Abstract
BACKGROUND:
Few studies characterize feeding performance in the NICU when predicting neurodevelopmental outcomes. Our objective was to investigate the relationship between time to full oral feeds (FULL-PO) and neurodevelopmental and feeding outcomes in the first 2 years in preterm infants admitted to the NICU.
METHODS:
This retrospective study included infants born between 01/01/2014-07/31/2017, gestational age < 32 weeks and/or birth weight < 1500 g. We examined feeding difficulties, cerebral palsy, and Bayley scores for those reaching FULL-PO at a post menstrual age (PMA)≤38.0 weeks (EARLY) vs.>38.0 weeks (LATE). Additionally, the oral feeding achieved at various timepoints between 36- and 42-weeks postmenstrual age (PMA) was measured to construct a timeline of oral feeding acquisition.
RESULTS:
Of 192 infants, 147(77%) achieved FULL-PO EARLY and 45(23%) LATE. Comorbidities and length of stay were higher and unadjusted Bayley scores were lower at 12 months corrected age (CA) and 24 months chronological age (CH) in the LATE group. Feeding difficulties were higher in the LATE group at 24 months CH. Infants born < 27–28 weeks GA were more likely to achieve oral feeding at a later PMA. Infants with bronchopulmonary dysplasia (BPD) had significant feeding and developmental delays.
CONCLUSIONS:
Establishing full oral feeds by 38.0 weeks PMA may be used as a predictor for feeding difficulties at 24 months CH. Infants born < 27–28 weeks GA and those with BPD are more likely to take extended amounts of time to achieve full oral feeding and need additional feeding support. Infants with BPD are high risk for neurodevelopmental delays.
Introduction
Extremely preterm infants’ survival rates have increased to as high as 36% as we have continued to learn how to optimize care through advances in research and technology [1]. However, the morbidity remains unchanged and concern for the presence of neurodevelopmental delays and cerebral palsy persists [1, 2]. Thus, we continue to seek a reliable means to predict outcomes and identify those infants who will benefit the most from early intervention.
An infant’s feeding ability is one of the earliest complex motor skills and with shared neural path-ways may provide insight into an infant’s neurodevelopmental capacity [3]. Initially, studies demonstrated an association between a history of feeding difficulties at follow-up and risk of language delay in preterm infants on neurodevelopmental assessment [4, 5]. Subsequently, it has been shown that feeding method at discharge alone can be associated with feeding issues and neurodevelopmental delays [6]. Specifically, the presence of a gastrostomy tube (GT) and medical comorbidities such as bronchopulmonary dysplasia (BPD), and intraventricular hemorrhage (IVH) in younger gestational age (GA) infants have been associated with delays [6, 7]. However, these studies have not investigated the specifics of oral feeding attainment such that we can predict the time at which preterm infants with and without certain comorbidities will achieve full oral feeding. This lack of knowledge results in frustration from the medical team and family as decisions regarding means for supplemental feeding and a prolonged hospital stay arise.
Recent studies are shifting focus to characteristics of infant oral feeding ability during NICU hospitalization to implement early interventions that result in improved neurodevelopment and feeding outcomes [8, 9]. With ELBW infants at a high risk for requiring GT placement, it becomes important to identify strategies for supporting oral feeding and motor skills in the NICU [7]. While the presence of speech language pathologists and occupational and physical therapists is required in high level NICUs and has shown overall benefit to patients, the most effective components of interventions for preterm infants are less clear [10, 11]. Additional research is needed to elucidate which oral feeding approach, therapies and frequency will impact long term outcomes.
The purpose of this study was to investigate if timing of oral feeding acquisition was a predictor for neurodevelopment and feeding difficulties in preterm infants; secondarily, we constructed models to demonstrate the time at which full oral feeding can be expected in preterm infants. We hypothesized that infants < 32 weeks and/or birth weight < 1500 g who did not achieve full oral feedings by 38.0 weeks PMA would have neurodevelopmental delays and feeding issues at 12 month CA and 24 month CH follow-up. The PMA of 38.0 weeks was chosen given prior studies that have shown attainment of full oral feeds of healthy preterm infants by approximately 34–38 weeks PMA and those with comorbidities having a wide range from 38 to 47 weeks PMA [12–16].
