Abstract
BACKGROUND:
One of the objectives of food industry is to seek new resources of oil that preferably presents nutritional values. For human health, fats and particularly vegetable oils are considered as an important source of energy when glucose is not available. In addition, the search for effective, natural compounds with antioxidant activity has been intensified in recent years to replace the synthetic products.
OBJECTIVE:
The aim of the present work was to determine the fatty acids profile (and variability) of the lipid fractions extracted from five different populations of the leaves of
METHODS:
The chemical compositions of tocopherols were also analyzed by HPLC for the first time for this plant part. The chemical percentage variability (presence of two main distinguished clusters) of the fatty acids was discussed using statistical analysis methods (Agglomerative Hierarchical Clustering “AHC” and principal component analysis “PCA”). The antioxidant activity of the dewaxed lipid fractions were investigated
RESULTS:
For the overall samples, the main saturated fatty acids components were capric acid (C10:0 = 2.49–13.88%), myristic acid (C14:0 = 4.71–9.12%) and palmitic acid (C16:0 = 5.31–9.03%). Alternatively, the main unsaturated fatty acids were oleic acid (C18:1w9 = 3.42–4.85%), linoleic acid (C18:2w6 = 10.94–16.99%) and most importantly α-linolenic acid (C18:3w3 = 20.92–48.92%), which is known for its multiple dietary, pharmaceutical and clinical benefits.
CONCLUSION:
The results of fatty acids methyl esters identification using GC and GC-MS showed a clear variability in the composition of fatty acids. The main result of this study illustrate the nutritional potential (richness in MUFA such as C18:1w9, presence of essential fatty acids such as C18:2w6 at all the stages of maturation, richness in omega-3 fatty acids such as C18:3w3. of the oil of
Introduction
The genus of
Actually, the food safety concern on human health resulting from excessive employment of synthetic additives have prompted the development of new sanitation methods to decrease the negative impact on human health and environment. One such possibility is the use of essential oils and plants products as antibacterial additives. Fixed oils are also known for their antimicrobial power related to fatty acids and phenol compounds.
Previous reports on the chemical composition of the fatty acids of
According to literature, and at best of our knowledge, there are only few reports dealing with the identification of the fatty acids of the leaves of
Materials and methods
Chemicals
Petroleum ether, anhydrous sodium sulfate, sodium and ascorbic acid were purchased from AnalaR NORMAPUR Prolab. Chloroform, absolute ethanol, activated charcoal (activated carbon),
Collection of plant material
Leaves of
Lipid extraction
The air dried leaves of
The chlorophyll removal was performed by reflux, using chloroform as solvent in the round-bottomed flask containing lipids and activated carbon for 2 hours. The ratio of activated carbon relative to initially air dried leaves was 1:5 (mass). The extract fraction was cooled to room temperature then filtered. At the end, the chloroform solvent was removed from the filtered solution in Rotavap under reduced pressure at 40°C.
The extracted lipid fraction was weighed to determine the total lipid content per dry weight of the plant, and then stored at +4°C for further analysis.
Fatty acids (FAs) composition
The FAs composition of the oils was determined after having converted the fatty acids FAs into fatty acids methyl esters (FAMEs) then analysed by gas chromatography (GC). The methyl esters were prepared by the following procedure: Lipids (0.5 g) were refluxed for 20 min in 10 mL of 2% sodium methylate (NaOMe), and then 20 mL of water was added. The fatty acid methyl esters (FAMEs) were extracted by
Chromatographic analysis of FAMEs
The analyses of the FAMEs were performed in a gas chromatographic GC apparatus (model CP-Varian 3800). This model was equipped with a FID detector and DB-Wax capillary column (60 m×0.32 mm internal diameter, 0.25 μm film thickness). 2 μL of the sample was injected in the Split injection mode (50:1). Oven temperature was programmed using four different ramps. The temperature started at 40°C, held for 0.5 min, then raised at a rate of 25°C/min to 195°C, then raised at a rate of 3°C/min to 205°C, then raised at a rate of 8°C/min to 230°C, then raised at a rate of 20°C/min to 260°C, and finally kept for 5 min. The injector temperature was set at 250°C and the detector temperature at 260°C, the flow rate of carrier gas (helium) was 1 mL/min. Identification and quantification of FAMEs was accomplished by comparing the retention times of peaks with those of pure standards purchased from Sigma and analysed under the same conditions. The results were expressed as total area percentage of identified individual fatty acids in the lipid fraction.
Gas Chromatography-Mass Spectroscopy (GC-MS) analysis of FAMEs
The GC-MS analysis was performed on an AGILENT 6890 GC/CMSD 5973 equipped with a capillary column UB-Wax (30 m×0.25 mm internal diameter, 0.25 μm film thickness) and a 70 eV EI quadrupole detector. Helium was the carrier gas, at a flow rate of 1 mL/min. Injector and MS transfer line temperatures were set at 250°C and 220°C, respectively. Column temperature was programmed same as the gas chromatography. 1 μL of samples were injected using split mode (50:1).
