Abstract
Type 2 diabetes mellitus (T2DM) and non-alcoholic fatty liver disease (NAFLD) harmonize and act synergistically in clinical practices. About 70–80% of diabetic patients develop NAFLD. At the same time, NAFLD existence increases T2DM development. Meanwhile, the presence of T2DM increases the progression to liver disease such as NAFLD, and to non-alcoholic steatohepatitis (NASH). The most prevalent chronic liver disease worldwide is a NAFLD. NAFLD and (T2DM) have a two-way pathophysiologic relationship, with the latter driving the development of the former into NASH. Nonetheless, NASH enhances the threat of cirrhosis as well as hepatocellular carcinoma (HCC), both cases in turn need transplantation of the liver. The only treatment for NAFLD is still lifestyle management because there are no FDA-approved drugs for the condition. In the current study, we review how curcumin (a naturally occurring phytopolyphenol pigment) treats NAFLD. Also we showed broad insights on curcumin-based therapy, by severe reduction of hepatic inflammation. Thus, our review showed that curcumin ingestion considerably decreased glycemic parameters (fasting blood glucose, glycosylated hemoglobin, insulin resistance index (HOMA-IR), and free fatty acids) and adipocyte-fatty acid binding protein (A-FABP), and adipokine released from adipocytes. Clinical trials are needed to evaluate the effects of curcumin and its specific dosage on liver enzymes, glycemic consequences, among NAFLD coexist with T2DM patients.
Keywords
Introduction
Over 400 million people are affected by diabetes globally [1], and type T2DM is the most common form of diabetes among diverse populations worldwide [2]. The Global Burden of Diseases [3] found that 22% upsurge in debility over previous 10 years was due to T2DM and its effects. Although glycemic control and diabetes treatment have made tremendous strides recently, the frequency of cardiovascular problems among T2DM patients continues to be a significant problem [4]. Despite the availability of numerous anti-diabetic medications managing hyperglycemia, therapeutic strategies addressing additional DM-related disorders as dyslipidemia and oxidative stress have also received significant attention. Oxidative stress may be the main reason for the emergence of T2DM. T2DM, upsurge oxidative species generation and decreased anti-oxidant capability have frequently been observed [5]. By increasing the flow of the polyol pathway, activating protein kinase C, changing the metabolism of eicosanoids, and inducing glucose autoxidation, hyperglycemia may contribute to oxidative stress and increase the production of reactive oxygen species (ROS). Diabetes can be brought on or made worse by ROS, which has many harmful effects, including reduced insulin secretion, decreased insulin synthesis, protein oxidation and fragmentation, DNA damage, production of free fatty acids, and augmented vascular permeability. Additionally, oxidative stress triggers the production of advanced glycation end products that promote the development of T2DM microvascular and macrovascular problems as well as endothelial dysfunction [6–8] (Fig. 1).

Schematic illustration of the pathophysiological mechanism through which T2DM and NAFLD cause an increase in oxidative stress and inflammation. One of the most common cardiac complications in diabetic patients, diabetic cardiomyopathy (DCM), is associated with oxidative stress, which results from a serious imbalance between the production of reactive oxygen species and/or reactive nitrogen species.
Anti-diabetic drugs, lifestyle modifications, frequent exercise, monitoring lipid profiles and arterial blood pressure are currently the most widely used treatments for T2DM. Effectual deterrence and medication are for diabetic patients. T2DM is a malady that can be avoided [9]. High risk T2DM subjects, pre-diabetes, and low glucose tolerance subjects must get a tight preventative strategy. Part of such prevention is changing the lifestyle, having balanced diet and consistent exercise.
Currently, pharmaceutical therapy, and lifestyle change are the most recent suggestions for diabetes control [10, 11]. The biological benefits of extracts or isolated polyphenolic monomers from diets, may improve metabolism, decrease oxidative stress and inflammation, and increase insulin resistance, have received a lot of attention recently [12, 13]. According to a growing number of randomized controlled studies [14–16], dietary polyphenols can improve a range of clinical indicators in NAFLD patients through gut, brain, liver, and their interconnected pathways.
