Abstract
Covert consciousness is a state of residual awareness following severe brain injury or neurological disorder that evades routine bedside behavioral detection. Patients with covert consciousness have preserved awareness but are incapable of self-expression through ordinary means of behavior or communication. Growing recognition of the limitations of bedside neurobehavioral examination in reliably detecting consciousness, along with advances in neurotechnologies capable of detecting brain states or subtle signs indicative of consciousness not discernible by routine examination, carry promise to transform approaches to classifying, diagnosing, prognosticating and treating disorders of consciousness. Here we describe and critically evaluate the evolving clinical category of covert consciousness, including approaches to its diagnosis through neuroimaging, electrophysiology, and novel behavioral tools, its prognostic relevance, and open questions pertaining to optimal clinical management of patients with covert consciousness recovering from severe brain injury.
Keywords
Introduction
Covert consciousness is a state of residual awareness following severe brain injury or neurological disorder that evades routine bedside behavioral detection. Patients with covert consciousness have preserved awareness but are incapable of self-expression through ordinary means of communication or behavior (Schiff, 2015; Bodien et al., 2022). Growing recognition of the shortcomings of standardized, bedside neurobehavioral examination techniques in reliably detecting consciousness, along with advances in neurotechnologies capable of discerning brain states or subtle signs indicative of consciousness, carry promise to transform approaches to classifying, diagnosing, prognosticating and treating disorders of consciousness (DoC), including covert consciousness (Edlow, 2018; Young & Edlow, 2021b). Here we explain and critically evaluate the evolving clinical category of covert consciousness, including approaches to its diagnosis through neuroimaging, electrophysiology, and novel behavioral tools, its prognostic relevance, and open questions pertaining to its optimal clinical management.
We begin with an approach to conceptualizing and operationalizing covert consciousness in research and practice, and explore how it problematizes prevailing taxonomies and diagnostic approaches in DoC that revolve predominantly around assessment of a patient’s overt behavioral repertoire. After describing and critically evaluating emerging methods of detecting covert consciousness, including neuroimaging, electrophysiology, and novel behavioral approaches, we describe issues pertaining to the clinical management of covert consciousness. We conclude with a discussion of underexplored questions in the diagnosis, prognosis and management of covert consciousness. Growing needs for consensus building, research and quality improvement efforts to modernize and sensitize faltering DoC nosology, diagnostic approaches, and prognostic paradigms to the possibility of covert consciousness are emphasized throughout.
What is covert consciousness?
Recognizing that there are currently no consensus-based definitions of covert consciousness or guidelines standardizing its diagnosis or management, and building upon the evolving use of the term in the scientific literature and clinical practice, we operationally define covert consciousness as a state of residual awareness following severe brain injury or neurological disorder that evades routine bedside behavioral detection.
This definitional attempt by no means intends to be definitive; it is instead a starting point for our analysis that reflects the term’s general usage in the scientific and medical literature. By ‘awareness’ here, we refer to the capacity to subjectively experience any aspect of the self or environment (Massimini & Tononi, 2018). It should be noted that the residual cognitive capacities of a patient with covert consciousness may or may not extend beyond awareness simpliciter to include higher-order cognitive capacities (Graham, 2021); this operational approach thus aims only to describe minimum conditions constitutive of covert consciousness (Hucklenbroich, 2014). The nature and scope of awareness in a patient with covert consciousness could conceivably range from elemental to elaborate (Bayne et al., 2020; Diserens et al., 2023).
Philosophers and scientists have described various forms that awareness can take, including perceptual awareness (i.e., experience of sense perceptions) (Aquila, 1974), emotional awareness (Mitchell, 2019; Stout, 2019), reflexive inner-awareness (Chaturvedi, 2022), self-awareness (Zahavi, 1999), bodily/interoceptive awareness (Nikolova et al., 2022), awareness of awareness (Armstrong, 1963; Montague, 2017), interpersonal awareness (Decety & Sommerville, 2003; Morin, 2006), among other forms beyond our present scope. Each of these states instantiate properties in virtue of which there is
The inherently subjective character of awareness and consciousness poses an inevitable epistemic challenge to attempts at assessing its presence or absence (Bernal Velásquez, 2011; Giacino et al., 2009; Nagel, 1974). Unlike cardiac or respiratory function, for instance, which can be directly visualized, measured, or auscultated, consciousness, except in the first-person instance, may only be reasonably inferred by closely examining its supervening manifestations, and to the extent possible, characterizing the integrity of what is known about its putative substrate and neural correlates (Chen & Yoshimi, 2023; Edlow et al., 2023; Plum & Posner, 1982; Young, 2018; Young et al., 2023).