Dewaxing of lipids
Prior to the subsequent determinations: antioxidant activity, quantification of total sterols and total tocopherols, the lipid fractions were dewaxed by the following procedure. Crude lipids were first dissolved in absolute ethanol, and then kept in refrigerator for a period of 24 hours in order to crystallize the waxes fractions; after that, the solution containing frozen waxes particles was filtered and finally, the solvent extracts were eliminated under vacuum pressure in rotary evaporator at 40°C.
Tocopherols analysis by HPLC
Tocopherols were analyzed by HPLC using an Agilent 1290 Infinity apparatus equipped with a Quaternary Pump Model 1260, an automatic liquid sample and a fluorimetric detector (FLD 1260). The detection was set at 295 nm for excitation wavelength and at 330 nm for emission wavelength. The separation column was a SI60 (250 mm×4.6 mm internal diameter, 5 μm film thickness, Lichrosorb, Merck KGaA Darmstadt, Germany). The mobile phase was
Determination of the total tocopherols content of P. lentiscus leaves
The total tocopherols content of the lipids was determined by the spectrometric method described in reference [34] with a few modifications. This method is based on the redox reaction between tocopherols and ferric iron (Fe3 +) which is reduced to ferrous iron (Fe2 +). The latter, in the presence of specific reagents such as
From the standard solution of α-tocopherol in dichloromethane we prepared aliquot solutions with different concentrations. To 2 mL of each solution was added 1 mL of 1,10-phenantroline reagent (0.4% in absolute ethanol), then 1 mL of ferric chloride reagent (0.12% in absolute ethanol) was added and the mixture was shaken for 5 min. The absorbance of the mixture was read at 510 nm. A blank was run, using 2 mL of dichloromethane, 1 mL of 1,10-phenantroline reagent and 1 mL of ferric chloride reagent. The above described procedure was followed by using sample solutions of lipids (dewaxed lipids). The total tocopherols in the dewaxed lipids were calculated from the regression equation of the standard curve.
The results were expressed as α-tocopherol equivalent in mg/(g of lipids).
Determination of the total sterols content of P. lentiscus leaves
This method is based on spectrophotometric absorption according to the Liebermann-Burchard test (or acetic anhydride test) [35, 36], based on a specific coloured reaction of the 3 β-hydroxysteroids having a double bond in position 5-6. The sterols form a stable complex with the β-acetic anhydride in an acid medium which absorbs in the visible at a wavelength of 550 nm (the Liebermann spectral reagent consists of 60 mL of acetic anhydride and 10 mL of concentrated sulfuric acid and 30 mL of acetic acid).
From a chloroformic solution of cholesterol with a concentration of 1 g/L, we have prepared a series of solution dilutions in order to plot a calibration curve linking the optical density to the concentration. 1 mL of each diluted solution was mixed with 2 mL of the Liebermann reagent, then kept for 25 min in order to allow reagent colour to fully develop and stabilize.
The above described procedure was followed by using sample solutions of lipids (dewaxed lipids). The total sterols in the dewaxed lipids were calculated from the regression equation of the standard curve. The results were expressed as cholesterol equivalent in mg/(g of lipids) and in μg/(g leaves “d.w.”).
Antioxidant activity
Radical-scavenging activity (DPPH assay)
The antioxidant activity was determined by DPPH method, which is based on the evaluation of the free-radical scavenging capacity. Briefly, 0.5 mL of the sample (dewaxed lipids) diluted in absolute ethanol was mixed with 0.5 mL of 250 μM DPPHepsfbox
Where A0 and A are the absorbance values of control and of the tested sample, respectively.
Antioxidants of reference (Vitamin E and ascorbic acid) were used for comparison. All experiments were performed in triplicate.
Bleaching ability of β-carotene in linoleic system
The inhibition of β-carotene bleaching by the lipid fractions was determined according to slightly modified method described in literature [37].
A volume of 5 ml of dichloromethane solution of β-carotene at initial concentration of 0.1 mg/mL was added to a round flask containing 20 mg of acid linoleic and 200 mg of Tween 80. Dichloromethane was completely removed at 40°C under vacuum, then 50 mL of distilled water was slowly added and vigorously shaken. Aliquots (40 μL) of different solutions concentrations were added to 1 mL of β-carotene/linoleic acid emulsion. A control preparation was obtained by adding 40 μL of ethanol to 1 mL of β-carotene/linoleic acid emulsion. Absorbance of the preparations was measured at 490 nm before and after 2 h of incubation in a water bath at 50°C. Antioxidative activity (AA%) in percentages was calculated using the following formula:
Where:
Ac0 is the absorbance of the control respectively measured before the incubation.
As120 and Ac120 are the absorbance of the test and the control respectively measured after 2 h of incubation.
Results were expressed in AA% corresponding to solutions concentrations of 2 mg/mL (relative to the 40 μL added). The results were also expressed as AA50, the concentration providing 50% β-carotene bleaching inhibition. Vitamin E was used as positive control. All trials were performed in triplicate.
Cluster analysis
Cluster analysis was performed using Agglomerative Hierarchical Clustering AHC (Ward’s technique) with Euclidean distance measure. The calculus was performed using a set of individuals composed of 5 different fatty acids samples (different plants from the region of Zeralda). The total number of adopted variables was 20 (representing simply the major identified components in all represented individuals, or the most influencing parameters that could make a difference
Principal component analysis (PCA)
The principal component analysis was performed using both the same individuals and variables adopted for Agglomerative Hierarchical Clustering method.
