Abstract
BACKGROUND:
Gentamicin is a commonly used medication in NICUs. It is known to have ototoxic & nephrotoxic side effects. To date there is no consensus about dosing regimen in different institutions. Our study aims to evaluate the Neofax® dosing regimen for gentamicin in neonatal early onset sepsis in relation to trough level before the second dose and its association with the incidence of gentamicin side effects, namely hearing impairment/loss and acute kidney injury.
METHODS:
Retrospective chart review of newborns admitted to Tawam hospital NICU (June 2019–May 2020) who received gentamicin for early onset sepsis (≤72 hours old). Trough levels before the second dose at 24,36 and 48 hours were reviewed (≥1 mg/L is high). Excluded patients with renal risk factors. Side effects (hearing impairment, acute renal injury) were also assessed.
RESULTS:
Total of 265 infants were included, among whom 149 patients received gentamicin at 24 hours interval, 99 at 36 and 17 at 48 hours interval. Trough level was high in 76% (P = 0.022), 65% (P = 0.127), and 53% (P = 0.108) of patients who received gentamicin at 24, 36, and 48 hours, respectively. Hearing screening was normal in 99.2% of patients, while 2 patients failed the test (Both with normal trough levels). No patients in our study developed renal injury related to gentamicin use.
CONCLUSION:
Neofax® gentamicin dosing often results in high trough levels, especially in late preterm/term infants. This study found no correlation between high trough levels and hearing impairment upon discharge or acute kidney injury. Further studies with larger sample size are recommended.
Introduction
Early onset neonatal sepsis, defined as positive blood or cerebrospinal fluid cultures within 72 hours post-birth, poses a life-threatening risk with an incidence of 3 to 4 cases per thousand live births. The absence of specific symptoms, especially in preterm infants, often leads to empirical antibiotic use while awaiting confirmatory cultures. Gentamicin is being commonly used to cover against gram negative bacteria. This study assesses gentamicin dosing and monitoring practices in neonates, contributing vital insights to optimize neonatal sepsis management.
Gentamicin is a bactericidal aminoglycoside antibiotic which is widely used in neonatal intensive care units against gram negative bacteria. It is active against many serious strains such as Enterobacter, Klebsiella, P. Aeruginosa, and Serratia. In combination with beta-lactams, it is the first-choice antibiotic regimen to treat early onset neonatal sepsis [1].
Pharmacokinetics of gentamicin is highly variable in newborns and throughout early infancy, with half-life ranging between 5.4–10 hours, clearance of 0.50 to 1.71 ml/h/kg and distribution volume from 0.4 to 0.7 l/kg. Typically, Half-life decreases by more than 50% in the first 7 – 10 days [1]. Preterm infants have a longer half-life than full-term infants, therefore it is necessary to measure trough levels whenever antibiotics are needed for more than 48 hours duration.
To date, there is no consensus on gentamicin dosing regimen, monitoring, or acceptable serum trough levels, with wide variety of practice in different units. Dosing is usually scheduled according to an empirical guideline, which is then adjusted based on serum levels prior to administration of the second dose [1].
Use of gentamicin is not risk free. Like other aminoglycosides, it is known to be an ototoxic and nephrotoxic medication. The smaller the baby, the higher the impact of sensorineural impairment and renal injury, hence serum levels should be monitored and maintained within standard therapeutic range. Renal injury is usually reversible, while sensorineural hearing loss can be permanent [1–3].
Controversy surrounding the use of gentamicin in newborns is not limited to dosing regimens but also extends to acceptable trough levels. In many centers, trough level below 1 mg/l is suggested to be safe to avoid toxicity, while other centers recommend keeping trough level below 2 mg/l to reduce toxicity. Overall, gentamicin toxicity is found to be less common in newborns compared to older children and adults [4]. Neofax® is a widely utilized, evidence-based medication reference within NICUs. Beyond offering dosage information, it also provides essential data on drug compatibility, pharmacokinetics, and potential adverse effects, all of which are indispensable for making informed decisions in the management of neonates.
In our center at Tawam hospital, we use Neofax® for gentamicin dosing, and check trough level before the second dose, targeting plasma level of less than 1 mg/l. Hearing screening (auditory brainstem response) is done routinely for all patients before discharge. If not done for any technical or logistic reasons, an outpatient appointment would be scheduled as soon as possible after discharge. We do check renal function at least once in the first 24 – 48 hours of age, and subsequent follow up repeats being individualized according to the clinical condition of the patient and associated risk factors. Urine output is strictly monitored for all patients within the first 48 hours of age.
Thus, we performed this study to evaluate our gentamicin dosing regimen for early neonatal sepsis as per Neofax® in relation to achieving acceptable trough level before the second dose, and to assess for incidence of side effects of the medication (ototoxicity and nephrotoxicity) compared to trough level as well.
This study is adding to the currently available pool of knowledge about gentamicin use in early onset sepsis in neonates. To our knowledge, there is no current literature from the region evaluating gentamicin dosing regimen recommended by Neofax® in this age group. Our study is aimed towards providing evidence about gentamicin dosing regimen, besides the timing and frequency of gentamicin serum level measurement.
Materials and methods
Retrospective study done in a single center in the United Arab Emirates (UAE), gained ethical clearance from Tawam Human Research Ethics Committee (T-HREC), UAE and none of the study’s procedures violated the main principles of the Declaration of Helsinki [5]. The need for written informed consent was waived owing to the retrospective nature of the study.
Study design and patients’ selection
A chart review was conducted to retrieve data of all newborns who were admitted to neonatal intensive care unit at Tawam hospital during the period between June 2019 and May 2020. Infants were included in this study if received gentamicin for treatment of suspected, clinical, or proven early onset sepsis (within the first 72 hours of life). We excluded patients who received gentamicin for late onset sepsis, patients with major congenital renal, cardiac, or metabolic abnormalities, or other congenital anomalies requiring surgery within the first 3 days of life, grade III – IV intraventricular hemorrhage on early scan, birth asphyxia requiring therapeutic hypothermia treatment, or concomitant use of medications that might affect renal function (inotropes and nephrotoxic medications).
Data collection
Data of eligible patients were collected using an electronic form and included the following: age, gender, birth weight, mode of delivery, blood culture, gentamicin trough level, hearing screen report, urine output, and creatinine level before and after gentamicin use. Gentamicin trough level of 1 mg/L or above is considered high. Acute kidney injury is defined as absolute, abrupt (within 48 hours) increase in serum creatinine of more than 0.3 mg/dL, or a percentage increase of > 50% and/or urine output < 0.5 mL/kg/hr. Hearing screen upon discharge was classified into either passed or failed.
Study outcomes
The primary outcome of this study is to evaluate gentamicin dosing regimen for early onset sepsis, as per Neofax®, in relation to gentamicin trough level before the second dose, while secondary outcomes aimed to assess the incidence of hearing impairment/hearing loss and acute kidney injury in relation to gentamicin use and/or trough levels before next dose.
Statistical analysis
Analysis was executed using Minitab software. Continuous data were described using measures such as mean and standard deviation (SD), while categorical or attribute data were summarized using counts and percentages. For proportion testing, the contingency table approach was applied, using the Chi-square test. The Test of Equal Variance was another primary method employed to assess the similarity in variances between different groups. For all hypothesis testing conducted in this study, the null hypothesis consistently assumed equality. The standard for statistical significance was set at a P-value of less than 0.05. Results below this threshold indicated a rejection of the null hypothesis. Through the application of these methodologies, we ensured our analysis was both rigorous and relevant to the study’s objectives.
Results
A total of 265 infants (154 males, 111 females) fulfilled our inclusion criteria, with a male to female ratio of 1.38 : 1 (58.1% males, 41.9% females). Majority of patients included were born via lower segment caesarean section – LSCS – (n = 174, 65.7%). Late preterms (LPT: 34 0/7 – 36 6/7 weeks) and early term infants (ET: 37 0/7 – 38 6/7 weeks) combined stands for more than 50 % of the sample size (n = 135, 50.9%), while moderate preterms (MPT: 32 0/7 – 33 6/7 weeks) represents 14.3% (n = 38), full term infants (FT: 39 0/7 – 40 6/7 weeks) 13.6% (n = 36), very preterms (VPT: 28 0/7 – 31 6/7 weeks) 12.8% (n = 34), extreme preterms (EPT:<28 weeks) 4.2% (n = 11), late term infants (LT: 41 0/7 – 41 6/7 weeks) 3.8% (n = 10), and post terms (PoT:≥42 0/7 weeks) only 1 patient (0.4%) – [Figs. 1, 2].