As the phenomenology of covert consciousness remains underexplored (Graham, 2019; Taylor et al., 2020), little is known about the first-person experience of covert consciousness and implications for quality of life and wellbeing (Graham, 2021; Graham & Naci, 2021) but it is likely that the contours of this experience vary considerably from person to person depending on the nature and extent of the culprit disorder, and the person’s pre-injury characteristics (Young, Bodien, et al., 2021). Critical ethical and clinical decisions, including those surrounding approaches to neurorehabilitation and the appropriateness of continuing or limiting life-sustaining treatment, may hinge on implicit or explicit assumptions about the nature and value (Bradford, 2022; Kriegel, 2019; O’Leary, 2021) of a patient’s level of consciousness, implications for wellbeing, and capacity for recovery (Glannon, 2016; Gosseries & Laureys, 2022; Graham, 2017; Lazaridis et al., 2021; Lee, 2019; Mertens et al., 2022; Tung et al., 2020; Young et al., 2022). The magnitude and at times life-or-death consequences of such decisions underscore the medical and moral imperatives to address these questions in clinical practice and research, which at minimum require clarifying whether a behaviorally unresponsive patient is covertly conscious in the first place (Ezer et al., 2020; Fins, 2015; Fins, 2016; Fins et al., 2020; Naci & Owen, 2022; Rohaut et al., 2019). As we later examine, covert consciousness may be identified through a variety of potential methods, including functional neuroimaging and electrophysiologic paradigms designed to detect volitional modulation of brain activity, brain-computer interfaces (Lulé et al., 2013; Pan et al., 2022; Xu et al., 2022; Young, Lin, et al., 2021), as well as through advanced neurobehavioral examination techniques (Mat et al., 2022) designed to capture conscious responses to which standard bedside assessment is insensitive.
‘Severe brain injury or neurological disorder’ is included in the operational definition above in order to exclude patients with conditions like psychogenic coma (Ludwig et al., 2016; Ryznar & Wilcox, 2019) or akinetic catatonia (Schnetzer et al., 2023). Although such patients might be considered covertly conscious if they are mistakenly regarded as unconscious at the bedside, these conditions are outside of the scope of this paper and inconsistent with evolving norms in the scientific literature which tend to limit application of this term to patients with severe neurological disorders or trauma. The potential neurological disturbances that may give rise to the syndromic phenotype of covert consciousness are varied (Fig. 1). While covert consciousness is typically invoked in the settings of severe brain injury, under this approach, a patient with stroke, toxic, metabolic, infectious or autoimmune leukoencephalopathy, advanced amyotrophic lateral sclerosis (ALS) or severe polyneuropathy such as Guillain-Barré syndrome (GBS/acute demyelinating polyneuropathy (AIDP)) with complete paralysis and anarthria may be considered covertly conscious if awareness is preserved but not evident at the bedside. In these conditions, weakness may progress to the point that a patient is no longer able to reliably communicate or respond to commands; patients with this profound degree of paralysis are more typically said to be in the “complete locked-in state” (CLIS); while harboring intact awareness and cognition, the capacity for self-expression is lost due to complete motor impairment with quadriplegia, anarthria and oculomotor paresis (Hochberg & Cudkowicz, 2014). This is distinguished from patients in an incomplete locked-in state, who retain the ability to reliably communicate at least a binary “yes/no” signal, for example through vertical eye movements (and in doing so can, with the assistance of a caregiver, spell out words using a manual letterboard) (Murguialday et al., 2011). While some favor the term CLIS when the condition is attributable to peripheral motor nerve or brainstem pathology and limit application of the term covert consciousness to circumstances where obstruction of self-expression is attributable to supratentorial pathology, or otherwise limit application of covert consciousness to circumstances where awareness is diminished but not altogether extinguished, others employ overlapping terminology, and there is no universally accepted etiological or localization-based approach to distinguishing these entities (de Jong, 2013; Medici et al., 2011; Norton, Graham, et al., 2023; Schnetzer et al., 2023). Complicating matters further, some have pointed out that it is a misnomer to classify some such states as ‘disorders of consciousness’ if it is the case that consciousness is preserved (and thus not inherently disordered, but rather limited in behavioral expression, as it might be in select instances of covert consciousness and behavioral MCS) (de Jong, 2013; Diserens et al., 2023; Hermann, Sangaré, et al., 2021). Whereas in conditions such as ALS and GBS the expectation is that consciousness and cognition will generally remain intact (Kotchoubey et al., 2003), in DoC following brain injury the expectation is that cognitive capacities will be impaired, which is why many of the assistive devices that have become available to manage motor impairments of other etiologies have been challenging to successfully apply in the DoC population. However, this dichotomy between conditions that impair versus spare consciousness and cognition is not always straightforward to apply in practice, considering the possibility for secondary complications of “pure” sensorimotor conditions that could result in alterations in consciousness (e.g., respiratory failure in GBS resulting in superimposed hypoxic brain injury), and in light of known variants such as ALS with frontotemporal dementia (ALS-FTD) or Bickerstaff brainstem encephalitis, a GBS variant, which may impact consciousness. Additionally, some contend that consciousness is affected by the transition to the CLIS as evidenced by changes in alpha band frequency and other electroencephalography (EEG) features (Colombo et al., 2023; Khalili-Ardali et al., 2021; Maruyama et al., 2021; Rosburg, 2019), theorizing that “an extinction in goal-directed thinking in completely paralyzed patients” could ensue if patients “lose the perception of the contingency between the required physiological behaviour . . . and its consequences” (Kübler & Birbaumer, 2008). While these findings may challenge the dichotomy between DoC and CLIS, some have argued that cognition may be maintained in CLIS through “imagined response-effect contingencies” and potentially restored through novel rehabilitative interventions, underscoring the clinical and ethical urgency to understand, identify, and manage these conditions (Soekadar & Birbaumer, 2015).
