Abstract
BACKGROUND:
Following a severe acquired brain injury, neuro-orthopaedic disorders are commonplace. While these disorders can impact patients’ functional recovery and quality of life, little is known regarding the assessment, management and treatment of neuro-orthopaedic disorders in patients with disorders of consciousness (DoC).
OBJECTIVE:
To describe neuro-orthopaedic disorders in the context of DoC and provide insights on their management and treatment.
METHODS:
A review of the literature was conducted focusing on neuro-orthopaedic disorders in patients with prolonged DoC.
RESULTS:
Few studies have investigated the prevalence of spastic paresis in patients with prolonged DoC, which is extremely high, as well as its correlation with pain. Pilot studies exploring the effects of pharmacological treatments and physical therapy show encouraging results yet have limited efficacy. Other neuro-orthopaedic disorders, such as heterotopic ossification, are still poorly investigated.
CONCLUSION:
The literature of neuro-orthopaedic disorders in patients with prolonged DoC remains scarce, mainly focusing on spastic paresis. We recommend treating neuro-orthopaedic disorders in their early phases to prevent complications such as pain and improve patients’ recovery. Additionally, this approach could enhance patients’ ability to behaviourally demonstrate signs of consciousness, especially in the context of covert awareness.
Keywords
Introduction
Spastic paresis
About a third of patients who suffer from a stroke or a traumatic brain injury will develop upper motor neuron syndrome potentially leading to spastic paresis (Urban et al., 2010; Wissel et al., 2010) and other neuro-orthopedic disorders. This syndrome can occur following any central nervous system lesion involving the corticospinal and parapyramidal tracts along the cortex, brainstem and spinal cord. Spastic paresis includes both a neurological and a muscular disorder (Gracies, 2015). The muscular disorder, called spastic myopathy, arises due to a lack of mobilisation of muscular groups placed in the short position. This will induce a cascade of molecular, biomechanical and physiological changes leading to spastic myopathy, which manifests clinically as muscular stiffness. It is important to stress that, as this disorder is due to a lack of mobilisation, it is (partially) avoidable if appropriate care is provided (Gracies, 2015). The spastic myopathy causes an increase in muscle tone without electromyographic muscle activity which (in contrast to spasticity) does not depend on the speed of mobilisation.
The second disorder is comprised of two distinct components: (1) spastic overactivity, which includes spasticity, spastic dystonia and spastic co-contraction, and (2) stretch-sensitive paresis (Gracies, 2015). Regarding spastic overactivity, the most known disorder is spasticity, which is often erroneously employed to name all mechanisms and related clinical symptoms linked to spastic paresis. A commonly accepted definition of the so-called spasticity is the clinical observation of an increase in velocity-dependent stretch reflexes in response to passive movement (Gracies, 2005b), which can be captured using scales such as the Modified Ashworth Scale (MAS) (Bohannon & Smith, 1987) or the Modified Tardieu Scale (MTS) (Boyd & Graham, 1999). Besides, spastic dystonia is described as an inappropriate muscle activation at rest, which is also sensitive to passive stretch. Another aspect is spastic co-contraction, which is the involuntary antagonist activation during agonist active mobilisation. The second component of the neurological disorder, stretch-sensitive paresis is characterised by a decrease in the central commands to the agonist muscle, leading to a reduction of the voluntary recruitment of agonist motor units (Gracies, 2015). Associated reactions can also arise, such as spastic muscle overflow, which corresponds to an inappropriate muscular activation distant from the initial muscle contraction (e.g., flexion of the elbow when standing up), or the irradiation reflex or stretch reflex (myotatic reflex) (Bhattacharyya, 2017), when a muscle is (over)extended. All these neurological phenomena will negatively impact the patient’s ability to move, thus increasing the lack of voluntary movement and risk of prolonged immobilisation. It is also important to note that the muscle disorder superimposes on the neurological disorder, exacerbating the symptoms of each. Despite the dramatic consequences of spastic paresis, the prevalence of these components in different aetiologies is still poorly investigated, and not studied at all in severely brain-injured patients with prolonged disorders of consciousness (DoC). However, a better characterisation of these different clinical manifestations in different aetiologies is fundamental to provide appropriate treatments (Thierry Deltombe et al., 2018).
The location of brain lesions, thus the aetiology, is likely crucial for the development of spastic paresis. In patients with cerebrovascular aetiology, the distribution of spasticity may involve one or both sides of the body, with the pathological flexor pattern at the upper limb, and extensor at the lower limb (Fig. 1). Patients who have suffered from an anoxic brain injury or a brainstem lesion may present mixed features of rigido-spasticity due to the dysfunction of the extrapyramidal pathways. These pathways extend from the brainstem to the basal ganglia, prefrontal and frontal brain regions and can result in pathological posture, such as decortication or decerebration (Fig. 2). Patients with traumatic aetiology may also present focal lacero-contusions and/or diffuse axonal injury, which generally consist of micro-haemorrhagic lesions, commonly affecting the white matter, the periventricular areas, corpus callosum, brainstem and cerebellum (Adams et al., 1989a). This distribution of cerebral micro-lesions may resemble the multifocal ischaemic encephalopathy and the consequent vascular parkinsonism. Other secondary parkinsonisms (Formisano et al., 2009) due to some neurosurgical complications like hydrocephalus, or meningo-encephalitis may also be associated to spastic paresis, as in post-traumatic or in post-encephalitic parkinsonism (Formisano & Zasler, 2014; Jellinger, 2004).
