Abstract
BACKGROUND:
Persistent pulmonary hypertension of the neonate (PPHN) is a serious disorder. The long pentraxin 3 (PTX3) plays an important role in angiogenesis, cell proliferation, tissue repair and cell regulation. The present study aims to assess the diagnostic and clinical value of PTX3 in PPHN.
METHODS:
The present case-control 60 full-term neonates diagnosed with PPHN by echocardiography within 72 hours of birth. In addition, there were 30 age and sex-matched healthy neonates who served as controls. All participants were subjected to careful history taking and complete clinical examination, Laboratory investigations included complete blood count, C-reactive protein (CRP), blood culture and PTX3 level. Radiological investigations included plain X- ray and two-dimensional transthoracic echocardiography (TTE).
RESULTS:
Comparison between patients and controls revealed that patients had significantly higher CRP (6.12±2.18 versus 3.69±1.25 mg/dl, p < 0.001) and PTX3 levels (2.07±0.67 versus 0.96±0.21, p < 0.001) when compared with controls. Patients with associated PDA had significantly higher PTX3 levels when compared with patients without (2.58±0.5 versus 2.02±0.51 ng/ml, p = 0.002). Also, patients with associated PFO had significantly higher PTX3 levels when compared with patients without (2.12±1.05 versus 2.05±0.46, p = 0.002). ROC curve analysis identified good performance of CRP and PTX3 levels in diagnosis of PPHN with PTX3 showing better performance.
CONCLUSIONS:
There is a significant association between serum PTX3 levels and PPHN particularly those with associated PDA or PFO.
Keywords
Introduction
Persistent pulmonary hypertension of the neonate (PPHN) is a serious disorder that is considered an important cause of neonatal morbidity and mortality. It is attributed to failure of change from fetal high-resistance pulmonary circulation to low-resistance circulation that develops after birth [1, 2].
PPHN occurs at a rate of 2 cases in every 1000 live births [3]. However, an Egyptian study identified the condition in 5.0% of their 640-neonate study [4]. PPHN may present in in late-preterm or term neonates and usually is not associated with birth anomalies. Affected neonates have severe dyspnea that needs mechanical ventilation [5]. Available treatment options included mechanical ventilation, nitric oxide inhalation and extracorporeal membrane oxygenation (ECMO). However, about 10–20% of diseased neonates die [6].
Considering the significant consequences of PPHN which include pulmonary vasoconstriction and remodeling with subsequent right to left extrapulmonary shunting and ventricular dysfunction [7], it is crucial to diagnose the condition as early as possible through identification of new biochemical markers. The long pentraxin 3 (PTX3) is a member of pentraxins family which is produced by endothelium cells, smooth muscle cells and fibroblasts and from innate immune cells during inflammation [8]. It plays an important role in angiogenesis, cell proliferation, tissue repair and cell regulation [9]. The present study aims to assess the diagnostic and clinical value of PTX3 in PPHN.
Materials and methods
The present case-control study was conducted in neonatal intensive care unit (NICU) at Al-Zahraa University Hospital, Al-Azhar University, Cairo, Egypt in the time from May, 2020 to April 2021. The study protocol was approved by the ethical committee of Al-Azhar Faculty of Medicine. The legal guardians of included neonates provided informed written consent to participate in the study.
The study included 60 full-term neonates diagnosed with PPHN by echocardiography within 72 hours of birth. Patients were excluded if they had major congenital heart disease, non-congenital heart diseases (e.g., endocarditis), sepsis, disseminated intravascular coagulation, low Apgar score and severe hypoxic respiratory failure. In addition, there were 30 age and sex-matched healthy neonates who served as controls.
All participants were subjected to careful history taking and complete clinical examination. Blood was obtained from the peripheral veins at admission.
Laboratory investigations included complete blood count, and blood culture. C-reactive protein (CRP) was assessed using semi-quantitative latex agglutination test (Omega Diagnostics, UK, Catalog no. OD023). Radiological investigations included plain X- ray and two-dimensional transthoracic echocardiography (TTE). Echocardiographic imaging was achieved Affiniti 30 ultrasound machine (Philips, USA) with multi frequency (2.5 MHz) matrix probe (S8-3). Comprehensive TTE M-Mode, Doppler, 2D (continuous and pulsed wave), mapping color flow in the standard and nonstandard views were performed according to American Society of Echocardiography guidelines by the same cardiologist. Pulmonary artery pressure (PAP) was assessed by measuring tricuspid valve regurgitation peak velocity with the use of the modified Bernoulli’s equation: P = 4 x V2 (P; pressure gradient (mmHg), V; blood velocity (m/s). PPHN severity was classified into: mild (pulmonary artery pressure (PAP)=35–50 mmHg, moderate (PAP = 51–70mmHg and severe (PAP > 70 mmHg [8, 9] (Fig. 1). PTX3 level was assessed using enzyme-linked immunosorbent assay (ELISA) commercial kits (Jiaxing Korain Biotech Co, China; Cat No: E1938Hu).