Patients and methods
This is a retrospective chart review. Data from premature infants admitted to the University Hospital Level IV NICU between January 1, 2014 and July 31, 2017 were evaluated. We included in-born infants with GA < 32 weeks and/or birth weight < 1500 g who subsequently completed at least 1 visit at our neurodevelopmental follow-up program at 12 months CA and/or 24±6 months CH. Exclusion criteria included infants born at an outside facility and those with major congenital or chromosomal anomalies, severe intrauterine growth restriction, or death prior to discharge. This study was approved by the Institutional Review Board of University of Texas Health San Antonio (San Antonio, TX).
Neonatal and maternal data were systematically collected from the electronic medical record. The data were entered into the secure Research Electronic Data Capture (REDCap) system. Prenatal characteristics including antenatal steroid use and maternal conditions were abstracted. Infant characteristics obtained included gender, GA, and anthropomorphic measurements. Infant morbidities collected were necrotizing enterocolitis (NEC, defined as Bell’s classification stage≥2), IVH (grade 3 or 4), periventricular leukomalacia (PVL), retinopathy of prematurity (ROP), postnatal steroid use, need for supplemental oxygen and BPD. BPD was defined as the need for oxygen supplementation at≥36 weeks PMA.
Nutritional characteristics obtained included day of life enteral and oral feedings were initiated and day of life full enteral and oral feedings were achieved. Full enteral feeds were defined as attaining a volume of 120 ml/kg/day. Achievement of full oral feeds was defined by the absence of gastric tube feeding for 24 hours in the presence of an oral intake≥120 ml/kg/day. This volume of oral intake is considered a standard minimum value at our site for the infant to consume by mouth and be able to achieve adequate hydration. Diet and feeding method at discharge were examined. Additionally, the day of life breastfeeding was initiated and presence of any breastfeeding at discharge were collected. The percent oral intake at 36-, 38-, 40-, and 42-weeks PMA were calculated until the infant was discharged. Infants achieving FULL-PO≤38 weeks PMA were categorized as the FULL-PO EARLY group. Infants achieving FULL-PO > 38 weeks PMA were categorized as the FULL-PO LATE group. Infants requiring a GT were included in the FULL-PO LATE group if the infant did not achieve an oral intake ≥120 ml/kg/day before NICU discharge.
The primary outcome evaluated was adverse neurodevelopmental outcome at 12 months CA and 24 months CH defined as the presence of cognitive, language or motor delays. The Bayley Scale of Infant Developmental testing, edition III (BSID III) was administered to determine cognitive, language and motor delays with composite scores of < 85 used as a threshold to define delay. Cerebral palsy is defined as a nonprogressive central nervous system disorder characterized by abnormal muscle tone in≥1 extremities and abnormal control of movement or posture. We included the diagnosis of cerebral palsy if it was described as present in the patient by the practitioner in the clinic note. Presence of feeding difficulties was collected at follow-up. Feeding difficulty was included if the provider documented gastric tube usage, diagnosis of oral aversion, feeding refusal, or coughing, choking or gagging with feeds.
Statistics
SPSS version 26.0 and SAS version 9.4 were utilized for statistical analyses. In order to detect a 20% decrease in the incidence of speech delay at 24 months CH with a type-I error of 0.05 and statistical power of 80%, 93 patients were required in each group for a total of 186 patients. Student’s t-test was used for analysis of continuous data and chi-square or Fisher exact test for categorical data. Multivariate analysis was performed to examine the relationship between oral feeding attainment and language, cognitive, and motor BSID III scores while adjusting for confounding variables using stepwise linear and logistic regression for continuous and categorical variables, respectively. Covariates utilized were GA, BPD, NEC, and postnatal dexamethasone days. Data is presented as mean±s.d., or as stated. P < 0.05 was considered significant.
Results
Study population
A total of 381 patients were born and admitted to our NICU January 1, 2014 to July 31, 2017 (Fig. 1). Three hundred forty-six infants qualified for inclusion. Of these patients, 153 were lost to follow-up or had incomplete developmental assessments and 1 infant was excluded for severe SGA. A total of 192 patients were included in the analysis. One hundred and eighty-eight infants were discharged on full oral feeds and 4 infants were discharged with GT feeds.