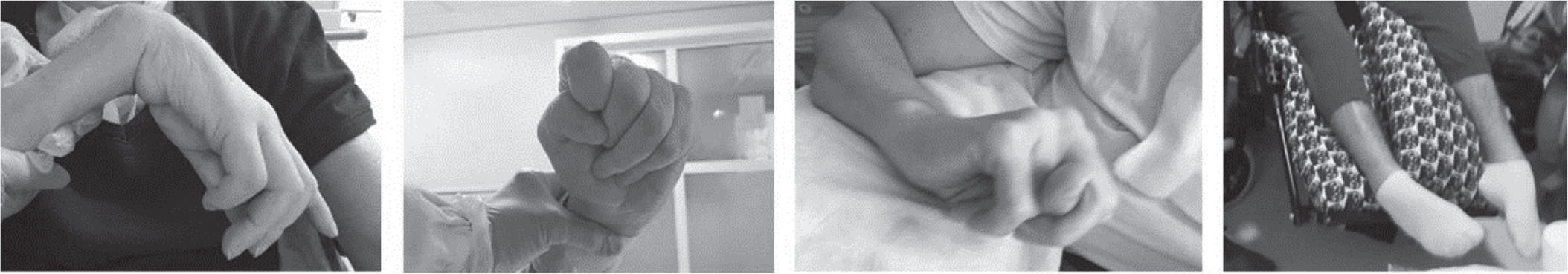
From left to right: Spastic and dystonic contractures, Klippel sign and spastic equinovarus foot.

The picture on the left represents a decortication posture, the one on the right, a decerebration posture.
Prolonged DoC include coma (no arousal and no awareness), the Vegetative State (VS)/Unresponsive Wakefulness Syndrome (UWS) meaning that the patient can display reflexive movements and eye opening without awareness of oneself or their surroundings (Laureys et al., 2010), and the Minimally Conscious State (MCS), a state in which patients show fluctuating purposeful behaviours without being able to functionally communicate (Giacino et al., 2002). Importantly, the severe disabilities present in patients with DoC integrally influence patients’ functional outcomes.
The recent European and American Guidelines (Giacino et al., 2018; Kondziella et al., 2020) focused on behavioural tools and advanced diagnostic techniques, which are recommended in patients with DoC. However, neuro-orthopaedic complications and motor disabilities were largely overlooked. These complex motor disorders can influence the diagnosis, prognosis, and therapeutic management of patients with DoC (Formisano, 2021). Indeed, the presence of severe motor disabilities may interfere with the motor output of those patients with DoC, who despite some partially preserved cognitive abilities, may feel imprisoned in their body without the possibility to interact with their environment through overt motor behaviours. This condition has been defined as cognitive-motor dissociation (Schiff, 2015), covert awareness (Owen et al., 2007), non-behavioural MCS (Stender et al., 2014), MCS* (Gosseries, Zasler, & Laureys, 2014; Thibaut et al., 2021) and functional Locked-in Syndrome (fLIS) (Bruno, Vanhaudenhuyse, Thibaut, Moonen, & Laureys, 2011; Formisano, Pistoia, & Sarà, 2011; Formisano, D’Ippolito, & Catani, 2013). However, recent reviews on covert awareness have highlighted the heterogeneity of the terms used in the literature and encouraged the unification of a common taxonomy (Schnakers et al., 2022; Zasler, Aloisi, Contrada, & Formisano, 2019). A better characterization of this state, including its motor component, is also warranted.
The main causes of severe motor disabilities in patients with DoC consist of diffuse spastic paresis (Thibaut et al., 2014; Winters et al., 2022; Zhang et al., 2021), commonly associated with pathological postures, such as decortication and decerebration (Dolce et al., 2002). Spastic paresis is often linked to pain perception and hyperpathia, which may impact patients’ quality of life (Bargellesi et al., 2018; Thibaut et al., 2014; Zasler, Formisano, & Aloisi, 2022) –see Figure 3. In this context, a thorough assessment of pain using standardised scales (such as the Nociception Coma Scale-Revised (Chatelle et al., 2012) should be systematically considered. This also leads to the conceptualisation of vicious circle involving spasticity and diffuse pain perception.