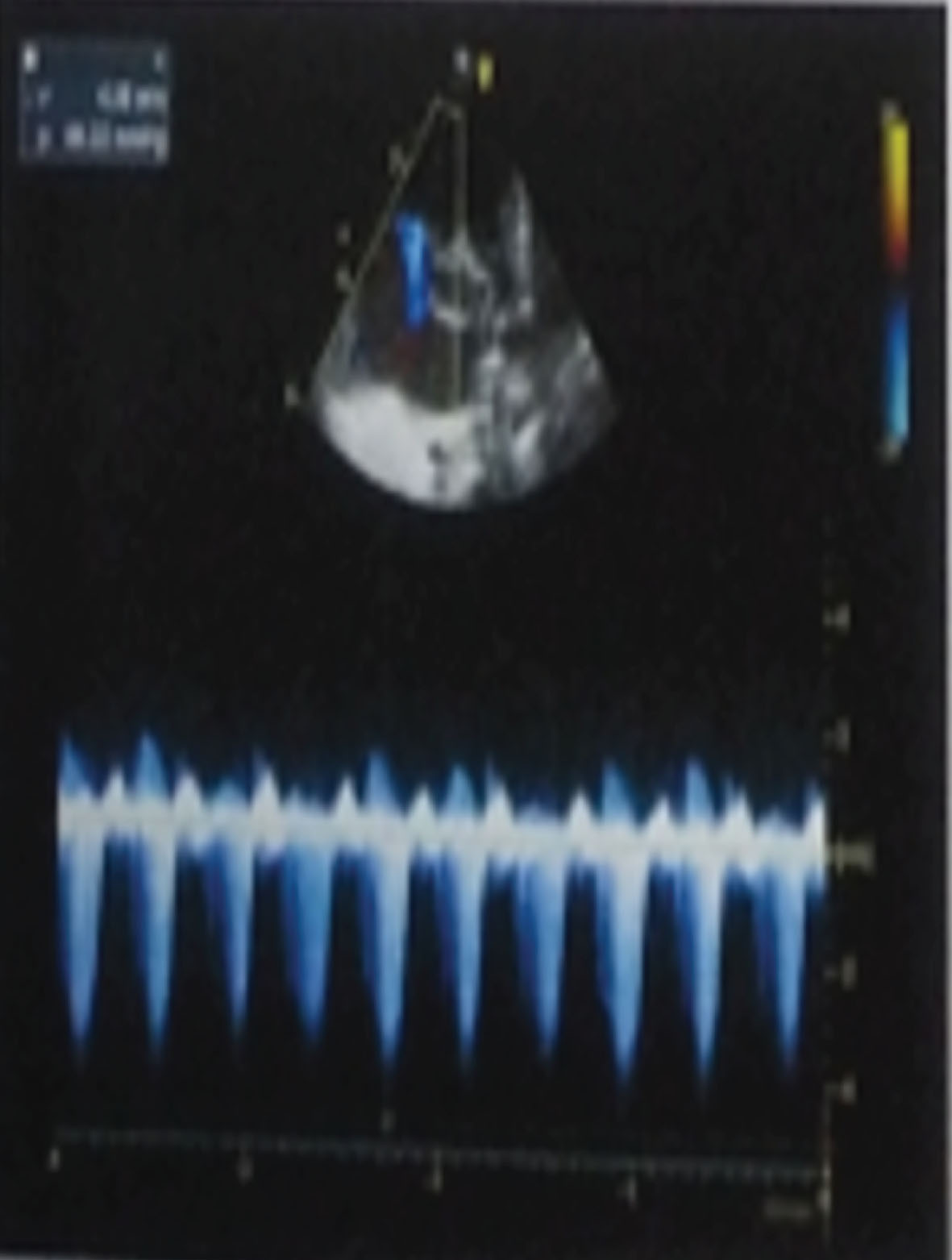
Apical 4-chamber view 2 dimensional with color and continuous wave Doppler showing severe pulmonary hypertension.
