Abstract
BACKGROUND:
Neonates affected by isoimmune hemolytic disease (HDN) are at risk of developing severe hyperbilirubinemia. Studies show that increasing levels of bilirubin impact neonatal neurodevelopment. To avoid complications associated with exchange transfusion, intravenous immunoglobulin G (IVIG) is used to treat hyperbilirubinemia. We included all infants who received more than two doses of IVIG treatment for isoimmune hemolytic disease. We analyzed the incidence of side effects associated with IVIG treatment and the rate of exchange transfusion.
METHODS:
A retrospective chart review performed between October 2011–October 2022 at East Carolina University Health identified neonates who received more than two doses IVIG for HDN. Neonates of postmenstrual age greater than 28 days old, receiving less than three doses of IVIG or received IVIG for other indications were excluded. The occurrences of adverse events, demographics and use of other medical therapies were reviewed.
RESULTS:
Eleven neonates were included in the case series. Most common cause of severe hyperbilirubinemia was attributed to ABO incompatibility. Six patients (54%) received three doses of IVIG, and five patients (45%) received four doses of IVIG with bilirubin levels decreasing below exchange transfusion. No treatment exceeding four doses of IVIG was reported, nor adverse events during treatment.
CONCLUSIONS:
In this cohort of neonates with HDN, bilirubin levels decreased after treatment with multiple doses of IVIG. Future research on recommendations of optimal total number doses of IVIG to reduce the risk for exchange transfusion.
Introduction
ABO or Rh incompatibility may cause severe hyperbilirubinemia and put the patient at risk of permanent brain damage [1]. When bilirubin exceeds the phototherapy (PTX) level and is near the exchange transfusion level, exchange transfusion would be considered. However, severe complications of this treatment have been reported, including thrombocytopenia, hypoglycemia, and death [2, 3]. Due to the disadvantages of exchange transfusion, intravenous immunoglobulin G (IVIG) has been used to treat neonatal isoimmune hemolytic disease (HDN) since 1987 [4–6]. However, IVIG therapy has been associated with other adverse side effects, including fever, hypotension, rash, vomiting, and potentially a higher rate of necrotizing enterocolitis (NEC) [7, 8].
The most recent neonatal hyperbilirubinemia guideline published by the American Academy of Pediatrics recommends that patients be treated with IVIG if there is a poor response to PTX and performing an exchange transfusion is challenging. Still, the dose of IVIG in HDN treatment is understudied and not addressed in the most recent guideline. Prior research has analyzed neonates receiving one or two doses of IVIG [9]. To our knowledge, there has been no research about patients who received more than two doses of IVIG treatment for neonatal isoimmune hemolytic disease [10, 11]. This is a case series including eleven neonates who received more than two doses of IVIG treatment for neonatal HDN. We analyzed the incidence of adverse events among neonates that required exchange transfusion after treatment with IVIG.
Methods
The study was approved by the Institutional Review Board at East Carolina University (UMCIRB 22-002433). The East Carolina University Health (ECUH) Maynard Children’s Hospital is a 198-bed tertiary-level regional referral hospital. This center serves a predominantly rural 29-county region in eastern North Carolina, with over 33,000 annual admissions. The ECUH Children’s Hospital routinely uses IVIG in HDN treatment. In conjunction with the clinicians’ assessment, the need for additional IVIG treatment is considered if the patient’s bilirubin remains close to exchange transfusion level after two doses of IVIG. We performed a retrospective chart review to identify the neonates who received IVIG treatment between October 2011–October 2022 at ECUH Children’s Hospital. Our review included all neonates who received more than two doses of IVIG for HDN treatment. All patients included in this study had a positive direct antiglobulin test (DAT). Neonates whose postmenstrual age was > 28 days old, who received fewer than three doses of IVIG treatment, and who received IVIG treatment for indications other than HDN were excluded.
We evaluated how many infants required exchange transfusion after more than two doses of IVIG treatment, and the occurrence of any adverse events. Specifically, we investigated whether the patients had any intolerance of IVIG treatment (i.e., change of mental status, fever, rash, or vomiting), and possible treatment side effects including NEC and aseptic meningitis. We also recorded the patient’s gestational age, race, gender, birth weight, and blood type; the mother’s blood type; types of isoimmune hemolytic disease; and whether any siblings were affected by jaundice. Data collected on the treatment of isoimmune hemolytic disease included PTX timing and duration, IVIG doses, and whether the patient received intravenous fluids.