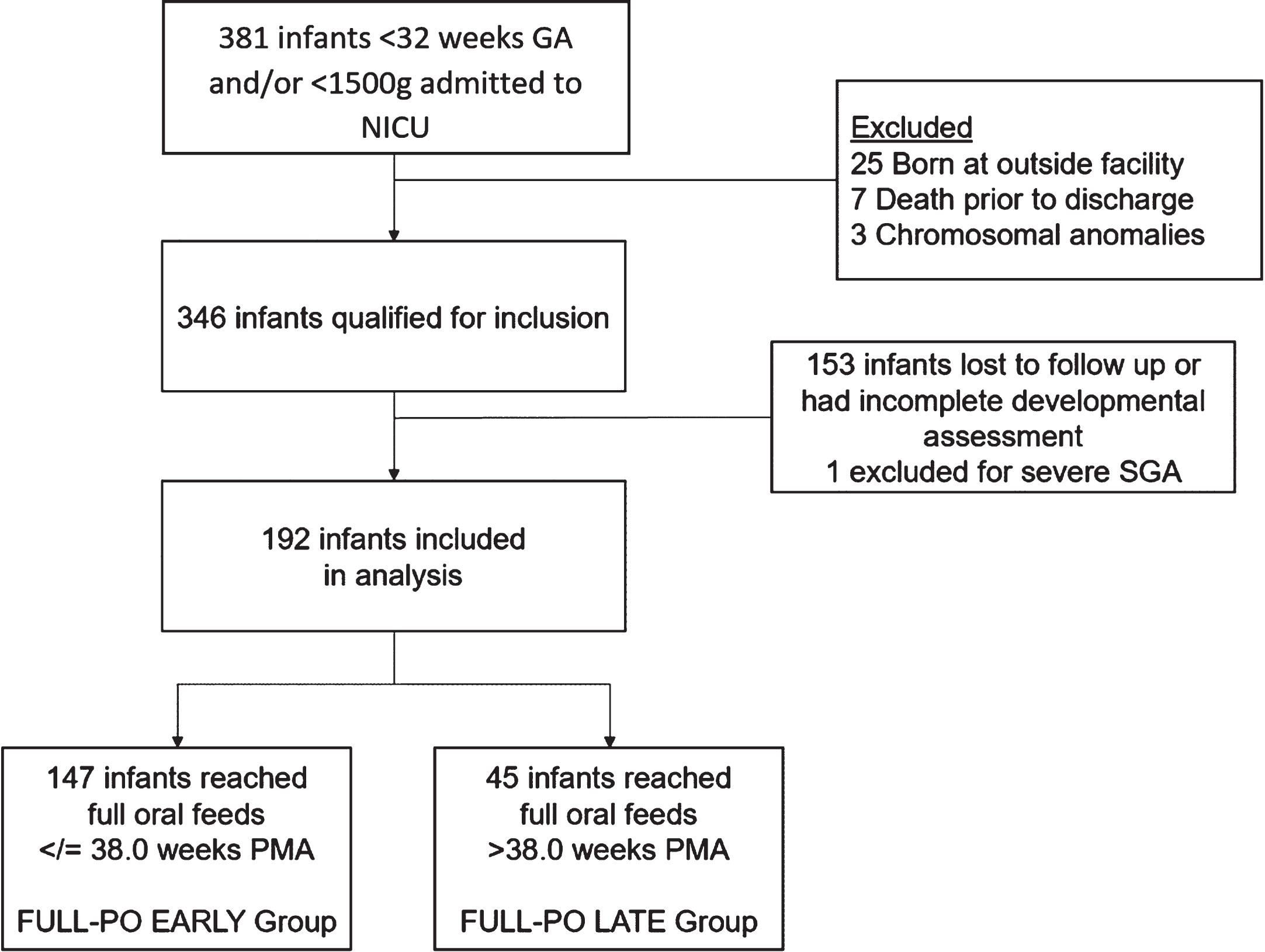
Flow diagram of infants selected for sample analysis. GA: gestational age, SGA: small for gestational age, PMA: post-menstrual age.
