Abstract
Formazans are well-known complexing agents and form highly stable complexes with verity of metals that are useful for number of pharmaceutical applications. The activity profile of formazans and their complexes are huge that include antibacterial, antifungal, antioxidant, anti-fertility, anti-tubercular, antiviral, anti-inflammatory, anticancer, anti-HIV, and anti-proliferative are among the biological activities covered by the formazans. The structures, synthesis, reactions, and spectral properties of formazans have been studied to highlight their potential applications in a variety of bioactive phenomena and analytical applications. The biological potential of the compounds will be greatly enhanced when substitution is carried out at R and R2. We observed that electron withdrawing groups at para position (if R, R1 & R2 = phenyl ring) are the best substitutions to achieve potent pharmacological activities.
Introduction
Formazan belongs to a significant class of organic molecules with two distinct functional groups on the same compound azo group(-N = N-) directly linked to imine group(-C = N-)[1]. Formazan compounds are highly colored due toΠ-Π* electron transfiguration in conjugated double bonds present among azo and hydrazo moieties [2]. Von Pechmann [3] and Bamberger [4] independently described their structure for the first time in 1892.
The chemical and biological activities of formazans are conflicted by tautomerism, particularly their chelating and color properties. The tautomerism in formazans was initially described by Pechmann and Runge [3] in 1894, although their findings were unsatisfactory. Hunter and Robert [5] (1941) demonstrated convincingly that the particular in each duo were identical for multiple pairs of formazans, despite previously being classified as tautomeric (
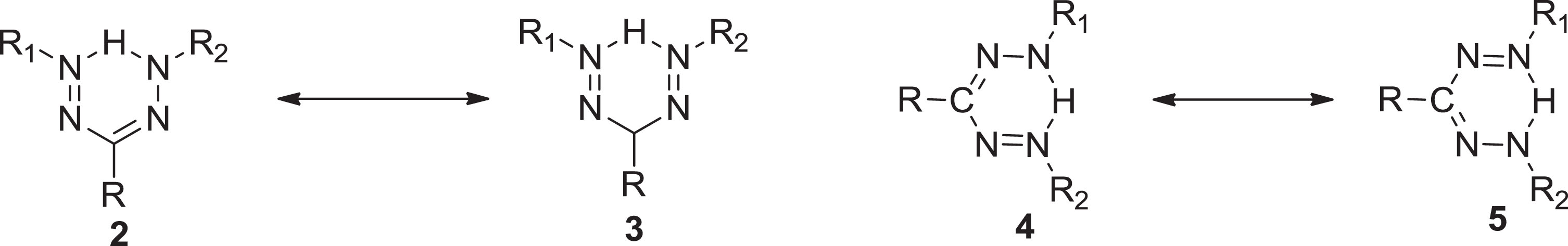
a) Tautomerism in formazan (
A formazan molecule acknowledged the persistence of four different configurations which were encountered by geometrical stereoisomerism around the two double linkages (C = N, syn-anti and N = N, cis-trans). The nomenclature syn/anti is used to describe the orientation around the (C = N) bond, while cis/trans denotation is used to represent the (N = N) bond. Ultimately, an exchange of the (C-N) interactions stated as s-cis or s-trans [6]. The alternatives to minimizing tautomerism for instant are the following (Scheme 2).
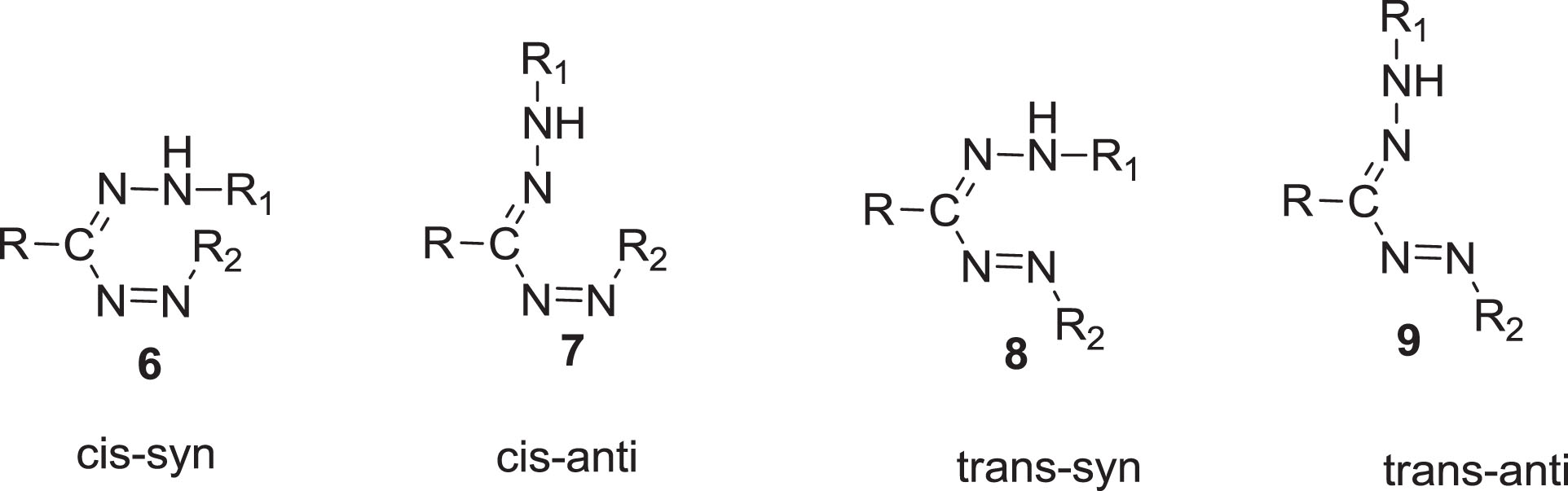
Geometrical forms
Formazans containing triphenyl rings are typically very soluble in organic solvents; their dissolution rate appears to be minimal in water because of the pigments of the solvent. The physical and molecular properties of these chromogenic compounds are heavily influenced by their terminal groups [7]. When formazans are oxidized, they produce tetrazolium salt. The formazan pair is a redox mechanism that functions as a proton receiver or oxidant in a range of natural sciences, including pharmacy, botany, toxicology, and immunology, although exclusively in biochemistry and cytochemistry [8, 9].
Their implementation in the chemistry field, due to its global scopes in pharmaceutical, medical, industrial and, chemical fields and synthesis of heterocyclic compounds has influenced the notice of many groups of the researcher [10]. Anti-fertility and anti-malaria activities have been shown by several formazans [11]. Tetrazolium salts are categorized as vitality promoters, and formazans have been discovered to have essential medical applications [12]. Owing their remarkable pharmacological and therapeutic efficacy as an anti-fertility [13], antibacterial and antifungal [14], antiviral [15], anti-microbial [16], anti-HIV and anticancer [17], anti-inflammatory agents [18], anti-tubercular agents [19], formazans and related functionalized molecular structure have received much attention. There are few publications on formazans and their derivatives for biomedical practices, with the majority of them focusing on coordination complexes of formazan [20]. Also, their derivatives and conjugates have recently been shown to possess promising anticancer action in cancer cells using photodynamic treatment [21]. The formazans, on the other hand, are more practical in determining the efficacy of anti-cancer medications [22] to determine tumor cell activity [23] and sperm viability. For the detection of paracetamol, a formazan derivative was used as an electrochemical sensor to modify a pencil graphite electrode [24].
In histochemistry, formazans are thought to be made from tetrazolium salts [25], but this is not the case; in reality, tetrazolium salts are made chemically by oxidation of the matching formazan. Friese [6] (1875) was the first to synthesize a formazan; however, he was unaware of the structure because this category of the compound was previously unknown. Von Pechmann [3] and Bamberger [4] (1892), independently identified the structure of the related compounds, and proposed the name “formazyl.” Von Pechmann and Runge [26] (1894) synthesized triphenyl tetrazolium chloride after studying the oxidation products of the formazan compounds. Nineham [5] (1955) listed 414 formazans that had been described and the current figures are probably more than 1000.
