Abstract
BACKGROUND:
To find the obstetrical and delivery associated risk factors of antenatal and postnatal grade III intraventricular hemorrhage (IVH) or periventricular hemorrhagic infarction (PVHI) in preterm neonates.
METHODS:
A retrospective study of obstetric and delivery associated risk factors included neonates (<35 gestational weeks) with severe IVH/PVHI (n = 120) and a prospectively collected control group (n = 50). The children were divided into: (1) antenatal onset group (n = 27) with insult visible on cerebral ultrasonography within the first 12 hours of birth or periventricular cystic changes visible in PVHI within the first 3 days; (2) neonatal onset group (n = 70) with insult diagnosed after initial normal findings or I-II grade IVH, and (3) unknown time-onset group (n = 23) with insult visible at > 12 h of age.
RESULTS:
The mothers of the antenatal onset group had significantly more bacterial infections before delivery compared to the neonatal onset group: 20/27 (74.1%) versus 23/69 (33.3%), (odds ratio (OR) 5.7 [95% confidence interval 2.1–16]; p = 0.0008) or compared to the control group (11/50 (22%); OR 11 [2.8–42]; p = 0.0005). Placental histology revealed chorioamnionitis more often in the antenatal compared to the neonatal onset group (14/21 (66.7%) versus 16/42 (38.1%), respectively; OR 3.7 [1.18–11]; p = 0.025). Neonates with neonatal development of severe IVH/PVHI had significantly more complications during delivery or intensive care.
CONCLUSIONS:
Bacterial infection during pregnancy is an important risk factor for development of antenatal onset severe IVH or PVHI. In neonates born to mothers with severe bacterial infection during pregnancy, cerebral ultrasonography is indicated for early detection of severe IVH or PVHI.
Keywords
Abbreviations
confidence interval
false discovery rate
germinal matrix hemorrhage
gestational week
intraventricular hemorrhage
interquartile range
magnetic resonance imaging
odds ratio
periventricular hemorrhagic infarction
score for neonatal acute physiology and SNAP perinatal extension
ultrasonography
Introduction
Germinal matrix hemorrhage (GMH) and intraventricular hemorrhage (IVH) remain common forms of brain injury in very preterm neonates with an overall incidence of around 25 % in children born before 32 gestational weeks (GW) [1–3]. The size of the germinal matrix decreases with increasing gestational age and therefore IVH is found in up to 36% of preterm infants at GW 22–23 and in only 1.2% at GW 30–31 [3].
In 8–15 % of cases, GMH-IVH can be complicated with periventricular hemorrhagic infarction (PVHI), referred to previously as grade IV IVH [1, 3–5]. The underlying mechanism of PVHI is venous obstruction induced by GMH-IVH, venous congestion leading to ischemia and secondary hemorrhagic infarction [4–8]. The higher the grade of GMH-IVH, the more likely PVHI develops [1, 5]. High intraventricular pressure due to large hemorrhage additionally affects the blood flow through the subependymal veins, increasing the size of the infarct [5].
Severe grade IVH (grade III or IV) or PVHI are often associated with adverse motor and cognitive neurological outcomes in preterm neonates, with higher scores of IVH/PVHI predicting worse outcome [4, 9–11]. Death occurs in 40% of preterm neonates with PVHI and cerebral palsy in 42% of survivors [10, 12]. The outcome of preterm neonates with antenatal lesions is extremely poor; in an early study by de Vries et al. 17/24 neonates died and none of the survivors had normal development [12].
Studies addressing the etiology and pathogenesis of severe IVH/PVHI have identified numerous maternal, pregnancy and perinatal risk factors with low gestational age as the most important single independent risk factor [2, 13]. Maternal transport to higher level hospital, antenatal steroids and improved resuscitation techniques have become the standard of care for women in preterm labor. However, improved tertiary care has not led to a significant decrease in the incidence of severe IVH/PVHI [3, 4]. The incidence of severe IVH has remained higher than 10% among neonates born before GW 28 for the past 10–15 years [14, 15]. Only a recent large retrospective cohort study has found a decrease in severe IVH with antenatal steroid exposure as the only factor associated with the lower odds of severe IVH [3].