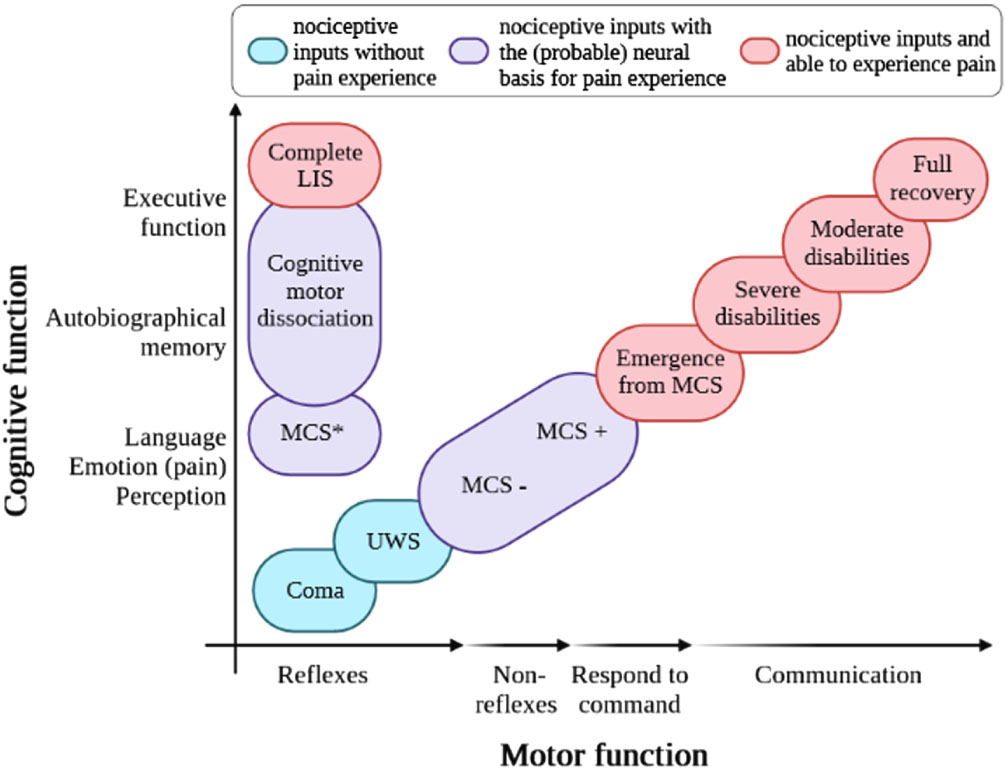
Variation in diagnosis of patients with pathological states of consciousness according to the level of recovery of cognitive and motor functions. UWS, unresponsive wakefulness syndrome; MCS, minimally conscious state; LIS, locked-in syndrome; in red, patients able to process nociceptive inputs and able to experience pain; in blue, patients processing nociceptive inputs but without evidence of pain experience; in purple, patients able to process nociceptive inputs and having the (probable) neural basis for pain experience (from Estelle A.C. Bonin et al., 2023b).
In summary, neuro-orthopaedic disorders can gravely impact DoC patients’ functional recovery, quality of life and possible diagnosis. In this narrative review, we describe the most frequent neuro-orthopaedic disorders in DoC (i.e., spastic paresis), before detailing additional symptoms and motor complications that may occur. Finally, assessments and treatment options, in the context of DoC, will also be discussed.
A literature review was conducted on PubMed using the following PubMed Advanced Search Equation: (“disorders of consciousness” OR “coma” OR “vegetative state” OR “unresponsive wakefulness syndrome” OR “minimally conscious state”) AND (“spasticity” OR “spastic paresis” OR “motor disorder” OR “hypertonicity” OR “neuro-orthopaedic disorders”). All English language studies that evaluated the prevalence of spastic paresis and/or the effect of a treatment on these disorders published in the last 20 years were included. Inclusion criteria were: (1) patients in coma, UWS/VS or MCS; (2) from traumatic or non-traumatic aetiologies; (3) spastic paresis assessed as a primary or secondary outcome and objectively reported MAS, MTS, or other related clinical scales. Given the scarcity of this topic all types of articles were considered (i.e., clinical trial, observational or open-label studies and case-reports). Additionally, a manual search within the bibliography of relevant papers and reviews was performed to complete the bibliographic search. All identified articles were screened for eligibility first based on their titles and abstracts and second, based on their full texts.
Results
PubMed database returned 181 articles wherein 158 were excluded for not matching our inclusion criteria. The remaining 23 articles are discussed in three main sections: (1) occurence; (2) assessment and (3) management of spastic paresis in DoC.
Spastic paresis in DoC
In patients with DoC, the proportion of patients suffering from spastic paresis (as measured with the MAS, thus not allowing to discriminate one symptom from another) is extremely high, ranging from 57 to 95% (Ganesh et al., 2013; Nakase-Richardson et al., 2013; Thibaut et al., 2014; Zhang et al., 2021). In addition, spasticity has been shown to be the most frequent complications, compared to dysautonomia, seizure, shunt placement or heterotopic ossification, in two large cohort cross-sectional studies on 68 and 122 patients with prolonged DoC (Ganesh et al., 2013; Nakase-Richardson et al., 2013). In another cross-sectional trial looking at the factors that could predict the severity of spastic paresis in a sample of 65 patients with prolonged DoC, no difference was found between etiologies nor diagnosis (Thibaut et al., 2014). On the other hand, it appeared that spasticity was positively correlated with time sinceinjury.
The high prevalence of spastic paresis might be linked to the extensive periods of immobilization these patients endure with very limited voluntary movements. Indeed, as mentioned, spastic paresis can be exacerbated due to disuse and immobilisation (Baude et al., 2019; Gracies, 2005a). Spastic paresis, immobilisation and muscle disuse will favour atrophy, loss of sarcomeres and accumulation of connective tissue and fat, thus enabling a positive feedback loop which limits the patient’s ability to voluntarily move. Patients with DoC often face all these situations together, which could lead to the exacerbation of spastic symptoms and further hinder the ability to express signs of consciousness, due the impairment of motor output. This hypothesis is in line with previous a finding showing that spasticity is not the only component explaining the increased MAS scores, muscle retraction and joint fixation observed in patients with DoC since no correlation between MAS scores and the Hmax/Mmax ratio (i.e., indicator of the severity of spasticity in stroke (Deltombe et al., 2018) was found in a cohort of 21 patients in prolonged DoC (Martens, Foidart-Dessalle, Laureys, & Thibaut,2017).