Motivation and purpose of study
Formazan derivatives are well-known complexing agents so lots of work have been done in this area which will be summarized by the researchers from time to time. Nineham [5] was the first that summarized the chemistry of formazans and tetrazolium salts in the year 1955. Further, in 1976, Altman [27] described the properties of formazans and illustrated how the tetrazolium salt-formazan reaction has been exploited to serve an extremely wide variety of functions. Furthermore, in years 1991, 2006, and 2019 all the information about the design and chemistry of formazans were compiled by Seidler [28], Sigeikin et al. [29] and Lipunova et al. [30]. However, in the year 2016 Shawali and Samy [31] summarized the pharmacological activities of formazans in well-decorated manner. Recently, in the year 2020, Gilroy and Otten [32] summed up details regarding formazan metal complexes in a very impressive way. After evaluating all the studies, the authors observed that there are no epitomized reports which cover the details of formazans and their metal complexes from the last three years. Therefore, the authors summarize the chemistry and biological potentials of formazans and their complexes from the year 2015 to till date.
General methods for the formation of formazans
Three synthetic approaches have been investigated for the synthesis of formazan [35]. The first method [35] was reported by Tezcan and Ozbek [36] which is based on the interaction of aryl diazonium salts with aldehyde and phenyl hydrazones in basic media (Scheme 3). On the other hand, the second, introduced by Katritzky et al. [5] was established on integration of aryl diazonium ionic species with reactive methylene groups (Scheme 4). Furthermore, Schiff bases are the fundamental units for the production of pharmaceutically relevant molecules for formazan analogues. Abu Elenien [37] claimed that the formazans were prepared by oxidizing hydrazidines with the appropriate hydrazine substituents (Scheme 5). The first method is the simplest and most practical; however, atoms of carbon substituents are confined to non-aromatic formations, hydrophobicity of the forum, reagent proportion, and set of substituents are all important in this reaction [38].
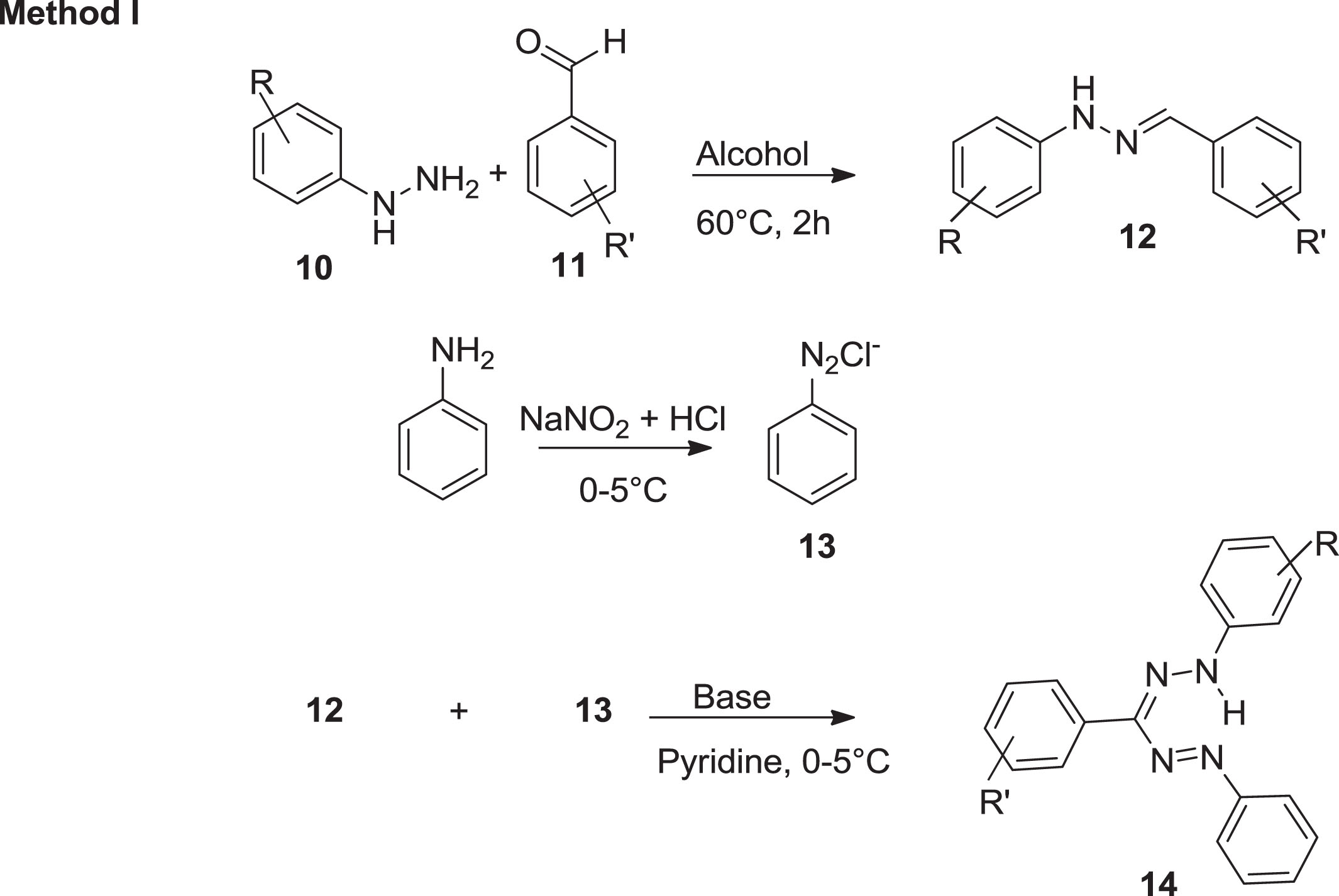
Interaction of aryl diazonium salts with aldehyde and phenyl hydrazones in basic media.
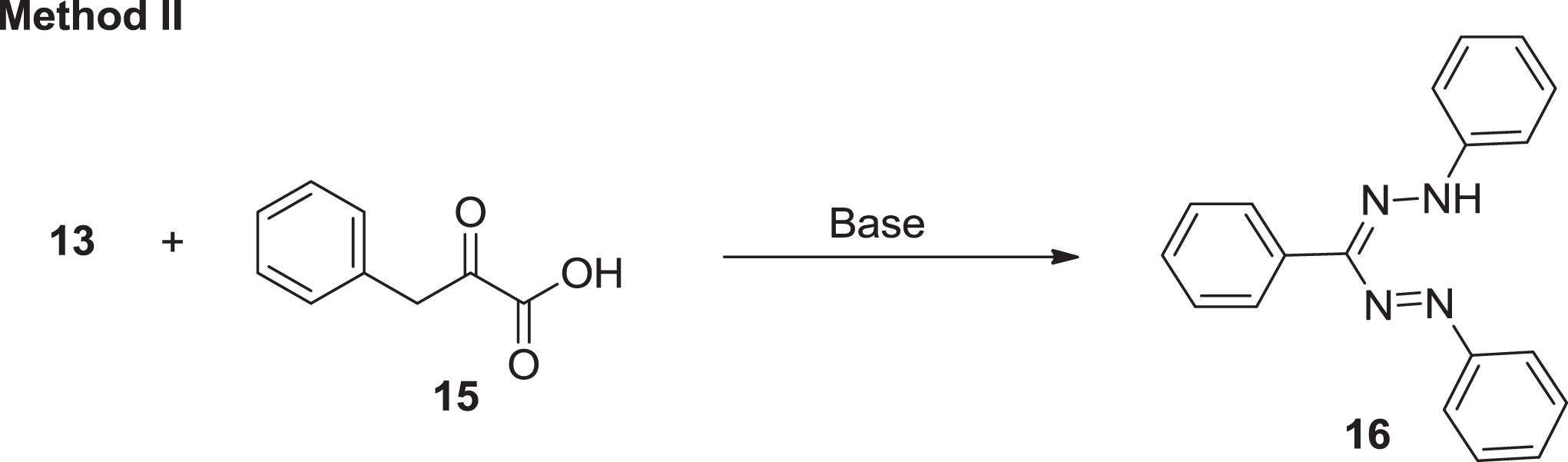
Coupling of aryl diazonium ions with active methylene groups.