In at least 50% of affected children the onset of GMH-IVH occurs on the first day of life, with rapid progression to severe grade IVH/PVHI within 1–3 days [5, 17]. However, in some neonates severe grade IVH/PVHI is detectable already during the first postnatal hours and may be of antenatal origin [5, 19]. Cerebral ultrasonography (US) is able to identify IVH/PVHI that has occurred before birth with residual findings such as ventricular dilatation, clots, strands and parenchymal cystic defects suggestive of antenatal origin [5, 12]. Earlier prolonged fetal stress, multiple pregnancy [12] and presence of thrombophilia [19] have been suggested as risk factors for early severe IVH/PVHI which may be antenatal in origin. Without a clear etiology, it is difficult to prevent antenatal IVH/PVHI carrying poor prognosis in severely preterm neonates.
The aim of the study was to elucidate pregnancy and delivery associated risk factors in preterm neonates with severe IVH/PVHI visible on early cerebral US within the first 12 h of birth. We hypothesised that the pregnancy and delivery associated risk factor profile is different in preterm neonates with antenatal compared to neonatal onset of severe IVH/PVHI.
Patients and Methods
Patients
The study was performed at a third level regional university hospital, which is one of the two pediatric referral centers serving about one third of neonatal cases in Estonia. The study subjects were identified from the hospital’s electronic database using the international classification of diseases 10th revision (ICD-10) [20] (diagnosis codes P 52.2., 0.3, 0.4, 0.6, 0.9), medical records of the neonatal intensive care unit and the Estonian Pediatric Stroke Database [21] between 1994 and 2019.
Inclusion criteria
Preterm neonates < 35 GW were identified from the clinical database if at least one of the following findings on cerebral US was present: (1) severe IVH (grade III or IV IVH) or PVHI, (2) residual intraventricular clots or strains (4) periventricular cystic changes within the periventricular stroke area [3, 12].
Exclusion criteria
Neonates were excluded if they had at least one of the following: (1) acute arterial ischemic stroke, (2) sinovenous thrombosis, (3) parenchymal hemorrhagic stroke in locations other than the periventricular area, (4) other diseases, congenital anomalies or genetic diseases involving the central nervous system, (5) second child of a twin pregnancy when both had severe IVH or PVHI.
Imaging data abstraction
GMH-IVH was graded based on the findings of cerebral US as follows: grade I –germinal matrix hemorrhage; grade II –intraventricular blood without distension of the ventricular system; grade III –blood filling > 50% of the ventricle and distending the ventricular system; and grade IV –haemorrhage involving periventricular parenchymal tissue, also known as PVHI [4, 23]. PVHI was defined on cerebral US as a parenchymal increase in echogenicity in the periventricular area, which can be fan- or globular-shaped and separate or partially communicating with the ventricle, progressing into a small periventricular cystic lesion or a larger porencephalic cyst with enlargement of the lateral ventricle [7, 24]. In case bleeds of different severities were documented, the most severe grade was assigned to the patient and used for analysis. All cerebral US studies during the study period were performed by pediatric radiologists. The initial US was done on the first day after the patient was stabilized, followed by imaging on the 2nd-3rd day and 5th–7th day and further once a week until discharge. If the status of the patient deteriorated and severe IVH/PVHI was clinically suspected, US was promptly repeated.
The diagnosis of PVHI was confirmed by magnetic resonance imaging (MRI) around the corrected age of GW 40 or later during infancy. On MRI PVHI was defined as mostly unilateral medullary venous territory infarction in the periventricular area, with porencephalic enlargement of the lateral ventricle and/or periventricular gliosis and/or cystic changes. Hemosiderin as a residue from hemorrhage, could be visible in susceptibility weighted images in the area of the germinal matrix, in the lateral ventricle and/or in the lesion on MRI [21, 26].
All radiological images were reviewed independently by neuroradiologists who were blinded to the clinical outcome of the patients. Cerebral US findings were available for review in all cases, MRI was available for review for 51/64 (80%) patients who survived and the parents agreed with MRI investigation. Autopsy was available for 47/56 (84%) patients who died, one patient (1.8%) had MRI before death. If MRI (n = 13) or autopsy (n = 6) revealed periventricular infarction in children with grade III IVH on cerebral US, the case was reclassified as PVHI. Details of the diagnostic criteria of PVHI have been published previously [21].
Study groups
Preterm neonates with severe IVH/PVHI were grouped based on the detection time of severe IVH/PVHI on cerebral US: Antenatal onset group with (a) acute grade III IVH or PVHI visible on the first cerebral US before 12 h of age or (b) periventricular cystic changes in the area of PVHI visible within 3 days of life, indicating antenatal development of PVHI [5, 12]. Unknown time-onset group with grade III IVH or PVHI visible during the first cerebral US performed later than 12 h of age, leaving the time of its development unknown. Neonatal onset group with initial normal cerebral US or grades I-II GMH-IVH with later progression of hemorrhage to grade III IVH or PVHI.