Regarding the evolution of spastic paresis, a recent longitudinal study including 19 patients with prolonged DoC (> 12 months post-injury), showed that both the aetiology and the diagnosis seem to influence the development of spasticity over time. Indeed, the authors found that in TBI patients, spasticity (upper limb) tended to increase in a delayed manner compared to non-TBI aetiologies (Winters et al., 2022). In addition, patients in UWS/VS demonstrated an increase in spasticity (lower limb) over a two-year period while it remained stable for patients in MCS. These preliminary findings, resulting from the first longitudinal study, highlight that the worsening of clinical signs of spasticity varies according to the type of brain lesion and the level of consciousness. However, the possible factors explaining such differences still need to bedetermined.
Finally, a link between the patients’ body mass index and the severity of spasticity has been highlighted in a recent cross-sectional study on 80 patients with prolonged DoC (Ippoliti et al., 2023). Indeed, a negative correlation was found between spasticity, as measured with the MAS and patients’ body mass index (i.e., the severity of spasticity was higher in patients with lower body mass indexes). Longitudinal studies are needed to explore the causal relationship between spasticity and nutritional status, as both impact patients’ quality oflife.
Assessment of spastic paresis
Spasticity is commonly assessed using the MAS and/or the MTS. The MAS specifically evaluates the level of resistance to passive movement but does not allow the quantification of the impact of velocity on passive movement (key component of spasticity), neither the angle of contraction outbreak nor possible tendon retraction (by means of range of motion amplitude –ROM). Indeed, this scale measures the resistance to passive movement adequately, but mainly evaluates a combination of contractures, spastic dystonia, in addition to spasticity itself (Gracies et al., 2010). On the other hand, the MTS takes into account these characteristics, thus could be considered more accurate to characterise the different components of spastic paresis (Yelnik et al., 2010). However, for patients with prolonged DoC, as most of them suffer from joint fixation to some extent (Thibaut et al., 2014), the use of the MTS is often limited as it is often not feasible to perform the assessments with the three different velocities nor the two ROM (i.e., angle of muscle reaction and full ROM). Ideally, a combination of the two scales could provide a more accurate characterisation of spastic paresis in patients with DoC. It should be noted that other scales to evaluate spastic paresis and its impact on patients’ functional outcome exist for other population of patients, such as the Patient’s Disability Scale (Brashear et al., 2002); however, they require the patient’s active participation, therefore cannot be used in patients withDoC.
Besides these two scales, electrophysiological assessments, such as electromyography, can be used to quantify spastic paresis in a more objective, sensitive and reliable manner. Several responses to electrical or mechanical stimuli exist, such as: (i) the M response (i.e., the direct motor response caused by stimulation of the alpha fibres), (ii) the Hoffmann reflex (H-reflex; allows assessment of the excitability of the alpha motor neuron), (iii) the F-wave (low amplitude wave that follows the M response) or (iv) the tendon reflex (T-reflex) (Voerman, Gregoric and Hermens, 2005; Bhattacharyya et al., 2017). The ratio of the maximal H-reflex amplitude and of the maximal M response amplitude is called Hmax/Mmax ratio, which reflects the percentage of excited motoneurons via the H-reflex in comparison to the direct activation of the motoneurons –M response (Katz et al., 1992). The Hmax/Mmax ratio can sometimes be an indicator of the severity of spasticity due partly to the hyperexcitability of the alpha motoneuron through sensitive inputs. This ratio has been shown to be correlated with the severity of spasticity in other neurological populations such as stroke (e.g., Deltombe, Jamart, Hanson, & Gustin, 2008). Recently, Martens and collaborators have investigated the correlation between the H/M ratio and the MAS scores in a cohort of 21 patients with prolonged DoC (Martens, Deltombe, Foidart-Dessalle, Laureys, & Thibaut, 2019). Interestingly, some patients with scores of 4 on the MAS (i.e., affected part in rigid flexion or extension) did not present an increased H/M ratio (i.e., ratio < 0.5), while two patients with an exaggerated ratio (i.e., ratio > 0.5) in the lower limbs, did not demonstrate any signs of spasticity (i.e., MAS = 0). These findings highlight that the Hmax/Mmax ratio does not seem to accurately reflect MAS scores in patients with prolonged DoC, supporting the fact that these patients suffer from a combination of motor disorders, which may include spasticity, muscle overactivity, spastic dystonia, spastic co-contraction, among others (Baude et al., 2019). That withstanding, there is a crucial need to better characterise the different component of spastic paresis in patients with DoC, in order to provide accurate treatmentoptions.
Management of spastic paresis
Physical therapy and rehabilitation approaches
Physical therapy (PT) is the first line treatment for patients with spastic paresis. Stretching is the most common approach used in patients with DoC, aiming to work on the viscoelastic properties of the muscle-tendon units and increase its extensibility, thus limiting the consequences of spastic myopathy. Despite its common use, to date, there is still no consensus on the optimal dosage of stretching to limit spastic paresis, especially in patients with DoC. A recent meta-analyses did not find enough evidence on its effectiveness in stroke (spasticity and range of motion (Gomez-Cuaresma et al., 2021)).
A first study looked at the effect of intensive physiotherapy including casting, splinting, standing, as well as botulinum toxin injections in 10 patients (both UWS and MCS) who remained in a rehabilitation centre for approximately 9 to 12 months (Wheatley-Smith et al., 2012). No significant improvement on spasticity nor range of motion was observed in this cohort of patients with prolonged DoC.