Data obtained from the present study were statistically analyzed using Statistical Package for Social Science (IBM SPSS) version 23. Qualitative data were expressed as number and percent and compared using chi-square test or Fisher’s exact test while quantitative data were expressed as mean and standard deviation (SD) and compared using t test or one-way ANOVA as appropriate. Correlation analysis was achieved using Pearson’s correlation coefficient. Receiver operating characteristic curve (ROC) analysis was used to predict the best cut off point for diagnostic marker, its specificity, sensitivity, positive and negative predictive values. P value less than 0.05 was considered statistically significant.
The present study included 60 neonates with PPHN and 30 healthy controls. Comparison between patients and controls revealed that patients had significantly higher CRP (6.12±2.18 versus 3.69±1.25 mg/dl, p < 0.001) and PTX3 levels (2.07±0.67 versus 0.96±0.21, p < 0.001) when compared with controls. No statistically significant differences were found between patients and controls regarding age, sex distribution, weight, Hb, HCT, RBCs, platelets, and WBCs (Table 1).
Clinical and laboratory data in the studied groups
Clinical and laboratory data in the studied groups
CRP: C-reactive protein, Hb: Hemoglobin, HCT: Hematocrit, PDA: Patent ductus arteriosus, PFO: Patent foramen ovale, PPHN: Persistent pulmonary hypertension of the neonate, RBCs: Red blood cells, WBCs: White blood cells.
Correlation analysis identified significant inverse correlation between PTX3 levels and patients’ weight (r = -0.261, p = 0.044). Also, there were significant direct correlations between pentraxin 3 levels and Hb (r = 0.317, p0.013) and HCT (r = 0.291, p = 0.024). No significant correlations were found between PTX3 levels and gestational age, RBCs, platelets, WBCs and CRP (Table 2).
Correlation between pentraxin 3 and clinical and laboratory data
CRP: C-reactive protein, Hb: Hemoglobin, HCT: Hematocrit, RBCs: Red blood cells, WBCs: White blood cells.
Interestingly, patients with associated PDA had significantly higher PTX3 levels when compared with patients without (2.58±0.5 versus 2.02±0.51 ng/ml, p = 0.002). Also, patients with associated PFO had significantly higher PTX3 levels when compared with patients without (2.12±1.05 versus 2.05±0.46, p = 0.002) (Table 3).
Relation between pentraxin 3 and echocardiographic findings
PDA: Patent ductus arteriosus, PFO: Patent foramen ovale, PPHN: Persistent pulmonary hypertension of the neonate.
ROC curve analysis identified good performance of CRP (AUC: 0.802, sensitivity: 70.0%, specificity: 100.0%, PPV: 100.0%, NPV: 62.5%) and PTX3 levels (AUC: 0.990, sensitivity: 100.0%, specificity: 93.33%, PPV: 96.8%, NPV: 100.0%) in diagnosis of PPHN with PTX3 showing better performance (Table 4, Fig. 2).