Illustrative cases of neonates with antenatal and neonatal development of PVHI are shown in Supplemental Figure 1.
Controls
The control group was based on an observational cohort of Happy Pregnancy Study (full name: “Development of novel non-invasive biomarkers for fertility and healthy pregnancy”, Principal Investigator Prof. M. L.), representing approximately 2/3 of all pregnant women receiving antenatal care at the Women’s Clinic of the third level regional university hospital between 2013 and 2015 [21]. Unselected pregnant women were recruited prospectively during their first antenatal visit (n = 2334). From this cohort, mother-child pairs with deliveries occurring before GW 35 were included in the control group with the exclusion of one mother-child pair in which case the child developed PVHI and was included in the study group.
Pregnant women with imminent preterm delivery were transported to the third level maternity hospital. Affected and control children were followed and delivered at the third level hospital. However, 26 of 120 (22%) premature neonates with IVH/PVHI were born at county hospitals and transferred to a tertiary neonatal intensive care unit by a specialized team including an anesthesiologist from the neonatal intensive care unit.
Data collection
Clinical data about pregnancy and delivery were extracted from maternal and neonatal, as well as from child’s medical records, and from autopsy and placental histology reports. Data about the parental lifestyle, risk factors and complications during previous and index pregnancies, as well as delivery data including neonatal resuscitaion of children with IVH/PVHI were requested from 14 hospitals, where antenatal care had been provided and/or delivery had taken place, and were reviewed retrospectively. For the control group, epidemiological data including reproductive history, parental lifestyle, and pregnancy risk factors were collected prospectively from three questionnaires completed at each trimester of pregnancy.
Data on in- and out-patient antibacterial treatment during pregnancy were retrieved from hospitals medical records (available for study patients) and from the population based Digital Prescription Centre (available only for the control group) along with the exact diagnosis of infection according to ICD-10 [20]. Only the infections treated with antibiotics were included. The infections of pregnant women were grouped based on organ systems: (1) infections of the genitourinary tract, (2) infections of all other organ systems including the respiratory and the digestive systems, (3) no bacterial infection.
Data about neonatal treatment and complications including initial neonatal physiology score for patients of the intensive care unit (SNAPPE II) [27] within 24 h of birth, time of diagnosis of grade III IVH/PVHI and autopsy findings and cause of death were collected from the intensive care units of the regional university hospital.
The definitions of diseases and a full list of collected findings are provided in supplemental material.
Ethics statement
The studies were approved by the Ethics Review Committee on Human Research of the University of Tartu. The Happy Pregnancy Study was approved by protocol no 221/T-6, 17.12.2012 and no 286/M-18, 15.10.2018 and all recruited participants signed a written informed consent. The PVHI study was approved by protocol no 294/M-18, 17.06.2019. As data about the risk factors for children with PVHI were collected retrospectively, the Committee waived the need for patient consent.
Statistical analysis
Prior to further analysis, normality of the data was evaluated using the Shapiro-Wilk test. Continuous data were summarized as medians with the interquartile range (IQR), and categorical data, as absolute counts and percentages. Differences between the groups were analysed by the nonparametric Kruskal-Wallis test for continuous variables. For dichotomous variables, univariate comparison was analyzed using multinomial logistic regression. All findings were adjusted for gestational age and year of birth, except for low frequency findings (in vitro fertilization, no intubation or hypothermia on admission). Odds ratio (OR) with the 95% confidence interval (CI) was estimated as the measure of association. All raw p values were two-sided. Multiple testing was corrected using the false discovery rate (FDR) linear step-up procedure [28]. The Benjamini-Hochberg critical values were calculated as (i/m)Q, where i is the rank in an ascending list of p values, m is the total number of tests, and Q is a false discovery rate of 0.1. The cut-off p value for significance of a single comparison for a set of risk factors was 0.026 between the antenatal versus neonatal onset group, 0.002 between the antenatal onset versus unknown time group, 0.009 between the unknown time-onset versus neonatal onset group, 0.021 between the antenatal onset group versus controls, 0.012 between the unknown time- onset group versus controls, 0.004 between the neonatal onset group versus controls.
Finally, a multiple logistic regression model was developed to describe the influence of the risk factors for IVH/PVHI for the study groups and controls. Variables with p < 0.2 in univariate comparison were entered in a stepwise multiple logistic regression model. Firth’s penalized likelihood approach was used for the models [29]. Model fits were examined by inspecting residual plots and calibration measures (Akaike information criterion). The output of the logistic regression model is presented as adjusted odds ratios (aOR) and the 95% CI. Statistical evaluation was performed using the statistical package SAS version 9.4 (SAS Institute, Cary, NC) and the R Statistical Software (version 4.0.2).
