Abstract
Photocatalysis using semiconductor metal oxide stands out as a highly effective and efficient method for eliminating organic pollutants from wastewater. This study aims to assess the photocatalytic capabilities of Mn doped ZnO nanocomposites in degrading methylene blue (MB) dye under ultra-violet light exposure. This study details the synthesis of ZnO photocatalysts through a straightforward one-step sol-gel method, incorporating varying levels of Mn-doping (0%, 2% & 4%). The structural and optical attributes were examined using techniques such as powder X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), transmission electron microscopy (TEM), energy dispersive spectroscopy (EDS) and UV-Vis diffuse reflectance spectra. X-ray diffraction analyses verified the presence of a hexagonal wurtzite crystal structure in all synthesized samples, exhibiting a high degree of crystallinity. In addition, an investigation into the impact of Mn impurities on the photocatalytic performance of ZnO catalysts was conducted in the context of methylene blue (MB) degradation. The experimental findings revealed that the Mn doped ZnO nanoparticles produced exhibited significantly superior photocatalytic performance compared to pure ZnO when used in breaking down methylene blue under UV-light exposure. This study proposes that these Mn doped ZnO could serve as a highly effective photocatalyst for treating water contaminated with certain chemically persistent synthetic organic dyes. The improved photocatalytic capabilities of ZnO nanostructures doped with Mn were ascribed to the synergistic impact of increased surface area in ZnO nanosphere and enhanced efficiency in charge separation resulting from optimized Mn doping. A potential explanation for the heightened photocatalytic performance of Mn-doped ZnO nanostructures is proposed tentatively.
Introduction
Water source contamination is currently among the most critical global issues, largely attributed to population growth and the release of waste materials by chemical and textile industries. The presence of synthetic dyes in these sectors significantly contributes to environmental pollution, as they possess high toxicity for both flora and fauna and exhibit resistance to natural breakdown processes. Additionally, the widespread use of heterocyclic dyes, like methylene blue (MB), in industrial contexts further compounds the problem due to their substantial toxicity, resulting in various detrimental impacts on the environment and human health [1, 2]. Numerous methods are employed to eliminate toxic substances from wastewater, encompassing biological treatment [3], adsorption process [4], and heterogeneous semiconductor photocatalysis [5–8]. Among these approaches, semiconductor heterogeneous photocatalysis can take place in the presence of solar energy, a sustainable and clean source and has demonstrated efficacy in converting harmful organic pollutants into CO2 and H2O [9, 10]. Semiconductor photocatalysts have been widely investigated as a primary focus for addressing the presence of organic pollutants in discharged water [11]. Semiconductor metal oxide photocatalysts play a vital role across diverse domains, especially in the spheres of environmental and energy applications. These substances possess distinct characteristics that render them highly valuable for leveraging solar energy to initiate chemical reactions, commonly achieved through the mechanism of photocatalysis. Semiconductor photocatalysts efficiently break down pollutants like dyes, detergents, and volatile organic compounds [12]. Their appeal lies in simplicity, mild reactions, and low energy usage [13]. Metal oxides like CuO, MgO, TiO2, Fe2O3, and ZnO are also explored for environmental pollutant reduction [14].
Recently, the utilization of ZnO particles for their UV absorption capabilities has captured the interest of a wide array of researchers. In addition, recent reports highlighted that the property of high photosensitivity in zinc oxide has led to significant attention, primarily due to its potential for degrading a variety of pollutants. Zinc oxide exhibits superior photocatalytic efficacy compared to other metal oxide, alongside favorable biocompatibility and environmental stability. It represents a semiconductor variety characterized by its wide direct band gap width (3.37 eV), substantial excitation binding energy (60 meV), and robust absorption in the deep violet to borderline ultraviolet (UV) range at room temperature [15, 16]. ZnO’s versatile properties, ranging from its semiconducting nature to its optical and antibacterial characteristics, contribute to its widespread use across these diverse fields [17–21]. Zinc oxide (ZnO) has risen to prominence as a multifaceted material with exceptional attributes, rendering it a central subject of investigation across diverse domains. Among its noteworthy applications is its utilization for piezoelectric catalysis. ZnO distinguishes itself as a significant piezoelectric catalyst. The imposition of mechanical pressure on ZnO crystals results in the creation of electric fields, a phenomenon that has the potential to amplify reaction kinetics and alter reaction trajectories, consequently contributing to the enhancement of catalytic efficiency. This distinct characteristic has captured interest in both environmental and industrial catalytic domains [22]. Apart from functioning as a piezoelectric catalyst, zinc oxide finds further utility in the realm of adsorption processes, particularly when coupled with other semiconductor materials. The combination of ZnO with different semiconductors leads to the formation of semiconductor composites, which display remarkable adsorption capacities. These composite materials harness the expansive surface area, adjustable surface chemistry, and photochemical reactivity of ZnO to efficiently adsorb a diverse range of pollutants from various mediums. The unique composite structure facilitates synergistic interactions between ZnO and the incorporated semiconductor, ultimately culminating in an improved level of adsorption efficiency and selectivity [23, 24]. However, the recombination of charge carriers in ZnO remains relatively elevated, limiting its broad utilization in photocatalytic reactions. To address this issue, a variety of approaches such as doping with other material, surface modification, making hybrid etc. have been employed. Doping with transition metals can also improve the materials optical absorbance and photocatalytic activity of ZnO nanoparticles [25–27]. It is feasible to introduce metal dopants such as Ag, Cu, Au, La, Sm, and Fe, as well as non-metal dopants like N, F, C, and S into the ZnO structure. Recently, S.H. Zyoud et al. synthesized Ni doped ZnO laser assisted chemical bath method and reported introduction of metal ions like Ni into ZnO through doping can enhance its photocatalytic activity by effectively narrowing its bandgap and mitigating the recombination rate of electron-hole pairs [28]. Similarly, Balasubramani et al. synthesized Cu and Co doped ZnO nanoparticles by citrate gel combustion method and reported that the improved visible-light photocatalytic efficiency of the prepared (Co, Cu)/ZnO arises from the incorporation of Co and Cu atoms into the ZnO structure, which results in the generation of photo-induced electron-hole pairs and an extended spectral response encompassing the visible region [29]. In their study, Kumaresan et al. synthesized nanoparticles of ZnO doped with Al and Cu. They found that the presence of Cu doping led to a notable enhancement in the photocatalytic capability [30]. This improvement was attributed to several factors, including the expansion of visible light absorption range, suppression of electron-hole recombination, and increased dye molecule adsorption on the surface of Cu-doped ZnO nanopowders. In this study, we have opted for the incorporation of Mn dopants into ZnO to enhance its photocatalytic efficacy. Manganese is being considered as a potential component for ZnO due to its wide range of electron states and strong solubility in the ZnO matrix. Manganese enhances the separation of photo-excited electrons from holes in a photocatalytic process, increasing photocatalytic activity [31]. Numerous techniques have been employed for synthesizing ZnO nanostructures, including non-aqueous methods and chemical precipitation, alongside the sol-gel process. Among these, the sol-gel approach has garnered significant attention owing to its straightforward operational procedure and cost-efficient pathway for synthesis. Consequently, the focus is on producing Mn-doped ZnO nanoparticles through a uncomplicated sol-gel route. The influence of Mn on the structural, morphological, optical, and photocatalytic characteristics of the ZnO nanoparticles has been methodically examined.