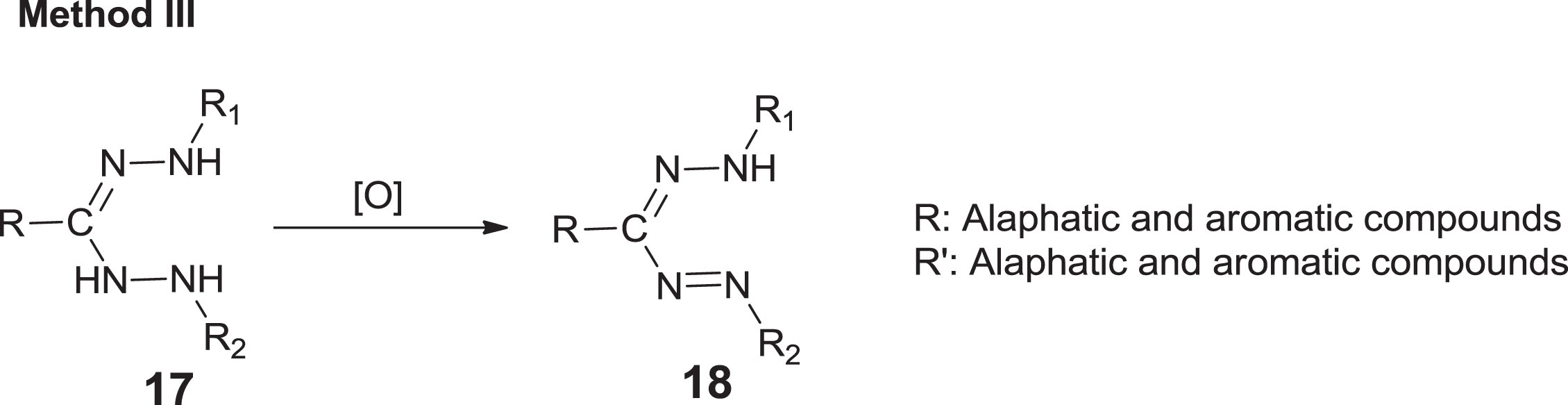
Oxidation of hydrazidines with the appropriate hydrazine derivatives.
Synthesis and biological activities of formazans
Formazans are employed in environmental monitoring as chemical and electrochemical sensing devices, as well as test equipment for rapid analysis, and as organic synthesis intermediates. A majorly effective approach for the bio-synthesis of formazans that adheres to conventional green chemistry standards was recently introduced against this backdrop [39, 40].
Lipunova et al. [30] defined the following scheme by a solvent-free mechanism of diazotization of aromatic amines 1
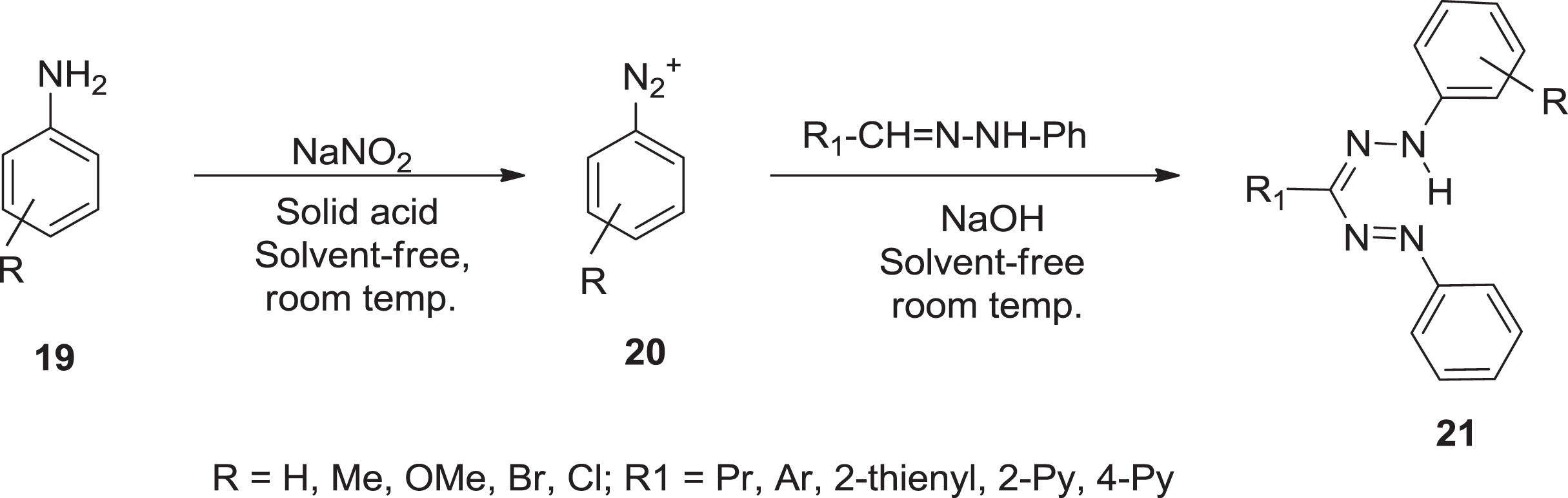
Synthesis of formazan from substituted amines via NaOH.