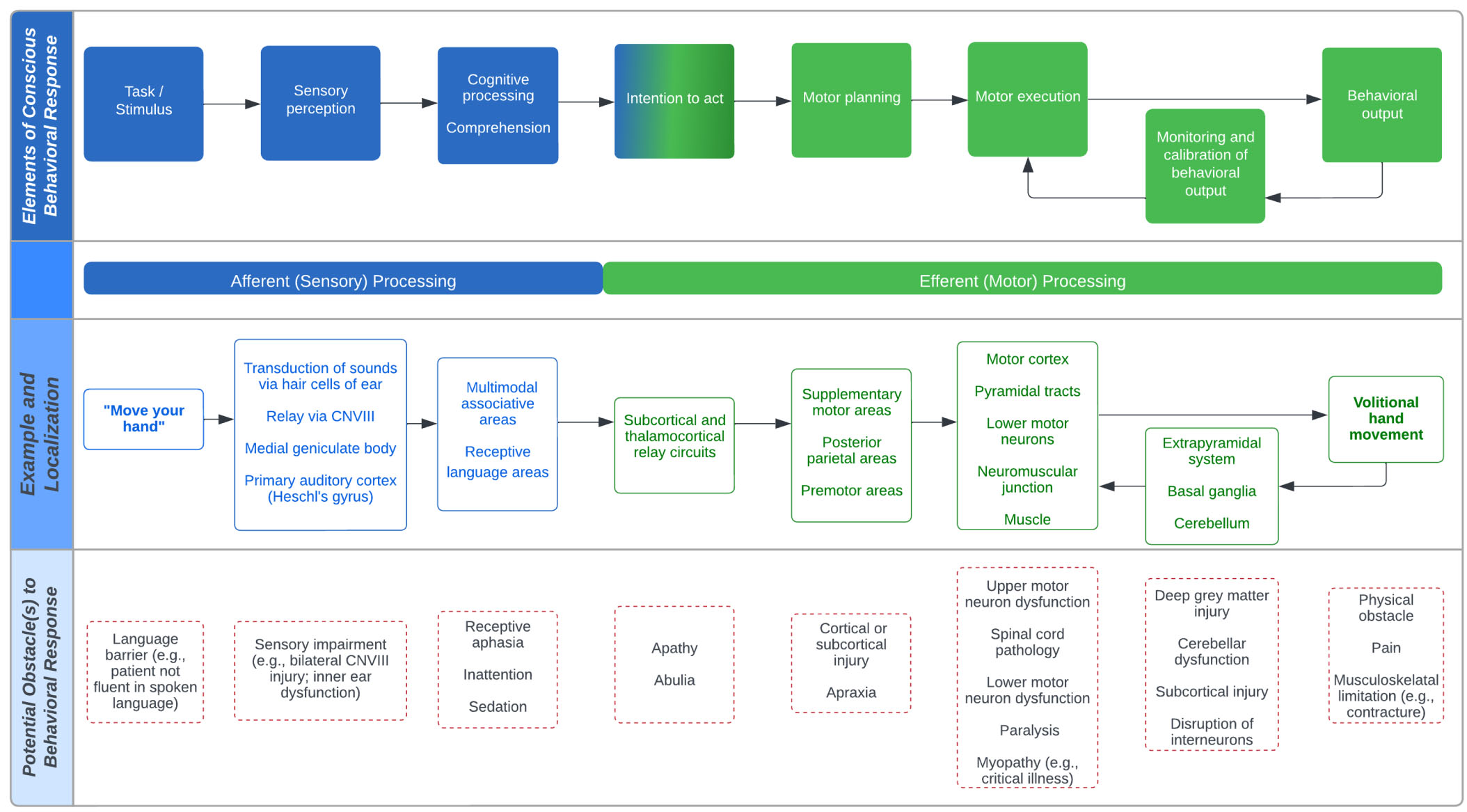
Modeling the potential pathogenesis of covert consciousness. Covert consciousness is a state of residual awareness following severe brain injury or neurological disorder that evades routine bedside behavioral detection. Because the sensitivity of neurobehavioral examination in detecting consciousness depends on intact afferent (sensory) and efferent (motor) processing, the basic elements of which are displayed on the top panel, a patient with deficit(s) affecting these pathways may be unable to appropriately respond at the bedside even if consciousness is preserved. The middle panel models an example of a common behavioral test used by clinicians to assess awareness at the bedside, “move your hand”, and simplifies the localization of the necessary steps for a patient to appropriately respond. The bottom panel row displays potential obstacle(s) to producing or detecting an appropriate behavioral response at each level of localization, which may cause or contribute to the pathogenesis of covert consciousness when awareness is otherwise preserved.