Materials and methods
Materials
Reagents including Zinc acetate dihydrate, Manganese acetate tetrahydrate, ethanol, and other compounds were procured from Sigma Aldrich Company, Mumbai. The acquisition of commercial methylene Blue (C16H18N3SCl) was made through local chemical suppliers. Distilled water served as the base for solution preparation. pH modulation was accomplished using hydrochloric acid or sodium hydroxide solution. The 99.5% pure reagents were used in the investigation. All of the equipment was thoroughly rinsed with tap water, followed by distilled water after being extensively cleaned in acid. All throughout process of this experiment, deionized water has been employed as the solvent.
Synthesis of manganese doped zinc oxide nanoparticles
Zinc acetate dihydrate (Zn (CH3COO)2·2H2O) was dissolved in ethyl alcohol to create a 0.1 M solution, forming the initial solution. This solution was then combined with the appropriate quantity of manganese acetate (Mn (CH3COO)2·4H2O) in deionized water, with varying concentrations of 0, 2, and 4 wt%. The companion solution comprised 10 mL of ethanol and 20 mM of NaOH. The first heating was conducted at 50°C for these three solutions. Subsequently, the manganese acetate solution was added to the round-bottom flask after heating the zinc acetate solution. The mixture of zinc acetate and manganese acetate was then stirred continuously and heated to 80°C for 30 minutes. Subsequently, a reflux process was conducted by introducing Zn (CH3COO)2·2H2O and Mn (CH3COO)2·4H2O into a solution, followed by the gradual addition of NaOH solution. The solutions were once again heated to temperatures of 60–65°C and stirred magnetically for a period of two hours. The solution was then gradually cooled to reach room temperature. After the formation of precipitates, a thorough rinsing with distilled water was carried out, followed by an ethanol treatment. This step aimed to eliminate undesired salts and contaminants through the precipitation’s ethanol wash. The final product was exposed to a muffle furnace maintained at 550°C for a duration of 5 hours. The identical process was replicated to produce pure ZnO nanopowders, omitting the use of a manganese source.
Photocatalytic experiment
A synthetic photocatalyst was evaluated on 30 ppm of the contaminant model methylene blue together with UV to demonstrate its effectiveness in examining the photocatalytic discoloration efficiency of the pollutant. To assess the effectiveness of un-doped and Mn-doped nanoparticles, UV light source (WSFSWL ultraviolet lamp with specified voltage 220 V, nominal wattage 30 W) was employed to effectively degrade the dye. ZnO nanoparticles, both un-doped and doped (0.2, 0.4M), were dissolved in 50 mL of 10 mg/L MB dye solution.For 30 minutes, the mixture was agitated in the dark to allow the molecules of MB dye to adsorb to the surface of the Zn1 - xMnxO nanoparticles. A Xenon lamp was used to irradiate the homogenous mixture (200 W). After being exposed to radiation for 10 minutes, 5 mL of the aqueous dispersion were sampled and centrifugation was used to separate the nonmaterial’s. An UV spectrophotometer was used to quantify the remaining MB in the supernatant liquid. The photo catalytic efficiency was determined by using the following formula [32]
where, C0 and Ce correspond to the initial and final concentration of MB before and after light irradiation.
Characterization
A Philips PW 137 X-ray diffractometer (XRD) with a Cu source (k = 1.5406) was used to measure the crystalline nature of the obtained products in the scan range (2 theta) of 20° to 80° at room temperature. SEM imaging is commonly carried out within the acceleration voltage range of 5 kV to 30 kV, employing a working distance that typically spans 5 mm to 15 mm. Additionally, a spot size in the range of 1 to 10μm is often chosen. These parameter selections collectively ensure an effective balance between resolution, depth of field, and sample preservation across various SEM application. Both HRTEM (TEM-2100 plus electron microscope) and EDAX (AZTEC/Xact) were selected. The CARY 5E UV-Vis spectrometer was utilized to explore optical properties in the wavelength range of 200–800 nm utilizing UV-Vis spectroscopy. Utilizing a 5DX FTIR spectrometer, the compounds’ functional groups were calculated. Using X-ray photoelectron spectroscopy, the chemical states of the elements included in the nanoparticles were identified (XPS).
Results and discussion
Analysis of X-ray diffraction
Figure 1 illustrates the XRD patterns of both Mn-doped and undoped ZnO nanoparticles. The XRD analysis was conducted within a 2θ range of 0° to 80° to examine the structural characteristics of the Mn-doped and pristine ZnO nanoparticles. The identified Miller indices, including (100), (002), (101), (102), (110), (103), (112), and (203), correspond to specific 2θ values: 31.95°, 34.61°, 36.43°, 47.73°, 56.78°, 63.05°, 68.13°, and 69.26°, respectively. These values correspond to distinct crystallographic planes within the hexagonal wurtzite structure, consistent with JCPDS Card no. 36-1451. Importantly, no evident peaks indicating the presence of impurities are observed in the doped samples. Figure 1 also illustrates the displacement of peak positions is a consequence of Mn atoms being integrated into the ZnO lattice, which introduces strain and distortion to the lattice structure. This perturbation caused by Mn doping impacts the distances between atoms, resulting in changes to the crystal arrangement and causing the observed shift in peak positions. The magnitude of this shift correlates with the quantity of Mn incorporated, with higher doping levels causing more pronounced lattice distortions. The usual crystallite size can be obtained utilizing Scherer’s formula [33]. The average crystalline size for pure ZnO is measured to be 49.5 nm, whereas in the Mn doped samples, the crystallite size decreases to 43.3 nm. It has been discovered that when Mn concentrations rise, the average crystallite size tends to reduce.