Results
Patients with severe IVH/PVHI (n = 131) were identified from the clinical database, 11 neonates were excluded from the study as they were the second twin of an affected set. The final study group consisted of 120 patients as shown in Fig. 1.
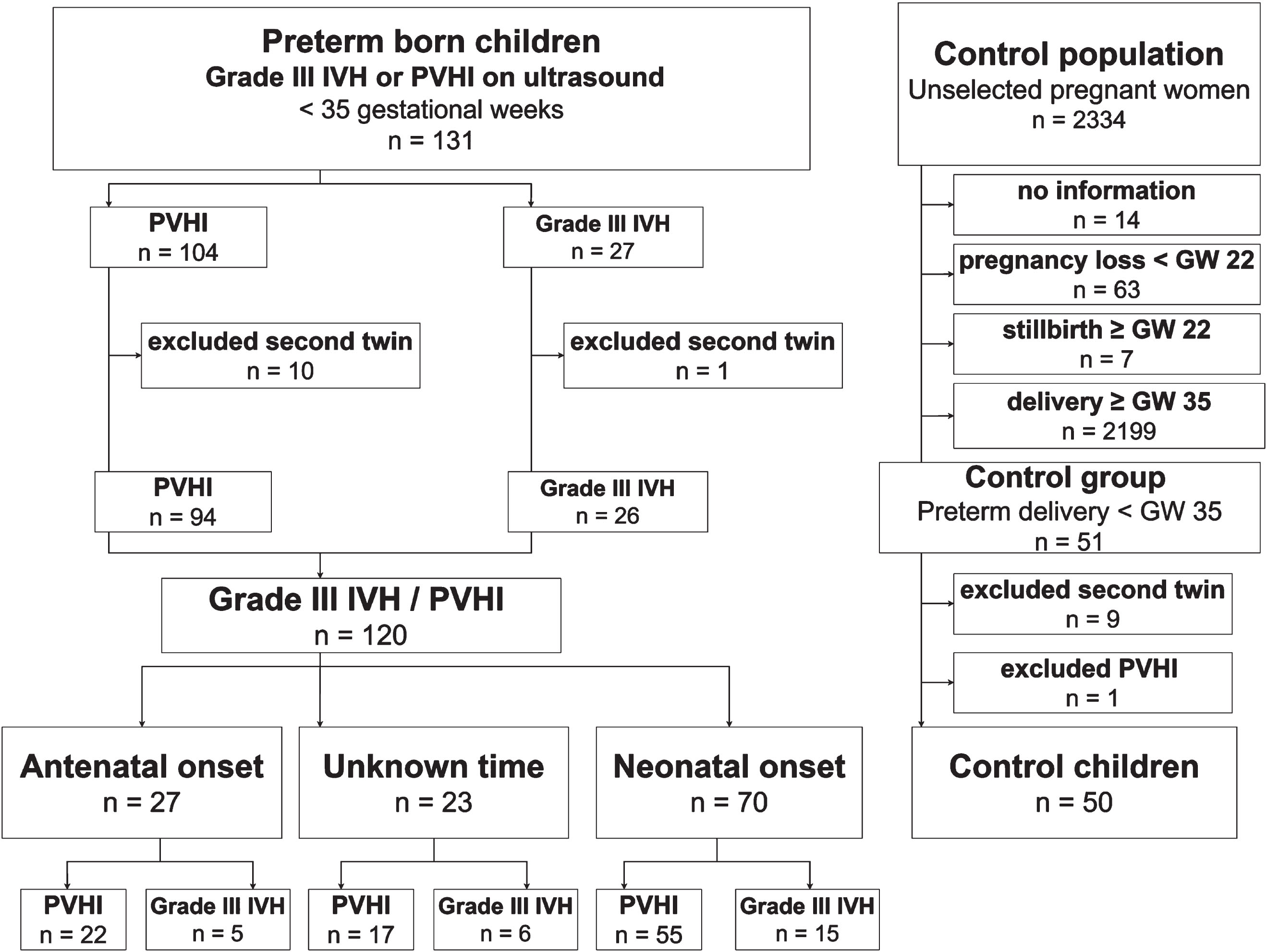
Study flowchart. (IVH-intraventricular hemorrhage, PVHI-periventricular hemorrhagic infarction, GW-gestational weeks.).