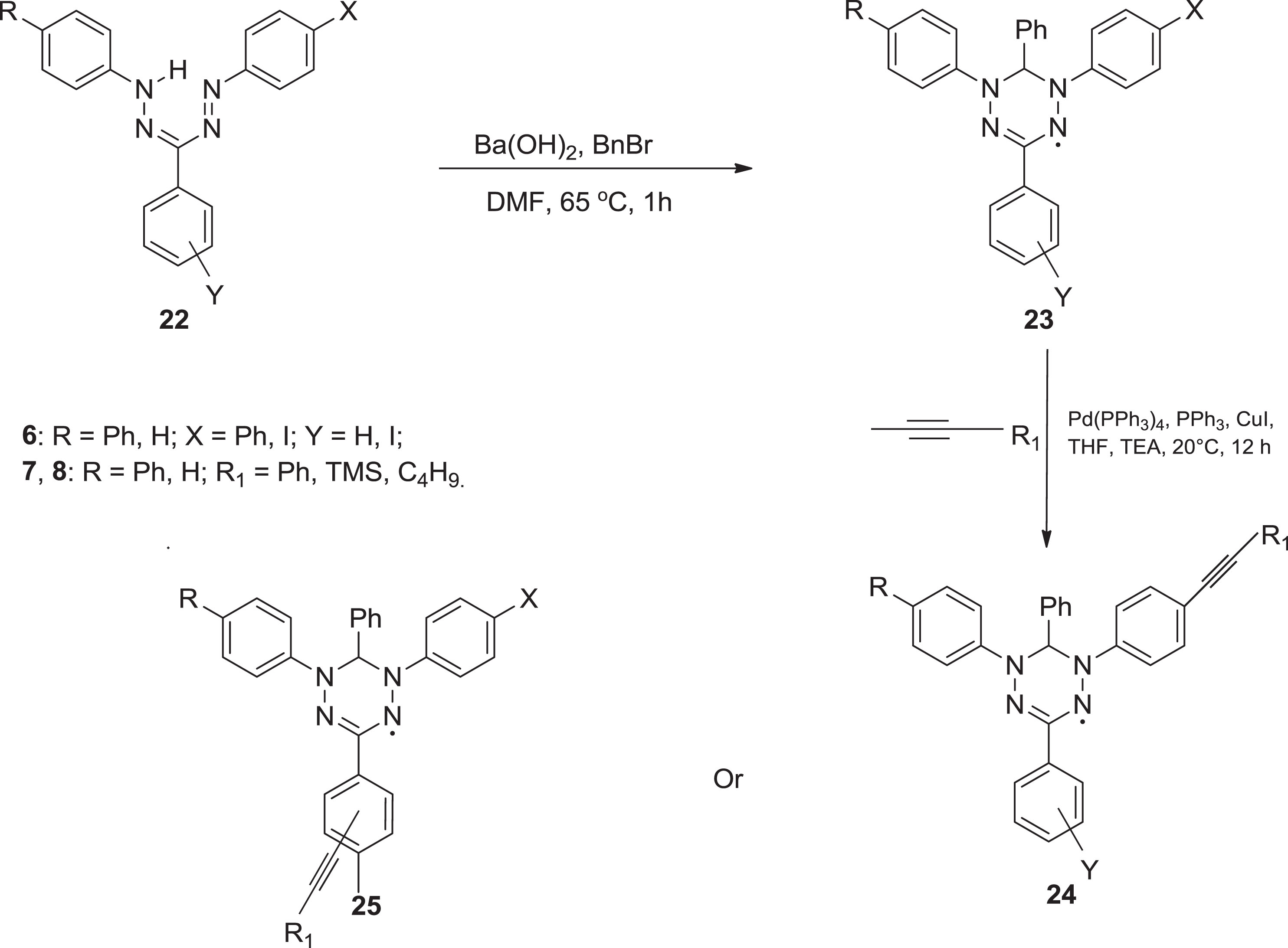
Synthesis of formazan derivatives via THF & TEA.
According to the literature, several formazans have been identified as having a wide range of biological actions and therapeutical applications [41] including antimicrobial [42, 43], antiviral [15, 44], anticancer [18], and anti-inflammatory [45]. The formazan derivative
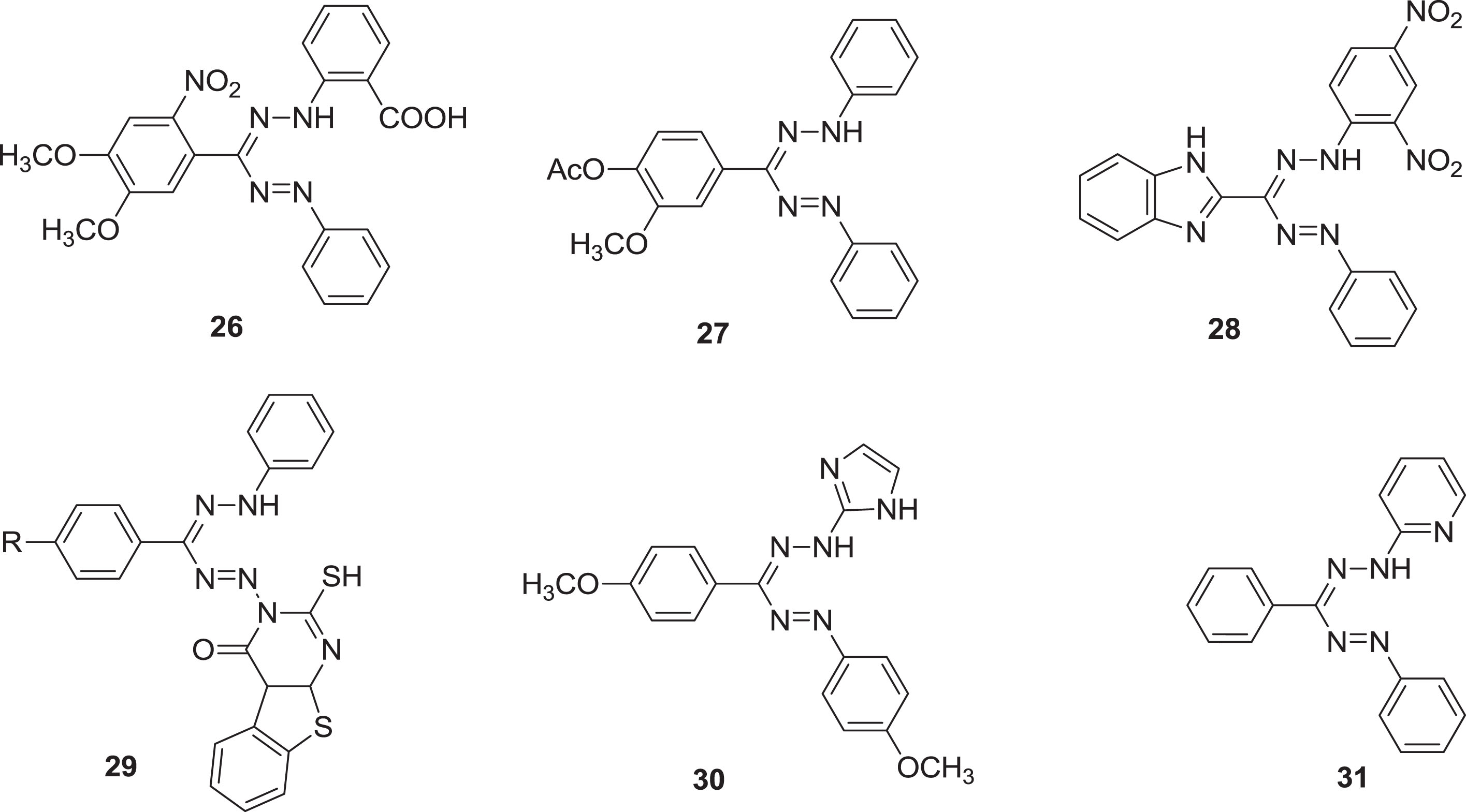
Structure of some antibacterial and antifungal formazan compounds.
After that Mahmoud et al. [14] successfully prepared herein a fresh series of nano-sized formazan analogues to investigate their potency as antifungal and antibacterial promoters in vitro. Dithiazone
The reaction of dithiazone with different hydrazonyl chlorides in the presence of EtOH.
Target compounds with an electron withdrawing groups were assessed in vitro for antibacterial and antifungal potential against two gram-positive bacteria, Bacillus subtilis, and Staphylococcus aureus; two gram-negative bacterial strains, Proteus vulgaris and Escherichia coli along with two fungal strains, Aspergillus flavus and Candida albicans using ketoconazole and gentamycin as a source of antibacterial and antifungal stimulants, respectively [49].
Against gram-positive bacteria, the majority of compounds showed moderate to better antibacterial activity. The acetyl derivatives had the best activity against S. aureus and B. subtilis, with inhibition zone values higher than gentamycin. In addition, most compounds appeared to have only minor antibacterial activity against E. coli gram-negative bacteria. The majority of the formazan analogues tested showed promising antifungal activity against the tested fungal strains [50].