Results and discussion
Lipid contents and fatty acids compositions of P. lentiscus leaves
The lipid contents and the fatty acids FAs composition of the
Fatty acid compositions of the lipids fractions obtained from the leaves of Pistacia lentiscus
ΣSFA: Sum of saturated fatty acids; Σ MUFA: Sum of mono-unsaturated fatty acids; Σ PUFA: Sum of poly-unsaturated fatty acids; Σ USFA: Sum of unsaturated fatty acids; U/S: ratio “unsaturated/saturated” fatty acids.
Fatty acid compositions of the lipids fractions obtained from the leaves of
ΣSFA: Sum of saturated fatty acids;
The results of gas chromatography GC and gas chromatography coupled to the mass spectroscopy GC-MS analysis of the fatty acids methyl esters FAMEs of
For better understanding of similarities and/or differences between the studied samples in term of FAs compositions, cluster analysis was performed using Agglomerative Hierarchical Clustering AHC. The results of AHC schemed in the dendrogram of the Fig. 1, showed two clearly distinguished clusters “or groups” of samples (cluster 1: Z1, Z5) and (cluster 2: Z2, Z3 and Z4). Which indicated an existing dissimilarity of the studied samples (proves the existence of some variability of the chemical composition). The comparison of the chemical composition of samples Z1 and Z5 indicates roughly close values of the percentages of both major and minor components. For samples Z2 and Z3, the distance between these two samples was very low (Fig. 1), which means a very close values of percentages in both samples, as confirmed by the data of Table 1. Sample Z4 was clustered with Z2 and Z3 but with some existing differences of some components. The most influencing difference is that C10:0 in Z4 was becoming a minor component (3.18%) at the opposite of Z2 and Z3 where it was considered as a major component (11.74%, 13.88%). The most influencing difference between cluster 1 and 2 is, the higher percentages of C18:3w3 (48.92%, 33.94%) in cluster 1 in comparison with cluster 2 where the percentages were important but lower (20.92–27.71%).
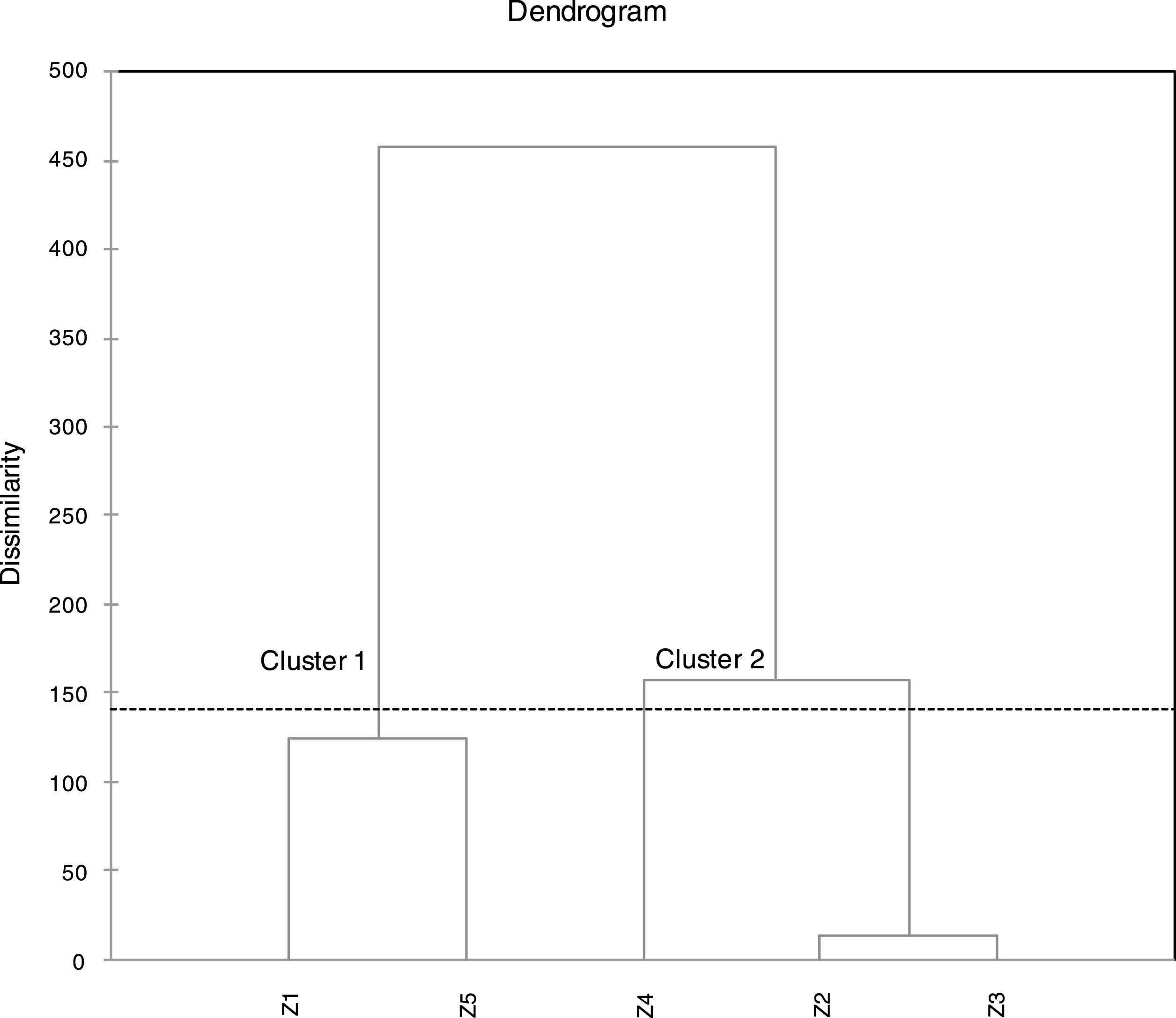
Dendrogram obtained from a cluster analysis of five samples of fatty acids of Algerian
In order to determine a more detailed similarities or differences between the two cluster and the samples, principal component analysis PCA method was performed to try to identify the most influencing components that can distinguish or regroup the studied samples.