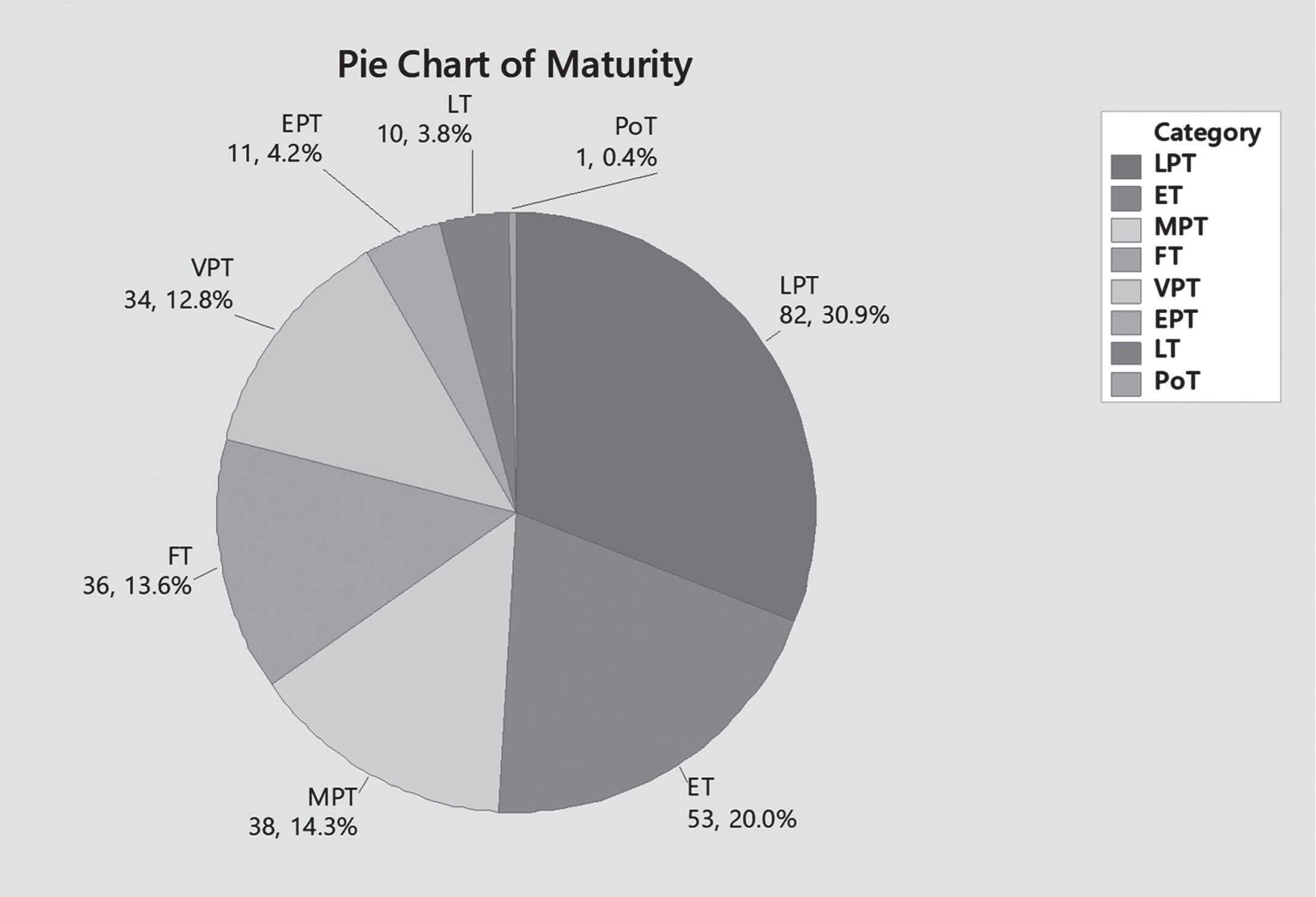
Maturity.
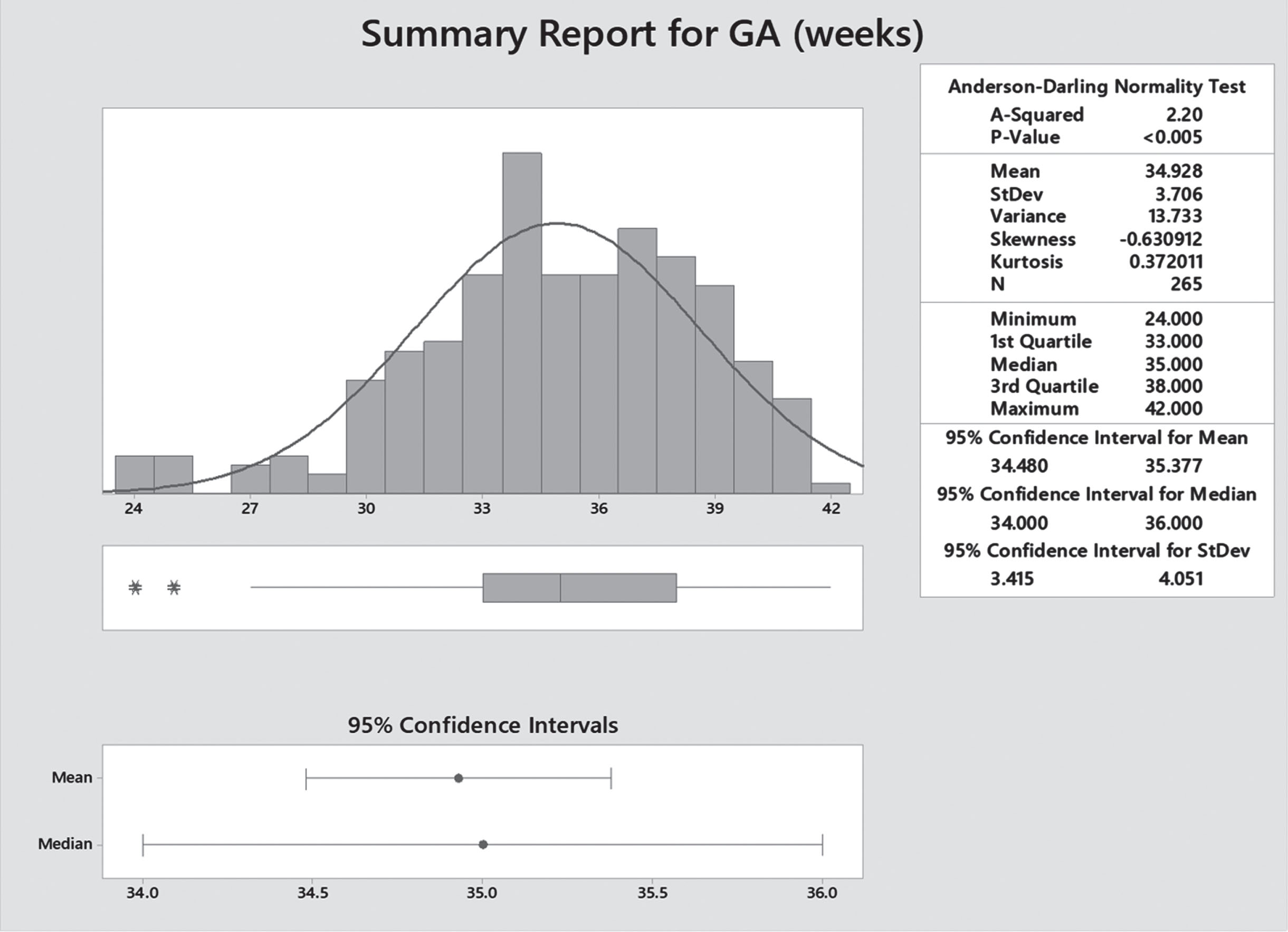
Gestational age distribution.