Among the preterm neonates with severe IVH/PVHI, 27/120 (22.5%) were considered antenatal in origin, including 20 neonates with IVH/PVHI visible on the first cerebral US (<12 h of age) and 7 neonates with cystic changes in the area of PVHI visible within 3 days of life. In 70/120 (58%) neonates severe IVH/PVHI developed during the neonatal period. All participants of this study were Caucasian. The demographic data for the study groups, as well as for the control group (n = 50) are provided in Table 1.
Maternal, pregnancy and delivery related risk factors. Data are presented as median (IQR) if not stated otherwise
Maternal, pregnancy and delivery related risk factors. Data are presented as median (IQR) if not stated otherwise
The data are presented as median and interquartile range [IQR] if not stated otherwise. Gravidity, total number of pregnancies including index pregnancy; Parity, number of births including index birth; GW, gestational weeks; N, number of subjects; n, number of observations; OR, odds ratio; CI, confidence interval for odds ratio; N/A, not applicable; g, gram; cm, centimeter, y, years, SNAPPE II, score for neonatal acute physiology and SNAP perinatal extension. Comparisons made with the Kruskal Wallis test for continuous variables and multinomial logistic regression for dichotomous variables. Statistically significant difference for a single comparison was set at 0.029 (FDR, false discovery rate). aAntenatal onset versus control. bUnknown time-onset versus control. cNeonatal onset versus control.
The mothers of the neonates representing the antenatal, the unknown and the neonatal onset groups were of similar age, had a similar number of previous pregnancies and deliveries, threatened pregnancy losses and imminent preterm deliveries. The neonates of all study groups with IVH/PVHI were similar regarding sex distribution; birth weight, head circumference and Apgar score, as well as SNAPPE score within the first 24 hours. The neonates of the control group had significantly higher gestational age, larger birth weight and head circumference and their mothers had a higher number of imminent preterm deliveries at GW≥22 compared to the unknown time-onset and neonatal onset groups.
Pregnancies in the antenatal onset group were characterized by a significantly higher prevalence of maternal bacterial infections compared to the neonatal onset group (p = 0.0008; aOR 5.7 95% CI 2.1–16)) (Fig. 2). The mothers of the antenatal onset group were more often diagnosed with genitourinary tract infection, including pyelonephritis, compared to the neonatal onset group (p = 0.005), but were also more often diagnosed with other infections (p = 0.006). After FDR correction, pyelonephritis alone was not significantly different between the antenatal and the neonatal onset group. Clinical signs of chorioamnionitis (p = 0.025) and histological chorioamnionitis (p = 0.025) were more often found in the antenatal compared to the neonatal onset group. There were no differences in other prenatal risk factors between the groups. The prevalence of pregnancy, delivery and neonatal risk factors and complications for all IVH/PVHI study groups and for the control group is provided in Supplemental Table 1.