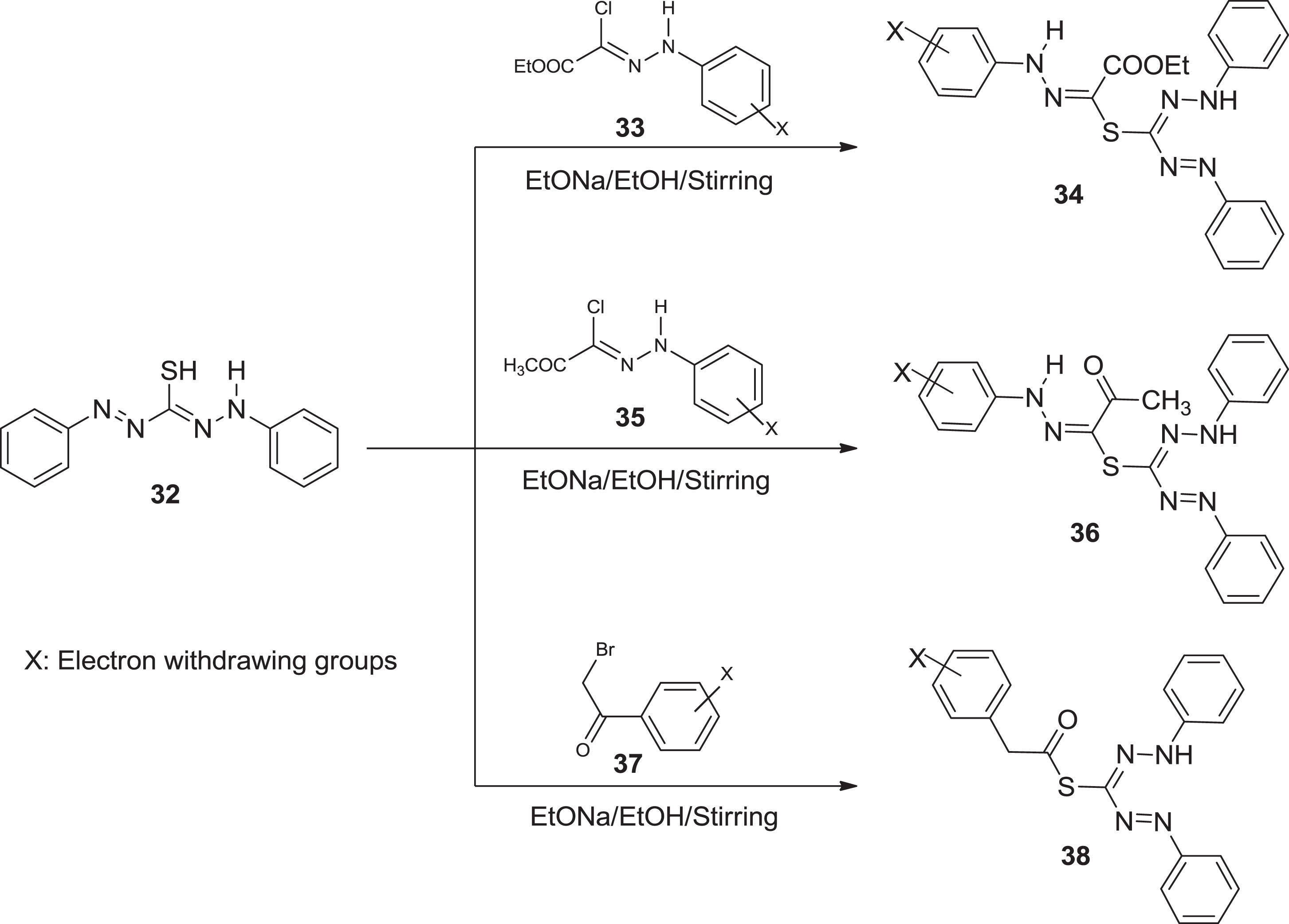
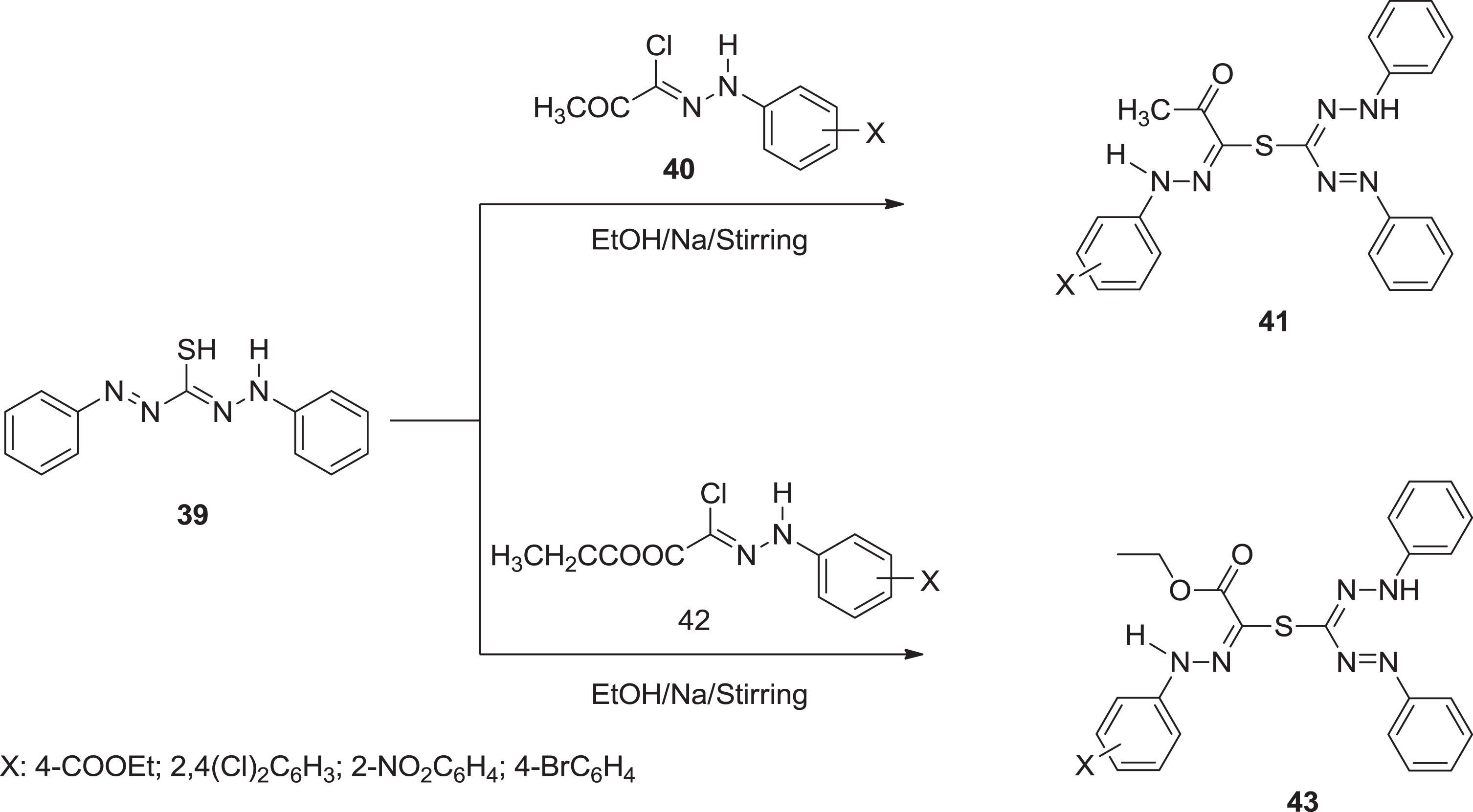
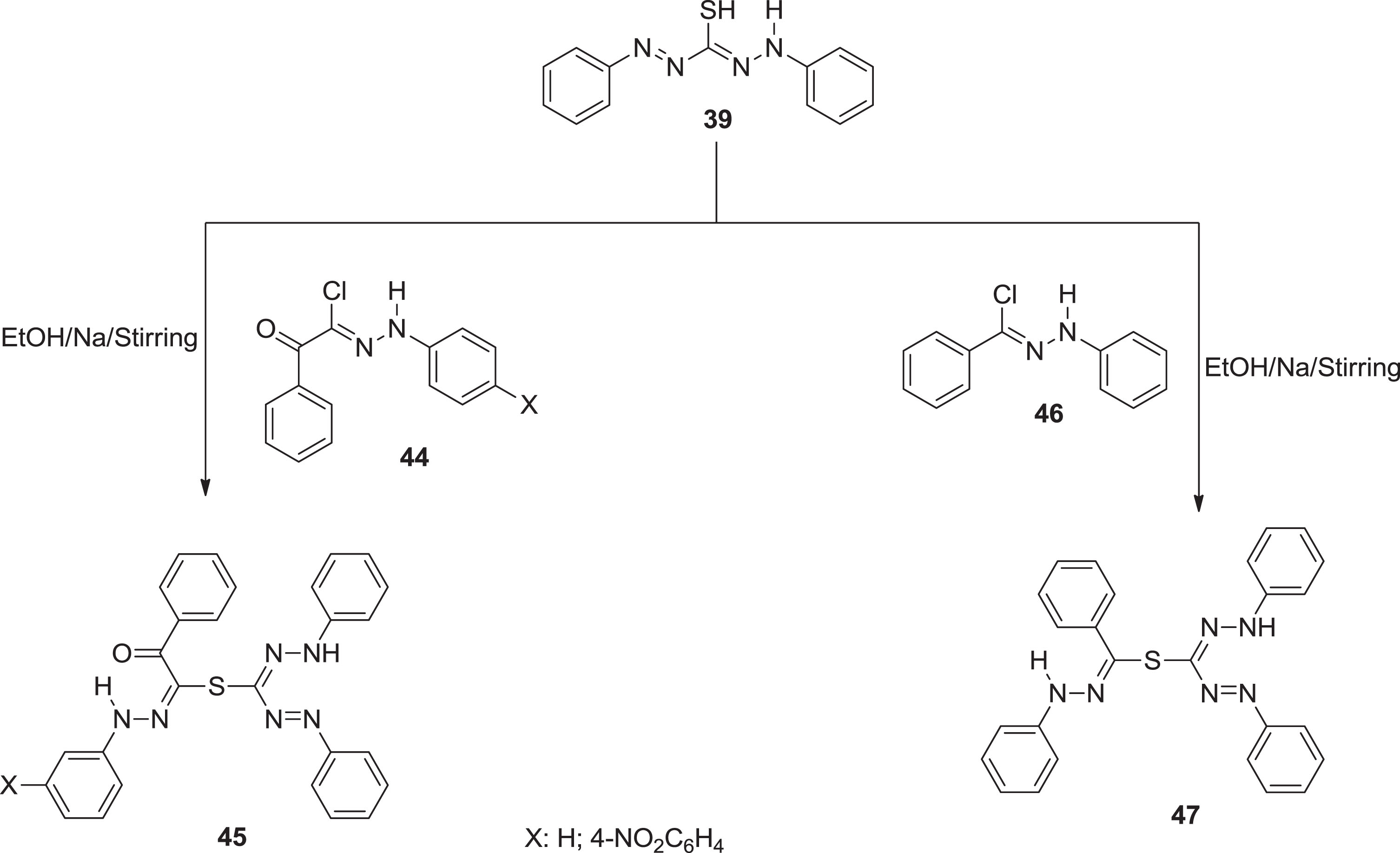
In continuation of our previous work of synthesis of nanosized formazan and investigation of their activity against COVD-19, as well as the promising data of these derivatives. Bayazeed et al. [51] interested in synthesizing a new series as an antimicrobial agent. All formazan analogous
Synthesis of formazan derivatives.
Synthesis of formazan analogues.
All new formazan compounds were synthesized and tested for antimicrobial activity and MIC values against three gram-negative and three gram-positive bacterial strains, as well as three fungal species. All compounds showed promising antimicrobial screening results, with MIC values ranging from 1.95 to 62.5 g/mL. Compounds 45 and 47 were effective against gram-positive bacteria