These above observations (occurrence of two clusters) were also confirmed by the results of PCA (Fig. 2), and which allowed us to discuss the similarities and the differences upon the chemical compositions. Loading factors for principal axes F1 and F2 (representing 71.24% of the total information), are given in Fig. 2. The F1 Axis, which represents 48.98% of the total information, is strongly and positively correlated with C23:0 (99.2%), C20:0 (97.8%), C22:2 (95.6%), C20:2 (95.1%), C20:1 (91.4%) and C20:3 (84.7%). This same axis is negatively correlated with C15:0 (– 87.7%), C16:0 (– 81.5%) and C14:0 (– 80.9%).
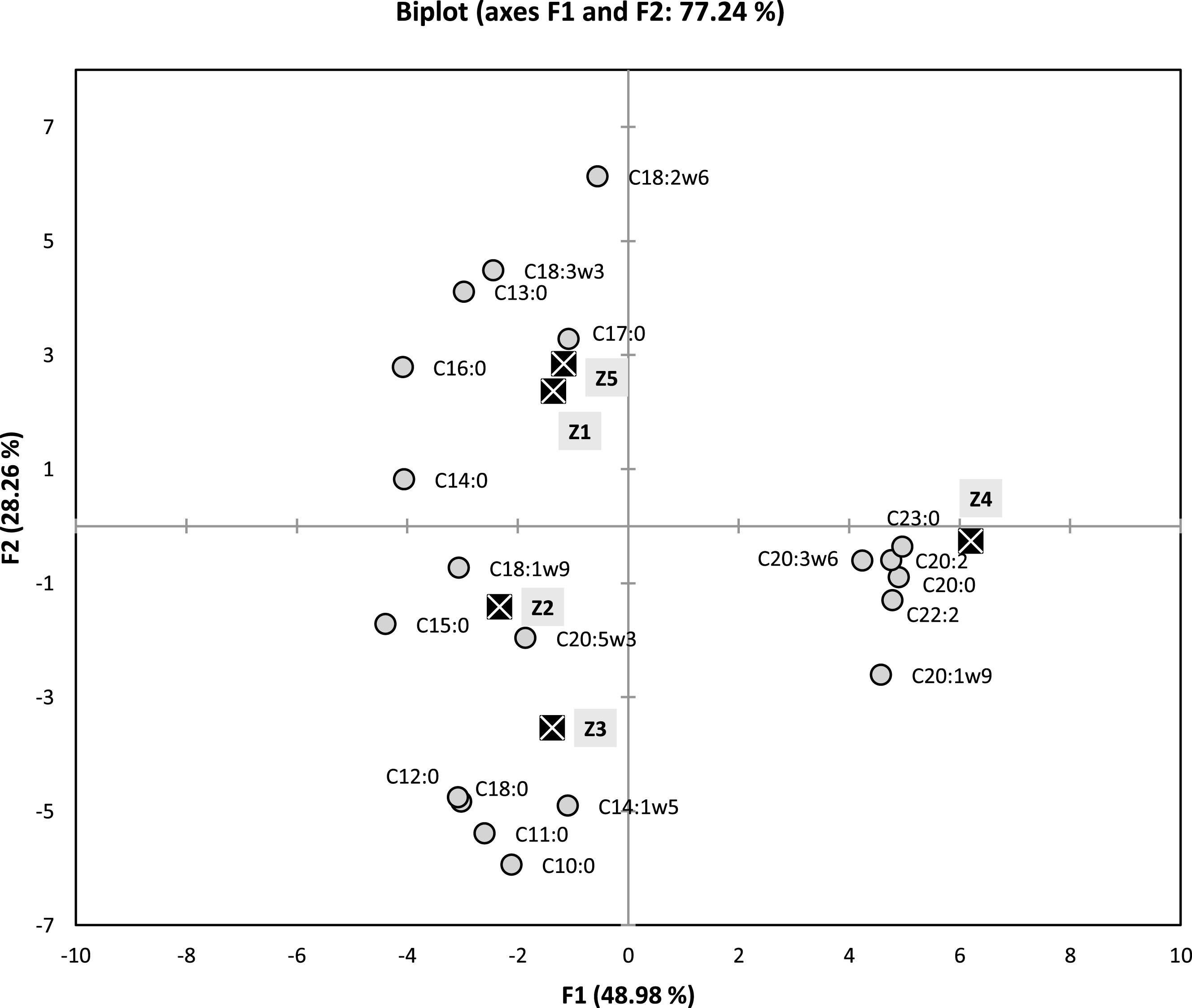
Two dimensional plot on axes F1 and F2 using PCA of five samples of fatty acids of Algerian
Axis F2, which represents 28.26% of the total information, is highly negatively correlated with C10:0 (– 90.0%), C11:0 (– 81.7%), C14:1 (– 74.4%), C12:0 (– 73.3%) and C18:0 (– 72.2%). This same axis is strongly and positively correlated with only one compound C18:2 (93.1%).