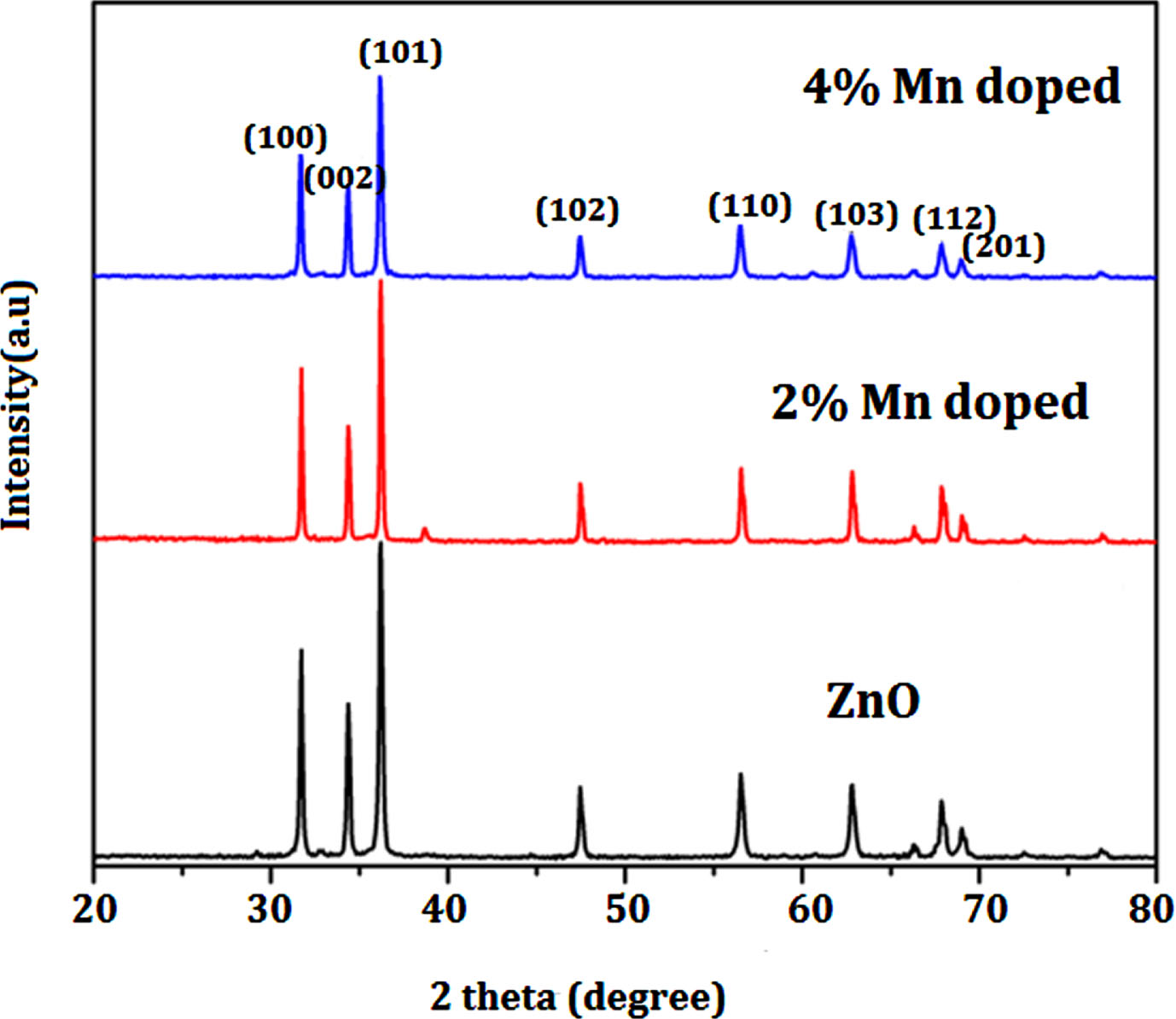
XRD pattern of Un-doped, 0.2% Mn/ZnO and 0.4% Mn/ZnO nanoparticles.
Using the KBr pellet method, FT-IR spectra were recorded in the solid phase, covering the 4000–400 cm–1 range. Figure 2 displays the FT-IR spectra for each individual sample. The alterations observed in the positions and strengths of IR peaks within Fig. 2 imply the potential incorporation of Mn into the ZnO host material. Notably, the IR peak at 632 cm–1 may be linked to the anti-symmetric stretching mode of Zn-O-Zn bonds, while the spectral range between 480 and 510 cm–1 corresponds to symmetric Zn-O-Zn and Mn-O bonds. Additionally, the appearance of a peak at 1120 cm–1 indicates the presence of hydroxyl-Zinc (Zn-OH) bond vibrations. The vibration detected at 1340 cm–1 can be ascribed to the C-O stretching mode associated with the citrate complex. In solid citrate, the deprotonation of carboxylic groups gives rise to the emergence of a peak at 1602 cm–1, which corresponds to the stretching vibration of C = O bonds. The presence of absorbed water becomes evident through prominent bands spanning the spectral range from 3430 to 1602 cm–1. It’s noteworthy that the provided values closely match those documented in pertinent literature, affirming a strong concurrence between established references and the present findings [34]. The characteristic peak at around 2300 cm–1 is typically associated with the presence of carbon dioxide adsorbed onto a sample. This peak corresponds to the asymmetric stretching vibration of the carbon-oxygen double bond (C = O) in CO2 [35, 36]. It indicates that carbon dioxide is interacting with the sample. This could be due to physical adsorption or chemisorption of CO2 molecules onto the surface. In this investigation, Mn ion replacement must cause the diagonal optical mode to swing downward and the peak to move to the doped sample’s vibrational modes are acceptable at a frequency of 480 cm–1 and above.
