Abstract
Introduction
Many lifestyle interventions use calorie restriction (CR), defined as a reduction in caloric intake without malnutrition, as a key tool to improve health. Indeed, CR appears to beneficially affect physiology and biomarkers related to cardiometabolic health, even in non-obese populations. A prime example of the cardiometabolic benefits of CR is the Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy (CALERIE) study trials, involving 25% calorie reduction over 6 months to 2 years [1–5].
CR is also well-known to extend lifespan in multiple non-human species [6, 7]. Despite the fact that delayed immune system aging is thought to be a major pathway through which this happens [8], the effects of CR on the immune system are less studied than the cardiometabolic system in
As adherence to long-term CR is one of its main challenges [16], we recruited participants through two international CR organizations (CR Society International and the CR Way) who all demonstrated successful long-term CR. This sample afforded a unique opportunity to test for changes in the immune system. Much like the larger human CR literature, others using a similar approach to ours have predominantly focused on cardiometabolic health rather than immune aging [17–19]. One study [20] did examine whether long-term CR had lower levels of insulin-like growth factor 1 (IGF-1), a factor implicated in cell-mediated anti-tumor immunity [21] and many types of cancer risk [22], but found no differences compared to Western-diet controls.
In the context of a 2-year randomized controlled trial of 25% CR, the CALERIE-2 study examined multiple immune-related outcomes [3, 24]. They observed lower levels of CRP and TNFα and slightly fewer white blood cells, lymphocytes, and monocytes in the CR group relative to ad-libitum eating controls, but no differences in vaccine response. As some have called into question whether a 25% CR for 2 years is sufficient to elicit processes activated in the rodent models [25], our first goal was to examine a sample averaging a longer period of restriction –10 years.
Our second goal was to augment the few prior studies of immune phenotype in non-obese human CR by expanding the scope of the immune measures [24, 27]. We conducted immunophenotyping to examine percentages of naïve and senescent T cells, hypothesizing that CR would have significantly greater percentages of CD28+CD57– and CD28+CD57–PD–1– T cells compared to normal weight and overweight/obese groups. We also examined telomere length, considered to serve as a marker of cumulative biological aging [28–32], in PBMCs as well as granulocytes, and the reverse transcriptase enzyme telomerase [33] in PBMCs. To our knowledge, the study presented here is the first to examine telomere length in humans with CR, and further, to examine telomere length specifically within these two main immunologic compartments. We hypothesized that the CR group would have longer PBMC (primary outcome) and granulocyte telomere length and greater expression of telomerase.
Materials and methods
Because individuals who have the ability to restrict their eating behavior for long periods may be phenotypically different from the average population, we matched the groups on self-reported age, race/ethnicity, gender, and educational attainment. Matched participants were recruited from the local area once the CR group was completed and their information on these matching variables obtained. Additionally, we recruited siblings of CR participants to attempt to control for genetic and environmental factors. Sample size was dictated by the maximum number of participants we were able to recruit in a predetermined data collection period of one year.
Participants traveled to the UCSF Clinical and Translational Science Institute Clinical Research Center (CCRC). To minimize confounds due to sleeping in an unfamiliar setting, participants spent two acclimation nights in the CCRC before the blood draw. To minimize confounds due to jetlag and diurnal activity patterns, participants woke up, ate meals, and slept according to their usual schedules in their home time zone. A subset (
Venous blood was collected immediately upon waking, after participants had fasted for a minimum of 8 hours. PBMCs were isolated by gradient density centrifugation by use of Ficoll-Paque (GE Healthcare, Piscataway, NJ, USA). Granulocytes were isolated from the red blood cell pellets after Ficoll separation by Ammonium-Chloride-Potassium solution. Genomic DNA was isolated from whole blood, PBMC and granulocytes using QIAamp DNA mini kit (QIAGEN).