Results
A total of eleven patients were included in the case series. Most of the patients were female, African American, and term-born. The median birth weight was 3,230 g (range: 1,585–3,835 g). The median reticulocyte count was 12.50% (range: 8.25% –18.21%). Patients’ characteristics are summarized in Table 1.
Patients’ characteristics
Patients’ characteristics
The median PTX start time was at three hours of life, ranging from zero to eleven hours. For 91% of the infants, PTX was started within six hours of life, and all infants had a total serum bilirubin above the PTX threshold when treatment was started. Most infants received six to ten days of PTX (73%). There were six infants (54%) who received a total of three doses of IVIG, and five (45%) who received a total of four doses of IVIG. No infants received more than four doses of IVIG. The details of the interventions are summarized in Table 2.
Interventions
Differences between the bilirubin level and exchange transfusion level after each IVIG dose are summarized using a box and whisker plot (Fig. 1) and Table 3. Overall, bilirubin levels decreased below the exchange transfusion threshold after each dose of IVIG treatment. One infant required an exchange transfusion due to the lack of response to the third dose of IVIG, with total serum bilirubin levels above the exchange threshold.
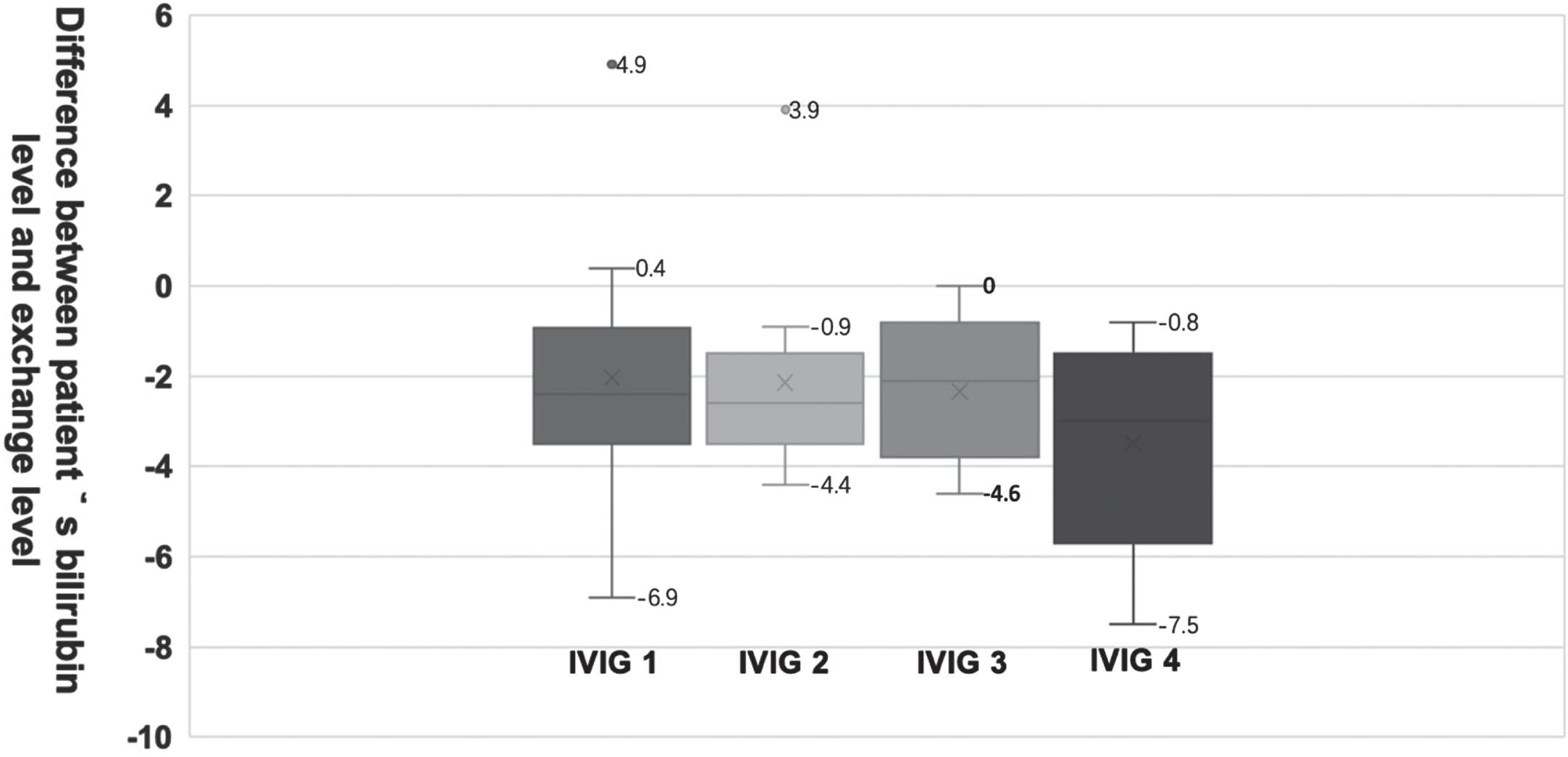
Differences between the bilirubin level and exchange transfusion level. The differences between the bilirubin level and exchange transfusion level before each dose of IVIG treatment are showed at Fig. 1. Zero is in Y axis is the exchange level.