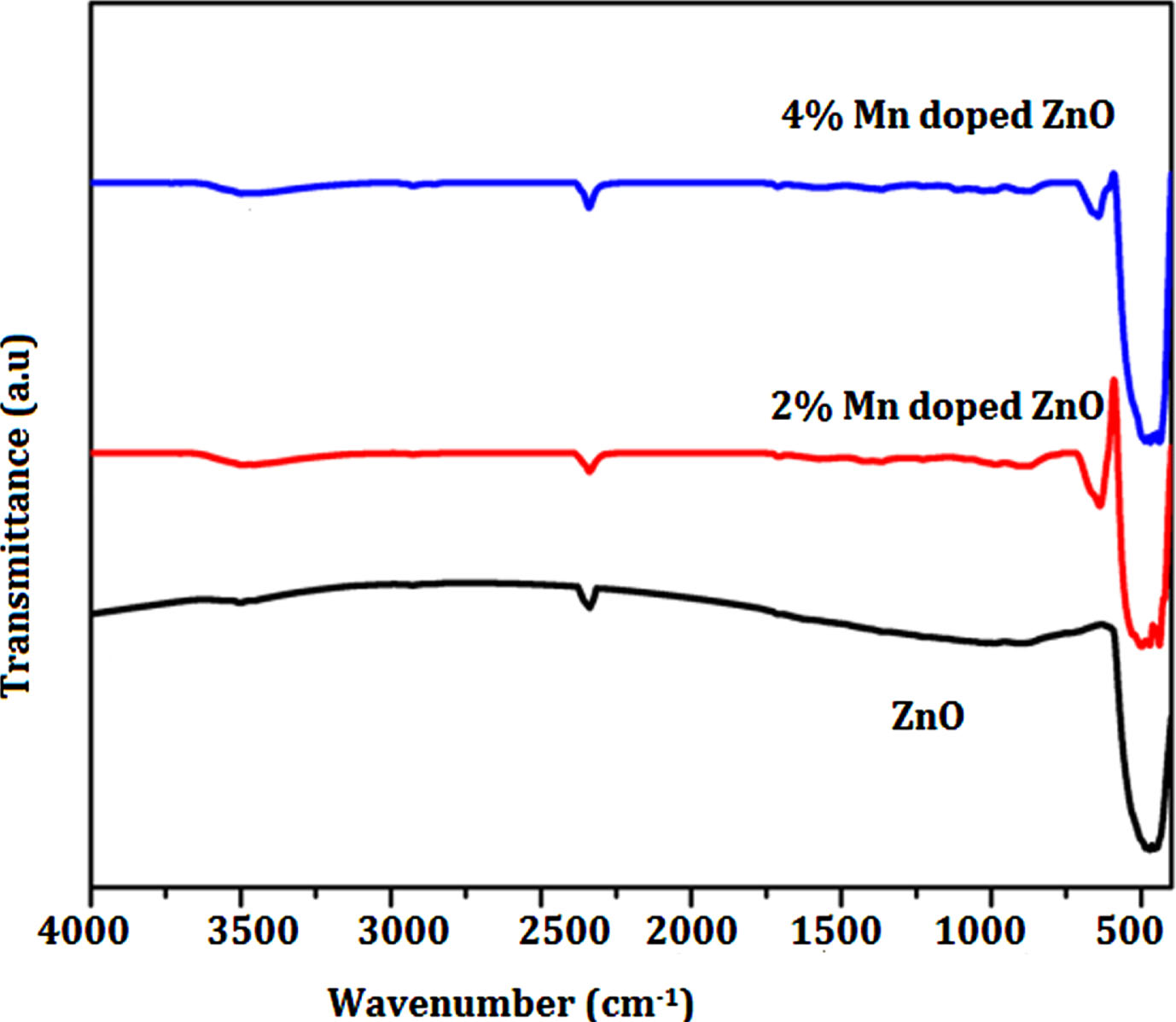
FT-IR spectrum of Un-doped, 0.2% Mn/ZnO and 0.4% Mn/ZnO nanoparticles.
The optical characteristics of the un-doped and Mn-doped ZnO nanoparticles were studied utilizing UV-visible spectroscopy. UV-Vis diffusion reflectance spectroscopy was used to make optical measurements in order to verify that Mn2 + has been swapped for Zn2 +. The KM plot was generated using the UV data of the samples to determine the band gap energy [37, 38]. The corresponding plots depicting the results are presented in Fig. 3. It has been found that the Mn doped ZnO’s optical band gap drops from 3.27eV to 3.20 eV, when the Mn concentration(0–4wt%) is increased. The cause of the Mn doped ZnO’s absorption wavelength range red shift most likely be attributed to the generation of a faulty energy level in the ZnO optical band gap between the valence and conduction bands.This might be as a result of the Mn2 + incorporating in the ZnO lattice causing a significant sp-d energy exchange between the s and p electrons in the conduction band of ZnO, as well as the d electrons of Mn allowing the conduction band’s location to move downward and reducing the energy of the forbidden band [39]. An efficient nano photo catalyst has been developed, as evidenced by the decreased band gap value of the final dopant Mn on ZnO semiconductors in this work.