Immunophenotyping was performed on cryopreserved PBMCs. The cells were plated in a 96-well v-bottom plate and stained with the following fluorescently-conjugated monoclonal antibodies from BD Biosciences unless otherwise noted: Brilliant Violet 605-conjugated anti-CD8 (SK1) and Brilliant Violet 711-conjugated anti-CD3 (UCHT1), Allophycocyanin (APC)-eFluor780-conjugated anti-CD28 (CD28.2) (eBioscience), Alexa Fluor® 647-conjugated anti-CD57 (HCD57), PE-conjugated anti-CD38 (HB7), Brilliant Violet 421-conjugated anti-PD-1 (EH12.1) and FITC-conjugated anti-HLA-DR (L243). LIVE/DEAD® Fixable Aqua Dead Cell Stain Kit (Invitrogen) was added into all stains to exclude non-viable cells. Staining was performed at 4°C for 30 minutes, then washed once with FACS buffer (Phosphate-buffered saline containing 0.5% bovine serum albumin and 1 mM Ethylenediaminetetraacetic Acid). Cells were then fixed in 0.5% formaldehyde and data was acquired on a BD LSR II Flow cytometer (BD Biosciences), with ≥200,000 lymphocytes collected for each sample. CPT beads (BD Bioscience) were used for instrument set up for each run and Rainbow beads (Spherotec) standardized instrument settings between runs. Aliquots of a control specimen were thawed with every run and assessed in triplicate to confirm run-to-run reproducibility. Fluorescence Minus One (FMO) controls were also prepared on control samples for each run, to check that gates were set consistently between runs. Data was compensated and analyzed in FlowJo V9 (TreeStar). CD3+CD8+ T cells were defined after standard lymphocyte, singlet and dead cell exclusion gates were applied to the data. FMO controls were used to define positive gates for expression of CD28, CD38, CD57, HLA-DR and PD-1 on CD3+CD8+ T cell populations.
The telomere length measurement assay was adapted from the published original method by Cawthon and colleagues [27, 34]. Tubes containing 26, 8.75, 2.9, 0.97, 0.324 and 0.108 ng of a reference DNA (from Hela cancer cells) were included in each PCR run so that the quantity of targeted templates in each research sample could be determined relative to the reference DNA sample by the standard curve method. The same reference DNA was used for all PCR runs.
Telomerase activity was measured in PBMCs by the Telomerase Repeat Amplification Protocol (TRAP) with a commercial assay (TRAPeze, Telomerase Detection Kit, Upstate/CHEMICON, Temecula, CA) using procedures described previously [35]. Two concentrations corresponding to 5,000 and 10,000 cells were assayed for each sample in order to ensure that the assay was in linear range. The assay reaction was performed in accordance with the TRAPeze kit manufacturer’s instructions, and radioactive products fractionated by 8% polyacrylamide–8 M urea sequencing gel electrophoresis. Telomerase activity is expressed as the equivalent of number of 293T cells per 10,000 PBMCs, and activity was quantified using the ImageQuant 5.2 software (GE Healthcare) as described previously [35]. Telomerase values were non-normally distributed and corrected via a natural log transformation.
Fasting serum samples were assayed by ELISA for qualitative detection of cytomegalovirus (CMV) IgG antibodies by the University of California, Los Angeles, Clinical Immunology Research Laboratories.
Analyses. The key outcome measure was PBMC telomere length. Statistical analyses consisted of ANCOVA models comparing the three groups (CR vs. normal weight vs. overweight/obese) all controlling for chronological age, and additionally covarying potential confounding variables when they were statistically significantly related to each respective outcome measure. These potential covariates consisted of self-reported gender and race/ethnicity, as well as CMV serological status due to its known effects on immunosenescence [36].
Results
Descriptive statistics of key study variables are summarized in Table 1. For the overall sample of 71 participants, the mean age was 55.01 years (SD = 14.53, range = 21–84), 54 were male, race/ethnicity was 88.6% white and 11.4% East Asian/Asian-American, and highest level of educational attainment was as follows: 6.7% high school, 6.7% some college, 20% bachelor’s degree, 20% master’s degree, and 46.7% doctoral degree. The CR group (
Descriptive statistics of study variables
Note: Mean (Standard Deviation) [Minimum-Maximum] except for Sex, Ethnicity/Race, Education, and CMV status, where N (%). Differing superscripts refer to statistically significant differences between groups with p < 0.05, two-tailed. 1Telomerase was non-normally distributed and therefore values reflect Median (Interquartile Range)[Minimum-Maximum]. Telomere and telomerase analyses control for chronological age.