Evidence supporting the effectiveness of using medical plant supplements for T2DM prevention and control is currently expanding [17–19]. The scientific world is becoming increasingly interested in curcumin (a medicinal herb). The rhizome of the turmeric plant, Curcuma longa, contains the active chemical curcumin. Turmeric contains a naturally occurring polyphenol molecule, which both in vitro and in vivo displays two crucial bioactivities [20]. First curcumin works as an antioxidant by scavenging ROS [21]. Curcumin also possesses hepato-protective ingredients which treat liver damage. According to numerous studies, curcumin may delay the onset of NASH by lowering inflammation and restoring the balance of the liver’s antioxidant systems. The second is curcumin, has an anti-inflammatory, antibacterial, and anti-carcinogenic compounds. As oxidative stress and inflammation are two major causes of insulin resistance and NAFLD, curcumin enhances natural peroxisome proliferator-activated receptor (PPAR) expression, which is essential for lowering them [22, 23]. The purpose of the present article is to provide evidence that curcumin reduces the severity of NAFLD by diminishing lipid accumulation, oxidative stress and inflammation.
Furthermore, our review explores the relationship between NAFLD and diabetes development in people with specific genetic variants and showed the pathophysiological relationships between T2DM and NAFLD.
NASH is a set of progressive liver disorders that vary from basic hepatic steatosis that is characterized by hepatocyte inflammation follow stop, Liver fibrosis conceivably leads to hepatocellular carcinoma (HCC) or end-stage liver disease progress in roughly represent 35% of NASH cases [24, 25]. NAFLD epidemic is widespread through western countries disarrays approximately 20–30% of the general populace and 45–75% of persons identified with T2DM [26, 27].
NAFLD is characterized by hepatic steatosis that is determined by histology/imaging with macrovesicular steatosis in more than 5% of hepatocytes in accordance with histological analysis. Hepatic steatosis also determined by proton density fat fraction, by proton magnetic resonance spectroscopy (MRS) or quantitative fat/water selective magnetic resonance imaging (MRI) with no secondary reason for steatosis [28].
Numerous medications may result in steatosis or steatohepatitis, which share clinical characteristics with NAFLD or alcoholic fatty liver disease. Primary steatotic trait can be utilized to categorize the steatosis by medicines (Table 1).
Drugs that cause obesity/insulin resistance, phospholipidosis,
steatohepatitis, macrovesicular steatosis, and microvesicular steatosis and
further drug-induced fatty liver disease and their theorized toxicity
mechanisms
Drugs that cause obesity/insulin resistance, phospholipidosis, steatohepatitis, macrovesicular steatosis, and microvesicular steatosis and further drug-induced fatty liver disease and their theorized toxicity mechanisms
The presence of tiny to large lipid droplets in the hepatocyte cytoplasm together with peripheral nucleus displacement is referred to as macrovesicular steatosis [29–31]. Although this type of liver damage is frequently curable, it may eventually progress to cirrhosis and possibly steatohepatitis. Excessive alcohol consumption and the use of glucocorticoids, total parenteral nutrition, methotrexate, and amiodarone as treatments are also linked to macrovesicular steatosis. The use of 5-fluorouracil, tamoxifen, irinotecan (IRI), cisplatin, and asparaginase during chemotherapy may also result in macrovesicular steatosis [32].
Hepatocytes undergo microvesicular steatosis when their nuclei are surrounded by a large number of tiny lipid vesicles. Microvesicular steatosis is associated with substantial impairment of fatty acid beta-oxidation in the mitochondria. Since mitochondria are unable to efficiently oxidize non-esterified fatty acids, they undergo enhanced esterification into triglycerides, which is the major lipid type that builds up under these circumstances. Since acute microvesicular steatosis develops quickly to either death or resolution, significant necrosis, cholestasis, and fibrosis are typically absent in this condition [33]. Drugs such as valproic acid, tetracycline, aspirin, ibuprofen, zidovudine, and vitamin A have been related to microvesicular steatosis.
The most common cause of chronic liver disease is thought to be NAFLD, which is also a growing public health problem with pandemic proportions [46]. Histology and proton magnetic resonance spectroscopy (MRS) [47] both detected 31% and 12.2% of NAFL patients, respectively, of which 5% had NASH. Although 20% of asymptomatic T2DM patients have biopsy-proven NASH and normal liver function tests, NAFLD is widespread in T2DM (59.67% ) [48]. Serum liver enzymes are less illuminating due fat deposition and the normal reference values for blood liver enzymes need to be altered. Armstrong et al. [49] found that 5%–7% of T2DM individuals without symptoms had advanced fibrosis (Table 2).