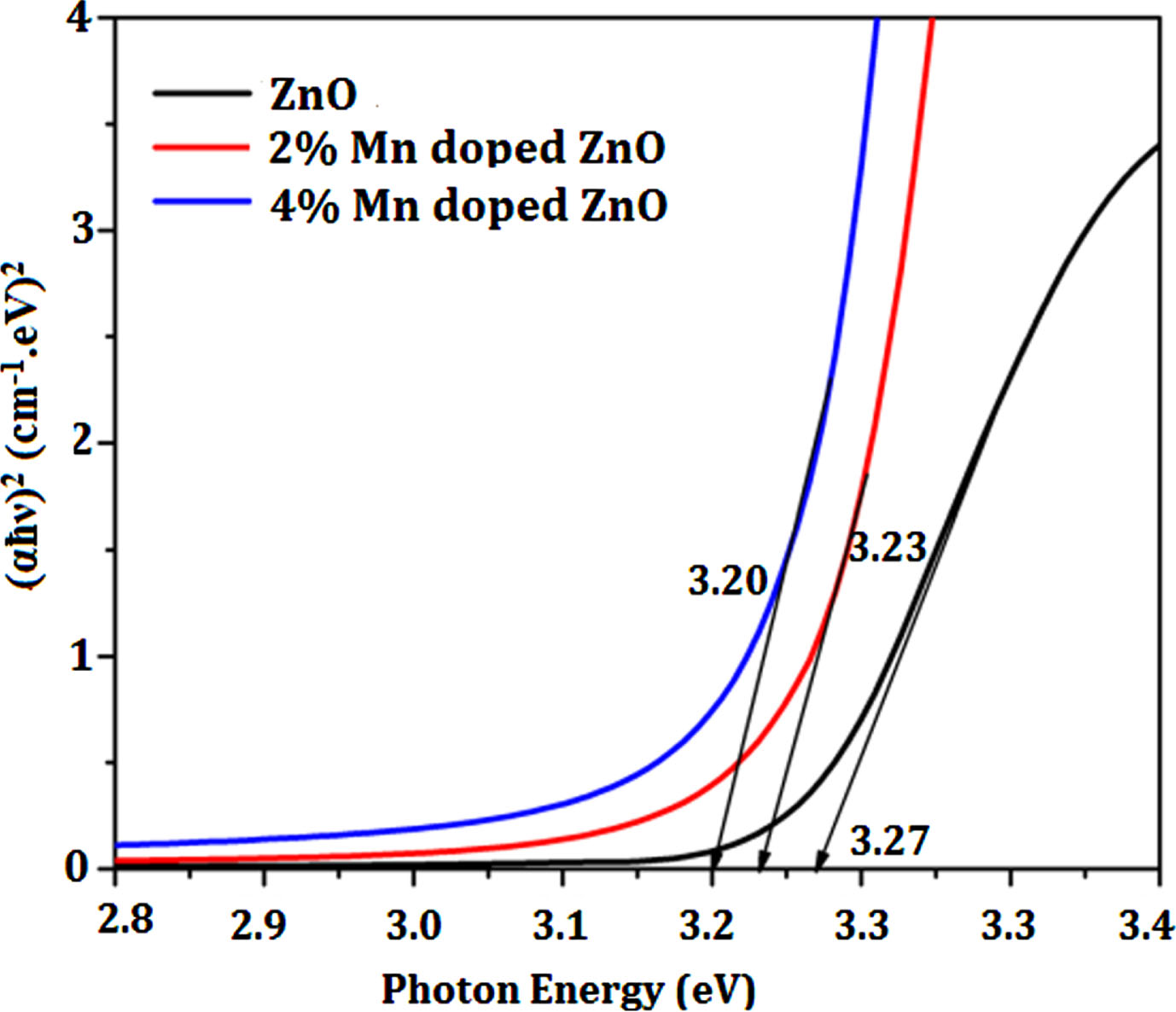
K-M Plot of Un-doped, 0.2% Mn/ZnO and 0.4% Mn/ZnO nanoparticles for band gap determination.
FESEM and EDAX is used to analyze the surface morphology and purity of as synthesized photocatalyst Mn-doped and pristine ZnO. From the SEM images, the variation in morphology in relation to the degree of doping concentration is clearly noticed. The images demonstrate that a significant portion of the particles has taken the form of finely dispersed spherical structures, while a portion exhibits an elongated spherical shape. The images extracted from Fig. 4 illustrate the presence of sizable clusters comprising uniform nanoparticles. Moreover, the doped samples display a higher degree of aggregation compared to the pure ZnO. The average size of pure ZnO nanoparticles measures around 50 nm. However, as the concentration of manganese doping increases, there is a noticeable reduction in the average size. This trend is also corroborated by the XRD measurements [40]. Notably, undoped ZnO exhibits a crystalline structure with a spherical morphology. In contrast, Mn-doped ZnO nanoparticles maintain a crystalline nature but adopt an elongated spherical flake-like structure. Through the examination of the EDX spectra of the Mn-doped ZnO, prominent elements identified encompass Mn, Zn, and O, accompanied by minor trace elements. These findings underscore the successful synthesis of Mn-doped ZnO, with an appropriate percentage of Mn incorporation, achieved using the sol-gel method. Additionally, both the atomic and weight percentages of these elements have been explicitly provided for enhanced clarity [41].

FESEM images of (a) Pure ZnO(b,c) 2% Mn/ZnO,4% Mn/ZnO (d) EDAX spectrum of 4% Mn doped ZnO nanoparticles.
Figure 5 depicts high-resolution transmission electron microscopy (HRTEM) image along with inserted selected area electron diffraction (SAED) patterns. In the case of pure ZnO nanoparticles, they assume a cubic spherical configuration with an irregular size of approximately 20 nm. These particles tend to agglomerate due to indistinct grain boundaries. As the doping concentration surpasses 0.2 and 0.4 M, a transition occurs in the grain size, rendering the image composed of spherical flakes, each measuring approximately 12 nm in width. Mn-doped ZnO nanoparticles exhibit a compact nanostructure, which significantly contributes to their heightened photocatalytic performance. Notably, these particles exhibit diffuse grain and grain boundary features. The insets accompanying each figure demonstrate how the SAED ring patterns mirror the ZnO nanostructure by exhibiting light with d-spacing values that correspond to the ZnO crystal arrangement. The ZnO nanoparticles (NPs) exhibit a structurally uniform nature, characterized by well-oriented lattice fringes with a d-spacing of approximately 0.25 nm. This d-spacing value closely aligns with the standard value associated with the (101) crystallographic plane. The selected area electron diffraction pattern of Mn-doped ZnO displays a combination of randomly scattered diffraction spots and ring patterns. These patterns indicate the presence of both single and poly-crystalline nano crystallites within the sample. The distinctive diffraction spots and rings have been successfully matched and identified using the reference data from the bulk ZnO JCPDS card 36-1451, facilitating the indexing process. The illustrations depict lattice spacing of ZnO and Mn-doped ZnO nanocrystals start out with random orientations from the SAED pattern. [42, 43].

HRTEM images of (a)2% Mn/ZnO (c)4% Mn/ZnO; (b&d) SAED pattern of pure and 4% Mn doped ZnO.
