms for early and delayed shunt infections may differ. Earlier infections are thought to be associated with gram-positive bacteria, while delayed shunt infections are associated with gram-negative bacteria [13]. Delayed shunt complications following intra-abdominal surgery due to gram negative infections are thought to arise from the development of a biofilm established at the time of surgery [13, 14, 15]. In this review, we have at least 2 year follow-up for the majority of our cohort.
Several groups have implemented risk-reduction techniques in order to reduce the risk of shunt failure in this patient population. Previously reported techniques include mechanical bowel preparation, sterilization of the patient’s urine, and externalization of the VP shunt prior to bladder augmentation surgery [13, 16, 17]. Mechanical bowel preparation was examined as a risk-reduction technique in a cohort of children undergoing bladder reconstruction [17]. In a series of 31 patients, 19 of whom did and 12 of whom did not undergo mechanical bowel preparation before reconstruction, there was no difference in the rate of shunt infections [17]. Colonization of sterile urine with bacteria at the time of bladder augmentation is a potential contributor to shunt infections. Matthews et al. report their practice of testing the urine for colonization and sensitivity prior to surgery in order to treat with appropriate antibiotics prior to surgery [13]. To our knowledge, however, this has not been investigated in either a retrospective or prospective study. In a series of 34 patients, Hayashi et al. reported their experience routinely converting VP shunts to ventriculopleural shunts prior to bladder augmentation [16]. In that series, there were no cases of shunt infection during a mean follow-up of 12 years [16]. The most common complication in that cohort was a pleural effusion, which occurred in 41% of patients.
As with any retrospective study, there are a number of limitations with this study. Patients in our cohort were identified using ICD-9-CM codes. As such, there is the potential for misclassification of patients due to miscoding within the database. Moreover, the exact indication for shunt revision or replacement is not readily available. Specifically, it is not possible to distinguish between shunt infection versus obstruction based solely on codes. In addition, there are variables that are not available for review in the PHIS database that may affect the results. For example, we were not able to assess the timing and administration of antibiotics relative to the time of surgery. In addition, in the case of a VP shunt infection, microbiology reports with the causative organism are not available for review. Also, the operative reports, which detail the indications for surgery and intraoperative findings, are also not available. We were also not able to control for other abdominal surgeries or failures that occurred at non-PHIS hospitals. The rate of VP shunt malfunction in the control group was surprisingly low, thus it is possible that patients in the control group were less likely to receive follow-up care at a children’s hospital. Finally, it is possible that some of the patients developed delayed shunt infections that were not captured within our study window. Despite these limitations, the PHIS database allowed us to perform a longitudinal study of a large cohort of patients managed at tertiary pediatric hospitals across the U.S, such that we had sufficient statistical power to demonstrate a clear association between bladder augmentation surgery and VP shunt malfunction in children with spina bifida.
In this large retrospective cohort study of children with spina bifida who have undergone VP shunt placement, approximately 7.7% of patients developed VP shunt malfunction within 2 years. After adjusting for a number of patient demographic and clinical characteristics, bladder augmentation appears to be associated with VP shunt failure. Identifying factors associated with shunt failure may allow for the development of interventions to decrease failure rates. Additional studies are necessary to better define this relationship and identify risk-reduction techniques.
Footnotes
Conflict of interest
None to report. None of the authors have any personal or financial conflicts of interest. The authors report no proprietary or commercial interest in any product mentioned or concept discussed in this article. This project is solely supported by intramural funding from the Section of Pediatric Urology at Nationwide Children’s Hospital and the Center for Surgical Outcomes Research at The Research Institute at Nationwide Children’s Hospital.