Prevalence of NAFLD and NASH Globally
NAFLD’s public health significance arises from its numerous effects on mortality, morbidity, and the use of medical services internationally [50, 51]. In general population, NAFLD and NASH fibrosis are linked to to liver-related mortality. Hepatocellular carcinoma, chronic liver disease, and cirrhosis of the liver are all frequently brought on by NASH and frequently develop in non-cirrhotic livers as well [52–54]. In the United States, the United Kingdom, and even in developing nations, NASH is the fastest-growing reason for liver transplantation.
NAFLD and T2DM are significantly predisposed as a result of obesity along with insulin resistance [59–61]. Clarifying whether the presence of liver disease in NAFLD upsurges the threat of developing T2DM is crucial from a clinical standpoint because it’s possible that treating NAFLD (particularly remedy intended to treat insulin resistance and obesity) could also reduce the threat of augmenting T2DM in addition to treating the metabolic risk factors linked to NAFLD. Contrarily, lifestyle modifications (such as a low-calorie diet and modest exercise) are acknowledged to significantly improve liver damage, fibrosis, and hepatic steatosis in people with NAFLD [62]. They are particularly successful at postponing T2DM in those who are at high threat for the illness [63, 64].
Genetics has been extensively studied and is the chief threat factor for the succession of NAFLD [65]. Romeo et al. [66] in a genome-wide association study (GWAS) highlight the critical part of (PNPLA3 I148M) patatin-like phospholipase domain containing 3 in NAFLD patients. They showed how PNPLA3 I148M contributed to the increased hepatic fat content and severe hepatic inflammation [67]. By using GWAS, Kozlitina et al. [68] identified the transmembrane 6 superfamily member 2 (TM6SF2 E167K) as a significant polymorphism site for the risk ofNAFLD.
The liver converts cholesterol into the 2 primary bile acids (Cholic acid and Chenodeoxycholic acid) that delivered into the intestine as conjugates of glycine and taurine. Subsequently, the intestinal microbiome transmutes primary bile acids into secondary bile acids like deoxycholic acid, litho-cholic acid, and urso-deoxycholic acid. These secondary bile acids then interrelate with a number of nuclear receptors in the intestine, involving Takeda G protein-coupled membrane receptor 5 (TGR5) and farnesoid X receptor (FXR). Bile acids interconnect with TGR5 and FXR receptors and regulate hepatic lipid and glucose metabolism.
TGR5 activation in enterocytes causes an increase in the amount of glucagon-like peptide 1 (GLP1) released from L cells. GLP1 release has an effect on plasma glucose level, which then invigorates the pancreatic islet to generate more insulin. The body expels insulin through the liver. Hepatic insulin extraction, which makes up 50–80% of insulin clearance, is decreased in NAFLD. By turning on hepatic FXR receptors, fatty acid and triglyceride production is reduced, potentially as a result of less hepatic lipid buildup [69, 70]. The formation of hepatic glycogen rises in addition to the production of hepatic gluconeogenesis falling as a result of FXR activation, that is shown in Fig. 2 are potential risk factors for T2DM.

Illustrates the association between the liver, bile, and gut that may lead to receptor alteration and change insulin metabolism and bile acid production. In non-alcoholic fatty liver disease (NAFLD), hepatic insulin extraction decreases, which accounts for 50–80% of insulin clearance. Fatty acid and triglyceride production is decreased by activating hepatic FXR receptors, possibly due to less hepatic lipid accumulation.
The precise NAFLD risk factors that increase T2DM risk are not known. However, it is well recognized that lipid accumulation is associated with hepatic insulin resistance and hepatic inflammation, both of which are significant NAFLD features. Therefore, by reducing hepatic fat accumulation in NAFLD, therapies can improve insulin sensitivity and chronic inflammation may help reduce the incidence of T2DM.
Turmeric is common name for the herbaceous plant Curcuma longa, which is consumed as a spice in many dishes. This plant species, was identified by its orange tuberculate rhizomes, and is extensively grown in South East Asia, where it has long been utilized as a natural medication for a broad range of pathological maladies. Curcumin has gained interest as a nutraceutical from scientists due to its anti-inflammatory and antioxidant activities as well as the safety of its pharmacological profile [71]. Curcumin aid in the deterrence and therapy of a wide range of maladies because of its broad spectrum of beneficial effects, including its antibacterial, anti-diabetic, antiviral, and anti-cancer qualities [72, 73].