All patients tolerated IVIG treatment without reports of adverse events. There were no documented episodes of hypotension, rash, fever, or other adverse events during IVIG treatment. Ten patients received enteral feeding during the IVIG treatment, while one patient did not receive enteral feeding because of severe fetal hydrops, and would not have been able to receive enteral feeding regardless of IVIG treatment. There was no NEC reported. No patient had documented aseptic meningitis.
Difference between patients’ bilirubin level and exchange level before each dose of IVIG treatment
*There are 5 patients received 4th dose of IVIG.
The standard therapy for HDN includes PTX, hydration, IVIG, and exchange transfusion. Hydration and PTX remain the safest options used as a standard practice. For infants who fail to respond to intensive PTX and develop significant hyperbilirubinemia, IVIG is a treatment with fewer side effects and complications in comparison to exchange transfusion. The decision to treat infants with IVIG at our center is provider-dependent when infants meet the criteria for escalation therapy. The current recommendation for escalation therapy is to treat with two doses of IVIG at 12-hour intervals [10]. For the 11 infants in our case series, a third and, in some instances, a fourth dose was given, preventing the need for exchange transfusion in ten (91%) infants.
Given the low quality of evidence, no clear recommendations regarding the use of IVIG were included in a recent Cochrane review [12]. However, a systematic review which included six randomized controlled trials showed a reduced need for exchange transfusions when using IVIG in HD [5]. Based on available data, the use of IVIG to treat HDN remains controversial [5, 14]. In our case series, the median bilirubin levels after each dose demonstrated a decrease in the serum bilirubin, thus preventing the need for exchange transfusion.
Although IVIG is considered safer than exchange transfusion, observational studies suggest that 2.2% of patients experienced NEC after IVIG treatment [2, 16]. There were no reports of NEC in our cohort; most infants continued to receive enteral feeds during and after IVIG infusion, with no feeding intolerance reported. Most of the infants in our case series achieved full enteral feeding while on treatment for HDN.
Blood group incompatibility occurs in 15% –25% of pregnancies [16, 17]. If an infant has A or B blood type, maternal anti-A or anti-B antibodies attach to the fetal blood cell, leading to cell destruction and the development of jaundice and anemia [16]. However, one in 150 infants develop mild hemolysis, and one in 3,000 develop severe disease [18]. Typically, affected neonates will usually be DAT-positive. The clinical picture of anti-A and anti-B antibodies in HDN usually results predominantly in hyperbilirubinemia without significant neonatal anemia. This is largely due to the relatively few groups A or B antigenic sites on neonatal red blood cells (RBCs), allowing the antibody-coated cells to remain in circulation longer [17]. In cases of severe hyperbilirubinemia, unconjugated bilirubin crosses the blood–brain barrier, resulting in bilirubin neurotoxicity and bilirubin-induced neurologic dysfunction (BIND), of which kernicterus or chronic bilirubin encephalopathy is the most severe and permanent clinical manifestation [19].
