Abstract
INTRODUCTION:
Retinopathy of prematurity (ROP) is one of the main reasons of preventable childhood blindness. In the development of ROP, MicroRNAs may be effective in the balance of factors that inhibit and activate angiogenic factors. We aimed to determine the changes in the blood levels of miR-146a, miR-143, miR-210, miR-21, miR-126, miR-211, miR-221, miR-106 and let 7f and to investigate their association with ROP. We hypothesed that the level of these miRNAs changed significantly in ROP cases.
MATERIALS AND METHODS:
This observational study was conducted prospectively in preterm infants with ROP. Serum levels of 8 miRNAs were measured. The relationship between disease stage and progression with miRNA gene expression was analysed. Preterm infants without ROP were taken as the control group.
RESULTS:
47 patients with ROP and 14 controls, were included in the study. In the ROP group, miR-210, miR-146a, miR-21 were statistically significantly lower. In the ROP group the expression level of miR-143 was insignificantly lower, miRNA-221 was insignificantly higher, and miR-106, miR-126 and let 7f were variable.
CONCLUSION:
It was observed that miR-210, miR-146a, miR-21 and miR-143 were significantly lower in patients with ROP compared to the control group. However, no association could be established between the type of miRNA and stage of ROP. These miRNAs may be used as adjunctive biomarkers for diagnosis of ROP.
Introduction
Retinopathy of prematurity (ROP) is one of the main causes of preventable childhood blindness. It develops after abnormal proliferation of retinal vessels, and although low gestational age and inadvertent oxygen usage have been reported as the main contributing factors, a large number of angiogenic factors such as vascular endothelial growth factor (VEGF), insulin-like growth factor (IGF-I), angiotensin converting enzyme (ACE), fibroblast growth factor (bFGF), tumor necrosis factor-α (TNF-α), and nitric oxide (NO) are also effective in the pathogenesis of ROP [1]. MicroRNAs (miRNAs) are small single-chain RNA molecules approximately 20-22 nucleotides in length that are not encoded and are involved in many cellular events and regulate gene expression post-transcriptionally. By this pathway, they affect the differentiation of cells, apoptosis, immune processes, and angiogenesis. They are highly expressed in endothelial cells and miRNAs may play a role in the balance of factors that inhibit and activate angiogenic factors and in the process of regulating vascular integrity and angiogenesis [2]. They are also effective in hypoxic conditions for the regulation of VEGF and other angiogenic factors. Many studies suggest that the effects or perturbations of miRNAs may affect the pathogenesis and/or treatment of retinal diseases in children and adults [3, 4]. miR-210 plays a significant role in vascular integrity and it is regulated by hypoxia. Many miRNAs have been found to be associated with retinal diseases in adults and children. An increase in the expression of miR-21 decreases the rate of proliferation and migration of endothelial cells as well as their ability to perform tubular structures and angiogenesis [5]. It is known that miR-146a is a proinflammatory agent effective in vascular formation though interleukin-1 and TNF-α [6]. MiR-221 blocks the proliferation and migration of endothelial cells and inhibits angiogenesis by induction of apoptosis. MiRNA inhibits cell growth and angiogenesis by regulating IL-6 and HIF-1a [7]. MiRNA-143 and MiRNA-126 are directly involved in retinal neovascularization. The let-7 family are expressed in retinal and choroidal endothelium cells and suppress angiogenesis [8]. Overexpression of miR-106a decreases HIF-1a and VEGF [9]. These effects are summarized in Fig. 1 [10]. Therefore, in this study, we aimed to determine the changes in the blood levels of miR-146a, miR-143, miR-210, miR-126, miR-211, miR-221, miR-21, miR-106 and let 7f and to investigate their association with ROP. We hypothesed that the level of these miRNAs changed significantly in ROP cases.