Curcuma longa, is a member of the Zingiberaceae family that is grown in tropical and subtropical climates where it has been used as a curative agent in Indian and Chinese traditional medicine [74]. The main curcuminoids in commercial curcumin are 77% curcumin (curcumin I), 17% demethoxycurcumin (curcumin II), 3% bis-demethoxycurcumin (curcumin III), and 3% cyclocurcumin (curcumin IV) [75, 76]. Turmeric also contains 69.4% carbohydrates, 6.3% protein, 5.1% fat, 5.8% essential oils, and 3–6% curcuminoids.
The biological and pleiotropic effects of curcumin are numerous (Fig. 3) [77–79] including: antibacterial [80, 81], antineoplastic [82, 83], ant proliferative [84], and anti-inflammatory agent [85–88]. Curcumin also offers therapeutic promise for treating diabetes mellitus [89–91], hepatic damage [92, 93], renal illnesses [94, 95], many disorders [96, 97], cardiovascular diseases [98, 99], and hepatic damage [100].
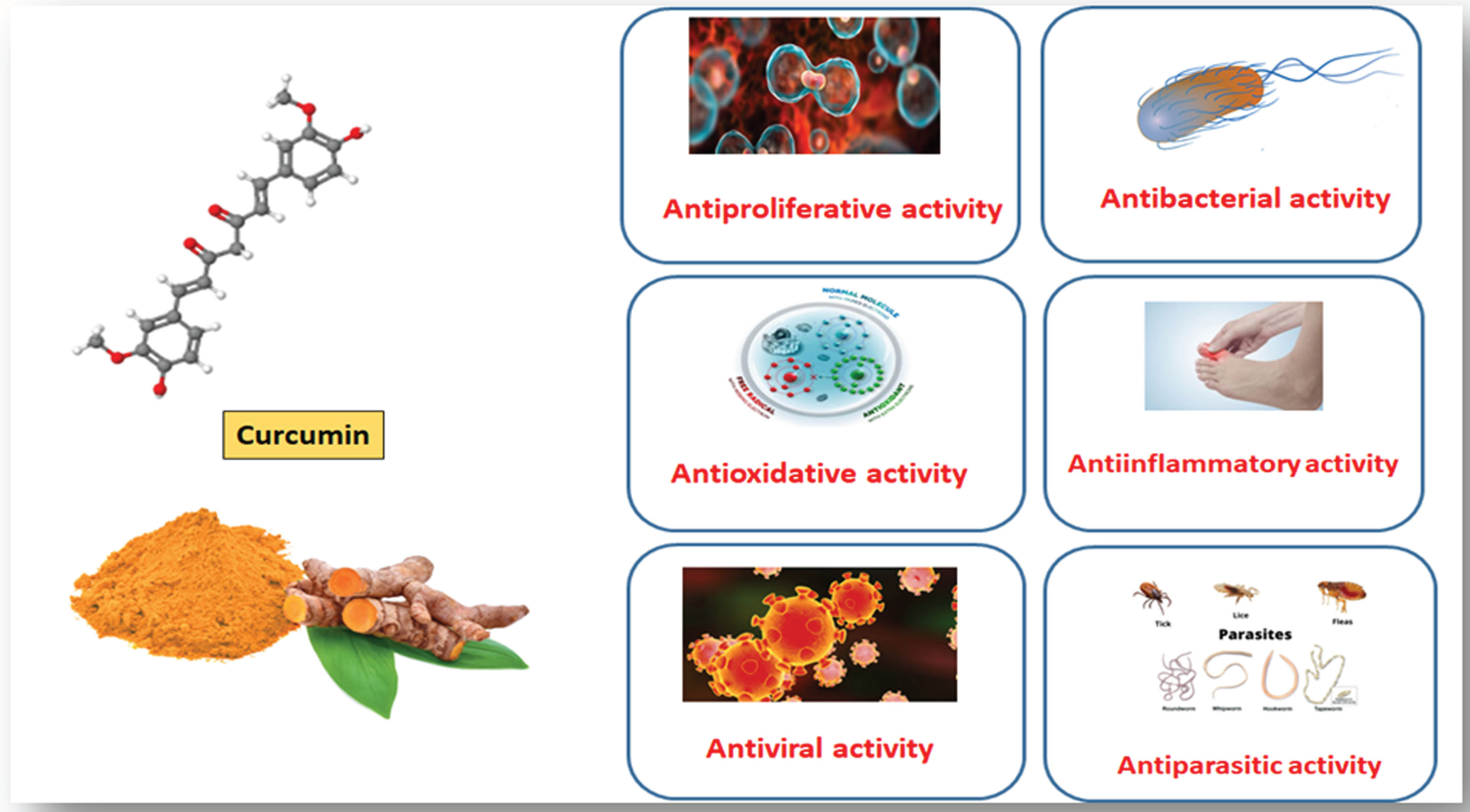
Depicts numerous pharmacological and pleiotropic benefits of curcumin including anti-inflammatory, anti-bacterial, antineoplastic, and antioxidant properties.