The vast majority of our patients were born with birth weight ranging between 1,500 – 3,999 gm (low birth weight to normal birth weight range) –86.5% (n = 229), while extremes of weight represented the rest of the sample size – less than 15% as shown in below chart [Fig. 3].
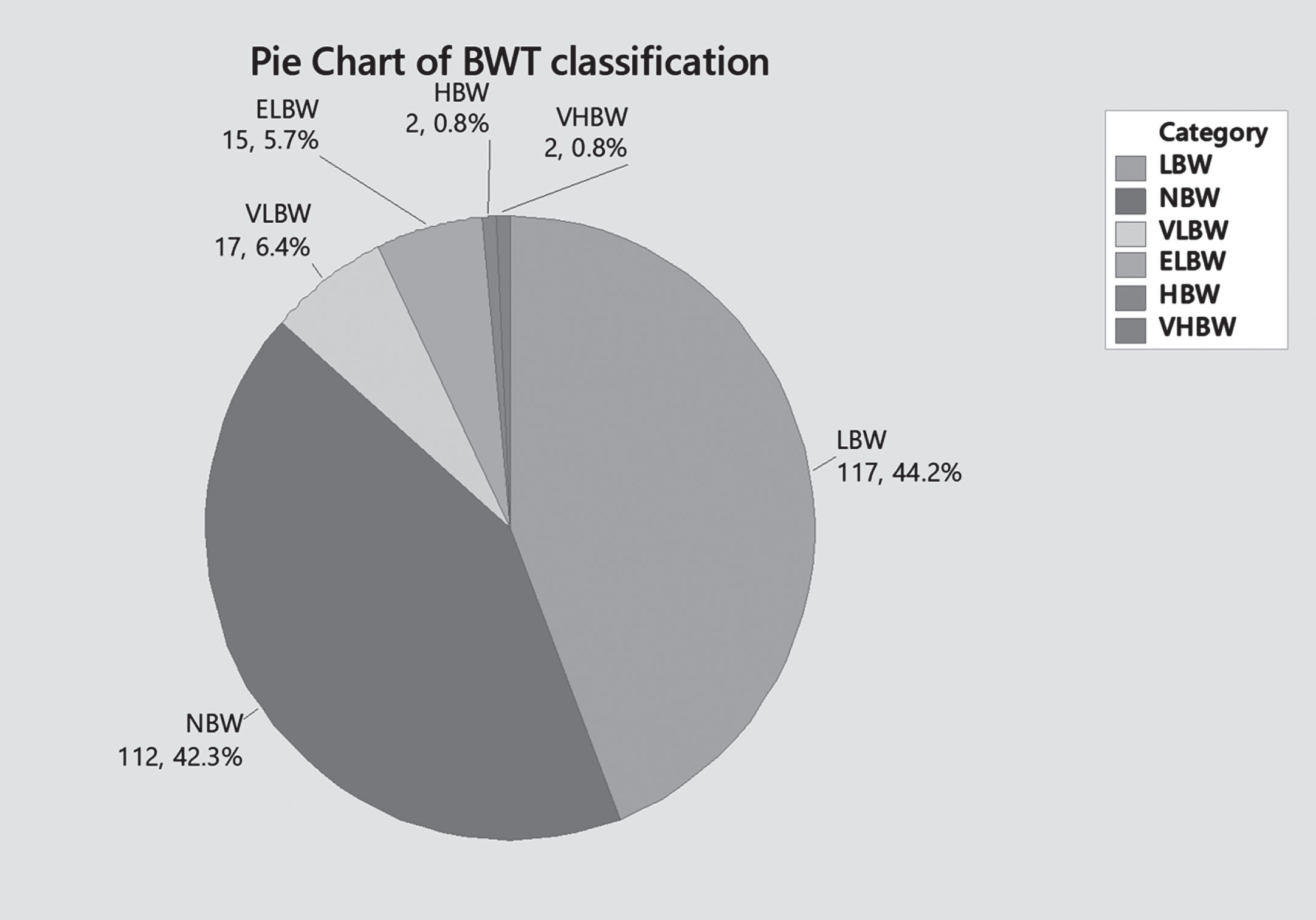
Birth weight.
Neofax® dosing regimen for gentamicin is based on gestational age at birth, hence 56.2% (n = 149) patients received once daily dose of 4 mg/kg (born at 35 0/7 weeks’ gestation and above), 37.4% (n = 99) who were born between 30 0/7 weeks and 34 6/7 weeks gestation received 4.5 mg/kg Q36 hours, and the remaining 6.4% (n = 17) received 48 hourly dosing regimen with 5 mg/kg [Fig. 4].
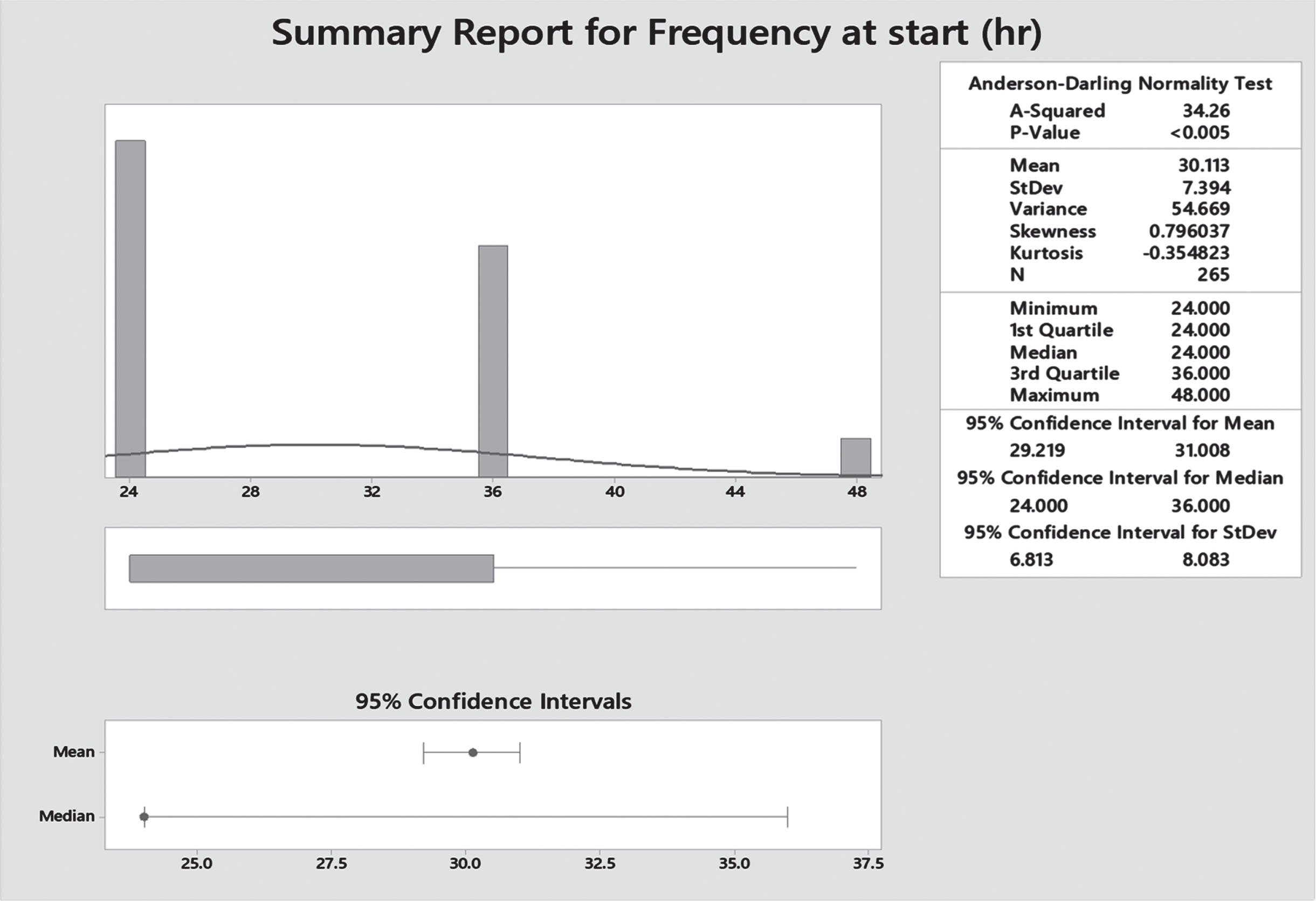
Gentamicin frequency.