“ . . . evades routine bedside detection” is used in the operational definition above to sensitize this approach to the possibility of covert consciousness being detected through non-routine behavioral techniques (such as video eye-tracking to immersive stimuli or other subtle motor/behavioral signs like olfactory responses). Thus, unlike several other proposed definitions, including those of cognitive motor dissociation (CMD), this approach does not necessitate the use of advanced neuroimaging or electrophysiologic techniques in diagnosing covert consciousness. For instance, Schiff describes CMD as a clinical syndrome of patients who “
Many different terms for covert consciousness have been used, sometimes interchangeably and at times with slight or substantial variations in meaning, CMD (Schiff, 2015), covert command-following (Bodien et al., 2017; Forgacs et al., 2014), preserved cortical circuitry (de Jong et al., 1997), hidden/preserved vitality (Shalit et al., 1970), loss of behavior with remaining conscious perception (Ingvar & Ciria, 1975), covert cognition (Cruse et al., 2012; Nachev & Hacker, 2010), complete locked-in syndrome (de Jong, 2013), covert activity (Sontheimer et al., 2017), covert attention (Morlet et al., 2023; Pan et al., 2020), covert awareness (Cruse et al., 2014; Fernandez-Espejo et al., 2014; Gibson et al., 2016; Graham et al., 2018; Graham et al., 2015; Owen et al., 2007; Peterson et al., 2013), covert narrative capacity (Naci et al., 2017), preservation of mental life (Naci & Owen, 2022), functional disconnection syndrome (Formisano et al., 2011; Laureys et al., 2006; Schiff, 2012), functional LIS (Formisano et al., 2013), functional minimally conscious, minimally conscious state star (MCS*) (Thibaut et al., 2021), among others (Schnakers et al., 2022). Meanwhile, prevailing clinical nomenclature (e.g., coma, VS / UWS, MCS), yoked to behavioral diagnostic criteria remains unsensitized to the potential findings of methods for detecting covert consciousness, and lumps together fundamentally different endotypes into the same rough behavioral categories, glossing over important diagnostic differences and risking mischaracterization of patients’ levels of awareness (Young & Edlow, 2021a; Young & Peterson, 2022). This irregular patchwork of terminology and heterogeneity in real-world uses underscores a pressing clinical and ethical need to develop a more organized approach to the taxonomy of covert consciousness (along with other DoCs), that is adapted to the range of potential findings of advanced testing modalities, and is explicit about etiology (for example, through a multi-axis or composite diagnostic approach). Such an approach stands to benefit clinicians, researchers and surrogate decision-makers alike. Efforts to describe endotypes of covert consciousness through objective, internal criteria are underway, and promise to aid in mitigating the forgoing challenges afflicting DoC classification schemes based solely on external behavioral or relational criteria (Hammond et al., 2021; Kondziella et al., 2021; Kondziella & Stevens, 2022; Zachar, 2002).
For nearly two decades, investigators have applied advanced neuroimaging techniques such as fMRI to identify awareness in patients with no signs of consciousness on the bedside examination (Schiff, 2015).
Prior to 2018, advanced neuroimaging techniques for detecting covert consciousness were used exclusively in the context of research. However, in recent years, clinical guidelines published by the American Academy of Neurology (AAN), American Congress of Rehabilitation Medicine (ACRM), National Institute on Disability, Independent Living, and Rehabilitation Research (NIDILRR) (Giacino et al., 2018a, 2018b) and the European Academy of Neurology (EAN) (Kondziella et al., 2020) have recognized the benefits of integrating functional neuroimaging into clinical practice. In these guidelines, national and international organizations recommend, for the first time, that functional neuroimaging tests be considered in the diagnostic and prognostic evaluation of some patients with DoC. The translation of these tools into clinical practice poses a challenge, as they require specific hardware and software, expertise in analysis, and nuanced interpretation (Bodien et al., 2023; Young et al., 2024). Nevertheless, over time we expect they will be integrated into a multimodal battery of tests that will increase the certainty with which level of consciousness is established. The development of a hub-and-spoke model system for DoC evaluation and management may aid in the process of democratizing access to advanced tools (Peterson et al., 2022; Young, 2022; Young & Edlow, 2021a). Below, we discuss the most common functional neuroimaging modalities used to assess patients with DoC and review how these modalities may be used to understand diagnosis and prognosis of severe brain injury.
Functional neuroimaging modalities can be used to directly assess covert consciousness by measuring the brain’s response to a specific task. Alternatively, data collected while the brain is in a resting state (i.e., no external stimuli or instructions are presented to evoke behavioral and/or neuronal responses), while the brain is passively stimulated (e.g., by auditory or visual stimuli), while the subject is asked to perform a task (e.g., target detection), or following administration of a chemical compound may inform the more nebulous “capacity” of consciousness, or grounds for possibility of conscious experience. Understanding the advantages and limitations of each approach and the range of possible interpretations of the data is critical to translating the results into a clinical tool.
Functional magnetic resonance imaging
Functional magnetic resonance imaging (fMRI) measures the brain’s hemodynamic response to a stimulus or cognitive process, or simply at rest. During periods of increased neuronal activity, the brain requires an influx of glucose and oxygen, which are delivered through cerebral blood flow. This bolus of blood contains more oxygenated versus deoxygenated blood, and, due to the ferrous MR signal-reducing properties of deoxygenated blood, contributes to an overall enhanced MR signal (Kwong et al., 1992; Ogawa et al., 1993; Ogawa et al., 1992). The Blood-Oxygen-Level Dependent (BOLD) response measured by fMRI is therefore an indirect measurement of underlying neuronal activity (Logothetis et al., 2001). However, during the resting state, in the absence of a stimulus or task, the BOLD signal fluctuates spontaneously at a low frequency. The correlation of the BOLD signal fluctuations in different brain regions is interpreted as functional connectivity (Fox & Raichle, 2007). One of the most widely studied resting-state networks is the default mode network (DMN), which is thought to mediate introspection and self-referential thought (Buckner et al., 2008; Levorsen et al., 2023; Menon, 2023; Wen et al., 2020).
Positron emission tomography
Positron Emission Tomography (PET) uses an intravenous radiotracer targeted towards brain metabolism or specific neurotransmitter systems to quantify brain metabolism. A decaying radiotracer emits a positron that interacts with an electron, resulting in the annihilation of both particles into photons. When these photons are detected, their original position can be calculated to localize the source of the annihilation, which results in an image of radiotracer uptake. The most common radiotracer is 18F-fluorodeoxyglucose (FDG), which measures brain metabolism (Sweet, 1951; Wrenn et al., 1951). Although PET is used most often with resting-state studies, it is possible to use FDG-PET (Jamadar et al., 2019) and 15O-water PET to probe covert cognitive function (Mamach et al., 2018).