As previously demonstrated with AHC method, there is appearance of the same formerly grouping of (Z1, Z5) and (Z2, Z3: with larger distance) and Z4, in which this time can be clearly distinguished (Fig. 2). The sample Z4 is strongly correlated with relatively higher percentages of C20:0 (7.07%
The comparison of obtained results here in this investigation with the previous previous reports from Germany [32] and Turkey [33] revealed very few similarities but lot of differences (Table 2). Starting with similarities, the samples of Algeria were presenting comparables ranges with those of Germany and Turkey for the following compounds C14:0, C18:0 and C18:3. When considering differences, there were lot of dissimilarities detected. The compound C16:1 which was not detected in Algerian samples was present as a minor compound in samples of Germany and Turkey. The percentage ranges of C16:0, C18:1 and C18:2, were higher in the samples of Germany and Turkey, in comparison with those of Algeria. A large number of detected minor compounds in Algerian samples (at least fourteen FAs) were not reported (or not identified) in those of Germany and Turkey. Finally, it is very useful to mention that the range variation of C18:3 in the Algerian samples were presenting larger arrays (for min. and max.) in comparison with those of literature samples. This is the first investigation than report the presence of C18:3 with a percentage that can reach 48.92%.
Comparison of the determined percentages of the fatty acids of the leaves of
The lipids of
Tocopherols composition analysis
Tocopherols analysis was performed for the five adopted samples. The results of the tocopherols percentages determined by HPLC analysis was exposed in Table 3. The data revealed that all investigated lipid samples contained a substantial amount of α-tocopherol “most important and requested vitamin”, ranging from 58.51 to 89.17% (with mean-value±sd = 76.14±11.20%). Furthermore, the γ-tocopherol was completely absent in all samples. For most investigated samples, the percentages of β-tocopherol were low (3.24–4.35% “minor compound”), except for Z5, where its percentage was relatively significant (14.02%). For δ-tocopherol, its percentage was moderately significant in the majority of studied samples (7.59–27.47%). The founded percentages values of tocopherols isomers suggested the presence a certain variability of the composition, which should be further clarified using a larger number of populations.
Tocopherols and sterols contents in the lipids fractions obtained from the leaves of Pistacia lentiscus
Percentages determined by integration of surface area.
Tocopherols and sterols contents in the lipids fractions obtained from the leaves of
Percentages determined by integration of surface area.
Recently, the cosmetic, drug and alimentary/nutraceutical industries have focused attention on low-cost renewable resources, rich in lipid-related compounds such as phytosterols which are an important part of the unsaponifiable matter of vegetable oils. The analysis of the sterols provides rich information about the quality and the identity of the oil investigated, and for the detection of oil and mixtures not recognized by their fatty acids profile [21].
The results of the quantification of the dewaxed lipid fractions obtained from the leaves of
The total contents of sterols were much higher than the content of total tocopherols. The range content of sterols was varying from 143.37 to 175.61 mg/“g of lipids”, which were presenting significant variations. The three samples Z2, Z3 and Z4 were presenting close values of sterols contents, and especially for Z3, Z4. The lowest content value was recorded for the sample Z5.
Quantification of total tocopherols of P. lentiscus leaves
Tocopherols (vitamin E isomers) are well known natural antioxidants whose presence in oils is often correlated with a relatively high abundance of USFA.