Trough level at 24 hours was high in 75.8% (n = 113), and normal in 24.2% (n = 36) of cases, P = 0.022. Whenever trough level is high, gentamicin is spaced out and trough level is repeated 12 hours later, unless antibiotic is discontinued before that [Figs. 5, 6].
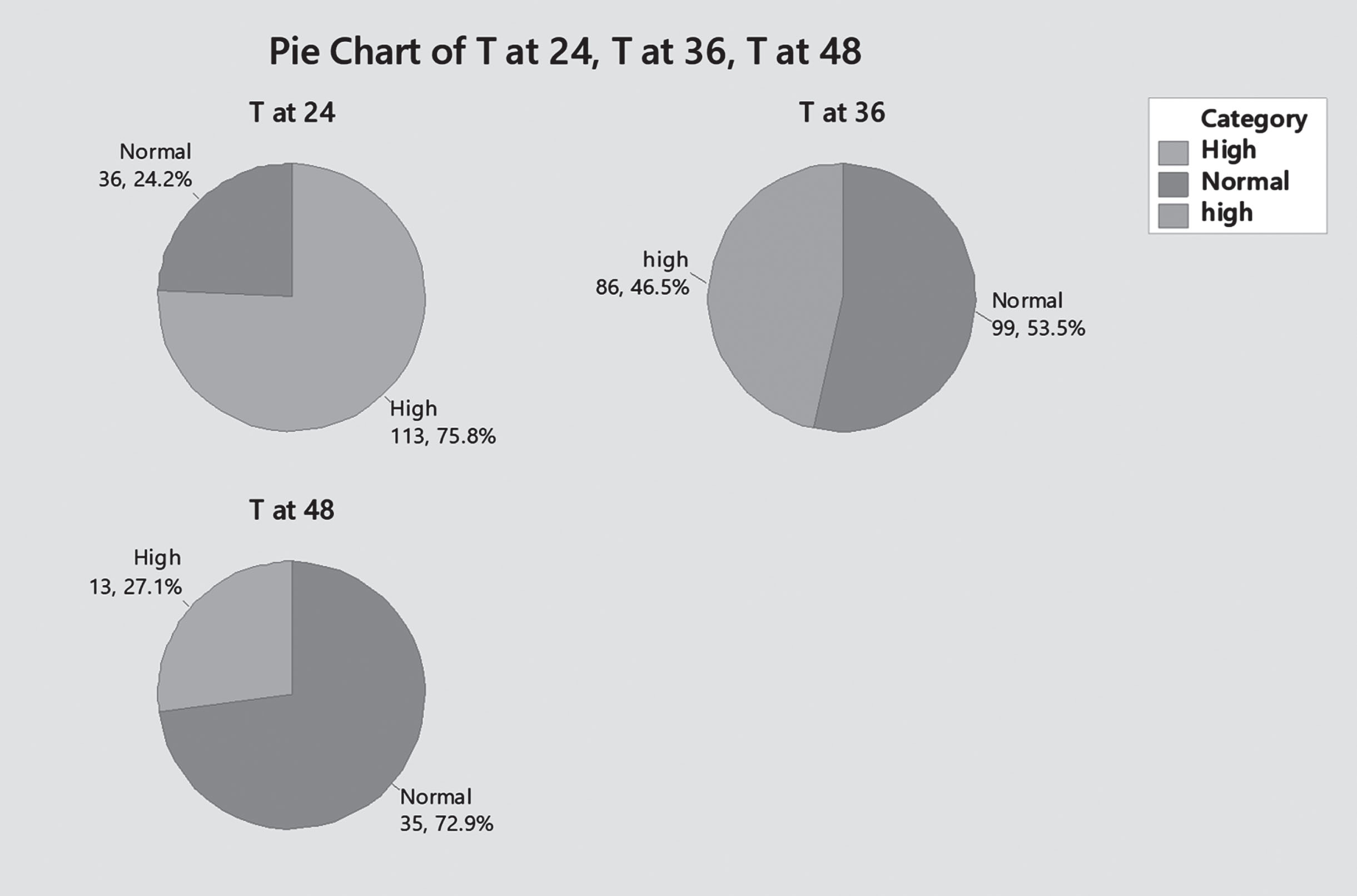
Trough level at 24, 26, and 48 hours (High/Normal).
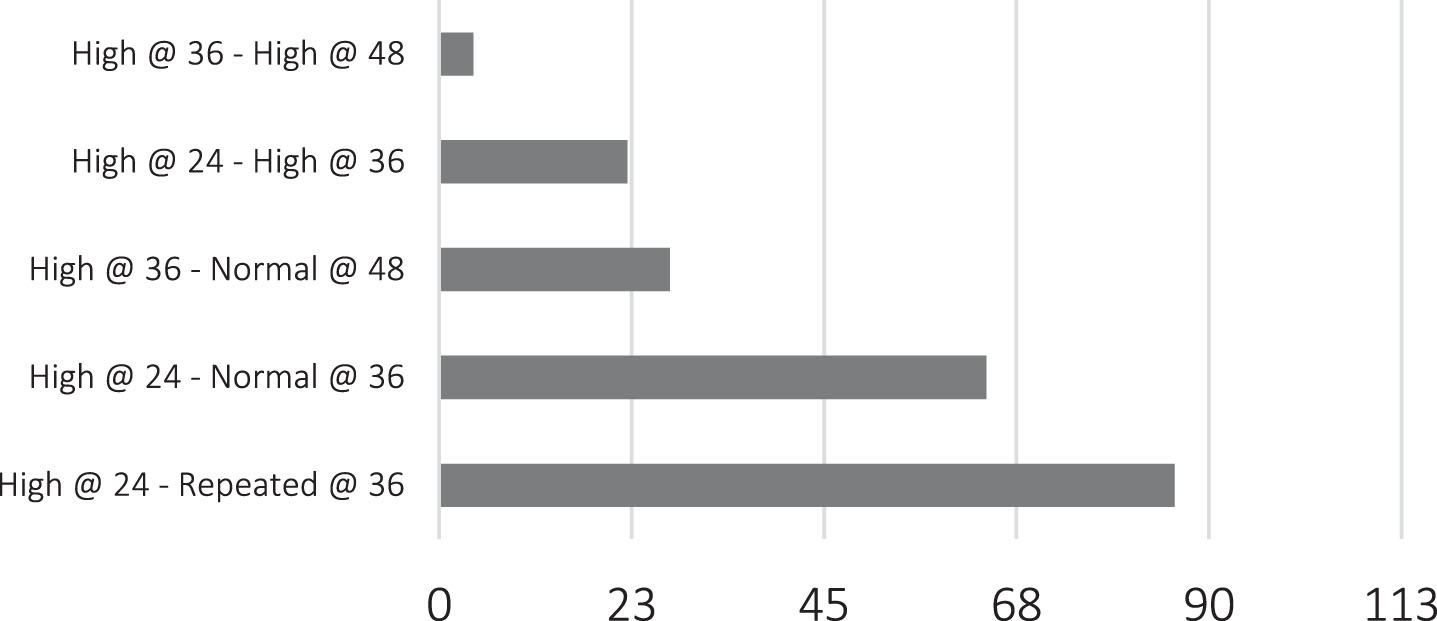
Repeated gentamicin trough levels.
Gentamicin trough level for patients scheduled at 36 hourly doses was high in 65% (n = 64) and normal in 35% (n = 35) of patients, P = 0.127. Taking in count the total number of trough levels done at 36 hours (99 patients scheduled at 36 hours+86 patients with high trough levels at 24 hours and repeated at 36 hours), 53.5% (n 99) had normal level – out of which –34.6% (n 64) had elevated level at 24 hours, and 46.5% (n 86) had high trough level – out of which 11.9% (n 22) had high level at 24 hours as well –P. value 0.008. [Figs. 5, 6].