A cross-sectional study in 109 patients in prolonged DoC evaluated the link between spasticity (as measured with the MAS), muscle contractures and the amount of weekly PT sessions patients received (Thibaut et al., 2018b), taking into account additional covariates, such as time since injury and medication. A negative correlation was found between the frequency of PT and both MAS scores and the presence of muscle contracture. In addition, time since injury and antispastic medication seemed to negatively and positively, influence the development of muscle contractures respectively (Thibaut et al., 2018b). Based on these findings, PT appears to be beneficial to reduce (or avoid dramatic increase of) spastic paresis in patients with DoC. This is especially important since the severity of spasticity has been shown to correlate with pain levels in patients with DoC, which might affect their quality of life (Bonin et al., 2022; Thibaut et al., 2014).
Beside PT and stretching, casting represents a valuable alternative to immobilise the limb in a stretch position for several hours per day. This technique is usually used to improve muscle length, increase joint range of motion and reduce contracture pain and spasticity. However, scientific evidences of efficiency is yet to support this method to reduce spasticity in various neurological disorders (Lannin et al., 2007). In addition, this technique can induce pain if not properly applied, therefore it should be used carefully in non-communicative patients such as those with DoC.
Complementary to PT, orthoses, or splints are a valuable alternative to casting to ensure proper limb positioning. Since there are limited risks of skin lesions or other mechanical types of pain being induced, orthoses are considered as very safe and can be placed for several hours per day, without any supervision from the medical staff. Despite the lack of practical guidelines, orthoses have shown some promising effects in a pilot study in patients with prolonged DoC. In a first RCT, the effects of a soft splint positioned in patients’ hands were tested to promote finger and wrist extension, and ensure hand opening, which is crucial for cares (Thibaut et al., 2015). Seventeen patients in prolonged DoC received either 30 minutes of soft splints or 30 minutes of manual stretching of the fingers and wrist’s extensors. Both interventions improved spasticity of the finger flexors, while hand opening increased only for the splint group. Even if the long-term effects of soft splints positioned along patient’s hands still need to be confirmed, this tool represents a safe, easy to use, and promising approach to manage spasticity of the upper limbs’ extremities. It is important to consider that casting may also provoke a rebound effect once removed or sometimes induce contact ulcers (when hard material is used), often resulting in increased spasticity and secondary pain sources.
Tilt tables are also often used by physiotherapists to promote physiological standing position. This approach, applied 1 hour per day for 3 weeks, has been tested alone or in combination with a stepping device in 50 patients with prolonged DoC. The authors found an improvement in signs of consciousness in patients in prolonged DoC for the group receiving tilt table alone and no effect on spastic paresis in both groups (Krewer et al., 2015). In addition, a RCT conducted in the acute setting, tested the effect of a robotic stepping verticalization protocol compared to conventional therapy performed in the intensive care unit. They demonstrated that this verticalization protocol could enhance short-term and long-term functional and neurological outcomes of patients with DoC (Frazzitta et al., 2016). Specifically, improving responsiveness as measured with the CRS-R and reducing the orthostatic hypotension, caused by standing, thanks to the muscular activation of the stepping device.
Another RCT on 47 patients tested the effects of 16 sessions over a 4-week period of robotic verticalization with functional electrical stimulation, without functional electrical stimulation, and a control group (conventional PT) (Rosenfelder et al., 2023). All three groups improved, and no difference could be identified between the three interventions.
In conclusion, PT is essential to avoid retraction and joint fixation in DoC but, to date, there are no scientific guidelines for the application of the different therapies in patients suffering from spastic paresis. Importantly, PT might be painful for patients with DoC and appropriate courses of pain management should be investigated.
Pharmacological treatment of spasticity
While pharmacological treatments are the first line interventions to reduce spastic muscle overactivity in various neurological conditions, only a couple of studies have investigated the effects of specific drugs in patients with prolonged DoC.
Interestingly, in a first cross-sectional trial of 65 patients with prolonged DoC, those taking anti-spastic drugs (all pharmacological interventions combined) had more severe spasticity than those not treated. Although it is logical that non-spastic patients do not receive anti-spastic drugs, this shows that the treatments provided are not sufficient to control severe spasticity. On the other hand, the same study highlighted that 33% of the patients showing spasticity, were not receiving any drugs. For those being treated, the most frequent medication was oral baclofen, prescribed in half of the patients (Thibaut et al., 2014). Only 4 patients (7%) received intrathecal baclofen therapy (ITB).
In addition, a retrospective cohort study on spasticity management in a cohort of 146 patients admitted to a DoC rehabilitation program (Zhang et al., 2021) reported that baclofen was the most commonly used oral treatment for spasticity in patients with DoC (32%). Benzodiazepines were used for medical problems other than spasticity (seizure, sympathetic storm, catatonia or myoclonus). Tizanidine and Dantrolene were only occasionally used. These treatments were discontinued in a large proportion of patients, which was correlated with an improvement of their state of consciousness.
Local injections (botulinum toxin or phenol neurolysis) were administered to the majority of a cohort of patients with DoC, in which, 56.9% received botulinum toxin and 51.4% received phenol, while some patients had both types of injection. The most frequent targets were the upper limb and neck muscles (Zhang et al., 2021).