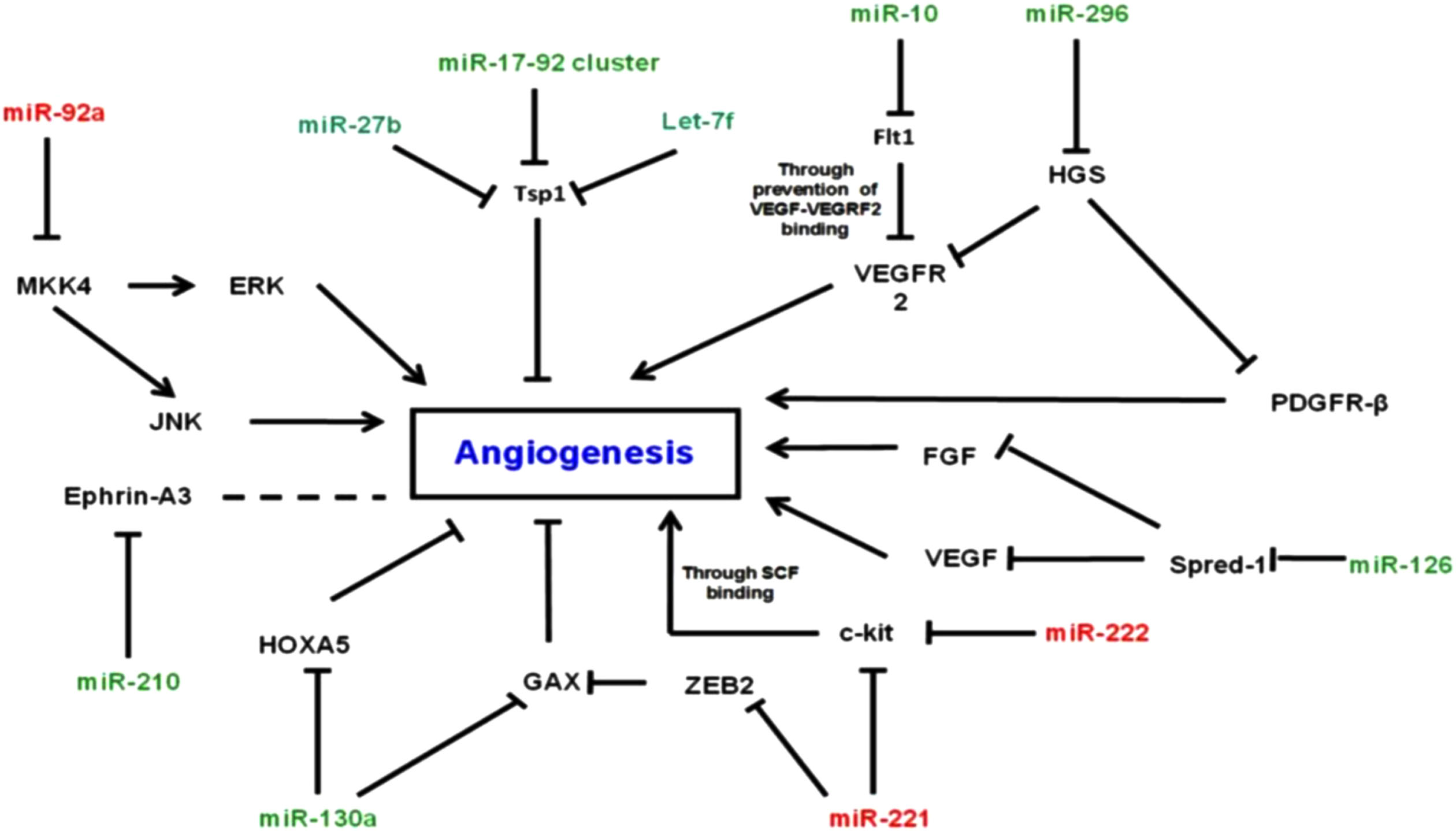
MiRNAs effective in the process of angiogenesis.