Gentamicin trough level for patients scheduled at 48 hours was high in 53% (n 9) and normal in 47% of patients (n 8), P = 0.108. Taking in count the total number of trough levels done at 48 hours (15 patients scheduled at 48 hours+31 patients with high trough level at 36 hours and repeated at 48 hours), trough level was normal for 72.9% (n 35) – out of which, 56.3% (n 27) had high level at 36 hours, and high in 27.1% (n 13) – of which 8.3% (n 4) had high trough level at 36 hours of age as well (P – value 0.611) [Figs. 5, 6].
In our study sample, hearing screening was done for most of patients before discharge –95.5% (n 253). It was normal in 99.2% (n 251) and failed in 2 patients. Hearing screening result was not related to gentamicin trough level in this study (Both patients who failed hearing screening had normal gentamicin trough levels) [Fig. 8].
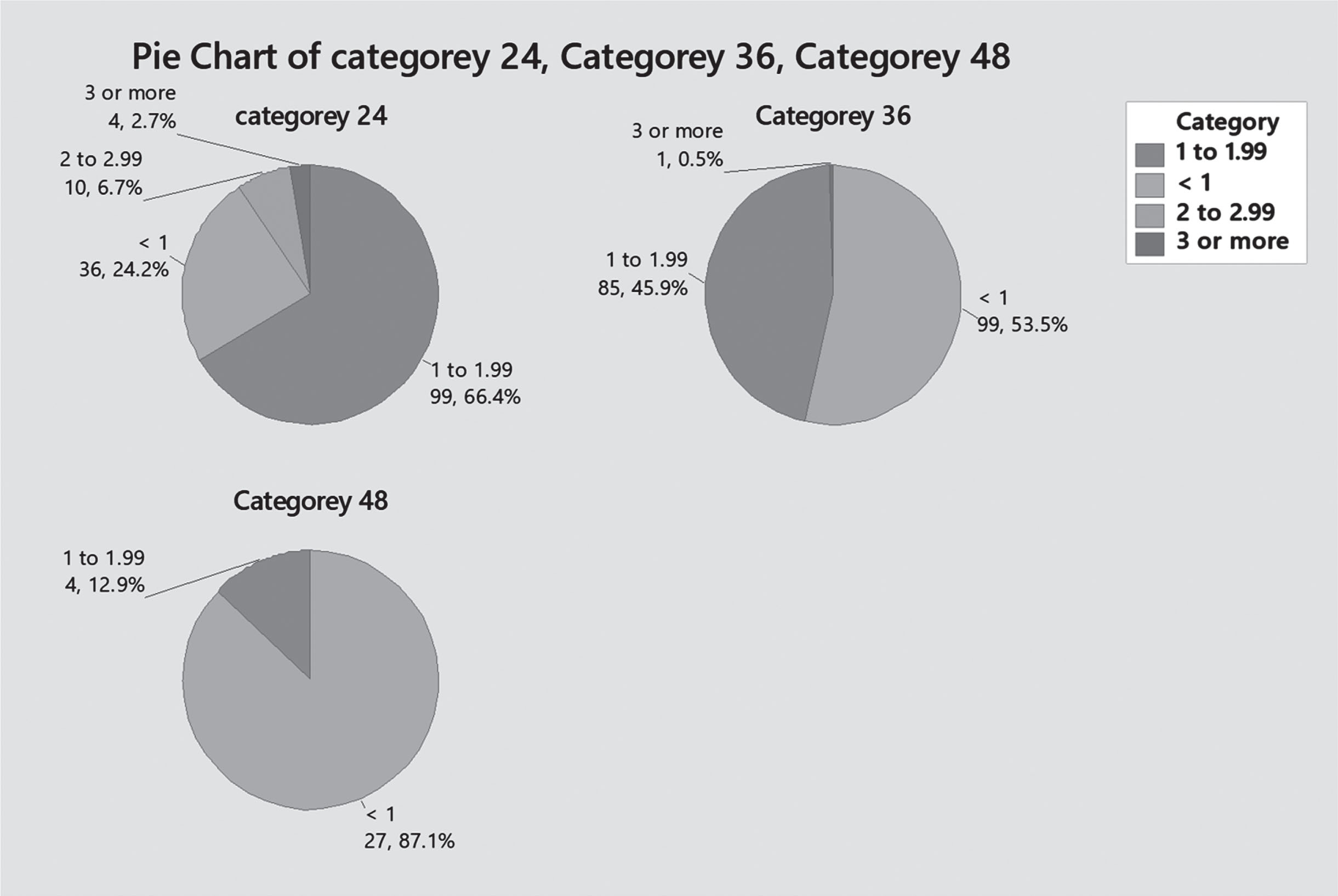
Trough level at 24, 26, and 48 hours (Level categories).
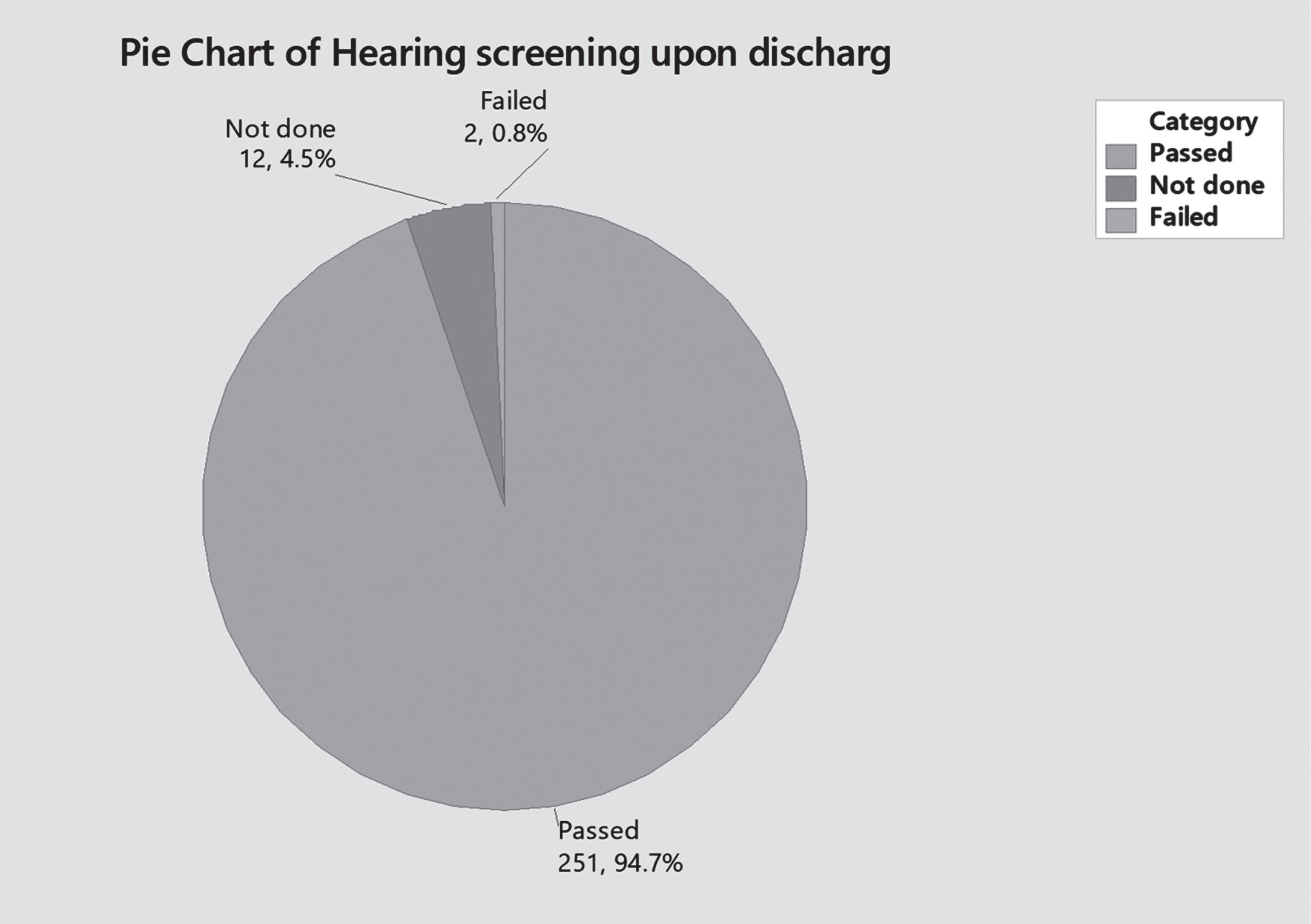
Hearing screening.