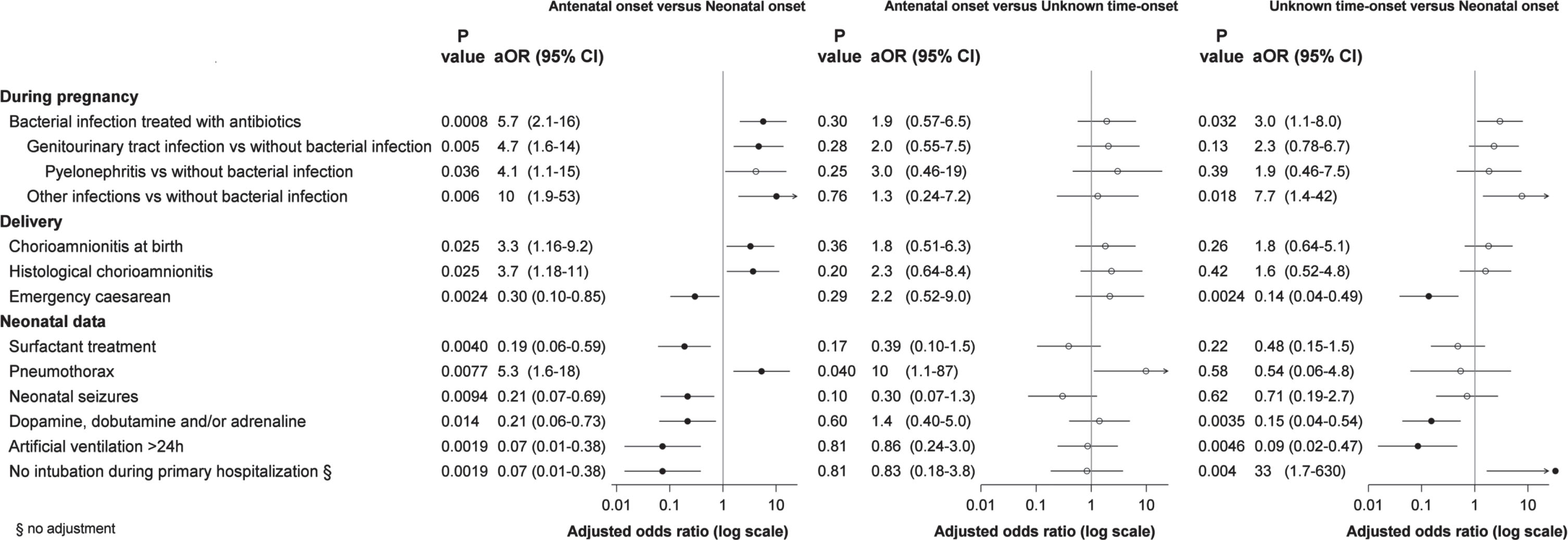
Pregnancy, delivery and neonatal risk factors in the antenatal, unknown time and neonatal onset IVH/PVHI groups. (aOR-adjusted odds ratio, CI-confidence interval, vs –versus, h-hours). The figure only presents statistically significant risk factors. All findings were adjusted for gestational age and year of birth, except for low frequency finding (no intubation). The cut-off p value for significance of a single comparison for a set of risk factors (FDR, false discovery rate) was 0.026 between the antenatal versus neonatal onset group; 0.002 between the antenatal versus unknown time onset group; 0.009 between the unknown time versus neonatal onset group. Only statistically significant characteristics are presented.
Compared to the neonatal onset group, the neonates of the antenatal onset group and the unknown time of onset group showed significantly less risk factors related to fetal or neonatal stress and complications, such as emergency caesarean sections, need for cardiotonic/vasoactive treatment and artificial ventilation > 24 h. Neonates in the neonatal onset group needed more frequent surfactant treatment and had neonatal seizures and persistent arterial duct requiring medical or surgical treatment (Fig. 2). Pneumothorax and no need for intubation during primary hospitalization occurred more often in the antenatal compared to the neonatal onset group (Fig. 2). Four of the 27 neonates (15%) in the antenatal onset group and 4/23 (17%) in the unknown time-onset group were never intubated, which is significantly more compared to the neonatal onset group.
Multiple logistic regression analysis adjusted for gestational age and year of birth revealed bacterial infection (p = 0.0013; aOR 5.8 95% CI (2.0–17)) and no need for surfactant treatment (p = 0.0054; aOR 6.1 95% CI (1.7–22)) as predictors for the antenatal onset IVH/PVHI compared to the neonatal onset group.
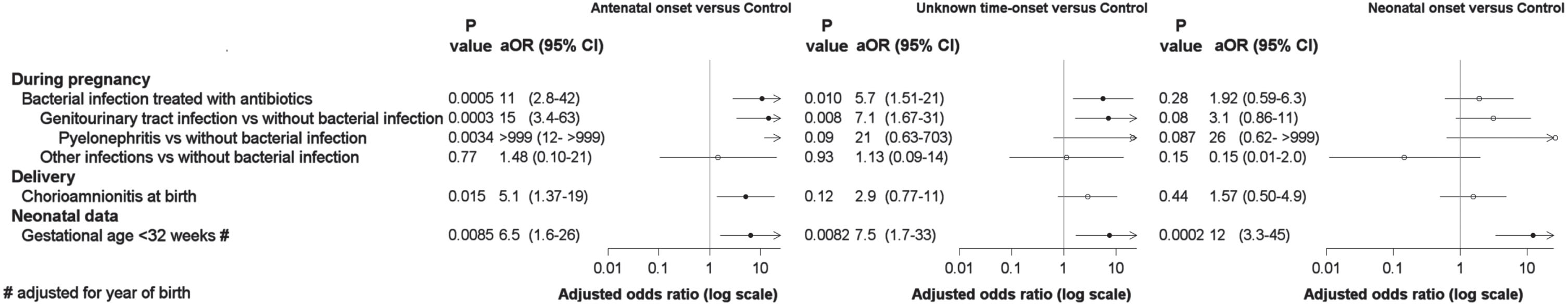
Pregnancy and delivery related risk factors in the antenatal, unknown time and neonatal onset IVH/PVHI groups compared to the control group. (aOR-adjusted odds ratio, CI-confidence interval). The figure only presents statistically significant risk factors. All findings were adjusted for gestational age and year of birth, except for the variable gestational age (adjusted for year of birth). The cut-off P value for significance of a single comparison for a set of risk factors (FDR, false discovery rate) was 0.021 between the antenatal onset group versus controls, 0.012 between the unknown time-onset group versus controls, 0.004 between the neonatal onset group versus controls.