The study was conducted prospectively in two university hospitals with preterm infants <32 weeks of gestation with the diagnosis of ROP at every stage during October 2019 and October 2020. ROP screening was done as per the Turkish Neonatal Society Guidelines [11]. Namely: all infants <32 weeks of gestation or <1500 grams were screened at the postconceptional 31st week or 4th postnatal week, whichever comes later. Infants who were deemed vulnerable according to the attending neonatologists were also screened at the same ages. Infants were evaluated according to the ICROP criteria by a consensus grader [12]. Infants with congenital heart diseases or other congenital anomalies were excluded from this study. When the diagnosis of ROP was made, two milliliters of venous blood was obtained from each infant during the ophthalmologic examination and was frozen to –80°C until the day of analysis. If the stage of ROP progressed, the analysed samples were taken during the worst stage of ROP. Therefore, the final phenotype and analysis were performed on the worst stage of ROP in every infant. Serum levels of 8 miRNAs were measured by real-time PCR. For this purpose, miRNeasy Serum/Plasma Kit (Qiagen, Catalogue no:217184) and miRNeasy Serum/Plasma Spike-In Control (Qiagen, Catalogue no: 219610) were used. The miRNA primers used are shown in Table 1. PCR cycles were repeated for 45 times. Gender, gestational age, birth weight, delivery pattern, morbidity, and ROP stages were recorded and compared. The relationship between the disease stage and progression and the miRNA gene expression was analyzed. Preterm infants without ROP were taken as the control group. Informed consent was obtained from the parents of all infants. The study was approved by the local ethics committee.
MicroRNA primers
MicroRNA primers
Statistical analysis was done using the SPSS version 17.0 program. For quantification of data obtained from RT PCR with SybrGreen dye for all reactions, Rotor-gene 2.1.0.9 software was used. Melt-curve analysis was done after each cycle and standarts developed by Tunçtürk et al. were used [13]. Analyses were done by dynamic tube and slope correct function. Fold regulation, fold change and significance were calculated online by using the gene globe data analysis center website. (at www.qiagen.com) Fold change is the ratio of gene expression in the sample to the controls. Fold regulation is the expression of fold change results in a biologically plausible way. p values <0.05 were deemed significant. Fold changes <1 were assumed to be “decreased” and fold changes >1 were assumed to be “increased”. Logistic regression analysis was done to determine the effect of these changes regarding birthweight and gestational age. Hosmer-Lemeshow goodness of fit statistics were used to assess model fit.
During the study period, there were 71 patients <32 weeks of gestation. Three infants with congenital heart diseases were excluded. Five patients were also excluded since their parents did not give consent for participation. A total of 63 infants including 48 patients with ROP and 15 controls were included in the study. The final analysis was performed in 61 infants, since sufficient data among the miRNAs could not be obtained in two samples (Fig. 2). All infants were Caucasian. Of the infants who had ROP, 29 (60.4%) were boys. Mean gestational age was 29.48±2.96 vs. 31.80±1.83 weeks and mean birthweight was 1312±454.93 g vs 2008±626.19 g respectively. 58 infants were delivered by cesarean section and 5 infants were delivered vaginally (Table 2). Mean days on mechanical ventilation, oxygen treatment, RDS, BPD, and red blood cell transfusions were significantly higher in the ROP group. In infants with respiratory distress syndrome (RDS), neonatal sepsis, bronchopulmonary dysplasia (BPD) and infants who received blood transfusions, ROP was significantly more common. The duration of oxygen therapy was also more prolonged in cases with ROP (15.60±11.83 vs. 4.67±6.94 days; p <0.001). However, there was not any correlation between miRNAs and duration of mechanical ventilation or oxygen treatment. There were no significant differences with regard to necrotizing enterocolitis and intraventricular hemorrhage in cases with or without ROP. Seven infants received anti-VEGF treatment and in 27 infants, ROP regressed. The rate of recovery was lowest in Stage 3 ROP (27.7%) and was higher in other stages (87.5% in Stage 1 and 57.1% in Stage 2). miRNA outcomes after the treatment of ROP were not analysed.