S. faecalis. Remarkable inhibitory activity was brought by compound 45 against gram-positive bacteria (MRSA). Furthermore, compounds 43b exhibited strong antibacterial potential towards E. coli. Molecules 45a & 47, on the other hand, were extremely effective against K. pneumonia. Compound 43b exhibited antifungal activity against Candid albicans. Although comparable to the reference standard ketoconazole, compound 45 inhibited the three fungal strains significantly. Furthermore, compound 41b outperformed A. niger, C. albicans, and Rhizopus species. Except for compounds 41b, 43a, and 47, all compounds demonstrated remarkable antifungal activity against Rhizopus species.
In continuation, Mehmet et al. [52] synthesized the (o, m, and p-CNF) formazans via coupling reaction.
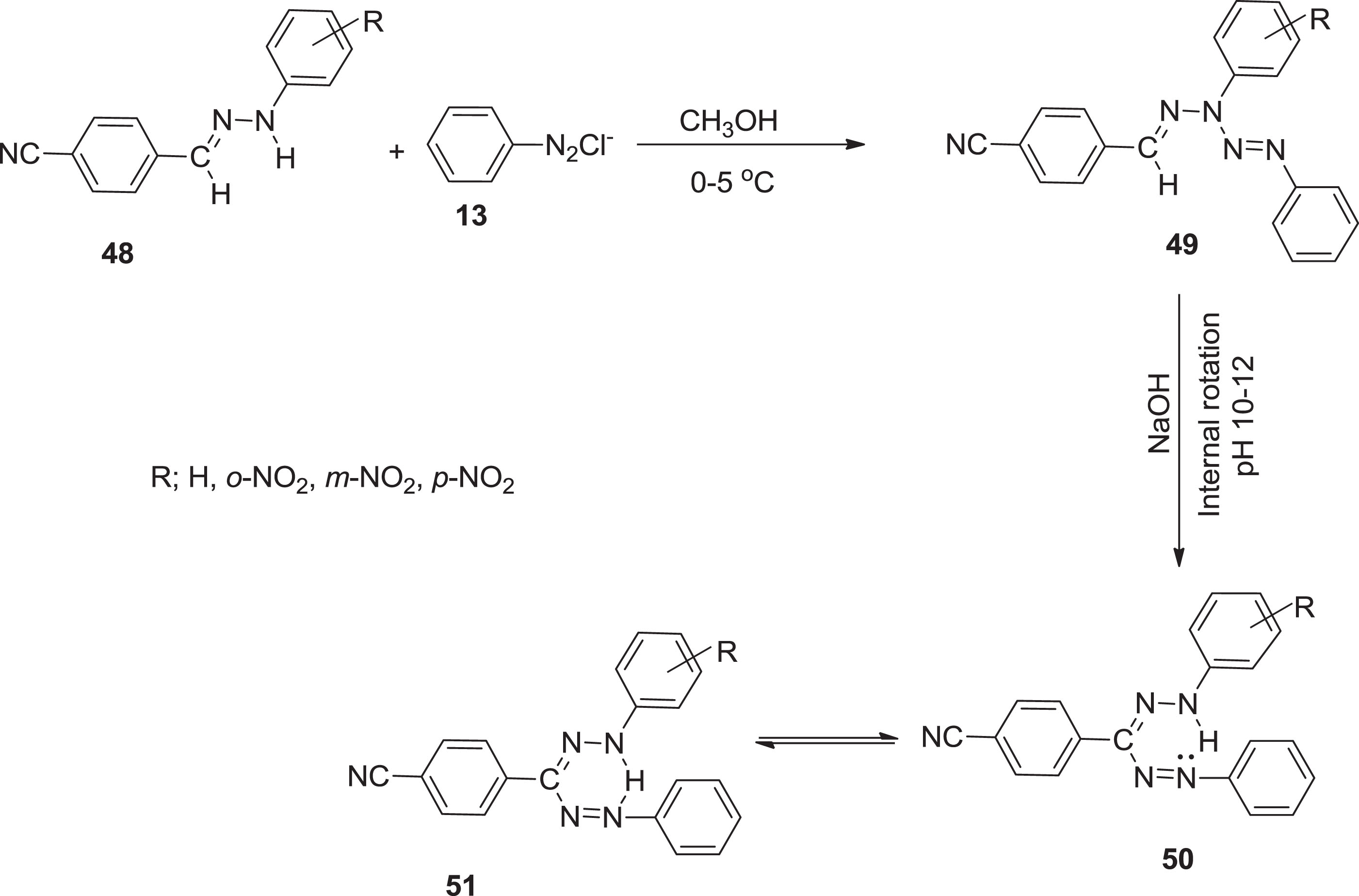
Synthetic route of formation of (o, m, p-CNF).