Regarding intratecal baclofen (ITB), the majority of clinical case reports in patients with DoC show a significant decrease in their MAS scores after a few months of treatment (Martens et al., 2017). In the study by Zangh et al, a third of patients (52/146) had an ITB trial with a 92,3% positive rate, and 70.8% of them were finally implanted. Other open-label studies on smaller samples reported similar improvement on spastic paresis severity in patients with prolonged DoC (Halbmayer et al., 2022; Margetis et al., 2014). It is worth noting that ITB has been shown to be linked to consciousness improvement in some open-label studies (Formisano et al., 2019; Sara, Pistoia, Mura, Onorati, & Govoni, 2009).
Interestingly, a case-report described an important reduction of spastic paresis of the upper limb in a patient in UWS who received levetiracetam, initially prescribed to treat seizures. The patient had received physical therapy, oral baclofen and botulinum toxin injection, which did not improve upper limb motor function (Pingue et al., 2020). The underlying mechanisms of levetiracetam on muscle tone reduction still need to be determined.
Novel therapies
Non-invasive brain stimulation approaches, such as transcranial current stimulation (tCS) and repetitive magnetic stimulation (rTMS) have been widely studied in the literature to promote motor function. Some studies, have also looked at its effects on hypertonia, especially in stroke (Alashram et al., 2023). In DoC, one RCT tested the effect of a single session of tCS (or sham) on spasticity (MAS), patients’ responsiveness (CRS-R) and brain activity (EEG at rest) (Thibaut, Piarulli, Martens, Chatelle, & Laureys, 2019) in 14 post-coma patients. Using a multichannel device, the anodes (excitatory electrodes) were placed over the prefrontal cortex (F3 & F4), the cathodes (inhibitory electrodes) were positioned on the primary motor cortex (C3 & C4) bilaterally. While no effect was found on the CRS-R, a reduction of spasticity was identified for the finger flexors right after the stimulation in addition to an increased connectivity in the beta band in the active compared to the sham group. This first pilot study highlights the potential benefits of tCS to reduce spastic paresis in patients with DoC.
The spinal cord can also be targeted to reduce spastic paresis. A pilot open label study evaluated the effect of epidural spinal cord stimulation targeting the cervical spine in 21 patients in prolonged DoC (Vorobyev et al., 2023). In addition to an improvement of signs of consciousness in some patients, almost half of them presented a reduction of spasticity severity.
Deep brain stimulation (DBS) targeting the central thalamus nuclei, known for its effects on the recovery of consciousness in a few case-reports and case-series studies (Vanhoecke & Hariz, 2017), was also associated with a reduction of the severity of spastic paresis in a recent multimodal case report (Arnts et al., 2022) and in a retrospective study conducted in a large cohort of patients with prolonged DoC (29 UWS and 11 MCS) (Magrassi et al., 2016).
Finally, acupuncture applied for a single session of 10 minutes enhanced the F-wave/H-wave ratio in patients with DoC compared to a controlled condition (Matsumoto-Miyazaki et al., 2016). However, the authors did not collect additional scales such as the MAS or the TMS to compare their neurophysiological findings with clinical outcome.
Spastic paresis and pain
In several large cross-section studies, it has been shown consistently that spasticity is positively correlated with pain in patients with DoC (for a systematic review see: (Martens et al., 2017)), especially in case of severe spasticity of the upper limb extremities (wrist and fingers’ flexors) and the hip adductors (Bonin et al., 2022), which are usually mobilized and stretched during a PT session. Although daily PT sessions might be efficient to manage spastic paresis in patients with DoC (Thibaut et al., 2018a), a recent clinical trial on 18 patients with prolonged DoC highlighted that patients presented higher pain scores during PT compared to baseline (i.e., tactile stimulation). More importantly, these scores during PT did not differ significantly compared to those observed during the nociceptive stimulation (i.e., pressure on the nailbed) (Bonin et al., 2022) –see Figure 4. Based on these results, appropriate pain management, especially before mobilizations and PT sessions should be provided to ensure patients’ comfort.