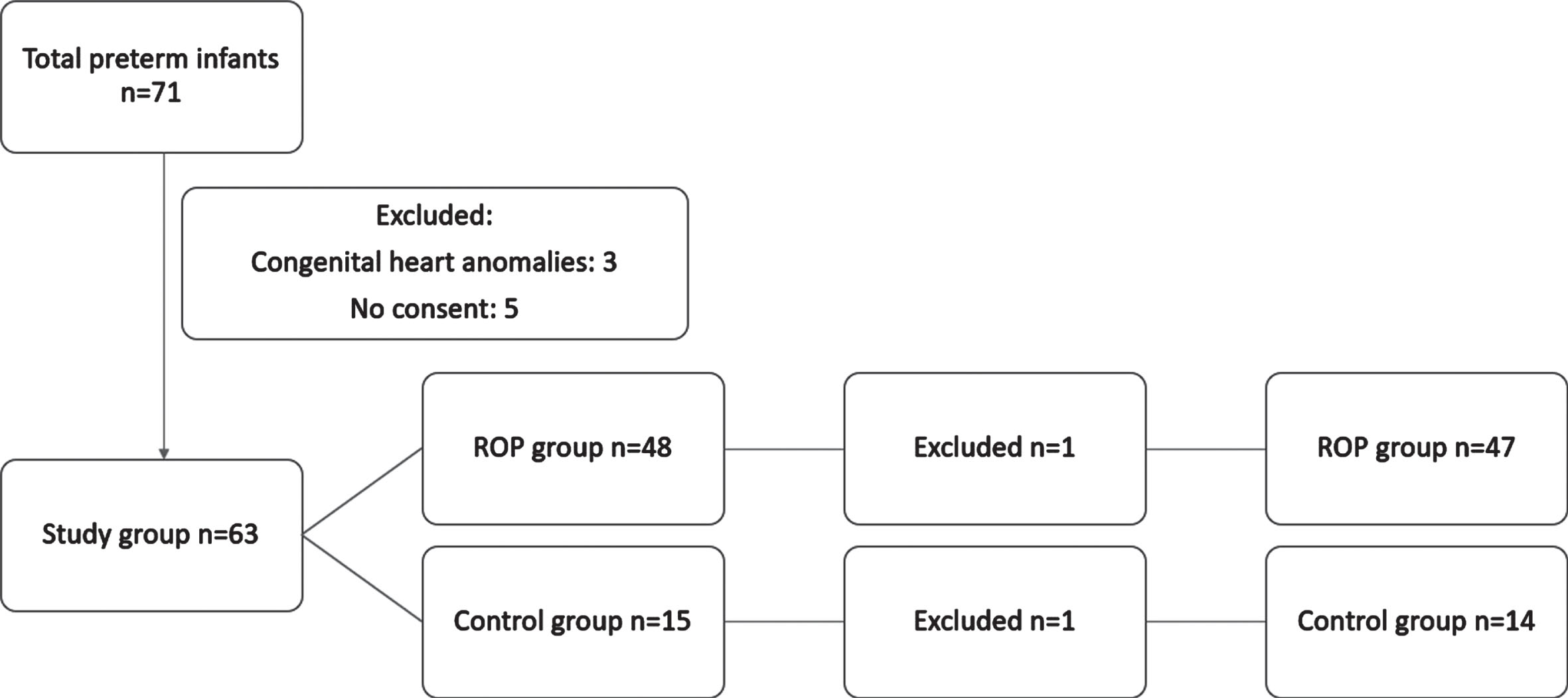
Flow diagram of patients included in the study.
Demographic data of groups
Most of the ROP cases were in Zone 2 and stage III, as shown in Table 3. Fold regulations in both groups are shown in Table 4. The clustergram figure is shown in Fig. 3.
Infants in different ROP stages
Fold regulation of miRNA
FR: Fold regulation; ROP: Retinopathy of prematurity *:Significant.