Under visible light exposure for several hours, the MB dye solution demonstrated remarkable stability, showing minimal alteration in concentration even after being illuminated in the absence of photocatalyst. The decolorization behavior of the MB dye was investigated under the influence of natural light using both freshly synthesized pure ZnO and Mn-doped samples at varying concentrations (2% and 4% Mn). During the photocatalytic degradation process of methylene orange (MB), both pristine and doped ZnO catalysts exhibit sufficient efficacy in breaking down MB molecules into smaller fragments. This phenomenon is substantiated by the reduction in absorbance intensities observed in the solutions following the photodegradation procedure. The undoped ZnO sample exhibited noteworthy photocatalytic performance, effectively decolorizing 63% of the MB dye solution within 120 minutes. In contrast, the presence of 2.0 wt.% Mn resulted in a degradation of 74% of the MB dye within the same time frame. Notably, the sample with 4.0 wt.% Mn showcased a significant enhancement in photocatalytic activity when compared to the pristine ZnO and 2% Mn-doped samples. This sample achieved an 86% degradation of the dye over the same time duration, showcasing considerable improvement. The kinetics data for the photocatalytic reaction using the prepared samples and Methylene Blue (MB) dyes are illustrated in Fig. 6. To analyze the reaction kinetics governing MB degradation, the experimental data were fitted to a first-order model expressed by the equation ln (Co/C) = kt. Here, “C” and “Co” represent the concentrations of Methylene Blue at final and initial times respectively, while “k” stands for the reaction rate constant. Notably, among all the photocatalysts examined, the 4% Mn doped ZnO sample exhibited the highest degradation rate. To assess the impact of catalyst dosage, initial concentration, and initial pH on the efficacy of removal, a series of methylene blue (MB) degradation experiments were conducted. Various weight concentrations of 4% Mn doped ZnO catalyst (0.01 g, 0.02 g, and 0.05 g) were employed, along with different concentrations of MB dye (10 mg/L, 20 mg/L, and 40 mg/L), all within a pH range of 5 to 11. The findings of the experiments revealed a correlation between removal efficiency and increasing catalyst weight. Specifically, as the catalyst weight increased, removal efficiency also demonstrated an upward trend. Notably, after a reaction time of 120 minutes, the highest percentage of MB degradation was achieved using 0.05 g of the catalyst. This observation underscores the notable removal efficiency associated with higher catalyst quantities. This phenomenon can be attributed to the fact that an increased number of material particles results in a corresponding rise in the number of photons and dye molecules absorbed. Consequently, the larger surface area of nanoparticles provides more active sites for the generation of radicals during the reaction. These results illuminate the intricate interplay between catalyst dosage and degradation efficiency, shedding light on the importance of surface area and active sites in catalytic processes.
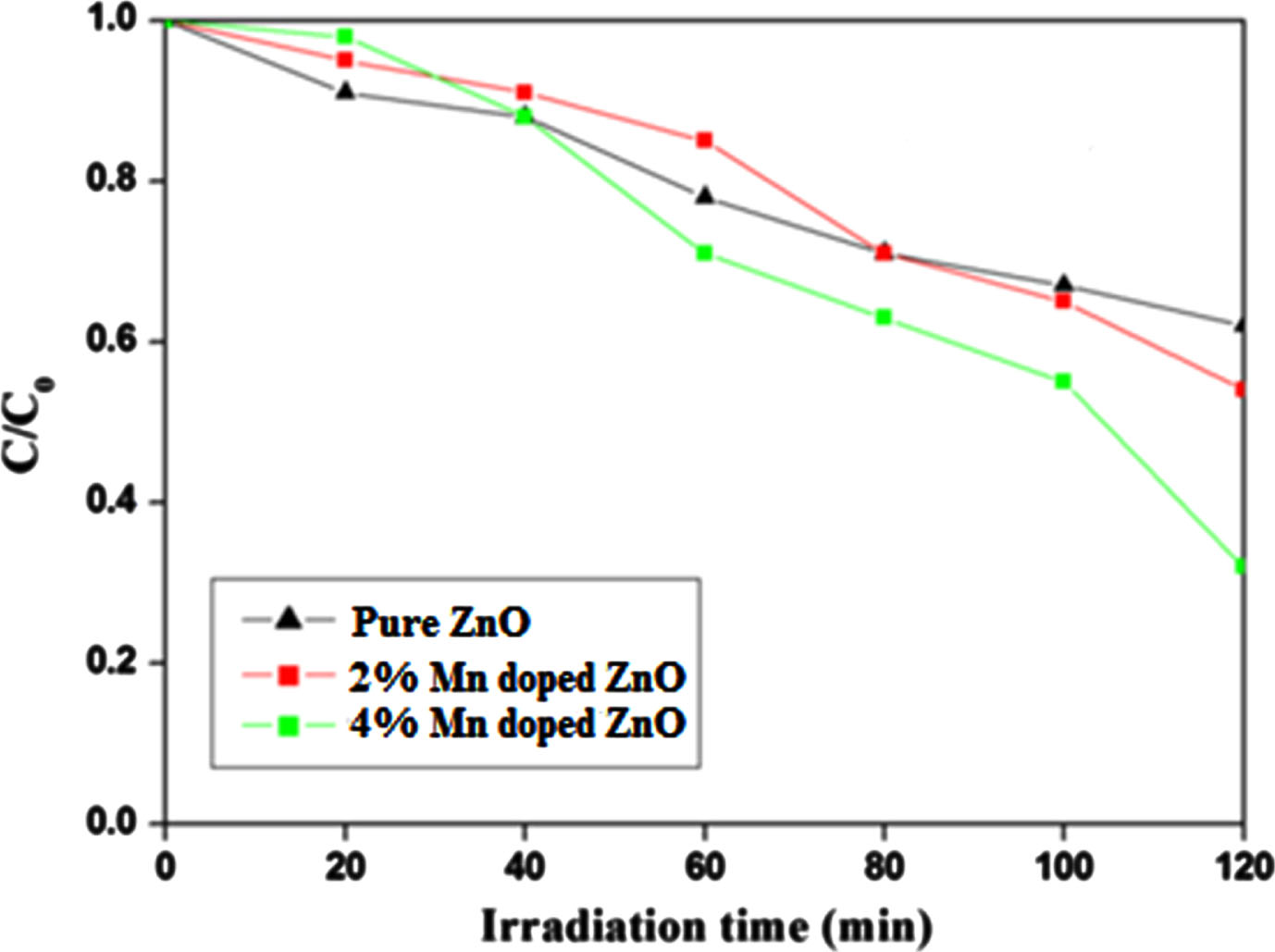
Photo catalytic degradation % of Methylene blue by un-doped ZnO and Mn doped ZnO.