Of the 192 infants, 147 (77%) achieved FULL-PO EARLY and 45 (23%) achieved FULL-PO LATE. There were no significant differences in maternal characteristics including antenatal steroid use, maternal diabetes, preeclampsia, and chorioamnionitis (results not shown). The infants in the FULL-PO EARLY group were of older GA and higher birth weight, length, and head circumference (Table 1). The incidence of NEC, ROP, BPD, days of postnatal steroid use, and need for supplemental oxygen at discharge were higher in the FULL-PO LATE group. Specifically, surgical NEC was higher in the FULL-PO LATE group. Length of stay differed between groups with the FULL-PO LATE group admitted for approximately twice as long as the FULL-PO EARLY group.
Infant characteristics, morbidities, and discharge outcomes by FULL-PO group
aValues shown as mean±SD or (percentage of total N).
Feeding characteristics are displayed in Table 2. Linear regression analysis demonstrates our sample of infants approximately < 27–28 weeks GA at birth were less likely to reach FULL-PO at ≤38 weeks PMA (Fig. 2).
Feeding measures in the NICU of infants by FULL-PO group
Feeding measures in the NICU of infants by FULL-PO group
aValues shown as mean±SD or (percentage of total N), b1 patient did not achieve full enteral feeds and was excluded from this analysis, c3 patients did not achieve full oral feeds before NICU discharge and were excluded from this analysis, EBM: expressed breast milk, PMA: post-menstrual age.
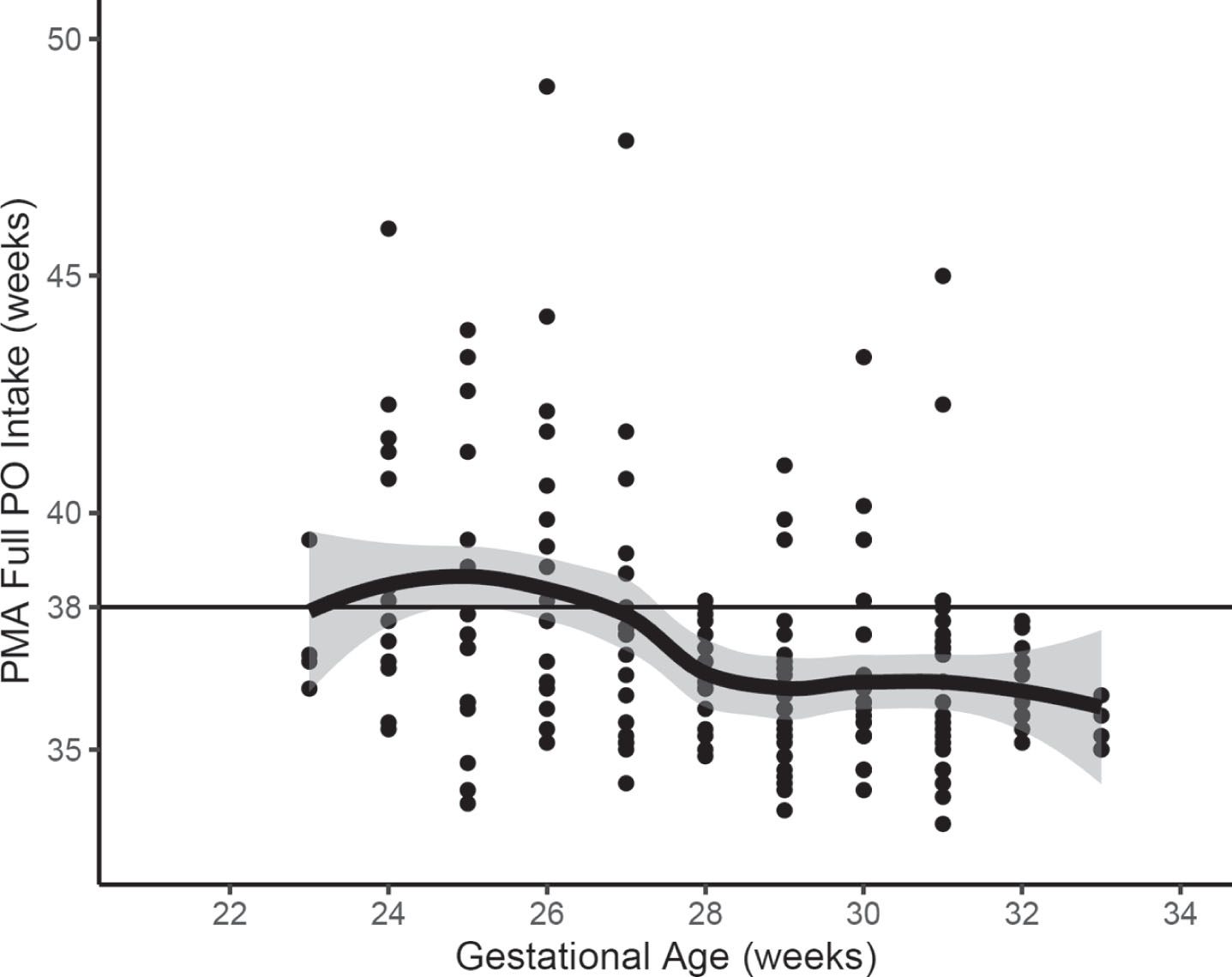