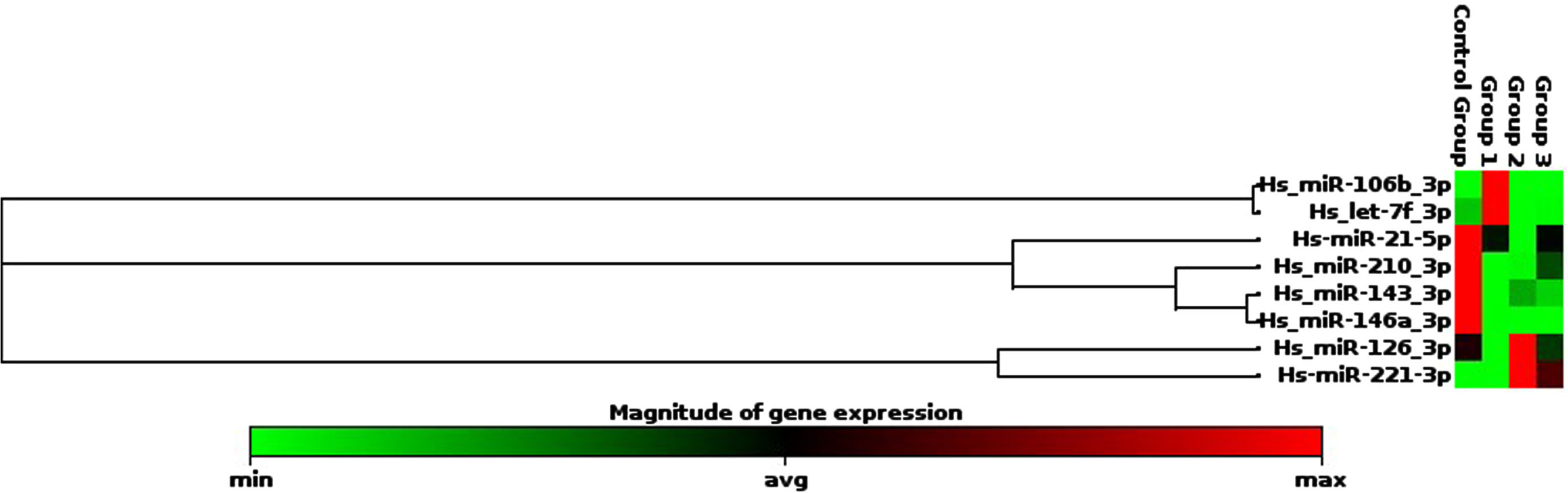
Clustergram of miRNAs.
In the ROP group, miR-210, miR-146a, and miR-21 were statistically significantly lower. In the ROP group the expression level of miR-143 was insignificantly lower, miRNA-221 was insignificantly higher, and miR-106, miR-126 and let-7f were variable. Fold changes with regard to ROP stages are given in Table 5. In multivariate analyses, birthweight and gestational age did not seem to affect the results for miR-210, miR-146a, and miR-21 (RR:0.97 (0.89-1,04)) and for miRNA-221 (RR:1,02 (0,95-1,09)).
Fold changes in different stages of ROP
CI: Confidence interval.
There are many environmental factors that affect the development of ROP. However, the role of genetic factors has not been studied extensively. Changes in the expression of various miRNAs have been implicated in the pathogenesis of ROP. MiRNA are not specific for the cells in which they are produced; and they may be translocated to other parts of the body by blood circulation. In many studies miRNAs were found to be effective in the development and differentiation of the eye. Karali et al. have reported that miR-9, miR-29c, miR-124a, miR-181a, miR-182, miR-184, and miR-204 are effective in the development of the eye — miR-184 in the lens and cornea and the rest in the retina [14]. During the normal development process of the eye, VEGF is the main molecule regulating the proliferation and migration of endothelial cells and the control of vascular permeability and vascular integrity. The association between miRNAs and ROP has been studied only in severe cases of ROP. Metin et al. have studied miRNAs in the development of Stage III ROP in animals [4]. To the best of our knowledge, this study is the first of its kind, evaluating the effect of miRNAs in all stages of ROP. However, since these tiny preterm infants have many other complications associated with prematurity and not specifically to ROP, these associations should be interpreted cautiously.