Fresh formazan signaling pathways with schiff bases nitro units as nitrogen-based hydro donors are designed and synthesized in this regard depending on the features of either the azo or hydrazo groups in the formazan schema [54]. Further, Turkoglu [7] synthesized fresh formazans
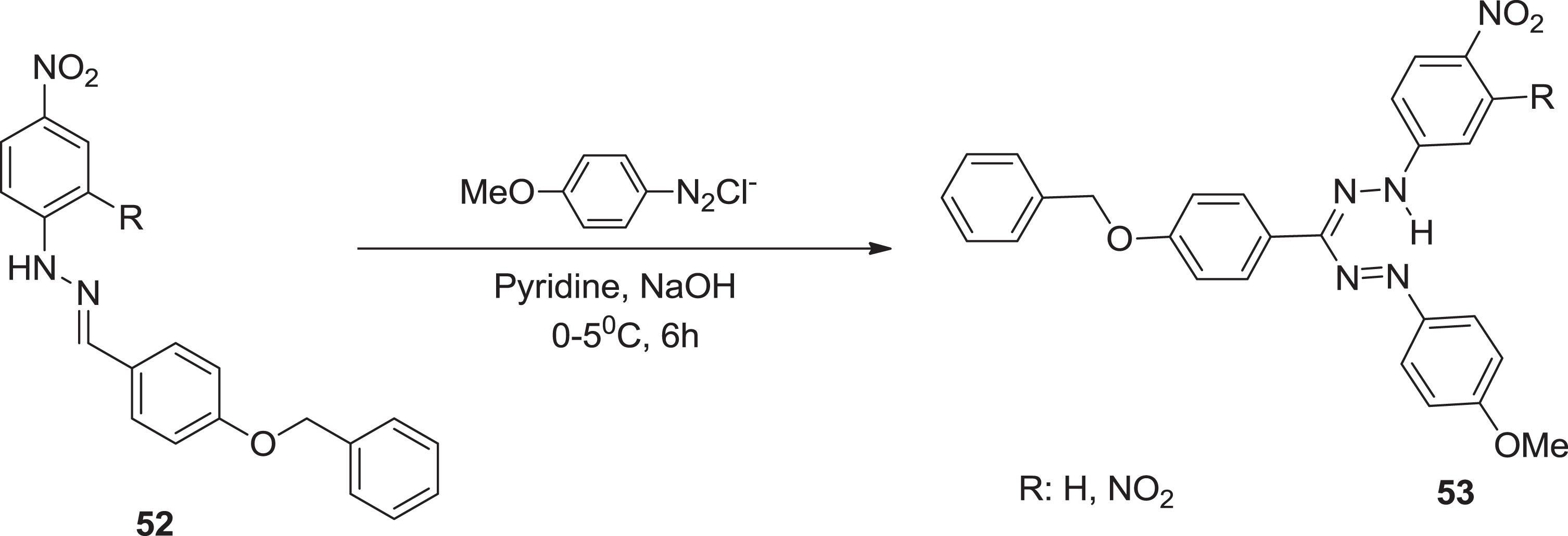
Synthesis of formazan sensors i.e. FNB and FDNB via coupling reaction.
Also, the diazyl formazans
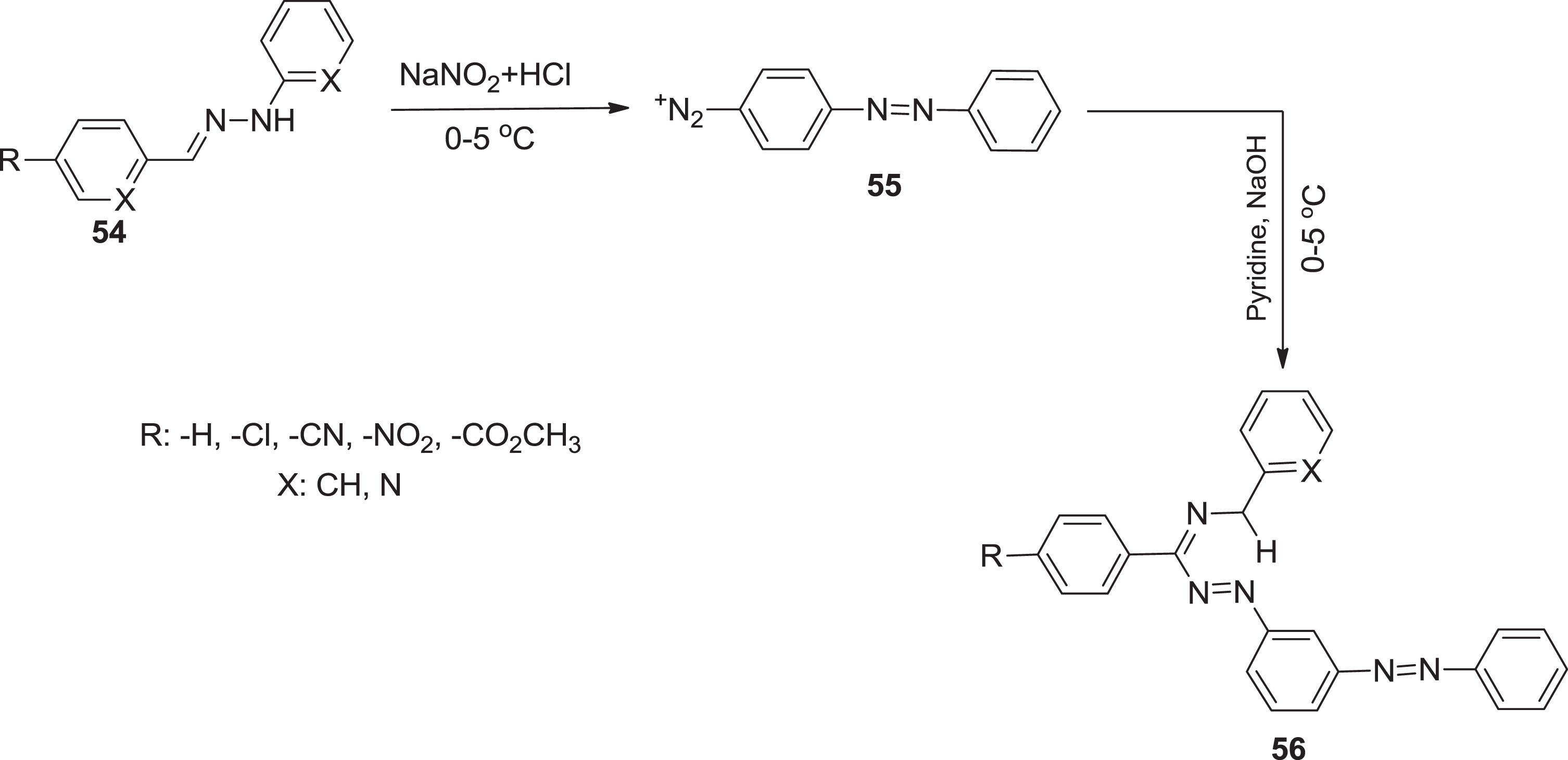
Synthesis of diazyl formazans.