Weeks PMA FULL PO was achieved in NICU by gestational age at birth. Mean gestational age±1 standard deviation shown as solid bold line with gray shaded area. 38 weeks PMA shown as horizontal solid thin line. GA: gestational age, PMA: post-menstrual age.
Examining the infants diagnosed with BPD which comprised almost 60% of the FULL-PO LATE group, we found time to oral feeding was achieved by 75% of the group at approximately 41 weeks PMA, whereas 75% of those not diagnosed with BPD did so by 37 weeks PMA (Fig. 3). At 36 weeks PMA the majority of infants without BPD had approximately 60% oral intake versus those with BPD had 25% oral intake.
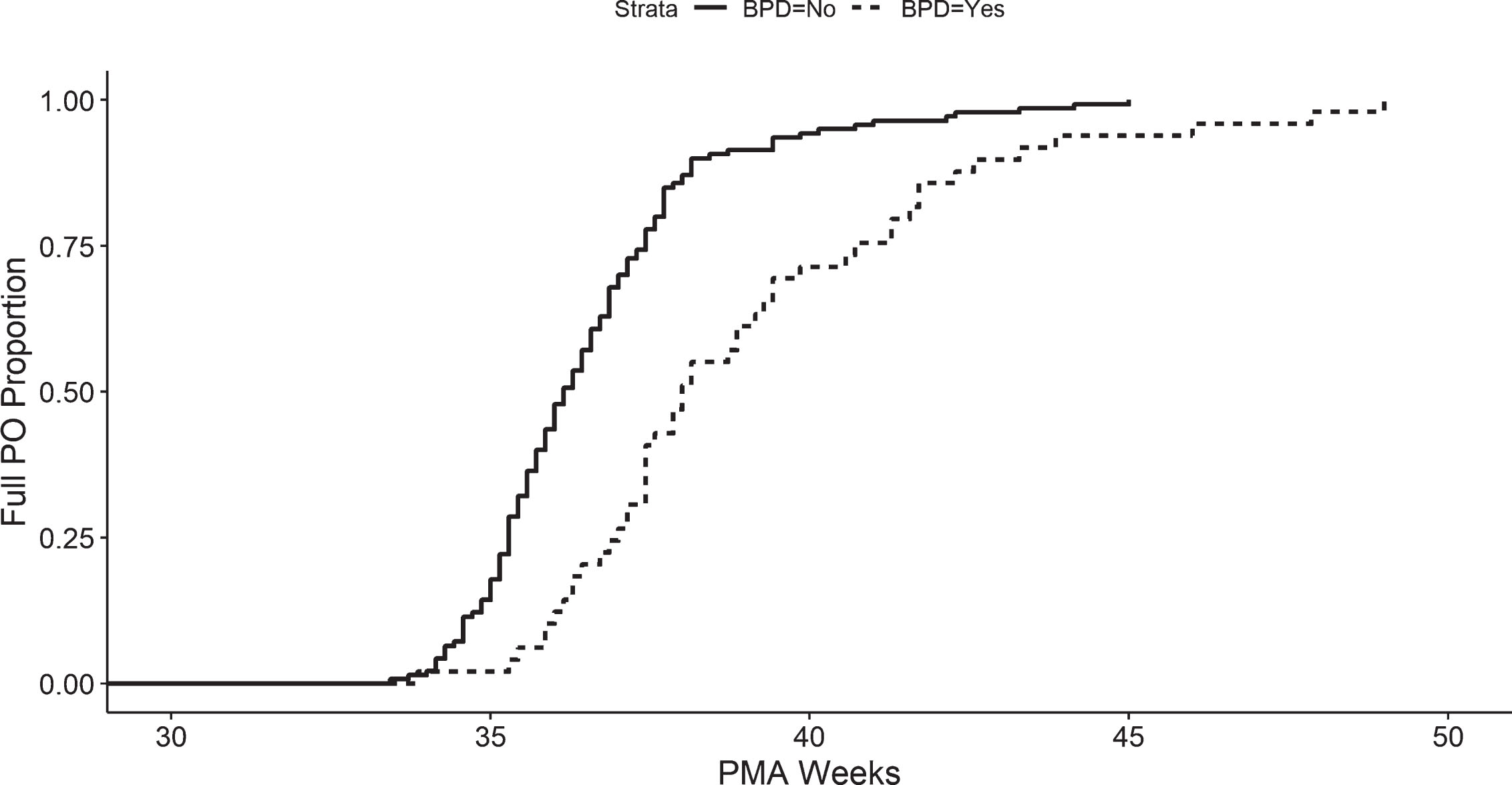
Proportion of infants without bronchopulmonary dysplasia (solid line) and with bronchopulmonary dysplasia (dotted line) achieving oral intake at given postmenstrual age in NICU. PMA: post-menstrual age.
Four patients (2%) had a GT placed before discharge in the setting of complex NICU courses. Mean time to GT placement was 41.7±9.8 weeks PMA in patients with a mean GA 26.5±1.9 weeks and birthweight 931.2±282.6 g. Mean PMA to commence oral feeding was 36.2±2.9 weeks PMA. Three patients had BPD and all patients had intestinal disease such as surgical NEC or spontaneous intestinal perforation. Two patients were no longer GT dependent at the 12 month CA visit. One of these 2 patients achieved FULL-PO before NICU discharge. The remaining 2 patients still required a GT at the 2-year follow-up.
There were no differences in incidence of feeding difficulties between groups at the 12 month CA visit (Table 3). Interestingly, 2 infants in the FULL-PO LATE group required GT placement after discharge. This occurred within the first year of life secondary to oral aversion. At the 24 month CH visit the FULL-PO LATE group had a significantly higher incidence of feeding issues (Table 3).
