Abstract
A circular shunt is a poor prognostic factor associated with Ebstein’s anomaly. Targeting the constriction of the ductus arteriosus (DA) in order to limit or resolve the circular shunt, has been shown to improve fetal outcomes. Prenatal non-steroidal anti-inflammatory drugs (NSAIDs) have been known to constrict the DA. Recently, prenatal NSAIDs have been used for that purpose in the treatment of circular shunt. Limited research shows that it may be an effective treatment leading to improved fetal outcomes. In this article, we did an extensive review of literature to describe this therapy’s effectiveness and outcomes. 82% of fetuses were able to achieve ductal constriction with prenatal NSAID therapy. For fetuses who achieved ductal constriction, fetal demise was less likely (6%) when compared to those who were unable to achieve the same (50%). Of all the fetuses with hydrops, 50% had resoluation of hydrops with prenatal NSAID treatment.
Introduction
Ebstein’s anomaly (EA) is a rare and complex congenital malformation of the tricuspid valve and myopathy of the right ventricle that has variable anatomic and clinical presentations. Although the incidence of EA is estimated at approximately 1 in 200,000 [1], its broad anatomic and clinical spectrum has been widely reported.
First described in 1886 by Wilhelm Ebstein, the anomaly consists of several anatomic components [2]. While the normal human heart has a tricuspid valve consisting of three coapting leaflets - anterior, posterior, and septal - the tricuspid valve leaflets in EA are abnormally displaced. EA is caused by failure of delamination of the septal and inferior/posterior leaflet leading to the apical displacement. This in turn leads to enlarged right atrium, right ventricular dilation and “atrialization” of the ventricle. In the normal heart, there is an apical displacement of the posterior and septal leaflets which is less than 8 mm/m2. Atrialization refers to non-trabeculated myocardium which extends to the functional tricuspid valve annulus in what would have otherwise been the trabeculated ventricle. Failure of the tricuspid valve leaflet delamination during embryonic life is thought to be the impetus for the anatomic malformation [1]. Atrial septal defects are a common finding with right to left shunting due to increased right sided pressures from tricuspid regurgitation and poor right ventricular compliance. The overall mortality of neonates with EA has been shown to be around 24% [3].
The presentation of EA is highly variable, ranging from fetal demise to incidental discovery of the anomaly in adulthood. In general, the severity of features directly correlates with the increased likelihood of fetal demise or neonatal death [4]. The circular shunt is an unfavorable consequence of severe forms of EA. In circular shunts, pulmonary and tricuspid regurgitation, in combination with a decreased functional right ventricle size, cause shunting of fetal blood across the foramen ovale to the left atrium. Blood subsequently leaves the left ventricle through the aorta but circumvents systemic flow through the ductus arteriosus (DA). Blood flow through the DA and into the pulmonary artery leads to further pulmonary regurgitation and insult (Fig. 1).