To evaluate the effect of gentamicin on renal function, we compared creatinine levels and urine output before and after use of the medication. Most patients in the study sample –92.1% (n 244) – did not have more than one creatinine level done, unless having other comorbidities which mandates their exclusion. In this study there was no cases with acute kidney injury that could be attributed to use of gentamicin or high trough levels, as per definition specified above [Figs. 9, 10].
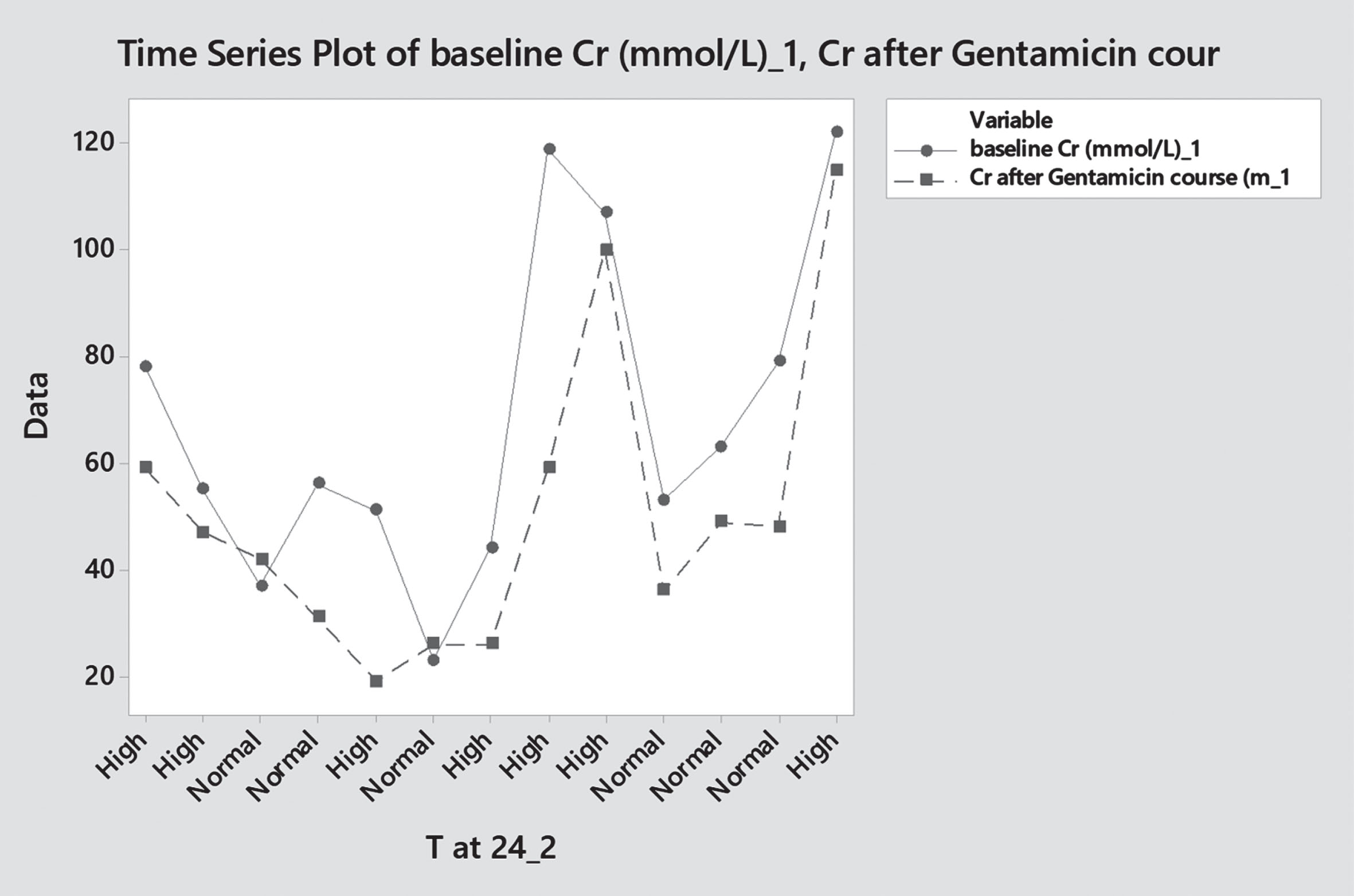
Pre and post Gentamicin creatinine levels – category 24 hourly dose.
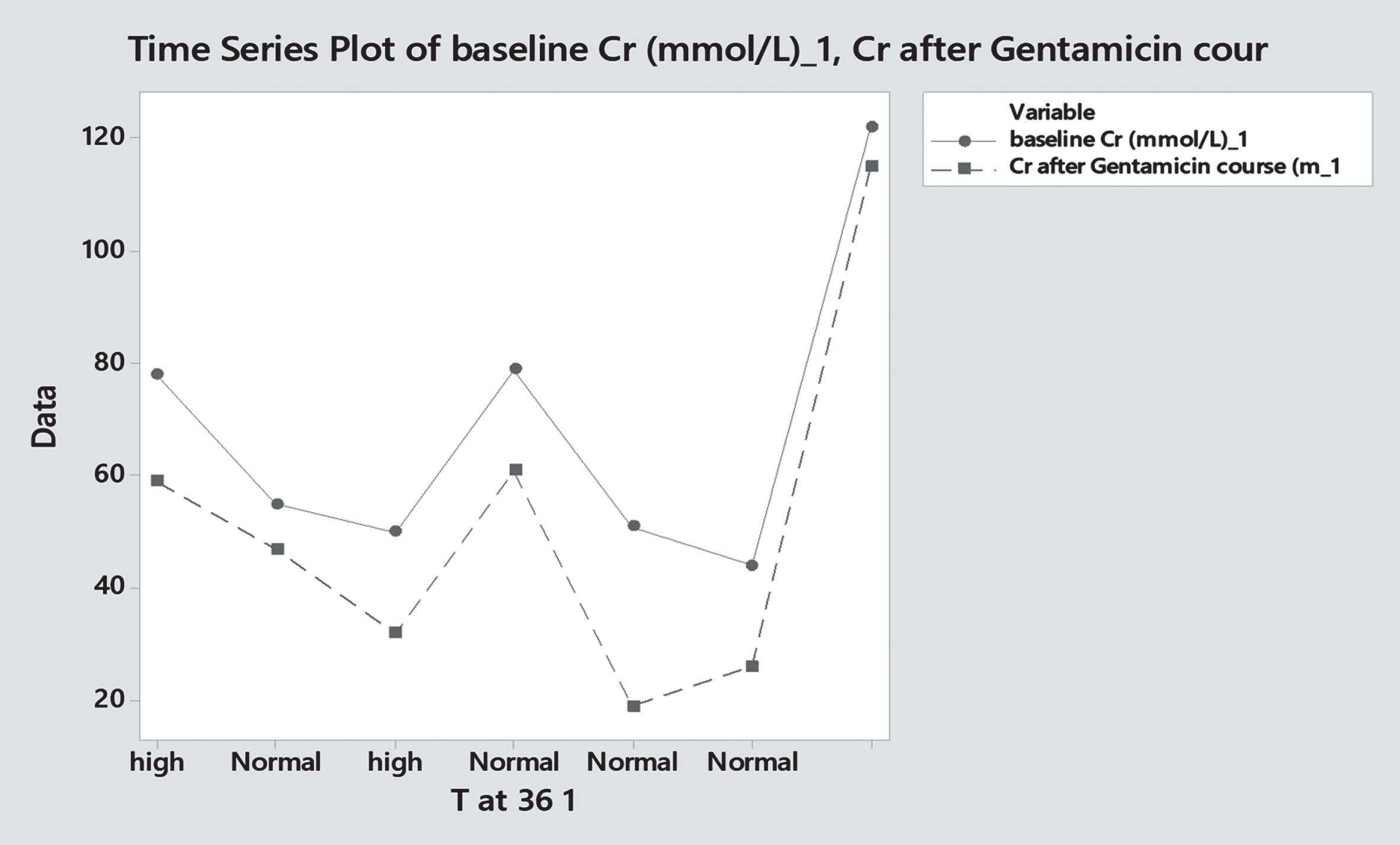
Pre – and post Gentamicin creatinine levels – category 36 hourly dose.