The results of the quantification of sterols and tocopherols in the dewaxed lipid fractions obtained from the leaves of
Antioxidant activity
DPPH assay (free radical scavenging activity)
The antioxidant activity of the lipid fractions was determined
Antioxidant activity (DPPH assay and β-carotene test) of the lipid fractions obtained from the leaves of Pistacia lentiscus
Antioxidant activity (DPPH assay and β-carotene test) of the lipid fractions obtained from the leaves of
Correlation between antioxidant activity (expressed as Antioxidant Capacity AOC = 1/IC50) and total tocopherols contents were investigated using linear equation model and estimated by minimum least square method. The found model gave a relatively good correlation factor “correlation coefficient
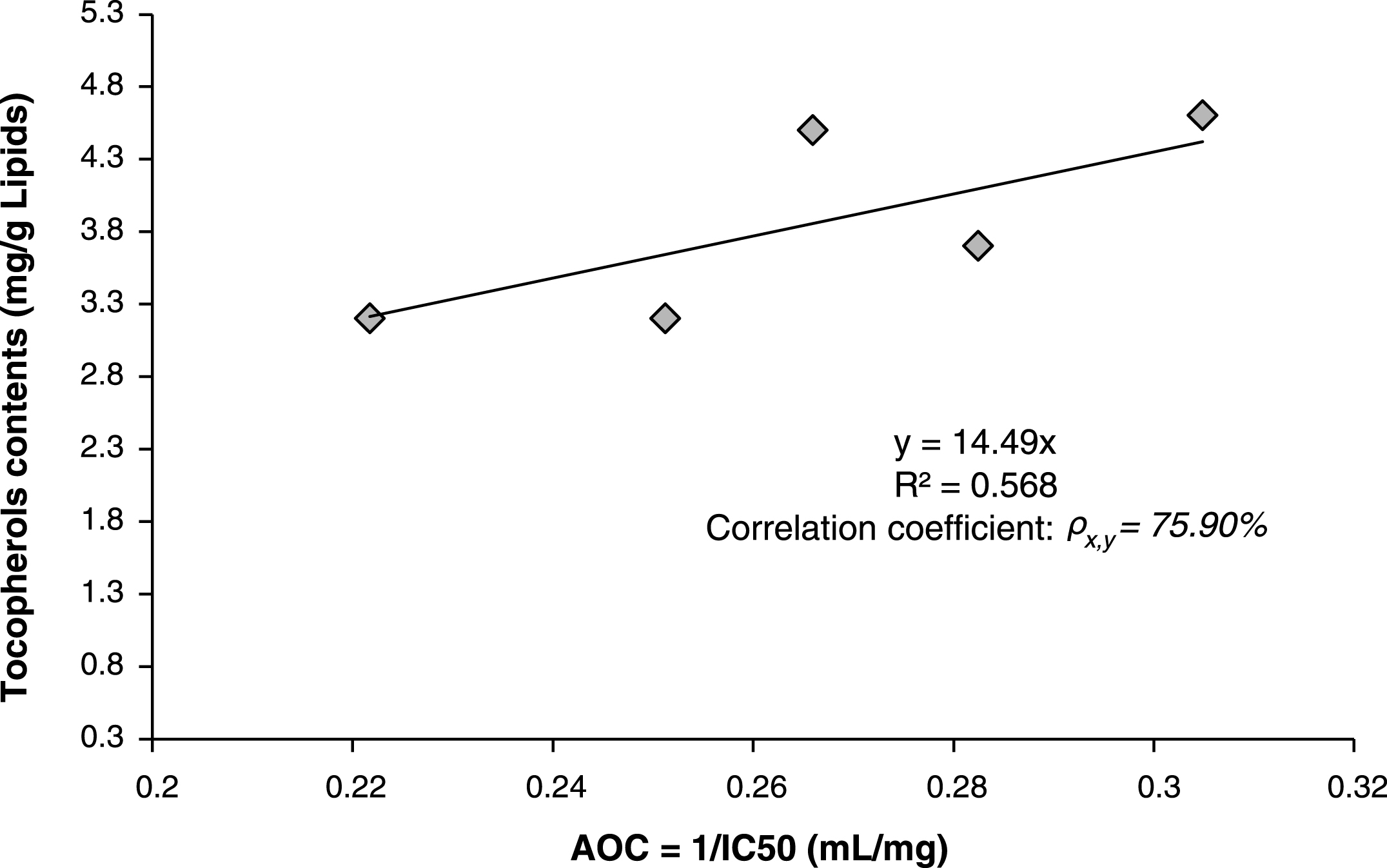
Linear correlation between antioxidant activity (Antioxidant capacity AOC) and total contents of tocopherols in the lipids of
Linear correlations between percentages of unsaturated fatty acids (C18:1, 2 and 3) and total contents of tocopherols were also investigated. When exception was made for the sample Z4 (excluded), Fig. 4, shows strong linear relationship between total tocopherols and the percentages of C18:1 or C18:2 or C18:3, or the sum of these components. Especially for both C18:3 and total C18: it was observed that higher content of tocopherols in the lipids is directly correlated with higher content of C18: (or sum of C18:) “Correlation coefficient
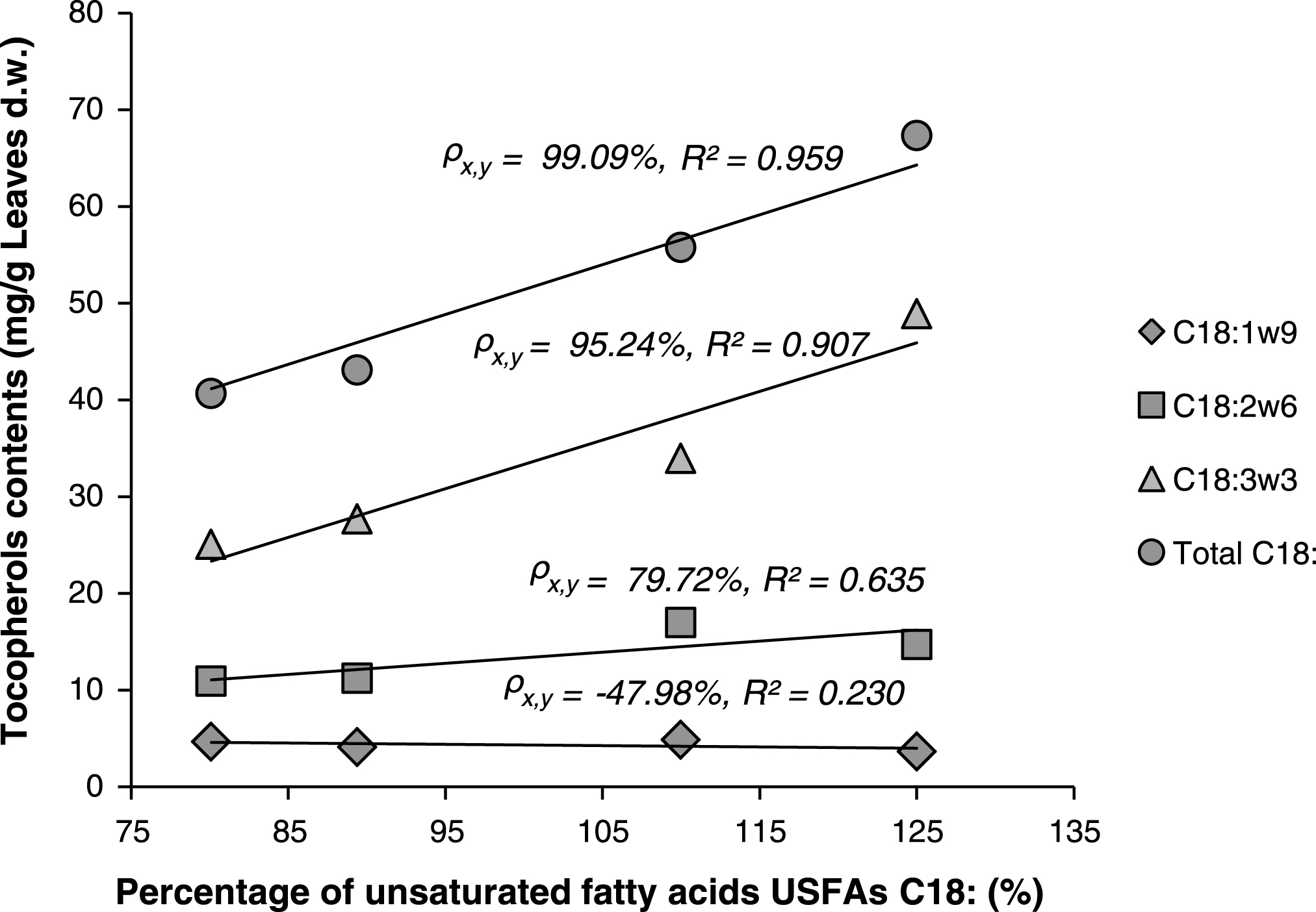