Functional near-infrared spectroscopy
Similar to fMRI, functional near-infrared spectroscopy (fNIRS) leverages the coupling between neuronal firing and the hemodynamic response to measure brain function (Jobsis, 1977). fNIRS relies on a light that, when emitted onto the surface of the head in the near-infrared spectrum of 700– 900 nm, passes through the scalp and skull, and is absorbed by oxygenated and deoxygenated hemoglobin. Changes in hemoglobin concentration are detected by an optode sensor on the scalp. In contrast to fMRI, fNIRS can be performed at the bedside, is less prone to motion artifact, and has fewer safety risks. fNIRS is highly portable and can be conducted at the bedside. However, the spatial resolution of fNIRS is limited to activity occurring within a cortical depth of 2– 4 mm.
Moving towards clinical implementation of functional neuroimaging techniques
Although the utility of clinical fMRI has been demonstrated in pre-surgical mapping for tumor resection and epilepsy surgery, and FDG-PET is used clinically for diagnosis of dementia, most functional neuroimaging tools have traditionally been considered investigational. However, there are now notable exceptions supporting the use of the techniques described here in clinical practice (Giacino et al., 2018a, 2018b; Kondziella et al., 2020). Currently, only a small number of specialty centers have the hardware, software, and expertise to implement these techniques and most have not undergone rigorous clinical validation (Young and Edlow, 2021). In almost all arenas, studies with large, well-controlled, patient samples recruited from multiple sites and with meticulous blinding procedures are needed to bridge this gap. Nevertheless, there is growing enthusiasm for clinical implementation of functional neuroimaging technologies because of the unprecedented opportunity to improve diagnostic and prognostic precision for patients with severe brain injuries (Comanducci et al., 2020; Monti & Schnakers, 2022).
Diagnosing covert consciousness with functional neuroimaging
Over the last 15 years, neuroimaging (and electrophysiology) studies have consistently shown that 10– 20% of patients who appear unconscious on behavioral examination retain conscious awareness when tested with fMRI (or EEG) using task-based motor imagery paradigms (Kondziella et al., 2016; Schnakers et al., 2020; Stender et al., 2014). Among the first reports of CMD was a 2006 case report of a patient with a behavioral diagnosis of VS/UWS (Owen et al., 2006). During an fMRI scan, when the patient was asked to imagine playing tennis, a BOLD response was observed in the supplementary motor area and when asked to imagine walking around her house, a BOLD response was observed in the parahippocampal area. This finding paralleled the result obtained in healthy control subjects. In 2010, a study of 54 patients confirmed the presence of CMD and fMRI was also used to establish a channel of yes/no communication in a single subject who appeared to fulfill criteria for the VS for 5 years prior to the assessment (Monti et al., 2010). Covert consciousness has also been reported in patients with acute severe TBI in the intensive care unit (Edlow et al., 2017). Covert consciousness has also been identified in the context of pediatric brain injury, but has been relatively underexplored in comparison to the adult population (Boerwinkle et al., 2023; Kim et al., 2022).
A substantial limitation of using fMRI to detect covert consciousness is the frequency with which individuals with a behavioral MCS diagnosis, and even healthy control subjects, fail to demonstrate the anticipated response to motor imagery paradigms. Motor imagery is a complex task requiring multiple cognitive processes including language comprehension, working memory and attention, to be intact. Some patients may be able to understand the command but fail to perform the task with the consistency required to generate a BOLD response. Fluctuations in arousal, sedating medications, and language impairment may also contribute to false negative findings. For this reason, a positive response on an fMRI motor imagery task in patients with a behavioral diagnosis of coma, VS, or MCS- is interpreted as CMD, but a negative response is ambiguous and does not suggest an absence of CMD. On a prognostic plane, covert brain responses detected by fMRI in acutely unresponsive patients following severe brain injury appear to portend greater likelihood of functional recovery, as measured by Glasgow Outcome Scale at 6 months (Norton, Kazazian, et al., 2023). Clinicians and researchers have emphasized the importance of facilitating responsible integration of fMRI in clinical practice (Monti & Schnakers, 2022; Young et al., 2023; Bodien et al., 2023).
PET has also been used to assess patients with CMD (Stender et al., 2014). However, because PET is acquired most frequently in the resting state (i.e., in the absence of a task that requires volitional cognition), consciousness cannot be directly inferred from this approach. Notably, FDG-PET was validated as a measure of capacity for consciousness against the perturbation complexity index (PCI; derived from combined transcranial magnetic stimulation (TMS) and EEG) (Bodart et al., 2017; Casali et al., 2013; Casarotto et al., 2016; Massimini et al., 2009). When combined with EEG, FDG-PET identified patients with a behavioral diagnosis of MCS with 94% sensitivity and 67% specificity suggesting that this method may identify patients with CMD (Hermann, Stender, et al., 2021).
Neither PET nor fMRI can be used at the bedside, and therefore may not be feasible tools for routine assessment of consciousness, especially in acute settings where respiratory requirements, adventitious patient movements, and medical instability may preclude prolonged recumbency in the scanner. fNIRS is a portable tool with fewer technical limitations, but to date, only a limited number of studies have used task-based fNIRS to detect CMD (Abdalmalak et al., 2021). In combination with fMRI and EEG, fNIRS may complement a multimodal battery of assessments aimed at detecting consciousness in severe acute brain injury (Kazazian et al., 2021). Leveraging fNIRS measurements in neurocritically ill patients, Bicciato et al. demonstrated covert responses to music in the left prefrontal area in some behaviorally unresponsive patients (Bicciato et al., 2022). More recently, resting-state fNIRS (rsFNIRS) has been used to identify putative “consciousness-supporting networks” which may indicate the capacity for consciousness in patients following severe brain injury (Chen et al., 2023; Liu et al., 2023).