Gentamicin remains one of the most frequently used antibiotics in neonatal units to treat gram negative infections, given its reliability and being familiar to most neonatologists. Till date there are multiple different dosing schemes being in use based on different pharmacokinetic studies and empirical recommendations.
A peak plasma level of 4 – 10 mg/L is considered a safe therapeutic range, while trough levels, just before the next dose, must be lower than 1 mg/L to avoid toxic effects (lower than 2 mg/L in some) [6]. Available evidence supports use of extended interval dosing regimen over multiple daily dosing for newborns [6, 7]. In our unit, we are using the standard dosing regimen adopted by Neofax® [8].
In this study we evaluated our dosing scheme of gentamicin, in comparison to achieving acceptable trough level of less than 1 mg/l. We found that in most cases, this regimen resulted in high trough levels before second dose, which is more evident with the more frequent doses (Daily dosing regimen were more likely to have high trough levels compared to patients receiving 36 hourly dosing regimen, who in turn had more incidences of high trough levels than 48 hourly dosing regimen).
Gentamicin has well known ototoxic and nephrotoxic side effects. Dose and frequency usually being adjusted after checking drug level, aiming to minimize such side effects. Cooper et al. Found that otoacoustic emission failure was almost 4 times higher in patients with low birth weight compared to normal birthweight infants when exposed to high gentamicin dose (43.1% vs. 9 %, respectively) [9].
Gentamicin trough level analysis
Gentamicin trough level analysis
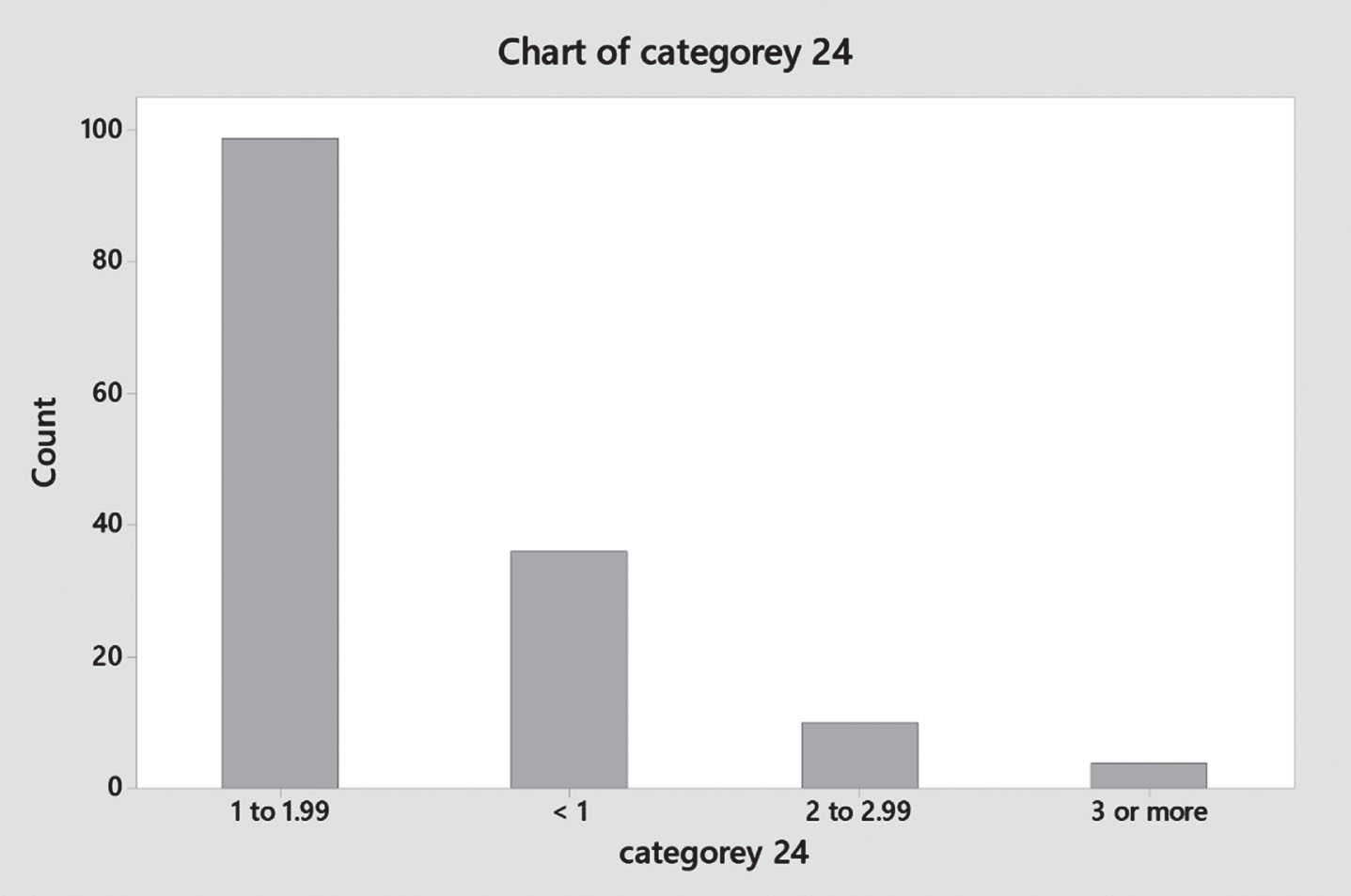
Gentamicin trough level distribution (Q24 hours).
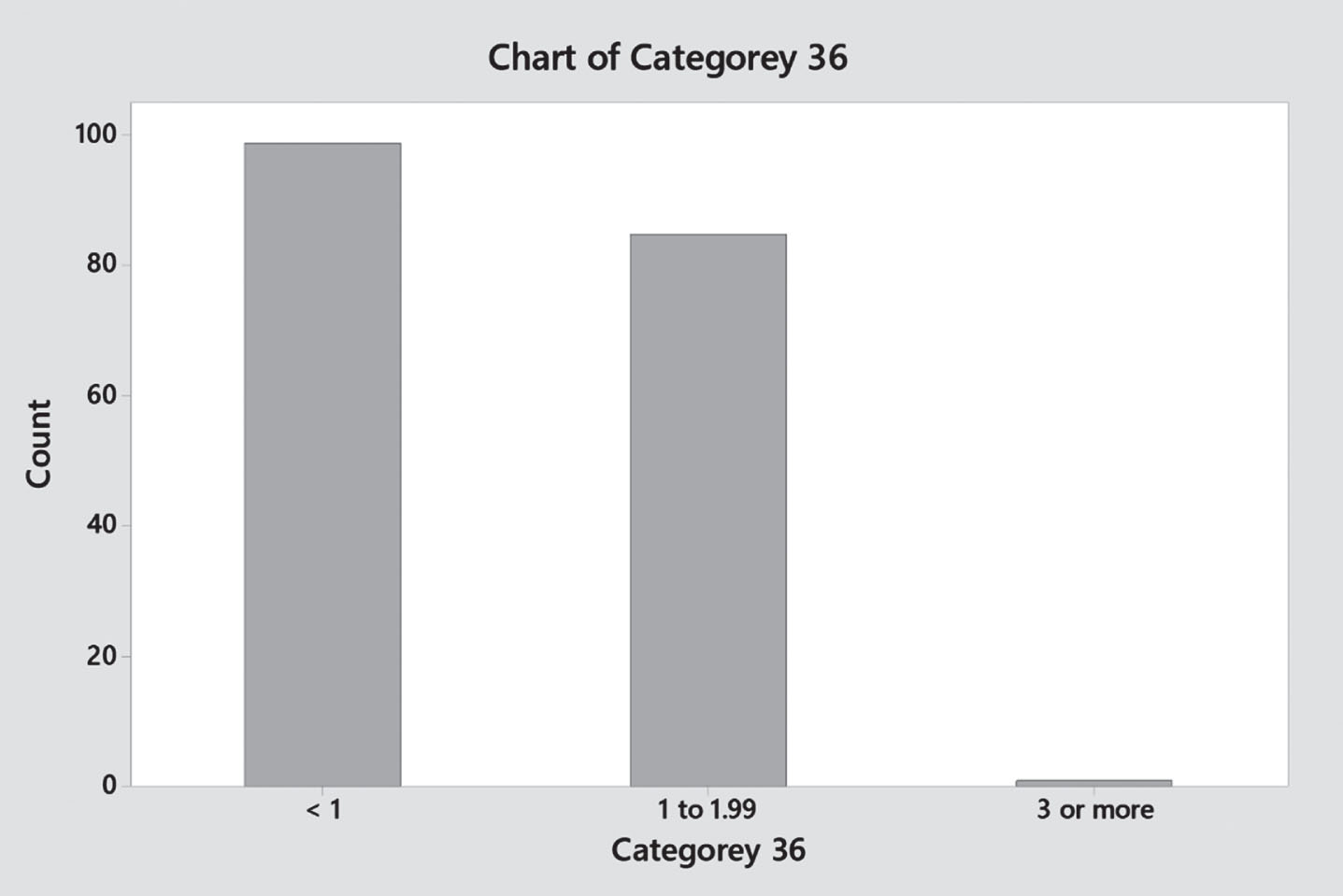
Gentamicin trough level distribution (Q36 hours).