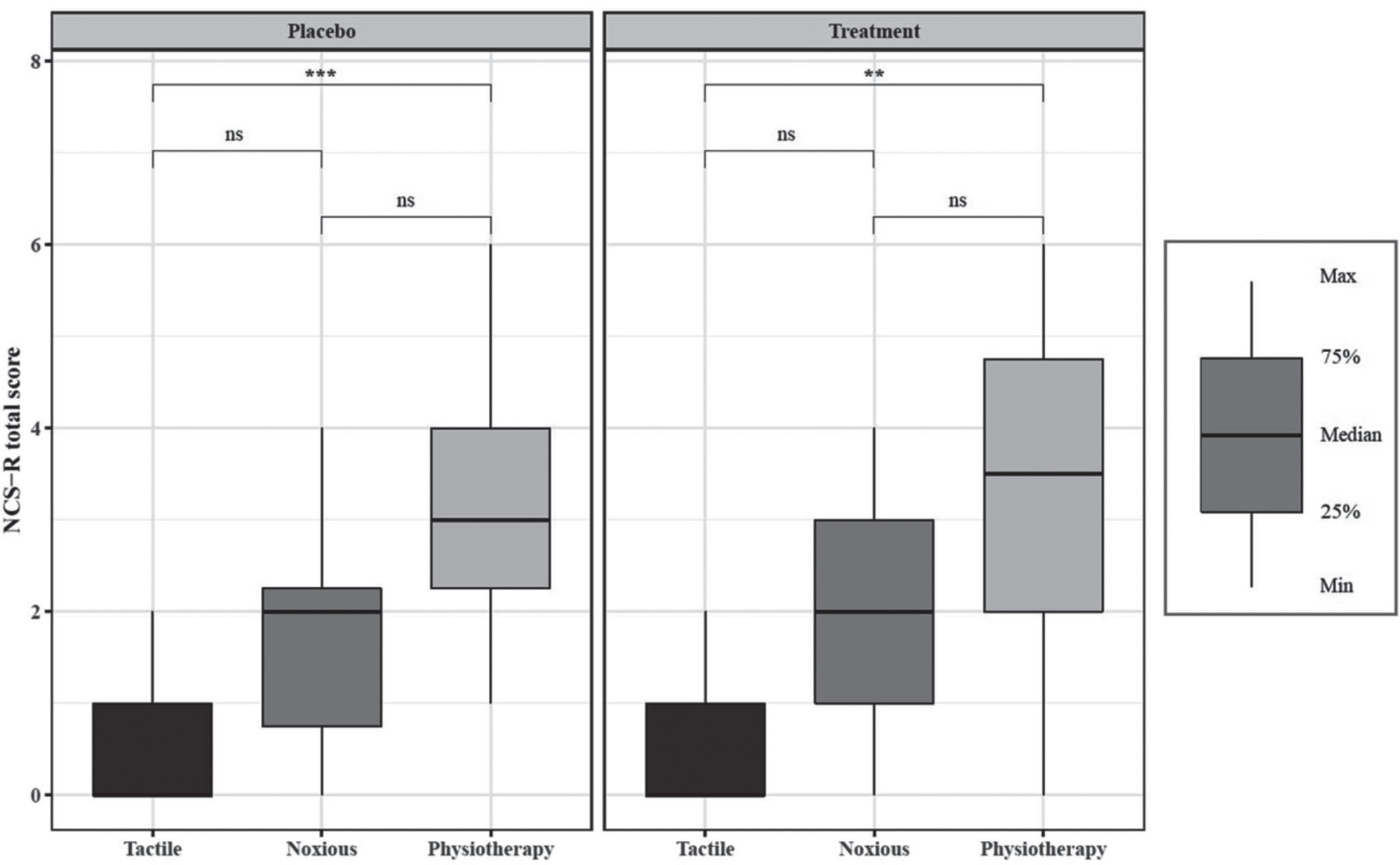
Changes in the NCS-R total score after tactile stimulation, noxious stimulation, and physiotherapy during the administration of a placebo (left panel) or an antalgic treatment (right panel). While analgesic treatment did not reduce pain levels, the scores on the NCS-R were significantly higher during physical therapy than the other two conditions. NCS-R, Nociception Coma Scale-Revised; ns, no significance, **p < 0.01, ***p < 0.001). Taken from (Estelle A.C. Bonin et al., 2022).
While it is well-known that spastic paresis can influence a patient’s quality of life and interfere with their rehabilitation, the current literature shows an alarmingly high incidence of spastic paresis in patients with prolonged DoC (Martens et al., 2017; Zhang et al., 2021). Since these patients are already limited in their range of movement, spastic paresis constitutes one of the most important disabling factors to be treated. In addition, chronic inactivity may enhance the severity of spastic paresis and the various other side-effects (e.g., ankylose, tendon retraction or joint fixation). This is in line with findings showing a positive correlation between severity of spasticity and time since brain injury (Thibaut et al., 2014). Moreover, disuse and paresis result in a decrease of cortical excitability of the motor cortical areas coupled with a decrease in motor representation of the immobilised body parts (Kaneko et al., 2003), which is especially important in the context of cognitive motor dissociation (Schiff, 2015). In conclusion, paresis and spasticity form a positive feedback loop which is exacerbated by prolonged immobilisation. It is, therefore, of upmost importance to break this cycle by managing these motor disorders from the first time the signs appear (a few days or weeks after the brain lesion) and by ensuring intensive mobilisation of the patient’s limb, even if no active movement is yet possible. In other words, efforts should be made to increase the intensity of mobilisations on the upper and lower limbs using, for example, a motorised movement trainer as soon as possible following brain injury.
Regarding pharmacological treatment, ITB have shown promising effects on spastic paresis as well as consciousness recovery, which makes this approach a treatment of choice for patients with prolonged DoC who often suffer from severe and generalized spastic paresis over the four limbs. Nevertheless, there is still a lack of controlled studies to confirm these benefits both on motor function and signs of consciousness. It is also crucial to stress that appropriate follow-up is primordial as serious side-effect can occur. Additional pharmacological approaches such as botulinum toxin injection seem to be commonly used in some institutions. However, the exact benefit and contra-indication as well as side effects specific to patients with DoC still need to be determined.
It is also important to stress that spasticity or spastic paresis has been shown to be associated with pain both in patients with DoC (Thibaut et al., 2014) and other neurological disorders such as stroke or multiple sclerosis (Van Schayck & Weiller, 2002). Therefore, the assessment and management of pain cannot be distinguished from the assessment and management of spastic paresis. Specific pain-related behavioural tools such as the Nociception Coma Scale-Revised (Chatelle et al., 2012) were developed and validated for patients with DoC. More recently, the use of a personalized painful stimulation has been proposed to be more sensitive compared to the standard painful stimulation consisting of the fingernail bed pressure (Formisano et al., 2020). Finally, while a lot of effort has been made to identify how patients with DoC experience pain, it has been suggested that (repeated) nociceptive stimuli, even without inducing a painful experience, could have dramatic physiological effects on the autonomous nervous system and should probably be taken into account as an independent factor (Bonin et al., 2023). Further studies aiming at identifying the consequences of such chronic nociceptive activity in patients with a DoC suffering from medical complications such as spastic paresis are mandatory, also for ethical reasons (Zasler et al., 2022). In this context, it is crucial to highlight the importance of treating any DoC patient equally with regard to their potential pain. This is especially pertinent in situations concerning nutrition and hydration withdrawal (Formisano & Zasler, 2020).