Functional neuroimaging is a powerful tool for understanding the pathophysiology of brain injury and may impact clinical diagnosis, prognosis and treatment efficacy. Its utility in research is in part due to the wide range of approaches available for both data acquisition and analysis. Functional neuroimaging techniques are typically more sensitive to brain network abnormalities than are structural imaging techniques, particularly in patients with mild brain injuries, and provide potential predictive and pharmacodynamic biomarkers for treatment studies. Although the clinical utility and generalizability of functional neuroimaging techniques continues to be limited by the lack of standardized methods and the absence of large validation studies, for some patients with DoC, functional neuroimaging is now indicated for diagnostic and prognostic purposes. Functional neuroimaging provides a wide range of possibilities for addressing scientific questions, but this same flexibility leads to high inter- and intra-individual variability that limits clinical interpretability. Moreover, it is often challenging to determine whether functional imaging findings are causal or epiphenomena in patients with cognitive dysfunction due to brain injuries. Results from large-scale international and collaborative studies such as Transforming Research and Clinical Knowledge in TBI (TRACK-TBI, ClinicalTrials.gov NCT02119182), Collaborative European NeuroTrauma Effectiveness Research in TBI (CENTER-TBI (Burton, 2017)), Late Effects of TBI (LETBI (Edlow et al., 2018)), Collaborative Neuropathology Network Characterizing Outcomes of TBI (CONNECT-TBI (Smith et al., 2021)), Traumatic Brain Injury Endpoints Development Initiative (TED (Manley et al., 2017)), the Enhancing NeuroImaging Genetics through Meta-Analysis Consortium (ENIGMA (Thompson et al., 2014)), Multimodal Resonance Imaging for Outcome Prediction on Coma Patients (MRI-COMA (Velly et al., 2018)), Data-driven neuroEthics for COnsciousness DEtection (DECODE, ClinicalTrials.gov NCT05010265), and the Neurocritical Care Society Curing Coma Campaign (Provencio et al., 2020), are poised to further expand translation of functional neuroimaging approaches into clinical practice.
Electrophysiology of Covert Consciousness
Elctrophysiologic techniques including active task-based electroencephalography (EEG), passive stimulus-based EEG, quantitative resting-state EEG, event-related potentials (ERPs), and perturbational approaches such as TMS-EEG have been investigated to aid in the assessment of consciousness and prediction of outcomes following brain injury (Comanducci et al., 2020; Curley et al., 2018; Edlow, Claassen, et al., 2021; Hermann, Stender, et al., 2021; Kondziella et al., 2016; Claassen et al., 2019; Egbebike et al., 2022; Young, 2023). EEG-based techniques carry several advantages over neuroimaging, including improved portability, lower cost, ease of administration at the bedside, and temporal resolution, but have a much larger repertoire of analytic approaches making methodologic standardization challenging (Schnakers et al., 2020). Of these techniques, only task-based EEG aimed at capturing volitional modulation of brain activity (i.e., command-following) currently provides evidence of covert consciousness. However, other electrophysiological techniques may provide informative data pertaining to the
Task-based EEG techniques are affected by similar limitations as task-based fMRI, insofar as they depend on a patient harboring residual motivation, cognitive and language capacities to reliably respond to a command, even if covertly, and carry similarly high false-negative rates (Formisano, Toppi, et al., 2019). These limitations have motivated the development of passive paradigms which do not require high levels of cognitive effort, such as passive, hierarchical language paradigms (Gui et al., 2020; Sokoliuk et al., 2021), music paradigms (Edlow et al., 2017; Coleman et al., 2009; Lord & Opacka-Juffry, 2016; Okumura et al., 2014; Wu et al., 2011), visual, tactile and olfactory (Pistoia et al., 2015; Schriever et al., 2017), and stimulation paradigms (Jain & Ramakrishnan, 2020). However, potential findings from passive paradigms are not dispositive of covert consciousness like finding of command-following, and are prone to similar confounds from sensory or processing disturbances. In light of the epistemic gap in discerning whether a patient who demonstrates intact passive responses to sensory stimuli is indeed subjectively aware and experiencing the stimuli presented or is merely exhibiting a reflexive, non-conscious brain response, the diagnostic label ‘covert cortical processing’ (Young et al., 2024) has been proposed in lieu of ‘covert consciousness’ among patients with intact cortical responses to passive stimuli but no discernible responses to active tasks (Claassen et al., 2021; Edlow, Claassen, et al., 2021). The TMS-EEG PCI has been proposed as potentially superior passive approach for the detection of capacity for consciousness that does not rely on intact sensory input and sensory processing, but has not yet been shown to be dispositive of covert consciousness (Edlow et al., 2023; Sarasso et al., 2014). These complexities recapitulate the need for a refined DoC nomenclature to more precisely classify endotypes of consciousness sensitized to notions of complexity, capacity and cortical processing.