Value of CRP and pentraxin 3 in prediction of PPHN
AUC: Area under the curve, CRP: C-reactive protein, NPV: Negative predictive value. PPV: Positive predictive value.
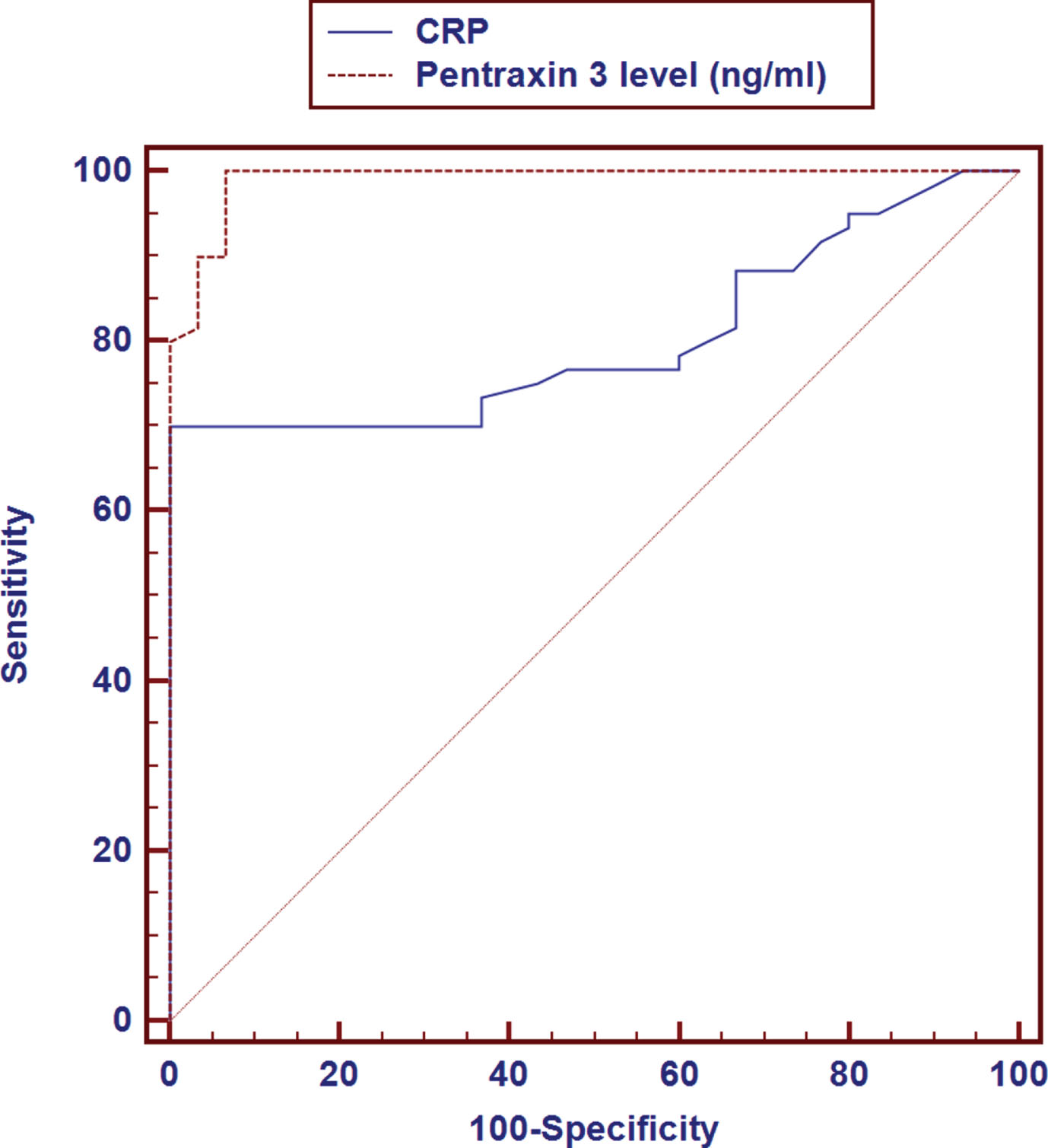
Receiver operator curve (ROC) curve for pentraxin 3 in persistent pulmonary hypertension of the neonate.
PTX3 is soluble pattern recognition molecule with essential roles in multiple biological processes involving innate immune response, regulation of tissue damage and remodeling, and balanced endothelial function [12, 13]. Dysregulation of PTX3 is reported in many pathological conditions affecting the cardiovascular system [14, 15]. In neonates, one study noted a significantly higher PTX3 in very preterm neonates in comparison to healthy term babies [15].
The present study found significantly higher PTX3 in babies with PPHN in comparison to healthy counterparts. These findings are supported other studies conducted on neonatal, pediatric and adult patients.
In neonates, the study of Farhadi et al., [16] assessed PTX3 in three groups of babies including those with pulmonary arterial hypertension, congenital heart disease without pulmonary arterial hypertension and healthy controls. Results showed that the former group had the highest PTX3. Similar conclusions were reported by the study of Wakeel et al [17].
Likewise, the study of Karakurt et al., [18] on older children, found that patients with severe pulmonary hypertension had significantly higher PTX3 and hs-CRP in patients as compared to controls. In adults, Tamura et al., [19] also noted a significantly higher PTX3 in adult patients with pulmonary hypertension in comparison to healthy controls.
Interestingly, the present study identified a significantly higher PTX3 in neonates with PPHN with PDA and PFO when compared with other neonates without associated anomalies. This may be explained by the pronounced inflammatory status observed in neonates with PDA. In one study, Meinarde et al., [20] recognized a significant association between elevated CRP levels and PDA.
Of note, the present study found that PTX3 showed a better performance as a diagnostic marker of PPHN when compared with hs-CRP confirming the suitability of PTX3 in diagnosis of PPHN. In conclusion, the present study found a significant association between serum PTX3 levels and PPHN particularly those with associated PDA or PFO. However, these conclusions may be limited by its cross-sectional design and being single-center study.
Footnotes
Acknowledgments
None.
Consent for publication
Informed consent was obtained from all individuals included in this study.
Competing interests
Authors state no conflict of interest.
Funding
None.