Beside spastic paresis, additional neuro-orthopaedic disorders may arise, however, the literature is particularly scarce on this topic. Other motor disorders such as pathological and dystonic postures, secondary parkinsonism and cerebellar syndrome may occur and interfere with overt behavioural responsiveness and impact the recovery of functional communication of patients with DoC.
Secondary parkinsonism may be present in patients with DoC, after severe TBI, but also after diffuse vascular or encephalitic brain damage or associated with hydrocephalus (Formisano & Zasler, 2014). This syndrome includes hypomimia, bradykinesia, sialorrhea and rigidity as in akinetic mutism, that is a condition, first described as secondary to diencephalic lesions, consisting of severe quadriparesis, mutism, akinesia, visual fixation and pursuit. It includes the complete or near-complete loss of spontaneity and initiation, such that action, ideation, speech and emotion are uniformly reduced, which could impact DoC patients’ ability to demonstrate signs of consciousness (Giacino, 1997, Formisano, D’Ippolito, et al., 2011).
Post-coma cerebellar syndrome may also be observed in brain injured patients, with clinical manifestations such as intention tremor, dysmetria, myoclonic jerks, and muscular hypotonia (Marks, 1993), which are often present in patients with DoC. However, to date, very few studies are available on the incidence and prevalence of post-traumatic cerebellar syndrome in patients with severe acquired brain injury. For patients with DoC, a cerebello-pyramidal syndrome may be diagnosed after brainstem or cerebellar damage, as well as after diffuse axonal injury (Adams et al., 1989b), but some of their clinical manifestations (e.g., intention tremor) only arise when patients emerged from MCS or can wilfully perform limb movements. Regarding myoclonic jerks, they have been reported in patients with DoC as a negative prognostic factor (Dolce et al., 2002). Additionally, a recent study found that 61% of patients with acute DoC had critical illness polyneuropathy and myopathy diagnosed with electroneurography-electromyography (Hakiki et al., 2022), which could worsen the severity of motor deficit, associated with diffuse muscle hypotrophy and neuropathic pain.
Moreover, the prolonged bed-rest syndrome (Scarponi & Zampolini, 2018), together with central and peripheral extreme motor deficits may be associated with neuro-orthopaedic complications, such as peri-articular ossification, also defined as neurogenic heterotopic ossification (NHO) (Bargellesi et al., 2018; Ippolito, Formisano, Caterini, et al., 1999; Ippolito, Formisano, Farsetti, et al., 1999), is often associated with tendon retractions, deformity and ankyloses (Mazzoli et al., 2019). In a recent multicentre observational study, 11.2% of a population of patients with DoC presented NHO, which were independently associated with functional disability, bone fractures and spasticity (Estraneo et al., 2021). In this latter study, NHO was also associated with a diagnosis of VS/UWS and traumatic aetiology. In another multicentre cross-sectional survey in post-acute rehabilitation setting, NHO involved one or more joints in 13.6% of the severe acquired brain injury patients, with a significant higher prevalence in young males, without any difference in relation to aetiology (traumatic versus non-traumatic) (Bargellesi et al., 2018).
To conclude, one could summarise the key message of this review article by stating that the car engine (consciousness) is crucial, but the chassis (motor output) is just as important to allow patients to express themselves, promote their functional recovery and enhance their quality of life. In this context, the preservation of mobility and limitation of neuro-orthopaedic disorders, should be a primary goal of rehabilitation as early as during the acute setting (e.g., acute setting). We encourage longitudinal studies investigating motor functions in DoC. Systematic reports of motor disorders severity as well as information on the patients’ rehabilitation pathways, should be included in all studies on patients with DoC as these factors might influence patients’ responsiveness and functional outcomes.
Conflict of interest
None of the authors have any conflict of interest to report.
Footnotes
Acknowledgments
AT thanks the members of the CSG and Professor Thierry Deltombe for the discussions on this topic. RF thanks the neurorehabilitation staff of Post-Coma Unit of Santa Lucia Foundation for the daily care of our patients with DoC and their caregivers. AT and NL also express their gratitude to the University and University Hospital of Liège, the patients and their families, the Leon Fredericq Foundation, the Belgian National Funds for Scientific Research (FRS-FNRS), FNRS Project No. PDR/BEJ T.0134.21, the University of Liège Conseil Sectoriel de la Recherche, and the European Union’s Horizon 2020 Framework Programme for Research and Innovation under the Specific Grant Agreement No. 945539 (Human Brain Project SGA3). NL is a FNRS post-doctorate fellow and AT is a FNRS research associate.
Appendix: Pharmacological treatments and surgical interventions in non-DoC populations
Given the paucity of literature on pharmacological treatments and surgical interventions to treat spastic paresis in patients with DoC, we summarize here those therapeutic interventions commonly applied in other patient populations that may also be useful in reducing spastic paresis-related disorders inDoC.