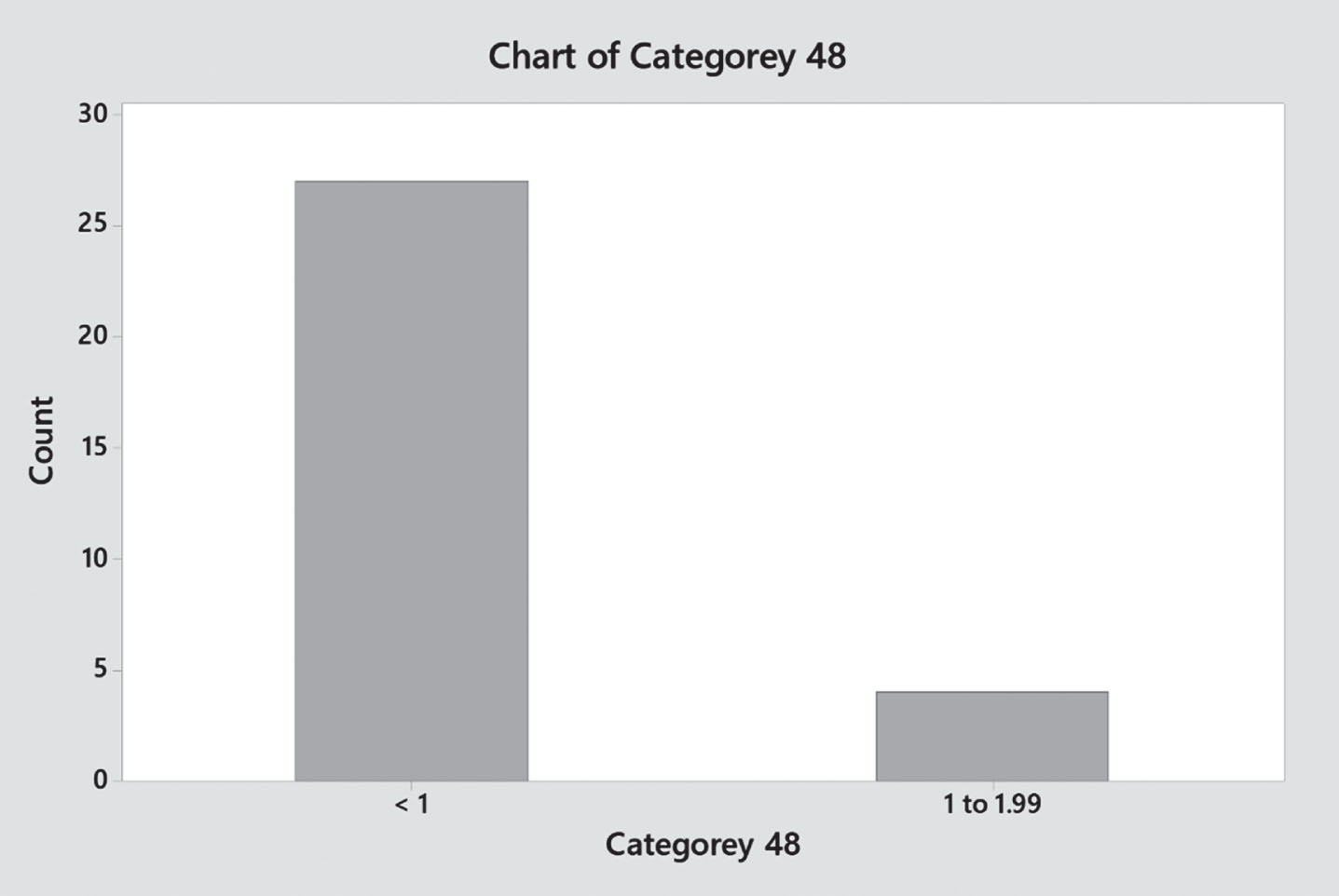
Gentamicin trough level distribution (Q48 hours).
On the other hand, Aust et al. Reported that, out of 134 patients received gentamicin in neonatal period, 8 of them had abnormal hearing screening studies. All of them having history of other risk factors for hearing loss (meningitis, asphyxia, significant jaundice, severe acidosis) [10]. Kohelet et al., Colding et al. and Finitzo-Hieber et al. described comparable results with no difference in auditory outcomes when corrected for the other risk factors in control group [11–13]. In this study we had equivalent results with no direct association between hearing loss and gentamicin trough level. Incidence of hearing loss in our study sample was very minute, less than 1% of the sample size –2 patients. Both patients with abnormal hearing screening upon discharge had normal gentamicin trough levels.
Gentamicin is also known to be nephrotoxic as well. Tugay et al. Reported that serum peak and trough levels were positively correlated with worsening renal function biomarkers, namely: serum creatinine, urine albumin/urine creatinine ratio, fractional excretion of sodium, fractional excretion potassium and urine calcium/urine creatinine. [14]. This effect can be potentiated in presence of other comorbidities, such as hypoxic ischemic encephalopathy, prematurity, and being small for gestation. Patent ductus arteriosus does affect half-life and distribution volume of Gentamicin, but there is no convincing evidence of increased nephrotoxicity so far [15–18]. In this study we used rate of creatinine raise and urine output to define acute kidney injury. In our sample we had a limited number of patients with repeated creatinine levels –7.9% (n 18) patients. None of our patients in the study developed acute kidney injury. Further studies with larger sample size are needed for further evaluation of this effect.
Limitations related to this study are mainly due to the retrospective nature of the study, with restriction of the source of information to the medical records, which in turn might lead to misclassification and recording bias. Another limitation is that the number of patients with repeated creatinine levels in the sample under study was limited. Thus, a larger number of patients might be helpful for more accurate results. However, scarcity of such data on common medications used in newborn period from the middle eastern geographic location cannot be denied, and hence on the positive side, our study does contribute to this aspect.
Conclusion
Based on the above results, we conclude that Neofax® standard gentamicin dosing regimen might not be the best reference for gentamicin dosing in early neonatal infections, frequently resulting in high trough levels above the targeted level, especially in late preterm and term infants with more frequent doses. In this study, high gentamicin trough level before second dose was not related to abnormal hearing screening upon discharge, though further follow up is needed. Also, we did not find any direct causative relationship between gentamicin use and acute kidney injury in our limited study. Further studies (possibly prospective) with larger sample size are recommended.
Footnotes
Acknowledgments
We would like to express our gratitude to all those who have supported and contributed to this research. We would like to thank our colleagues for their valuable insights and expertise, and our deepest appreciation goes to our patients whose data been used in this study. We are deeply grateful to everyone who has helped make this research possible.
Disclosure
The authors have no conflicts of interest to disclose. The study was conducted independently and without any financial or other support that could bias the results. The authors have not received any funding or other financial support for this research and have not received any honoraria or other forms of compensation related to the publication of this paper.