An International Federation for Clinical Neurophysiology (IFCN) Expert Group comprehensively reviewed decades of literature on neurophysiology in evaluation of patients with DoC (Comanducci et al., 2020), and proposed a “[m]ultimodal graded neurophysiological assessment in patients with prolonged DoC. The operational stepwise workflow include[s] multiple steps of instrumental evaluation with increasing complexity starting from conventional neurophysiologic measures (standard EEG and SEPs) to ERPs and finally advanced approaches (qEEG analysis, TMS/EEG and active EEG paradigms). This general scheme might help direct behaviorally unresponsive patients towards different lines of evaluation based on objective markers of thalamo-cortical integrity” (Comanducci et al., 2020). Each of these paradigms require a high level of expertise to perform and interpret, and the current paucity of standardized performance and analysis pipelines limits widespread diffusion in clinical practice.
The IFCN Expert Group also emphasized caution in the development of training datasets for machine-learning [ML] classifiers, especially if “the classifier is initially trained to differentiate VS/UWS from MCS patients based on behavioral labels [as the] clinical diagnosis may fail to recognize brain-injured patients who are conscious but disconnected and unresponsive [, and hence in these circumstance,] the true state-of-affairs (i.e. conscious versus unconscious subjects) necessary for a correct training remains unknown, engendering a circularity problem with potential impact on the accuracy and interpretability of the results.” The IFCN Expert Group suggests that to remedy this issue “the problem of circularity can be addressed by refining the diagnostic labels by means of additional paraclinical markers” while pointing out that “[a]nother potential disadvantage of ML-based approaches to multivariate data is that, given their black-box nature, they do not necessarily provide direct mechanistic insights into the underlying neuronal processes” (Comanducci et al., 2020).
The 2020 EAN DoC guidelines issued several recommendations surrounding the use of electrophysiological techniques in assessing some patients with DoC. Delineating the relative utility and evidence-base for different electrophysiological techniques, the EAN guideline provides modality-specific recommendations around clinical standard EEG, sleep EEG, quantitative high-density EEG, active paradigms with high and low density EEG, and cognitive evoked potentials including P300, and TMS-EEG as part of multimodal assessment of consciousness (Kondziella et al., 2020).
The EAN guideline concludes that “standardized clinical rating scales such as the CRS-R and the FOUR [(Full Outline of UnResponsiveness)], including careful inspection of voluntary eye movements, EEG-based techniques and functional neuroimaging (fMRI, PET) should be integrated into a composite reference standard. This means that a given patient should be diagnosed with the highest level of consciousness as revealed by any of the three approaches (clinical [(i.e., behavioral)], EEG, neuroimaging)” (Kondziella et al., 2020; Wijdicks et al., 2005).
In a similar vein, the AAN/ACRM/NIDILRR DoC guidelines describe that “ . . . injury sequelae (such as severe hypertonus) may confound behavioral assessment and compromise diagnostic accuracy. Additionally, diagnostic findings may remain ambiguous despite serial assessment due to the inconsistency or subtlety of the behavioral evidence. The largest functional neuroimaging study conducted to date in patients with DoC reported that ambiguous or erroneous findings clouded clinical diagnosis in 33 of 126 (27 percent) of cases [(Stender et al., 2014)]” and recognizing these challenges, recommend (2e) that “[i]n situations where there is continued ambiguity regarding evidence of conscious awareness despite serial neurobehavioral assessments, or where confounders to a valid clinical diagnostic assessment are identified, clinicians may use multimodal evaluations incorporating specialized functional imaging or electrophysiologic studies to assess for evidence of awareness not identified on neurobehavioral assessment that might prompt consideration of an alternate diagnosis” (Giacino et al., 2018a, 2018b). Unlike the European guideline, however, the AAN/ACRM/NIDILRR guideline does not distinguish between various types of electrophysiologic studies in its recommendation, as different methodologies were used to establish the recommendations.
Novel behavioral markers of covert consciousness
While assessment of covert consciousness is most commonly tied to the use of advanced neuroimaging and electrophysiologic techniques such as those discussed above, it is important to note that approaches to detecting covert consciousness span beyond neuroimaging and electrophysiologic tools to include novel behavioral examination techniques designed to elicit and capture signs of awareness that might ordinarily evade routine bedside detection. These include covert eye tracking detected through wearable eye trackers (Alkhachroum et al., 2023), olfactory sniffing (Arzi et al., 2020; Wang et al., 2022), heart-rate variability (Liuzzi et al., 2023; Machado-Ferrer et al., 2013; Riganello et al., 2010; Riganello et al., 2018), auditory localization (Carrière et al., 2020), startle habituation (Hermann et al., 2020), nociceptive responses (Cortese et al., 2021), resistance to eyelid opening (van Ommen et al., 2018), quantitative analysis of subtle facial expressions (Chatelle, Hauger, et al., 2018; Obayashi et al., 2021), volitional swallowing tasks (Mélotte et al., 2023), and alternative structured behavioral exam techniques such as the Motor Behavior Tool Revised (MBT-R) (Jöhr et al., 2020; Pincherle et al., 2019), Sensory Modality Assessment and Rehabilitation Technique (SMART) integrating visual, sound, tactile, olfactory, and gustatory modalities (Gill-Thwaites & Munday, 2004), and the Music Therapy Assessment Tool for Awareness in Disorders of Consciousness (MATADOC) (Magee, 2018; Magee et al., 2023; Magee et al., 2014); (Carrière et al., 2022; Diserens et al., 2023; Liuzzi et al., 2023).