MiRNA-210 has been shown to increase in hypoxic response in cases of diabetic retinopathy, hypoxic-ischemic encephalopathy, cerebral ischemia, and ROP [15]. In our cases it decreased linearly in Stage I through Stage III ROP. MiRNA-210 is a counter-regulatory agent that acts to confine the hypoxic insult. MiRNA-210 has been found to have increased in end-stage diabetic retinopathy. However, the proliferative phase of diabetic retinopathy is similar to the hypoxic phase of ROP and the decrease in the miRNA-210 levels may indicate that the hypoxic phase has been stabilized and proangiogenic processes have started [16]. Therefore, it acts to decrease new vascular proliferation, which may be stimulated by other agents. Therefore, this decrease may be used as a biomarker of poor prognosis. The decrease in miRNA-210 levels was less pronounced in Stage I ROP, which may be used as a biomarker for the development of ROP. Li et al. have demonstrated that in patients with hypoxic-ischemic encephalopathy, miRNA-210 expression increased in the first few hours but decreased thereafter. This decrease was associated with the suppression of apoptosis in microglial cells, hence decreasing brain injury [17]. Zhao et al. have also shown that miRNA-210 can be used as a biomarker in acute cerebral ischemia [18].
MiRNA-21 expression is associated with tumor progression in several cancers, and it has a pro-oncogenic, pro-fibrotic and anti-apoptotic effect. Usui-Ouchi et al. have shown that it is also associated with the proliferative stage of diabetic retinopathy, which resembles the second stage of ROP [19]. In Stage I ROP MiRNA-21 decreases but it increases thereafter until Stage III, suggesting that it is correlated with the degree of fibrovascularization, showing vascular proliferation, apoptosis, and cell migration. On the other hand, increased expression of miRNA-21 has been found to be associated with myocardial infarction. Although it cannot be concluded that it is specific for stage III ROP, it might still be used as a biomarker of severe ROP due to severe hypoxia and elevated levels may be a preventive mechanism acting to limit ischemic injury, as shown by Buller et al. in ischemic brain lesions [20].
MiRNA-146a suppresses the activity of endothelial cells and NF-κB signals and is associated with inflammatory response and vascular remodeling in atherosclerosis and ischemic stroke [21, 22]. Sutyawan et al. have shown that NF-κB activation in ROP is a mediator of endothelium survival in retinal vessels. The study of Kotb et al. showed an anti-inflammatory effect through astrocytes and decreased the levels of cyclooxygenase 2 [23]. In our study, it decreased significantly in ROP cases in Stages I through III as a preventive marker of endothelial cell death through NF-kB activation. Therefore, it may be used as a biomarker of progressive ROP. On the other hand, it may be used in the future as a therapeutic agent to treat microvascular complications or to prevent ROP.
MiRNA-221 is also anti-angiogenic and inhibits endothelial cell proliferation and migration through the eNOS pathway. De la Torre et al. have shown that its expression is increased in diabetic retinopathy and its levels are correlated with angiotensin II and VEGF [24]. In our study, it increased insignificantly in ROP Stage I through III. On the other hand, this increase was parallel to the duration of oxygen therapy in ROP patients. Metin et al. have also shown increased levels of miRNA-221 in Stage III ROP patients with plus disease [4].
Changes in miRNA-143, miRNA-126, miRNA-106, and let-7f were insignificant. These results are consistent with other studies, although the results of some studies are insignificant [4, 25]. The effects of birthweight and gestational age were insignificant.
Our study has several limitations. One major limitation is the small number of patients, which might lead to type I statistical errors. We were not able to analyze more miRNAs that might be related to angiogenesis and ROP due to financial constraints. There were no Stage 4 and 5 ROP patients in the study group, therefore we could not analyze miRNA levels in more severe ROP cases.
However, our results indicate that the expression of ROP-specific mRNAs, especially miRNA-210, miRNA-146a, and miRNA21, may be used as a biomarker or as an adjunct in the diagnosis of ROP. miRNA was highly associated with stage III ROP, but a clear association could not be established with the stage of ROP and the level of other miRNAs. Studies on the treatment of ROP with miRNA are very rare and preliminary but promising for future. These results should be confirmed by further studies in larger cohorts.