The photocatalysts undergo repeated collection and usage, including the Mn (4 wt%)-doped ZnO samples, which are also assessed for their suitability for multiple applications as photocatalysts. As depicted in Fig. 7, the efficiency of the Mn-ZnO samples in photocatalysis experiences a slight reduction after seven cycles of Methylene Blue degradation. This decline is attributed to losses during washing and incomplete regeneration. The Mn-ZnO samples emerge as a strong candidate for photocatalytic purposes, given their stability and significant potential in water treatment. Notably, the photocatalytic performance is profoundly impacted by various factors like band gap energy, particle size, shape, and modifications to the catalyst’s surface. Moreover, in order to confirm the stability of Mn doped ZnO after the photocatalytic reaction, XRD spectra was recorded for recovered catalyst and the results shows that the crystalline structure of the catalyst remains largely unchanged after the photocatalysis, indicating that the catalyst retains its stability under the experimental conditions employed. A potential mechanism is put forth in Fig. 8 to elucidate the enhanced photocatalytic activity of ZnO subsequent to the introduction of Mn doping, despite the tendency of Zn to create a distinct ZnO phase. On a pristine ZnO sample, the photocatalytic process occurs via the photo-excitation of electrons transitioning from the valence band (VB) to the conduction band (CB). As a result, photogenerated charges in the form of electron-hole pairs are generated. Under the influence of UV photons, ZnO crystals become stimulated, resulting in the generation of electron-hole pairs. Ordinarily, these photo-induced electron-hole pairs exhibit a tendency to recombine; nevertheless, a subset of these pairs manage to reach the surface. Here, they engage in interactions with oxygen and water, leading to the creation of liberated superoxide and hydroxy radicals. The generated radicals exhibit strong reactivity, enabling them to engage in decolorization by attacking dye molecules. Rapid recombination, however, has the potential to diminish the effectiveness of pristine ZnO. Hence, the anticipation is for notable photocatalytic activity as long as electron-hole separation persists. Incorporating Mn into the ZnO lattice enhances the concentration of dye molecules on the catalyst’s surface, consequently amplifying the quantity of reactions occurring per unit area at the catalyst’s surface. A prior study [44] examined how the photocatalytic activity of ZnO is influenced by its crystallite size. The findings indicated a consistent correlation: a reduction in the size of ZnO consistently corresponds to heightened activity, attributed to surface area effects. In simpler terms, diminishing the dimensions of ZnO leads to an augmentation in the overall surface area of the specimen, thereby elevating its photocatalytic activity by virtue of the substantial increase in accessible active sites. A comparable mechanism was previously suggested by Siva Karthik et al. [45] and Madhan et al. [46]. Finally, upon conducting a comparative analysis with previously documented investigations focusing on transition element-doped ZnO photocatalysts, it becomes evident that our Mn-doped ZnO photocatalyst excels in terms of photocatalytic activity under UV light. This heightened efficacy can be attributed to the minimized recombination rate between photogenerated electron-hole pairs. This phenomenon underscores the paramount importance of achieving an optimal crystalline structure as a key factor in augmenting photocatalytic performance. Furthermore, these findings strongly suggest that Mn doping offers a promising and effective avenue for elevating the efficacy of photocatalytic treatment. Incorporating this insight into our manuscript serves to accentuate the distinct strengths of our material and its potential significance in diverse applications.
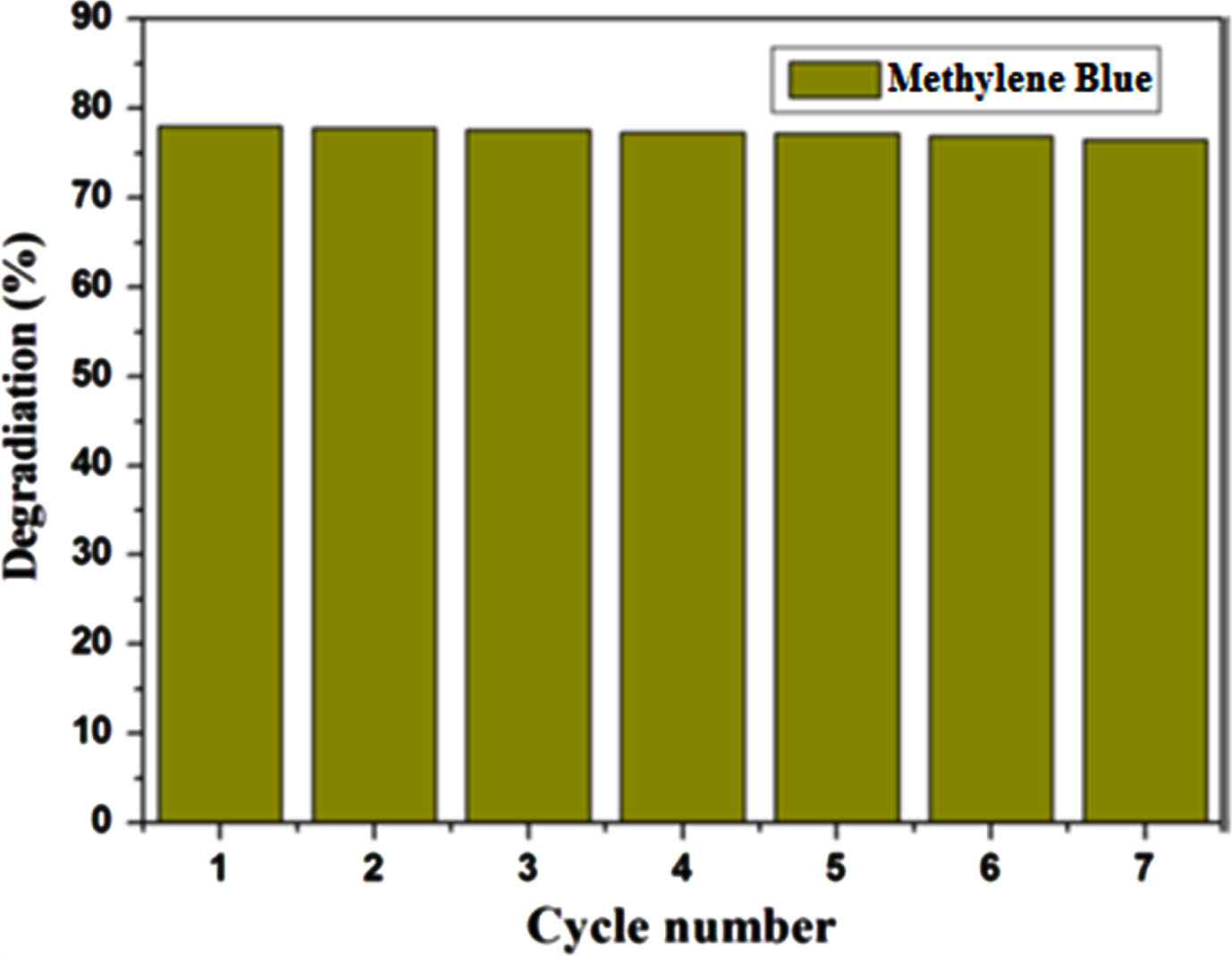
Reusability Test of 4 % Mn doped ZnO.