Some have advocated for greater inclusion of caregivers in the diagnostic process for patients with DoC, capitalizing on their potentially greater attunement to subtle or idiosyncratic cues of awareness that might be overlooked by standard techniques (Hermann et al., 2019; Moretta et al., 2017; White, 2006) with one study suggesting that involving primary caregivers with emotional stimulation may positively modulate patient performance on the CRS-R (Formisano, Contrada, et al., 2019). The Social and Family Evaluation (SAFE) scale has been proposed as a structured method for integrating family and caregiver input into the diagnostic assessment of consciousness, but requires validation (Formisano, Contrada, et al., 2019). Substantially lower costs, ease of dissemination and fewer barriers to clinical implementation make novel behavioral tests for covert consciousness promising prospects for widespread translation and adoption.
Clinical management of covert consciousness
After receiving a diagnosis of covert consciousness, clinicians and surrogates may naturally ask “what now?”. Many open questions pertaining to optimal clinical management of patients with covert consciousness recovering from severe brain injury remain. The 2018 AAN/ACRM/NIDILRR guideline on DoC recommends that “[i] situations where there is no behavioral evidence of consciousness on clinical examination but functional neuroimaging or electrophysiologic testing suggests the possibility of preserved conscious awareness, frequent neurobehavioral reevaluations may be conducted to identify emerging signs of conscious awareness and decisions to reduce the intensity of rehabilitation treatment may be delayed for those individuals receiving active rehabilitation management, with the length of time over which these are done determined by an agreement between the treating clinician and the health care proxy given the lack of evidence to provide guidance” (Giacino et al., 2018a, 2018b). In addition to conducting more frequent reevaluations and delaying decisions to reduce intensity of rehabilitative treatment, the finding of covert consciousness should prompt closer examination of potentially treatable barriers to behavioral self-expression, including sensory, motor or cognitive impairments, or sedating medications (see Fig. 1, bottom row). Opportunities to leverage and engage patients’ covert capacities through personalized neurorehabilitative approaches where possible should be recognized and strengthened (Thengone et al., 2016). Pharmacologic therapies including neurostimulant medications to promote potential recovery and expression of consciousness should be carefully considered on a case-by-case basis (Barra et al., 2022). Techniques such as the Individualized Quantitative Behavioral Assessment (IQBA) may be considered to closely track and contextualize changes in volitional responses over time (Giacino & Smart, 2007).
Therapeutic decisions should ideally be made in multidisciplinary collaboration with a patient’s physician(s) and clinical team, including physical therapy, occupational therapy, speech and language therapy, and neuropsychology, with planning to meticulously monitor changes in patient status before and after each therapeutic intervention or modification. If concordant with a patient’s preferences, values and goals, consideration may also be given to clinical trial enrollment where appropriate, with careful attention to the ethical nuances of research involving patients with DoC (Edlow, Sanz, et al., 2021; Lewis et al., 2023; Thibaut et al., 2019; Young et al., 2022). Efforts to develop brain-computer interfaces (BCIs) to restore communicative capacity to patients who are unable to speak but may be able to leverage preserved abilities to volitionally modulate brain activity are underway, but remain nascent (Chatelle, Spencer, et al., 2018; Lugo et al., 2020; Rohaut et al., 2019; Xu et al., 2022; Young, Lin, et al., 2021). Emerging evidence highlights a potential role for multisensory stimulation in enhancing recovery in CMD (Attwell et al., 2019), however more study is necessary.
Since the recovery trajectories of patients with covert consciousness following severe brain injury may be more favorable than behaviorally unresponsive patients without covert consciousness (Egbebike et al., 2022), some have suggested that this finding could inform counseling of surrogates and determinations of who might optimally benefit from structured rehabilitation settings. While positive findings might reasonably inform such decisions, negative findings (i.e., the absence of evidence of covert consciousness) are not as clearly actionable given limitations associated with false negative rates and low sensitivity of available techniques. In other words, the absence of evidence of covert consciousness does not constitute evidence of absence of covert consciousness. This important dimension of diagnostic uncertainty should be carefully explained to caregivers and clinicians when disclosing results of tests for covert consciousness and deciding on best next steps in management (Young et al., 2024).
Covert consciousness: Remapping the clinical and philosophical landscape
In
In the absence of a ‘gold standard’ test for covert consciousness (owing to the inherently subjective character of conscious experience), combining multimodal techniques will likely be required to advance precision assessment of consciousness. There are critical and growing research opportunities to determine optimal clinical management of covert consciousness and to improve the rigor and reliability of clinical assessment by aggregating diagnostic evidence across different modalities. Similar opportunities and challenges have been recognized in the process of aggregating validators for psychiatric diagnoses (Solomon, 2022). The establishment of a global registry for patients with covert consciousness may be a natural opportunity to foster these goals. It is clinically and ethically imperative that current systems of classifying and diagnosing disorders of consciousness be updated through consensus-based methods to sensitize clinical practice to the possibility of covert consciousness among patients who are behaviorally unresponsive. These efforts could be buttressed by multilateral initiatives to facilitate responsible implementation of professional society guidelines to improve care for this vulnerable population around the world.
Footnotes
Acknowledgments
The authors acknowledge and dedicate this work to patients affected by disorders of consciousness and their devoted caregivers, including members of the Patient and Family Advisory Board (PFAB) of the Lab for Neuroimaging of Coma and Consciousness (NICC).
Conflict of interest
The authors have no conflicts of interest to report.
Funding
This work was supported by the NIH BRAIN Initiative (F32MH123001), NIH Director’s Office (DP2HD101400), National Institute on Disability, Independent Living and Rehabilitation Research (NIDILRR), Administration for Community Living (90DPTB0011, 90DPTB0027, 90DP0039), Chen Institute MGH Research Scholar Award, and the American Academy of Neurology (AAN) Palatucci Advocacy Award.