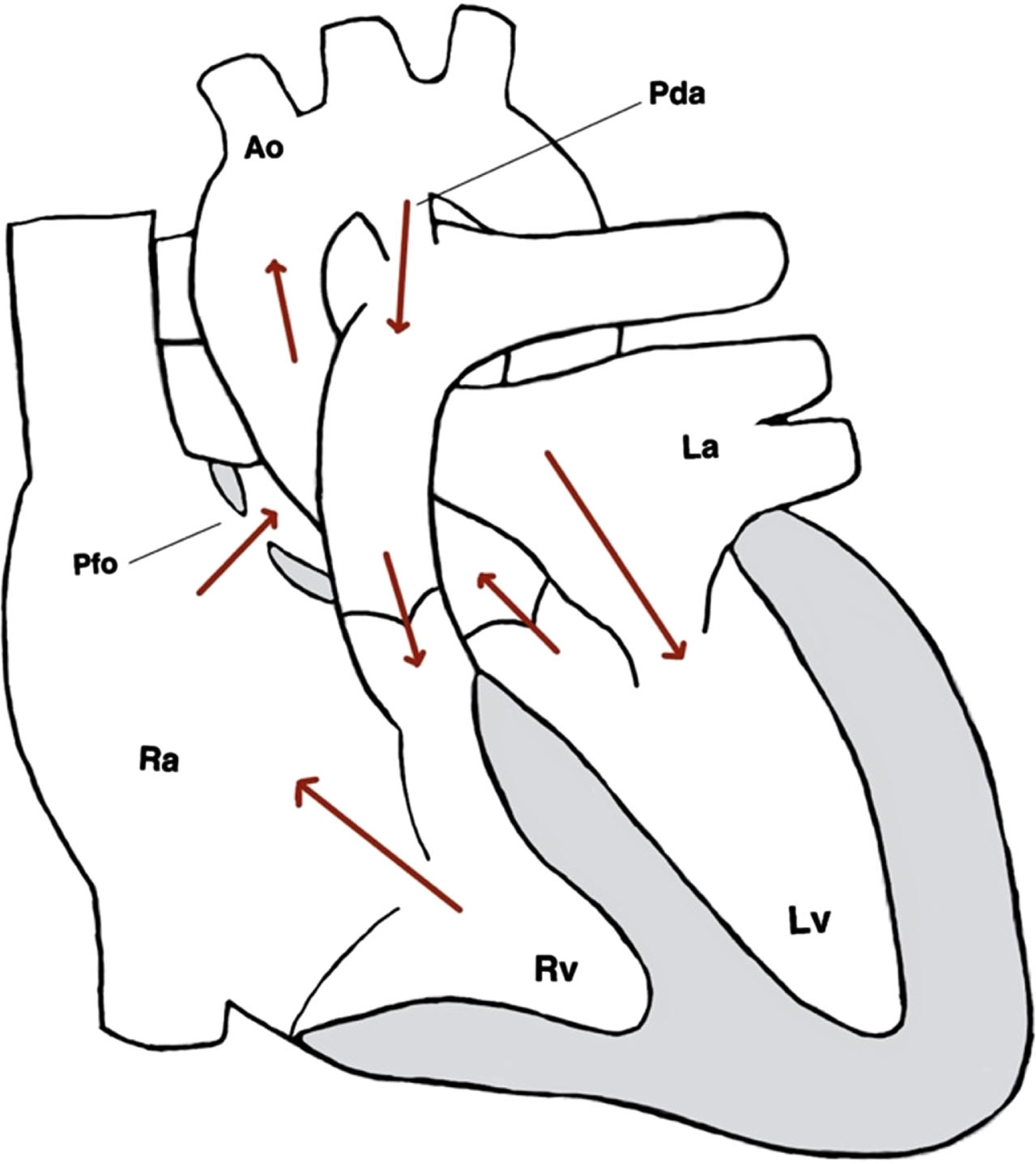
Direction of blood flow in circular shunt.
A circular shunt is associated with a high perinatal mortality. One study found the overall perinatal mortality of EA to be 45%. However, fetuses with EA and pulmonary regurgitation, indicative of circular shunt, were almost twice as likely to experience fetal demise or neonatal death compared to those only experiencing EA [5].
Due to the high perinatal mortality, the disruption of the circular shunt may be helpful in improving outcomes in this population. Targeting the DA and causing its constriction may limit the effects of circular shunt and lead to improvement in outcomes [6]. Nonsteroidal anti-inflammatory drugs (NSAIDs) have long been known to cause ductal constriction and closure in preterm infants and third trimester fetuses [7, 8]. This body of evidence makes prenatal NSAIDs a crucial next stage of inquiry in the treatment of circular shunt.
There are a number of studies and case reports that have used prenatal NSAIDs to cause ductal constriction in infants with circular shunt physiology. These reports have demonstrated ductal constriction and improvement in fetal outcomes. However, there are currently no recommendations or established guidelines on the use of prenatal NSAIDs for treatment of circular shunts. Our study aims to systematically review the available literature on the treatment and outcomes of prenatal NSAIDs use on fetal EA with circular shunt and establish its effectiveness and potential side effects.
A MESH search was performed for Ebstein anomaly, circular shunt, prenatal NSAIDs in the published databases, Pubmed and Embase, in English between January 2000 to June 2022. The articles were selected in accordance with PRISMA guidelines [9]. We included prospective studies where NSAIDs were given prenatally in the treatment of fetal circular shunt. Studies that did not fit these criteria were excluded. 137 articles were obtained, screened and no duplicates were found. The articles were screened independently by two authors. The primary outcome was fetal death versus livebirth and secondary outcome was resolution of fetal hydrops. The outcomes measured were whether or not there was ductal constriction after use of NSAIDs, if oligohydramnios was noted at any time, presence of hydrops and whether it resolved with NSAIDs therapy, live birth versus fetal death, and whether indomethacin, the original therapy, was able to be transitioned to ibuprofen or discontinued altogether while maintaining ductal constriction. Data was extracted from the studies by two authors independently and compared. Risk of bias was assessed independently by two reviewers and agreed upon by consensus. Missing variables or data were excluded from data analysis.
Results
Out of 137 articles, 5 articles met our inclusion criteria (Fig. 2). These articles included a total of 22 fetuses with circular shunt in which NSAIDs were used to attempt ductal constriction [10–14] (Table 1). Through screening, 120 articles were removed due to irrelevance. Seventeen full-text articles were assessed and twelve were excluded due to being undesired study design or did not meet our eligibility criteria of NSAIDs use for intervention.