Linear correlations between percentages of unsaturated fatty acids (C18:) and total contents of tocopherols in the lipids of
The result of β-carotene bleaching test was summarized in Table 4. In addition, kinetic evolution of antioxidants of reference (BHA, BHT, vitamin C, vitamin E) in β-carotene/linoleic system is presented in Fig. 5. The antioxidants of reference showed different responses in term of their activity at 2 mg/mL. Explicitly, vitamin C presented very weak activity in comparison with the rest of the antioxidants, whereas BHA and BHT were very effective antioxidants. Furthermore, vitamin E was also exhibiting practically lower but close activity to both BHA and BHT. The comparison of the antioxidant activity AA% of the lipid samples with those of antioxidants of reference shows some variability of this activity ranging from 37.24 to 71.72%, the highest value was recorded for sample Z2 (this value was close to recorded for vitamin E) whereas the lowest one was belonging to sample Z3. In terms of AA50, the antioxidant data values revealed the presence of two clusters of activities. The first cluster, which involves Z1 and Z2 samples, showed the highest activities in term of AA50 (1.53 and 1.43 mg/mL). The second cluster (Z3,4,5) exhibited a slightly lower activities compared to the first one with AA50 range variation of 2.21–2.52 mg/mL. As a conclusion the lipid fractions presented good antioxidant activities in comparison with those of antioxidant of reference.