Descriptive statistics of study variables
Note:
For all measures of immune activation and immunosenescence in CD4+ T cells, no statistically significant differences were observed between the three groups (data not shown). Table 2 displays results for CD8+ T cells, where a statistically significant group difference emerged in activated CD8+ T cells defined as CD38+HLA-DR+ cells,
Group differences in frequencies of CD8+ T cell activation and maturation phenotypes
Note:
The CR group had shorter PBMC telomere length (
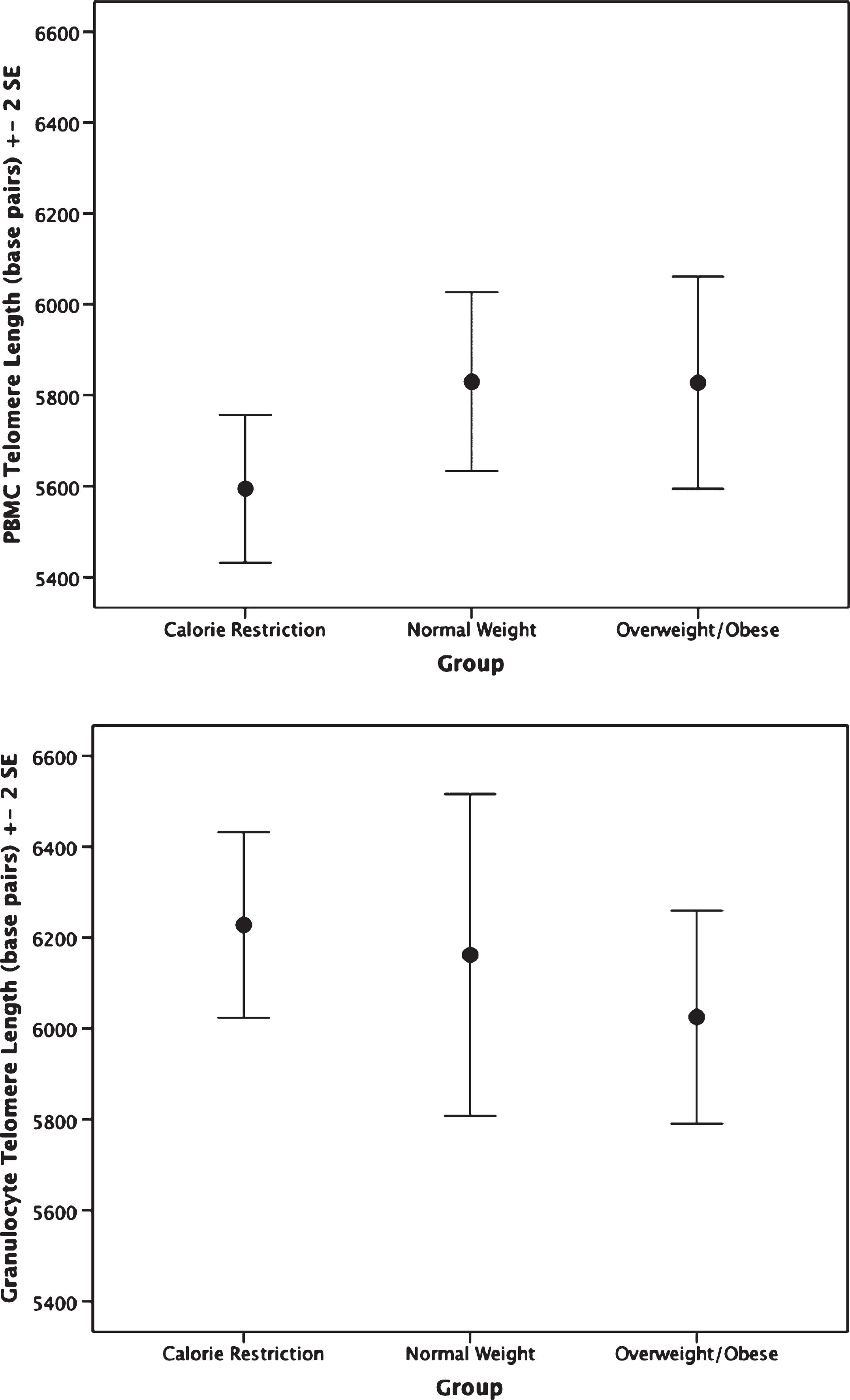
Telomere length by group. Top: Peripheral blood mononuclear cell telomere length. ANCOVA analyses indicate the calorie restriction group has significantly shorter telomere length compared to the other groups;

Telomerase activity by group. Groups are not significantly different,
In the largest study to date of long-term human calorie restrictors averaging over a decade of calorie restriction, we set out to examine whether CR is associated with enhanced immunosenescent and cellular aging profiles of T cells. Although research has observed beneficial effects of CR on the cardiometabolic system in non-obese populations [4, 5], or beneficial effects of CR on cancer risk in populations with excessive weight [37], we did not observe these benefits in this context. As delayed immunologic aging is purported to be a main mechanism of CR’s lifespan-extending effects, we expected to see a strong and consistent pattern of lower senescent and higher naïve cell percentages as well as longer telomere length in the CR group compared to the normal weight and overweight/obese groups. In contrast, we observed very few differences between the three groups in markers of immunosenescence, and the CR group had shorter PBMC telomere length than the comparison groups. In fact, the telomere length in the CR group was significantly shorter when compared to the obese group.