Ductal constriction was confirmed by echocardiography across all 5 studies. Gill et al. and Torigoe et al. also utilized fetal cardiac MRI to evaluate for ductal constriction. Freud et al. defined ductal constriction as visible narrowing and flow acceleration with a peak velocity > 2 m/s and/or increase in diastolic flow with pulsatility index < 1.9. Gill et al. defined it as visible DA narrowing and normalization of the umbilical artery flow. Torigoe et al. defined it as visible narrowing as well as increased doppler flow gradients from the baseline. Finally, Lopes et al. and Powel et al. did not specify criteria used to define constriction of the DA. All the above-mentioned criteria were used to adjust NSAIDs therapy based on individual fetal response and disease progression.
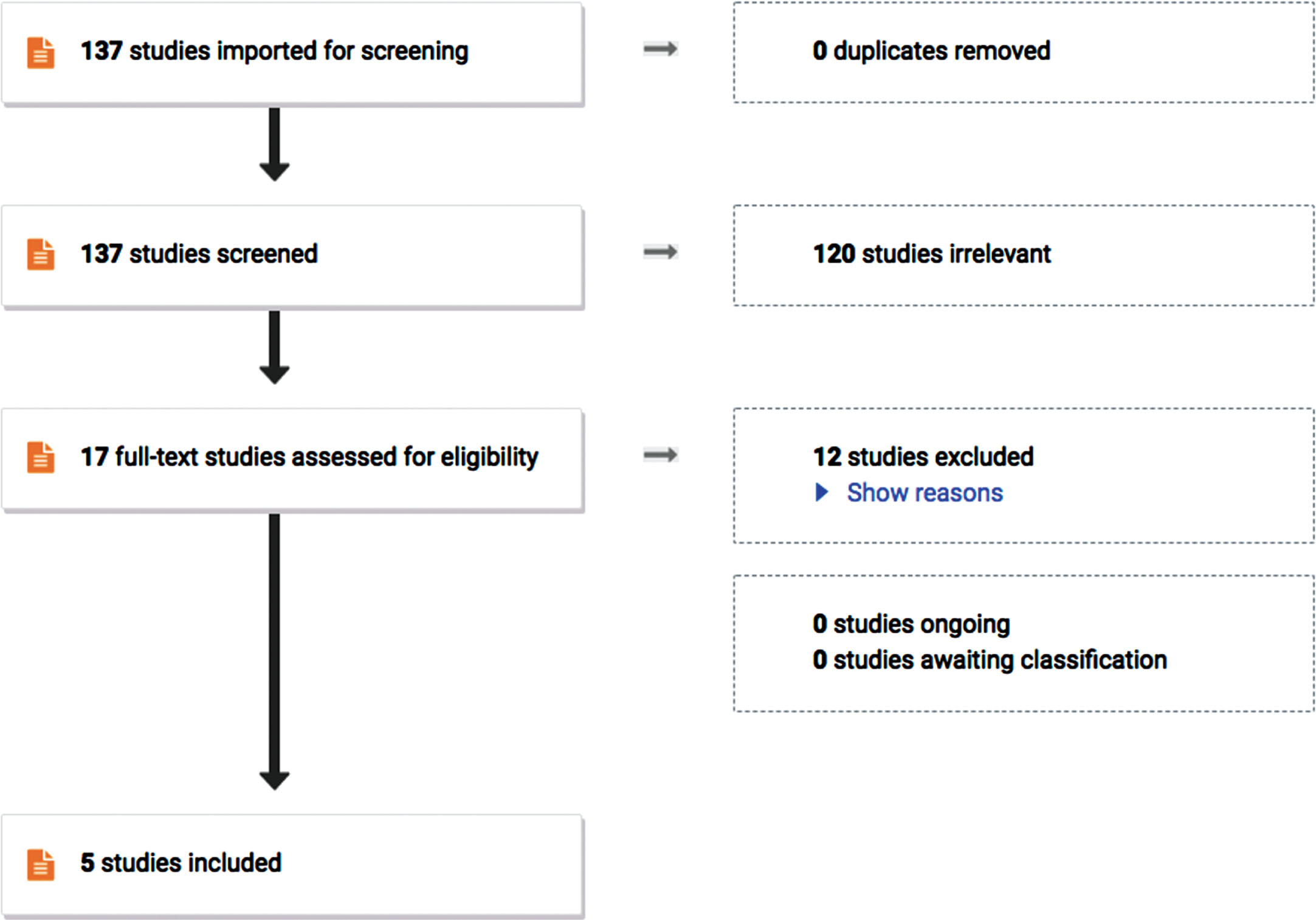
PRISMA flow chart representing how studies were screened and selected for analysis.