Neurodevelopmental and feeding outcomes at 12 and 24 month follow-up by FULL-PO group
aValues shown as mean±SD or (percentage of total N).
A total of 144 (75%) infants completed both 12 month CA and 24 month CH follow-up visits. Details of outcomes are seen in Table 3. There were significant differences between cerebral palsy and BSID III scores seen among groups. Additional logistic regression analysis of cognitive outcomes at 12 month CA and 24 month CH were not significantly influenced by milk type (expressed breast milk or donor milk) received in the first month of life (data not shown).
A multivariate logistic regression model was constructed for the primary outcome with adjustment for GA, BPD, NEC, postnatal dexamethasone days (Table 4). Presence of BPD was associated with an increased odds of language delay at 12 month CA and 24 months CH. Achieving feeds > 38 weeks PMA was not statistically significant for language delay, but did trend towards higher likelihood for language delay. In similar logistic regression models BPD was a primary predictor of cognitive delay at 24 months CH. All other variables were not found to be statistically significant in remaining models for cognitive and motor outcomes.
(a) Logistic regression models for language (a) cognitive (b) and motor (c) domains at 12- and 24-month visits
(a) Logistic regression models for language (a) cognitive (b) and motor (c) domains at 12- and 24-month visits
BPD: bronchopulmonary dysplasia, NEC: necrotizing enterocolitis.