It is unclear why we observed differences in PBMCs and not granulocytes, but it is important to examine them separately as PBMC and granulocyte populations have different roles in immune function, and reflect different immune responses. PBMC telomere length is affected by both history of infections (which cause more replicative turnover) as well as the biochemical environment of the blood [38]. Due to their rapid turnover, granulocyte telomere length is thought to more directly represent the common myeloid progenitor stem cell compartment,[38, 39]. Thus, it is possible that the short lived granulocytes may have less variance in turnover, between people, and may be less influenced by environmental exposures, like dietary factors, in the blood. PBMC telomere length may be more influenced by biochemical and lifestyle factors.
We do not know why CR was associated with shorter PBMC telomere length, but we can offer speculation. Given the pattern of findings here, one possibility is that the CR diet as practiced left those individuals more vulnerable to infection, despite that CMV seropositive status was no different between the groups. CMV exposure typically occurs during early childhood, such as during daycare [40], and therefore was likely acquired before CR started. However, there could be differences in how well people are suppressing reactivation of the CMV virus, which we did not measure and would require CMV viral load analyses as well as specific immune responses such as IgG concentrations or CMV-specific T cell responses.
There may be differences in the immune cell populations that require greater subset measurement to observe than we undertook here. T cells make up 60% of the PBMCs [41], and so differences in T cell telomere length could have a large influence on PBMC telomere length. The difference in PBMC telomere length could potentially be due to a skewing in major memory T cell subsets (i.e. effector vs central memory) in the CR group due either to increased subclinical infections in this group or how their immune system maintains latent viral infections such as herpes viruses (e.g. CMV, Epstein Barr Virus). This however would have required more complex phenotyping to detect.
These overall null findings add to a sparse, but already mixed, literature in animal and non-human primate species. In mice, CR led to fewer senescent intestinal crypt enterocytes and liver hepatocytes and better telomere maintenance in these cells [42]. However, in primates, CR did not lead to changes in telomere length in leukocytes or other tissues [43].
In the context of mixed evidence of the relationship between CR and immune aging in model organisms such as mice and macaques, it perhaps is not surprising that such a relationship would be difficult to observe in human CR. Compared to model organisms, humans eat an extremely variable diet. This means human CR can be practiced in infinitely variable ways with similarly low caloric intake but different macro and micronutrient content, eating patterns, and consistency. Most humans find it difficult to adhere to intense long-term CR [6, 16], and indeed, our research has demonstrated that this group of long-term CR practitioners have a distinct psychosocial profile that suggests a predisposition to the ability to sustain long-term CR [44]. Moreover, CR can put individuals at risk for malnutrition [6, 16], calling into question whether CR should be pursued as an intervention for immune system aging for non-obese humans. However, our results should be interpreted with caution in light of conflicting findings from Meydani and colleagues, who found beneficial effects on circulating inflammatory markers in the context of a randomized trial of CR [24].
This study had the following limitations: Although this is the largest study of
Instead of a strong pattern indicating beneficial effects of human CR on T cell immune activation and immunosenescence, we observed no clear evidence that CR may delay immune aging compared to normal BMI and overweight/obese matched individuals. In fact, the few results emerging from our study suggested potentially negative effects of CR, particularly in the context of PBMC telomere length. Delayed immune aging, therefore, must be further studied before it is put forth as a major mechanism of CR’s lifespan-extending effects, at least as it is most faithfully practiced in non-obese humans.
Footnotes
Acknowledgments
Funding for this project was provided to AJT by the Robert Wood Johnson Foundation Health and Society Scholars Program, the Appleby Foundation, the RWJF/UCSF Health Disparities Working Group, and the UC Berkeley Population Center. The flow cytometry was made possible with help from the University of California San Francisco-Gladstone Institute of Virology & Immunology Center for AIDS Research (CFAR), an NIH-funded program (P30 AI027763). This project was also supported by the National Center for Research Resources and the National Center for Advancing Translational Sciences, National Institutes of Health, through UCSF-CTSI Grant Number UL1 RR024131. The contents of this paper are solely the responsibility of the authors and do not necessarily represent the official views of theNIH. The authors gratefully acknowledge invaluable support from Paul McGlothin, President of The CR Way Longevity Center and Vice President for Research of the CR Society Intl. and Meredith Averill, Chairman of the Board of The CR Society Intl. and Treasurer and V.P. of The CR Way Longevity Center. We also gratefully acknowledge the guidance of Elizabeth Blackburn, Cynthia Kenyon, and Anne Brunet. Finally, we are extremely grateful for the participants for providing their time, energy, and dedication to the study.