Summary of study data including treatment regimen, side effects, and fetal outcomes. LB indicates live birth; FD, fetal death; NC, no constriction; NH, no hydrops
Of the 22 fetuses that received NSAIDs therapy, 82% achieved ductal constriction. Freud et al. were able to achieve ductal constriction in 12 of 15 (80%) fetuses receiving therapy. In a case series by Torigoe et al., three of four fetuses were able to achieve the same outcome with NSAIDs. Cumulatively, 4 of the 22 fetuses were resistant to NSAIDs therapy, even with high doses of indomethacin. Of the 18 fetuses able to achieve ductal constriction, only 1 experience fetal death (about 6%). By comparison, of the 4 fetuses unable to achieve ductal constriction, 2 (50%) experience fetal death. The average gestational age at NSAID initiation of survivors was 29.6 compared to an average of 30.6 for nonsurvivors. Of the 22 fetuses, at least 59% experienced oligohydramnios at some point during therapy. However, in the case report by Gill et al., the presence of oligohydramnios was not stated.
The therapeutic regimen for each study is summarized in Table 2. However, it is important to note that therapy response differed in each fetus and was titrated accordingly. Of the fetuses able to achieve ductal constriction, 56% were unable to wean therapy to ibuprofen and needed to continue indomethacin to maintain ductal constriction. Of those able to achieve ductal constriction, 8 were able to discontinue indomethacin therapy while 14 were unable to do so.
General treatment regimen for each study. It is important to note treatment regimen differed for each fetus based on disease progression and response to therapy
Of the 22 fetuses identified in these studies, 10 (45%) presented with hydrops at some point. The main point of treatment was aimed at reduction of this unfavorable outcome. Notably, of the 10 fetuses with hydrops, 50% had resolution with prenatal NSAIDs therapy (Table 1).
NSAIDs can be an effective means to constrict the DA leading to improvement in outcomes in fetuses with circular shunt physiology. Hydrops fetalis is a known complication of severe EA with and without circular shunt. It is defined as two or more fluid collections of fetal body compartments. Hydrops is related to poor prognosis and outcomes [5]. Of those able to achieve ductal constriction, 50% had resolution of hydrops fetalis. There was also improvement in fetal death for those able to achieve constriction (6%) versus 50% in those unable to achieve constriction.
Currently there are no guidelines on using NSAIDs as a treatment for circular shunt. While NSAIDs treatment regimens differed, all were initially treated with indomethacin (Table 2). Indomethacin is thought to cause poor fetal urine output and subsequent oligohydramnios [15]. Due to risks associated with indomethacin, each study attempted to wean therapy to a lower dose of indomethacin and transition to ibuprofen while maintaining ductal constriction. Once ductal constriction is achieved, weaning therapy to lower doses of indomethacin or ibuprofen may be attempted in order to decrease associated risks such as oligohydramnios and renal dysfunction. It is important to note that therapy response differed in each fetus and was titrated accordingly. While NSAIDs therapy carries an increased risk of oligohydramnios and renal dysfunction, it is potentially a more favorable consequence when considering the high mortality associated with a circular shunt. However, 64% of fetuses required the continued use of indomethacin in order to maintain the ductal constriction. Data is limited due to the rarity of EA and circular shunt. More research is needed to maximize treatment regimen effectiveness while minimizing side effects.
Interestingly, Torigoe et al. noted changes in fetal middle cerebral artery (MCA) flow during NSAIDs therapy. Circular shunt can frequently cause absent or retrograde blood flow of the MCA, which can significantly impact neurocognitive outcomes in survivors [16]. Fetal evaluation revealed a decrease in MCA pulsatility index and improvement in anterograde flow after NSAIDs therapy compared to baseline. These findings could indicate improved cerebral perfusion and possibly lead to improved developmental outcomes.
Limitations
Although we have performed an extensive literature review, our study data is limited due to rarity of EA and circular shunt. There are only 22 subjects which were analyzed and majority of the subjects are from one study, Freud et al., which contributed 15 of the total 22 subjects. Another limitation is lack of the postnatal data. Since the majority of fetuses were obligated to continue indomethacin without weaning therapy to ibuprofen, it would be beneficial to study the postnatal outcomes including any evidence of renal impairment. It would be interesting to further investigate the difference in the outcomes based on the gestational age with NSAIDs therapy as well. Large multicenter studies are needed to better understand the long-term sequelae and to evaluate the fetal and maternal safety along with the postnatal outcomes.
Footnotes
Acknowledgments
All authors made significant contributions to the study’s concept and design. Faith Peña, Tiffany Emanuelson, Ryan Jones, Steven Todman, and Siddharth Mahajan performed reference collection, data collection, and data analysis. The original manuscript draft was completed by Faith Peña and Tiffany Emanuelson. All authors agree to the final draft of the manuscript.
Disclosures
Competing interests
The authors have no financial or non-financial interests to disclose.
Funding
No funding was received for the completion of this manuscript.