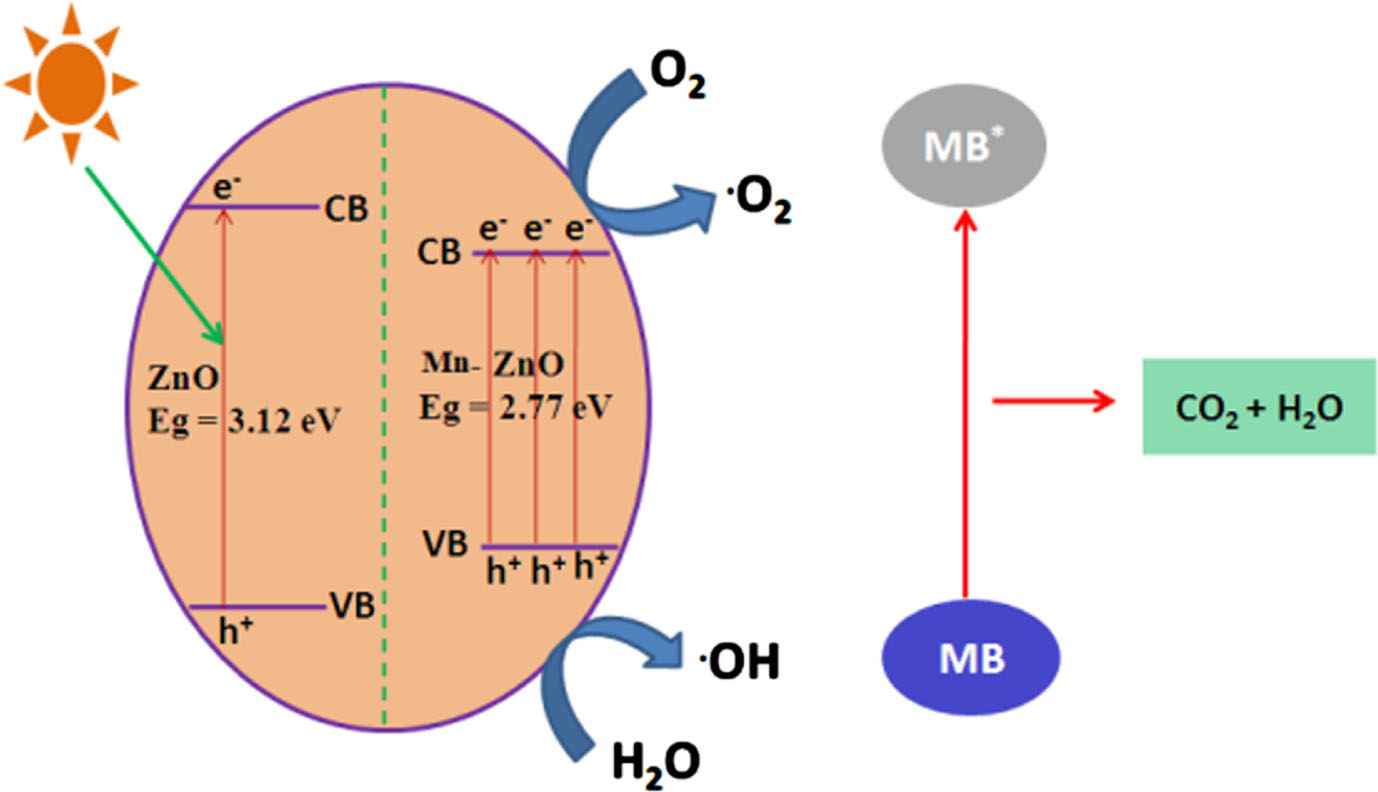
Mechanism of photocatalytic degradation.
Comparing photocatalytic degradation performances of ZnO based photo catalysts utilized in degradation of pollutants in aqueous solution
The pure and Mn doped ZnO nanoparticles have been successfully prepared by sol-gel technique. Based on the powder XRD investigations, the samples exhibited a hexagonal wurtzite structure, and there were no signs of impurity phases being detected. Through the analysis of UV-Vis spectra, a noticeable red shift and reduction in the bandgap energy were observed, transitioning from 3.27 to 3.20 eV. UV investigations also unveiled that Mn-doped ZnO exhibited photo absorption that extended into the visible light range. The introduction of Mn dopant was found to elevate the impurity levels between ZnO’s conduction and valence bands, leading to a reduced band gap and heightened absorption of visible light. The TEM images consistently revealed a spherical morphology for all nanoparticles, with an average size of around 20 nm. The photocatalytic efficiency of pure and Mn-doped ZnO catalysts was evaluated by exposing MB to visible region The best photocatalytic activity is displayed by Mn-doped catalysts when compared to un-doped ZnO. Notably, the sample containing a 4 wt.% Mn dopant demonstrated outstanding efficacy, achieving an 86% degradation of MB within 120 minutes at pH 6. The recycling assessment demonstrated a stable degradation rate for the Mn-ZnO catalyst throughout the first four cycles, without any noticeable decline in its effectiveness. This finding implies that Mn-doped ZnO catalysts have potential utility in the realms of wastewater treatment and pollution alleviation, offering significant efficacy as photocatalysts.
Footnotes
Acknowledgments
We are grateful for the lab facilities offered by the management of Panimalar Engineering College, Chennai-600123, India.
Funding
No funding from any source
Conflict of interest
The author declares that the work reported in this publication was not influenced by any known conflicting financial interests or personal connections.