Recently published guidelines from the American Academy of Pediatrics recommend administering IVIG (0.5–1 g/kg) if the total serum bilirubin rises despite intensive PTX, or the bilirubin level is within 2–3 mg/dL (34–51μmol/L) of the exchange level [10]. If necessary, a subsequent dose can be given in 12 hours. Monitoring for rare adverse effects of IVIG infusion is recommended [10, 18]. There is no contraindication to subsequent dosing. However, prior to this case series, there have been no reports in the literature regarding the use of a third or fourth dose of IVIG to prevent exchange transfusion in infants diagnosed with ABO incompatibility who experienced increasing total bilirubin despite intensive PTX.
The hallmark of treatment for hyperbilirubinemia is PTX. Intensive PTX can lower the serum bilirubin faster, shortening the overall length of treatment. Intensive PTX requires a narrow-spectrum light-emitting diode (LED) blue light with an irradiance of at least 30 mW/cm2 per nm at a wavelength of around 475 nm [10]. For 10 (91%) infants in this case series, PTX was started within 6 hours of life, with the number of lights ranging from double to quadruple PTX, with or without the PTX blanket. Despite lacking Food and Drug Administration (FDA) approval, IVIG has been used to manage different clinical conditions in fetuses and neonates [4]. Our case series focused on effectively treating blood group (ABO) incompatibility with IVIG to prevent the need for exchange transfusion. However, there is no consensus in the current literature; prior studies describe multiple factors affecting response to IVIG, including age upon admission to the hospital, duration and strength of PTX, and dose and timing of IVIG administration [8, 21].
One case report describes a newborn infant with severe hyperbilirubinemia due to anti-C antibodies, treated with IVIG on the day of life (DOL) 14 after being diagnosed with severe anemia [8]. She received one dose of IVIG due to worsening anemia, and an exchange transfusion was performed, a few hours after which the patient developed NEC. Only one dose of IVIG was given, but the authors hypothesized that IVIG was associated with the development of NEC, by causing an increase in blood viscosity that may have promoted the formation of microthrombi and reduced mesenteric blood flow [8, 15]. Their assumption was based on a previous retrospective study that included 167 newborn infants who received IVIG, of whom 11 (2.2%) were diagnosed with NEC. In that sample, more infants had Rh incompatibility (51%) than ABO incompatibility (30%) [17]. Newborns were treated with a dose of IVIG (500–1000 mg/kg over 2–4 hours) when total serum bilirubin level plus two points reached 85% of the cutoff value for performing an exchange transfusion [15]. These newborns had severe hemolytic anemia and higher bilirubin levels, requiring more exchange transfusions than infants treated with PTX alone (19% vs. 7%;
Another retrospective study compared single and multiple doses of IVIG to treat HDN [7]. In this study, all 39 infants in the cohort had ABO incompatibility and treated with LED PTX. The cohort was divided into Group I, who received a single dose of IVIG; Group II, who received two doses; and Group III, who received no IVIG. One infant each in groups I and II required an exchange transfusion, but no side effects of IVIG (i.e. allergic reactions, renal insufficiency, wheezing, and aseptic meningitis cases) were reported [7]. There was no report of NEC in this study cohort and no difference among groups in length of PTX treatment. A more recent study described no significant difference in outcomes among 46 infants with ABO incompatibility treated with IVIG, as compared to 68 infants exposed to PTX only; and no adverse effects during IVIG treatments (including no cases of NEC) [22].
Conclusions from our case series are limited due to its retrospective nature. Furthermore, the information recorded regarding use of new-generation light-emitting diode (LED) PTX lights did not include the specific irradiance measures at the bedside to understand if changes in the total bilirubin level corresponded to the body surface area exposed. Another limitation of our report was the lack of data from long-term follow-up visits, which could have been useful for ascertaining neurodevelopmental outcomes.
Despite these limitations, our case series reports novel data regarding the use of higher-dose IVIG (1 g/kg/dose, with 3–4 doses administered) to decrease the total bilirubin level below the threshold for exchange transfusion. Compared to previous studies of single-dose IVIG administration, the rate of exchange transfusion in our cohort was significantly lower, with only one of eleven infants requiring exchange transfusion. More importantly, in contrast to conclusions drawn from previous studies, no infant in this cohort developed NEC, an adverse event that drastically increases the morbidity and mortality of infants admitted to the NICU. Additional multicenter analyses and, if feasible, randomized controlled trials, could provide more definitive data on the safety and efficacy of repeated IVIG dosing.
Footnotes
Acknowledgments
The authors thank Meredith Chanas for assisting with data collection.
Financial disclosure statement
The authors certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