This retrospective study evaluated the relationship between timing of oral feeding acquisition and neurodevelopmental outcomes in a preterm population at a level IV NICU. We elected to examine the time point of 38.0 weeks PMA given studies have shown healthy preterm infants achieve full oral feeds by an estimated 36 weeks PMA [9, 18]. Similarly in our population of infants the majority reached full oral feeds 36.1 + 1.1 weeks PMA. Research has demonstrated preterm infants not reaching full oral feeds by 40.0 weeks PMA to be at risk for neurodevelopmental delays [8]. Therefore, we concentrated on a potentially earlier critical time at which preterm infants would be expected to orally feed.
We found infants not reaching full feeds orally by 38.0 weeks PMA are at increased risk for feeding difficulties later in life. This was not seen at 12 months CA, potentially because it was underreported as infants are beginning to experiment with solids, or these concerns develop as a child is expected to eat a larger variety of solid foods. Additionally, there are limitations in the study as the clinic providers may not have inquired if feeding issues were present. We have since adjusted our own practice to probe further on feeding issues at all NICU follow-up visits.
Similar to our findings, Patra and Greene found “feeding difficulty” was the strongest predictor of cognitive and motor delays at 8 months, but not at 20 months CA [19] Conversely, Lainwala et al. reported preterm infants born at < 32 weeks GA with full oral feeding by 40.0 weeks PMA was associated with decreased risk of adverse neurodevelopmental outcomes including cerebral palsy at 18 to 26 months CA [8]. While we did not find differences in all areas tested on the BSID III, nor did we see a continued difference in cerebral palsy between our groups, we speculate this may be secondary to a smaller sample size and using a different time point of 38.0 weeks PMA for reaching full oral feeds.
As prior studies have shown, we too demonstrate infants at greatest risk appear to be smaller, younger GA infants [4, 12]. In addition, there was a higher percent of males in the FULL-PO LATE group. Specifically, infants with significant gastrointestinal or pulmonary disease demonstrated high risk for needing supplemental routes of enteral feeding and prolonged TPN use. It is likely the pulmonary and gastrointestinal disease is associated with the smaller, younger, and predominantly male population. Overall, oral feedings started at a PMA approximately 2 weeks later and it took up to 3 weeks longer to achieve full oral feeds in the FULL-PO LATE versus FULL-PO EARLY group. Similarly, for infants in the FULL-PO EARLY group breastfeeding began at an earlier PMA and was more often maintained at time of discharge. The prolonged length of stay the FULL-PO LATE group required is concerning and indicative of the economic burden that occurs when infants struggle to orally feed.
Hwang et al. documented infants < 28 weeks GA reach full PO feeds at a later time point than infants 28–32 weeks GA [12]. Our unique model shows with additional precision the transition point for our population to be 27–28 weeks GA. We found infants born at approximately < 27–28 weeks GA achieve FULL-PO at 38.5±3.1 weeks PMA. This may influence the time at which supplemental feeding routes are considered in this population, which is of great importance when counseling families on feeding outcomes and discharge planning.
In this study BPD was a strong predictor for language and cognitive delays at 24 months CH even after adjusting for covariates of significance. Other studies have found later attainment of FULL-PO at 40.0 weeks PMA in infants with increased incidence of BPD and more ventilator days [8]. Subsequently, this group of infants had increased risk for neurodevelopmental delays. Our study indicates that even 38.0 weeks PMA may serve as a marker for infants with BPD having poor feeding and neurodevelopmental outcomes. Moreover, our study captures the shifted time course infants with BPD learn to orally feed. Infants with BPD initiate feeds later and slowly increase oral intake. If full oral feeds are achieved, they do so up to 6 weeks later than the FULL-PO EARLY group. Park et al. also demonstrated in a cohort of infants ≤28 weeks GA those with mild, moderate, and severe BPD were delayed 2.2, 5.7, and 7.5 weeks in their achievement of full oral feeds as compared to infants without BPD who did so in the range of 35–37 weeks PMA [14].