The mothers of the antenatal and unknown time of onset groups had significantly more bacterial, including genitourinary, infections, compared to the mothers of the control group (p = 0.005 and p = 0.01, respectively). The mothers of the antenatal onset group had also significantly more often pyelonephritis and clinical signs of chorioamnionitis at birth compared to the mothers of the control group, but pyleonephritis did not remain significant after FDR correction. The other prenatal and delivery rated risk factors were not different between the study and control groups.
Multiple logistic regression analysis adjusted for gestational age and year of birth revealed bacterial infection (p = 0.0067; aOR 14 95% CI (2.1–98)), maternal chorioamnionitis at birth (p = 0.021; aOR 15 95% CI (1.5–143)), no signs of imminent preterm delivery (p = 0.039; aOR 8.2 95% CI (1.1–61)) as independent predictors for antenatal onset IVH/PVHI compared to control. Emergency caesarean section (p = 0.03; aOR 6.5 95% CI (1.2–36)) remained the only independent predictor for neonatal onset IVH/PVHI compared to control.
The commonest bacterial infection in the mothers of both the study and the control groups was genitourinary infection, accounting for at least 63.6% of all infections during pregnancy in all groups. There were no differences between the proprotions of genitourinary and other infections across all study groups (Table 2).
The mothers of the control group had infection significantly earlier compared to the mothers of the antenatal and neonatal onset groups. Most of the mothers with infection in the control group (81.8%) had bacterial infection at GW < 21 compared to only 15.8% in the antenatal onset group, 35.7% in the unknown time-onset group and 13% in the neonatal onset groups (Table 2).
Data on bacterial infections in the study groups adjusted for birth year and gestational age
Data on bacterial infections in the study groups adjusted for birth year and gestational age
GW, gestational week; IQR, intequartile range, N represents number of bacterial infections treated with antibiotics during pregnancy/all patients. Data for infection are presented as n (%) of all infections in the group. *No adjustment. Statistically significant difference for a single comparison was set at 0.009 (FDR, false discovery rate). aAntenatal onset versus control. bNeonatal onset versus control.
Our study provides evidence that antenatally and neonatally developing severe IVH or PVHI in premature neonates may have different etiologies. The antenatal environment, especially maternal bacterial infection and/or chorioamnionitis, may be a risk factor leading to antenatal development of severe IVH or PVHI, visible already within the first 12 hours of birth. We found significantly more bacterial infections (both of the genitourinary tract and other infections) and histologically proven chorioamnionitis in the mothers of children with antenatal severe IVH or PVHI, compared to the mothers of children with neonatal development of severe IVH/PVHI. Inversely, children with normal initial cerebral US findings or grades I-II IVH with later progression to severe IVH/PVHI had significantly more often acute complications during delivery or postnatal intensive care.
In recent years, considerable attention has been paid to the likelihood of antenatal onset of intracranial lesions, which can lead to development of cerebral palsy during infancy [12, 30–32]. In our study of neonates with severe IVH/PVHI, the pathology was visible within the first 12 hours in almost a quarter of cases and was considered antenatal in origin. Ischemic cerebral injury has been found in 40% of stillborn infants [33, 34] and periventricular white matter was the main site of damage associated with placental infarcts in 62 of 70 cases [34]. In a prospective study of infants with PVHI born before GW 34, 11.7% died and 40.3% of the patients who died had a large brain lesion considered to be of antenatal onset in 26.9% of the cases [12].
Several meta-analyses have shown significant association between histological chorioamnionitis and cerebral palsy [30, 35]. Acute chorioamnionitis is associated with neutrophilic inflammation of the placental tissues thought to result from an ascending bacterial infection [32]. Our findings indicate that besides chorioamnionitis, other antenatal maternal bacterial infections, especially genitourinary infections, can be an important risk factor for development of severe IVH/PVHI already before birth. Maternal bacterial infection was found to be an independent predictor for antenatal onset IVH/PVHI compared to neonatal onset IVH/PVHI in multiple regression analysis in our study, and bacterial infection, maternal chorioamnionitis at birth and no imminent preterm delivery were independent predictors for antenatal onset IVH/PVHI compared to control. In at least 30–40% of cases preterm delivery occurs due to maternal or fetal infection and can initiate fetal inflammatory response leading to damage of the developing brain [32, 36]. Understanding the temporal effects of antenatal infection in relation to onset of neurological injury is crucial for promotion of neurotherapeutics and prevention strategies for preterm infants [12, 32].