The primary roles of the protein hormone insulin are balancing between nutrients and glucose [101]. In accordance with high blood sugar levels, the islets of Langerhans liberates insulin, which is then traveled into its target tissues, like liver, adipose tissue, and skeletal muscle [102]. Blood sugar levels return to normal as a result of insulin’s stimulation of adipose tissue and skeletal muscle resulting in the absorption of glucose as well as suppression of the liver’s endogenous production of glucose [103].
The process of triggering insulin’s mode of action is by the attachment of insulin to its receptor, which is present on the target cell membrane [104]. The receptor tyrosine kinase gets activated and instigates the insulin receptor substrate (IRS) to be phosphorylated [105]. Further downstream processing, the lipid kinase-like phosphatidylinositol-3 kinase (PI3-K) along with the serine/threonine kinase as well as Akt/PKB are stimulated [106]. In adipose as well as muscle cells, the GLUT4 is glucose transporter which is translocated from cells internal side to the exterior cell membrane, resulting in enhanced glucose uptake. By inhibiting glycogenolysis and gluconeogenesis, insulin signaling and action reduce endogenous glucose production (Fig. 4) in liver cells [107].

The pharmacological actions of curcumin on insulin receptor activation and glucose absorption. Protein kinase-B, Phosphatidylinositol-3,4,5-triphosphate and 4,5-bisphosphate (PIP3 & PIP2), respectively; Insulin receptor substrate 1 (IRS1), tumor necrosis factor (TNF), AMP-activated protein kinase (AMPK), Nuclear factor kappa-light-chain- activates B-cells (NF-kB), Peroxisome proliferator-activated receptor gamma co-activator 1 (PGC-1), Acetyl-CoA carboxylase (ACC) and for free fatty acids (FFAs).
Defects in the insulin signaling system are responsible for both insulin resistance, which is outlined by lower responsiveness of circulating levels of insulin in target tissues [108], and (T2DM) [109]. T2DM along with insulin resistance have been linked to ageing, obesity, inflammation, and a deskbound lifestyle [110]. Hyperglycemia brought on by insulin resistance over time damages macro- and microvascular tissue and results in conditions such, retinopathy, cardiovascular disease, nephropathy and neuropathy [111]. Increased plasma-free fatty acids (FFAs) diminish insulin’s capacity to lower hepatic glucose synthesis along with promotion of glucose absorption by fat and muscle cells, which is associated with insulin resistance and obesity [112, 113].
Asia has a long history of using Curcuma long medicinally, especially in South East Asia and China [114]. The chief naturally occurring polyphenol found in Curcuma longa and other species of curcuma were curcumin. It is identified as diferuloylmethane [115]. 1, 7-bis(4-hydroxy-3-methoxyphenyl)-1,6 heptadiene-3,5-dione makes up this substance. Two methoxylated phenols, an enol form of diketone, and two methoxylated phenols make up the structure of curcumin, with the latter serving as the active site for the former’s antioxidant activities [116]. The amount of methoxy groups on their aromatic rings is the only structural distinction between curcumin and other curcuminoids, like de-methoxy-curcumin and bis-demethoxy-curcumin. The potential anti-atherosclerotic, anti-cancer, anti-inflammatory, hepatoprotective, immunomodulatory, nephroprotective, and anti-diabetic properties of turmeric and curcumin have been proven by scientists.