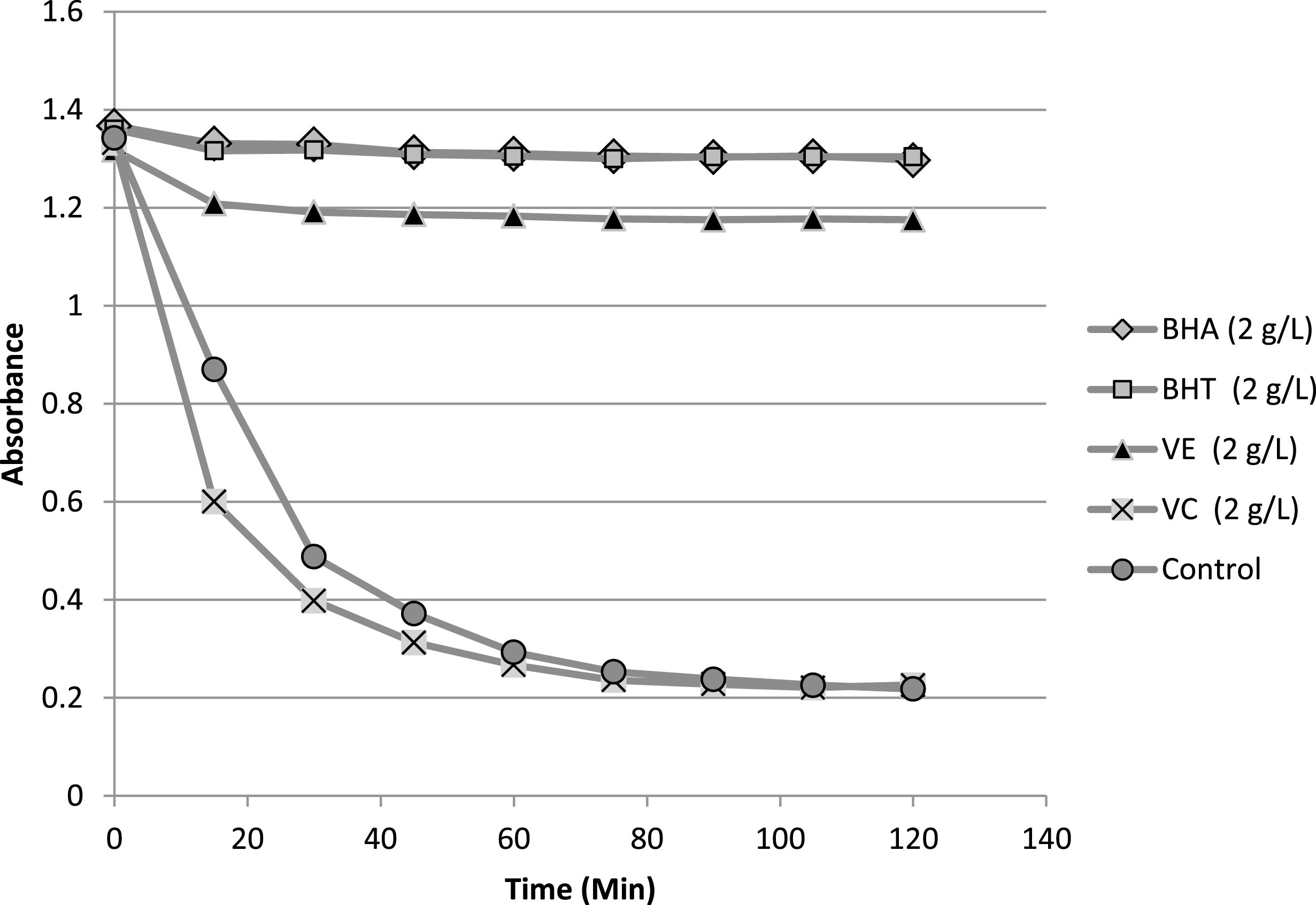
Evolution of the absorbance of antioxidants of reference in β-carotene/linoleic system as function of time.
In order to determine possible existing relations between antioxidant activity (two assays) and the main important chemical components of the fatty acids (C18:1,2,3) and tocopherols (α,β,δ-Toc), correlations between these three groups of variables were determined using Excel internal statistical function “COEFFICIENT.CORRELATION”. The results were gathered in Table 5. Strong positive correlation was found between IC50 (DPPH assay) and the two isomers β,δ-Toc. Alternatively, strong negative correlation was obtained between IC50 and α-Toc Moreover, good correlation was also found between IC50 and USFAs C18: except for the case of C18:3 where low correlation value was noticed (15.61%). This means that the higher antiradical power (lower IC50 values) is primarily correlated with high percentages of α-Toc (strong negative correlation), and in the same time with low percentage of C18:1,2 and β,δ-Toc. For β-carotene assay, the results indicated no meaningful correlation between the three investigated variables. Furthermore, correlations between the percentages of the two sets of variables C18:1,2 and α,β,δ-Toc were also determined. First, C18:2 was highly positively correlated with β,δ-Toc, and at the same time highly negatively correlated with α-Toc. Secondly, tocopherols isomers were exhibiting some strong correlations within each others
Correlations between antioxidant activity (DPPH assay and β-carotene test), C18: and tocopherols
Correlations between antioxidant activity (DPPH assay and β-carotene test), C18: and tocopherols
One of the objectives of food industry is to seek new resources of oil that preferably presents nutritional values. For human health, fats and particularly vegetable oils are considered as an important source of energy when glucose is not available.
The lipids of leaves of
The main result of this study illustrate the nutritional potential (richness in MUFA such as C18:1w9, presence of essential fatty acids such as C18:2w6 at all the stages of maturation, richness in omega-3 fatty acids such as C18:3w3) of the oil of