Maternal genitourinary infection was the most prevalent infection in all PVHI groups and in the control group in our study. Still, the mothers of the control group had asymptomatic bacteriuria mostly during the first half of pregnancy, while none of them developed pyelonephritis. In an earlier study a reduction in the incidence of pyelonephritis was seen after a screening program of asymptomatic bacteriuria was introduced [37]. Untreated asymptomatic bacteriuria frequently leads to symptomatic or recurrent urinary tract infection and these mothers have up to a 20–30% risk of pyelonephritis later in pregnancy [38]. Acute antepartum pyelonephritis was found to be an independent risk factor for preterm delivery [39].
Children with neonatal development of severe IVH/PVHI had significantly more often distress at birth and were born in a critical condition compared to children with antenatal PVHI. All infants in the neonatal group were intubated at birth and most of them needed artificial ventilation for more than 24 hours and required cardiotonic/vasoactive drugs. Multiple logistic regression analysis revealed emergency caesarean section as an independent predictor for neonatal onset IVH/PVHI compared to control, referring likely to fetal distress. Neonates with antenatal onset and unknown time-onset IVH/PVHI had generally milder symptoms after preterm birth and some never needed intubation and artificial ventilation, suggesting that these children had survived the acute period and were stable by the time of birth. These children would have been easily missed unless screened by cerebral US. Similarly, some fetuses with antenatal stroke may survive in utero, stabilize and be born at term without neurological symptoms during the neonatal period [21]. Our earlier study showed that the mothers of term born children with presumed antenatal periventricular venous infarction had significantly more infection, especially pyelonephritis, during pregnancy between GW 24–32, demonstrating once more the role of infection as an important cause of antenatal IVH/ PVHI [21].
Poor outcome of neonates with severe IVH/PVHI has an enormous familial, societal and financial burden [2], despite the implementation of both pharmacologic and care-oriented prevention strategies for preterm neonates. In order to further reduce the incidence of IVH and thereby cerebral palsy in the preterm population, prevention strategies must include active measures to prevent and immediately treat all maternal bacterial infections. The awareness of infection related risks in pregnant women, particularly in the early second half of pregnancy, should be raised among obstetricians and other specialists who regularly treat bacterial infections in pregnant women.
Our study demonstrates that all preterm neonates born after any kind of antenatal maternal bacterial infection and/or chorioamnionitis need early cerebral US to detect antenatal development of IVH/PVHI. Unlike term born children with perinatal stroke, preterm born children do not typically present with seizures [40]. Some pretem born children with antenatal infection and IVH/PVHI may have only mild symptoms after birth and their antenatal onset of severe IVH/PVHI can easily be missed. Routine cerebral US assists early diagnosis of antenatal IVH/PVHI [40] and prompts early rehabilitation.
The study has several limitations. The study period was long involving interventions with a major effect on neonatal outcome, like asymptomatic bacteriuria screening of mothers, prenatal transfer of risk mothers to higher-level hospitals, and antenatal steroid use and surfactant treatment instituted during that period. In addition, although the level of intensive care and socio-economic standard improved, the influence of pregnancy related risk factors remained unchanged. To minimize possible bias, risk factor analysis was adjusted for year of birth and gestational age, and an approach incorporating standardized definitions of the risk factors was employed. Considering that the data about all prescribed and purchased antibiotics for the control group during pregnancy was available, there were still significantly less mothers with infection in this group compared to the IVH/PVHI groups. If anything, we could have underestimated the true prevalence of infections in the IVH/PVHI groups due to missing data for outpatient antibiotic treatment or bacteriuria screening.
Conclusion
Bacterial infection during pregnancy is an important risk factor for development of antenatal onset severe IVH or PVHI. In neonates born to mothers with severe bacterial infection during pregnancy, cerebral ultrasonography is indicated for early detection of severe IVH or PVHI.
Footnotes
Acknowledgments
The authors thank all pregnant women, patients and their parents, as well as the staff of the archives of the hospitals, for their contribution.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed by the Estonian Research Council grants [PUT 148, PRG1021 and PRG1912] and grant of the Tartu University Hospital PR 143/2022.
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest.