According to literature reports, curcumin (20μM) was subjected to 3T3-L1 cell line and primary adipocytes for 6 days. It resulted in the browning of white adipocytes and mitochondrial growth [117]. After curcumin administration, PGC-1, PPAR, (PRDM16)-PR domain containing 16, Ucp1 protein and levels of mRNA upsurge. Mitochondrial proteins CPT1 and cytochrome-c were also augmented by curcumin therapy [118]. Curcumin therapy increased both the total and phosphorylated levels of AMPK. PGC-1, PRDM16, and Ucp1 protein levels reduced when curcumin and the AMPK inhibitor dorsomorphin were supplied concurrently, demonstrating that curcumin’s actions are brought on by AMPK activation [119]. This study unequivocally shows that curcumin therapy has controlled obesity, insulin resistance, and T2DM.
The likelihood of developing T2DM, IR, NAFLD, obesity, and other metabolic disorders is amplified by genetics, and gender [120]. Lifestyle, nutritional, and circadian rhythm adjustments have a long-term positive impact on the clinical and Para clinical outcomes of NAFLD and T2DM [67]. Curcumin was given to overweight/obese T2DM [121] at a dose of 300 mg/day, and Na et al. demonstrated that curcumin decreased body mass index (BMI), fasting blood glucose, glycosylated hemoglobin, insulin resistance index (HOMA-IR), and free fatty acids. Glucose drop was only 18% and the glycosylated hemoglobin drop was only 11% from baseline. Later, the same group found that curcumin reduces adipocyte-fatty acid binding protein (A-FABP), an adipokine released from adipocytes [121, 122] that coordinate lipid-mediated processes [123] that decrease free fatty acid levels.
The possibility of reaching its target has been one of the key issues with curcumin/curcuminoids’ potential as medicinal agents. Because of their poor stability and pharmacokinetic characteristics, curcuminoids’ therapeutic benefits have recently come under scrutiny [124]. Curcumin has limited bioavailability since it is poorly absorbed and readily destroyed [125]. Docking studies are needed to understand its pharmacokinetic and pharmacodynamics activity of curcumin.
Conclusion
Globally, T2DM affects thousands of people and has a complex pathophysiology. Changes in lifestyle, food quality, and increasing physical activity and curcumin supplements can improve diabetes control. Although research is needed to understand the optimal amount of curcumin that can be used. Curcumin is a natural anti-inflammatory and anti-diabetic drug that offers a secure and affordable alternative for treating diabetes. To establish the role of curcumin in the treatment of T2DM, we advise conducting randomized controlled clinical trials. In conclusion, it is challenging to discern between the cause and effect of NAFLD and T2DM, since the interaction between them is so complicated. While NAFLD may increase the risk of T2DM, T2DM offers the best metabolic environment for NAFLD advancement. Numerous elements are similar in both clinical conditions—such as an accumulation of fatty acids, pro-inflammatory cytokines, oxidative stress, that are implicated in the disease’s progression. Treatment options for NAFLD and T2DM may become stronger with a complete understanding of their underlying mechanisms. The restricted solubility and low bioavailability of curcumin require numerous strategies to overcome. These include creating innovative curcumin formulations, such as sustained-release tablets, liposomal encapsulation, nanoparticles, emulsions, and synthesizing curcuminoids. The development of a “super curcumin” in the near future is awaited for result from curcumin’s improved bioavailability and clinical trial findings, boosting this promising natural product to the vanguard as a therapeutic remedy for diabetes. This article provides insights for researchers to further carry out docking studies to have clear-cut comprehension of curcumin’s potential to cure diabetes.
Footnotes
Acknowledgments
Manar Atoum is thankful for The Hashemite University for Support, KRP is thankful to the Department of Biotechnology, Sri Padmavati Mahila Visva Vidyalayam (Women’s) University, Tiruapti-India and KRD is thankful for Sree Balaji Dental College and Hospital, Bharath Institute of Higher Education and Research (BIHER) Bharath University, Chennai, Tamil Nadu.
Fund
No fund because it is a review article.
Author contributions
Manar Atoum draft manuscript preparation, revision and editing of the article.KR Padma contributed in draft manuscript preparation, writing, drawing figures.
KR Don contribute drafting this review article and drawing figures.
Conflict of interest
No conflict of interest between the authors.