Reasons for this are likely multifactorial. Practitioners and speech pathologists may be apprehensive to attempt oral feeding in the presence of moderate levels of respiratory support. Moreover, it has been shown infants with BPD have great difficulty coordinating swallow and respiration patterns [20, 21]. While channeling the majority of their energy towards respiration, little endurance remains for successful oral feeding attempts.
During the study period it was our NICU’s practice to mainly use humidified high-flow oxygen delivery via nasal cannula as a step down from nasal intermittent positive pressure ventilation. Oral feeding attempts began when the patient was stable on 2–3 liters per minute of oxygen. When oral feeding attempts began infants progressed from attempting oral feeding once a day to attempting oral feeding at all feeds in a stepwise fashion. The infant’s number of oral feeding attempts was increased when deemed appropriate by the medical team, typically once the infant could complete the number of oral feedings ordered at that time. Our unit has since transitioned from this provider-based model to an infant-driven feeding model. Patients receive neonatal speech, physical and occupational therapy support during their NICU stay.
We also found that infants with multiple major comorbidities such as intestinal injury and BPD are high risk for GT dependence upon discharge. How to optimally support the oral feeding skills of infants with aerodigestive complications in the NICU requires more investigation.
Four patients underwent GT placement prior to discharge, with 2 patients no longer requiring GT feeds by the 12 month CA visit. Moreover, 2 patients who were taking full volume by mouth developed oral aversion and required GT placement by 6–8 months corrected age. All patients receiving GTs were unable to attain full oral feeds by 38.0 weeks PMA. Additionally, the FULL-PO LATE group had an increased risk of feeding difficulties by the 24 month CH follow-up. This calls attention to the likelihood of a critical time period at which a preterm infant should be expected to acquire sufficient skills to consume at least 120 ml/kg/day volume and thrive. It raises question to the optimal time period for GT placement in young infants, as well as encourages practitioners to remain vigilant when assessing oral feeding success in the outpatient setting.
Limitations of this study include the retrospective design and inability to achieve sufficient power due to smaller sample sizes of each group. There are also limitations in reliably identifying certain outcomes such as cerebral palsy and feeding difficulties as it was dependent on provider documentation. We included patients who had visits between 18–30 months. It is possible patients assessed before 24 months CH may have improved or declined in developmental and feeding areas if they had been assessed at an older age. Additionally, we recognize the definition of feeding difficulty is limited in this study and certainly can include more specific behaviors. But it was the decision of the authors to maintain a broad definition. It was not within the scope of this study to examine ventilatory needs, amount of oxygen supplementation, swallow studies, outcomes of upper airway procedures etc. potentially required by the infants with aerodigestive complications. These factors and oral feeding characteristics of infants with BPD and intestinal injury requires further investigation so we can better predict an individual child’s success of orally feeding and allow for timely feeding therapy interventions.
Further research is needed to define oral feeding trajectories and qualities for various NICU infant populations. More information regarding gestational age subgroups and determination of whether specific early interventions result in attainment of oral feeds despite the presence of comorbidities would aid in clinical decision making. Therapy type, program specifications, and frequency that result in improvements for infants with feeding difficulties and neurodevelopmental delays is not well established. Much remains to be learned about how to feed infants with complex conditions such as chronic lung disease, intestinal malformations, and upper airway disorders.
Conclusion
In conclusion, infants not reaching full feeds orally by 38.0 weeks PMA are at increased risk of feeding difficulties at 24 months CH. Infants with BPD are at highest risk of speech and cognitive delays during the first 24 months of life. Infants born 27–28 weeks or with BPD or intestinal complications initiate and attain full oral feeds much later than other preterm infants and are at high risk for needing supplemental feeding devices. Therefore, premature infants who have not achieved full oral feeds by 38.0 weeks PMA or infants with BPD should receive early childhood intervention and additional therapies targeted to improve cognitive, speech and feeding delays. Following NICU discharge primary care providers and neurodevelopmental follow-through providers need to maintain awareness of the challenges these infants face as they appear to be at increased risk for feeding issues and should be monitored closely.
Footnotes
Acknowledgments
The authors of this manuscript thank the staff of the University Hospital neurodevelopmental follow-up clinic, PREMiere, and the Neonatal Nutrition and Bone Institute program for their care and dedication to improving neonatal outcomes and supporting families.