Here, Mohammed and Dahham [56] synthesized formazan derivatives
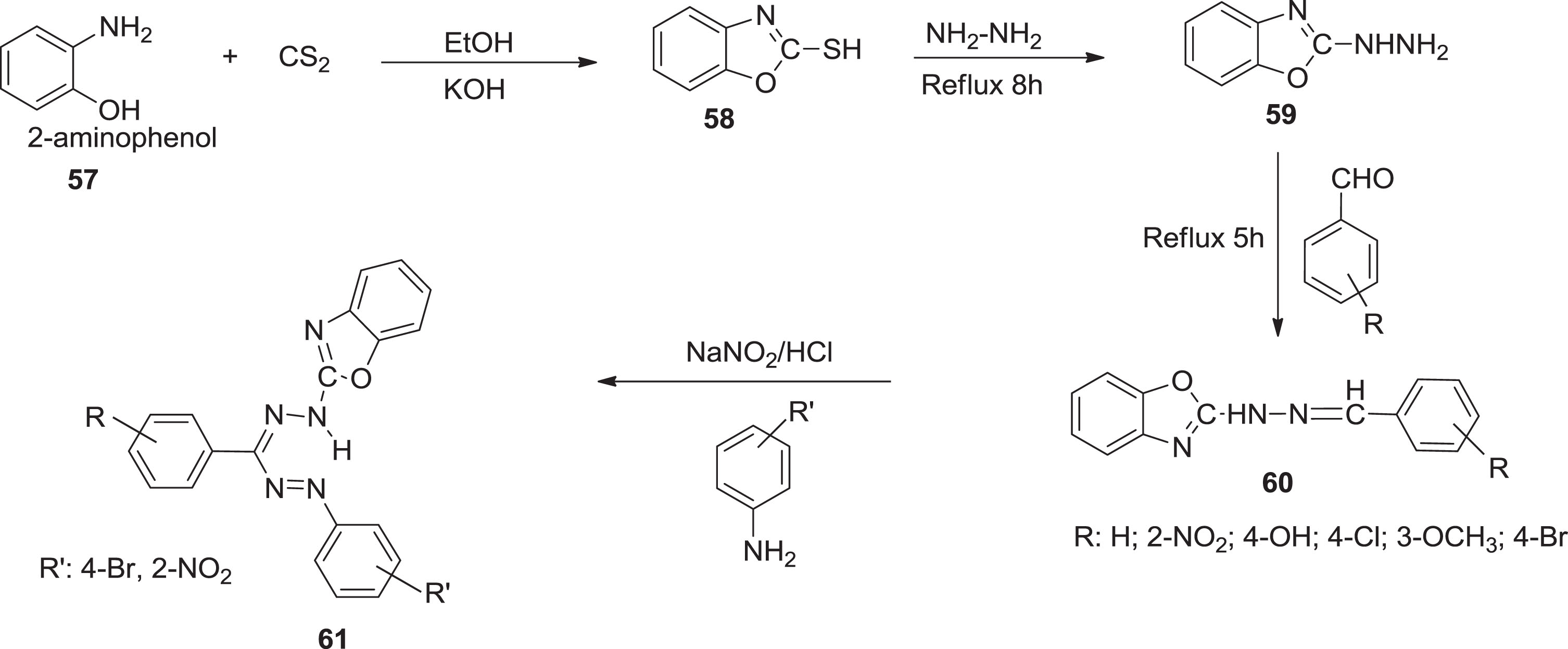
Pathways for synthesized formazans.
Furthermore, a sequence of new heterocyclic comprising formazan counterparts
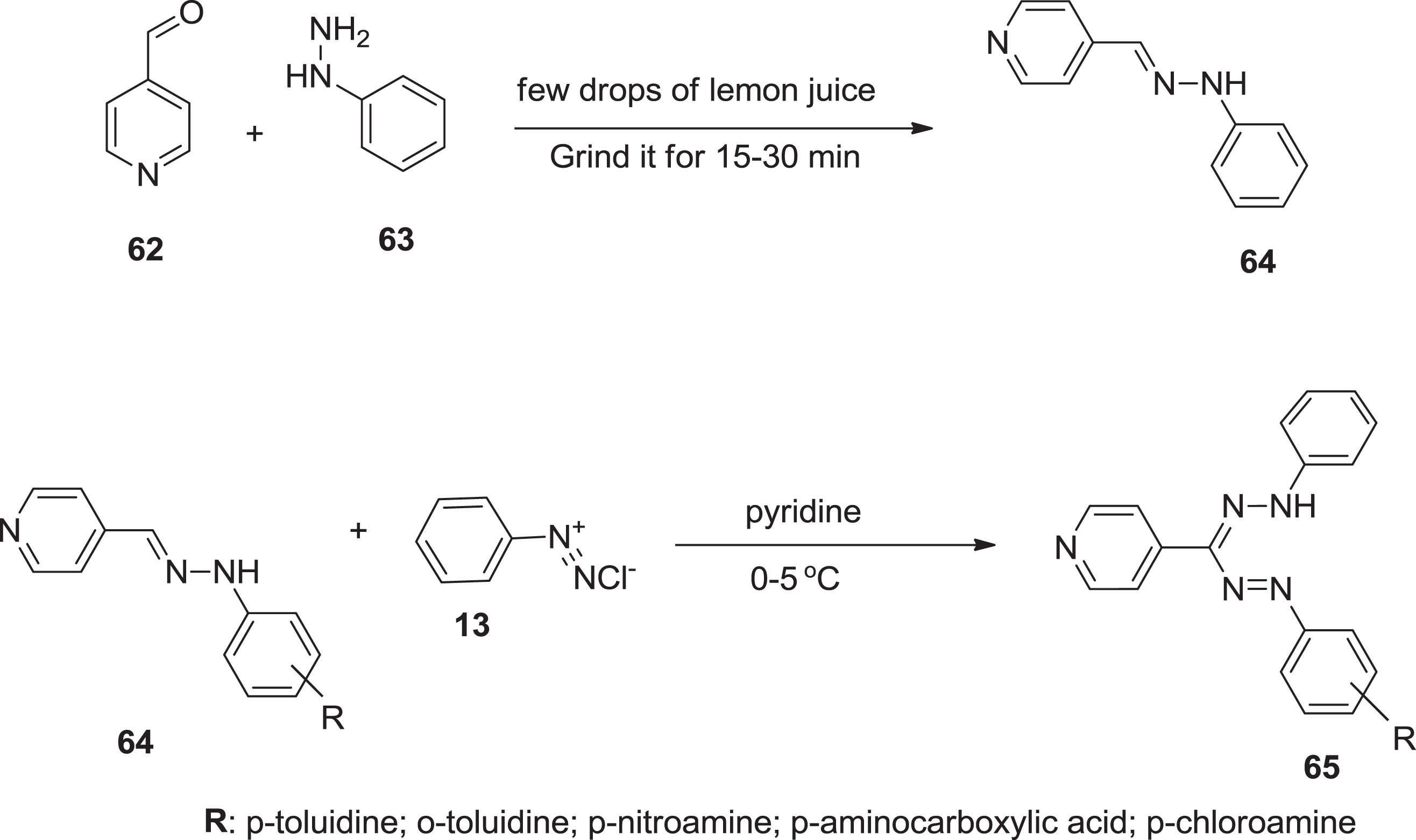
Preparation of formazan compound using organic catalyst.
In addition, Solanki and Marjadi [58] prepared formazan derivatives
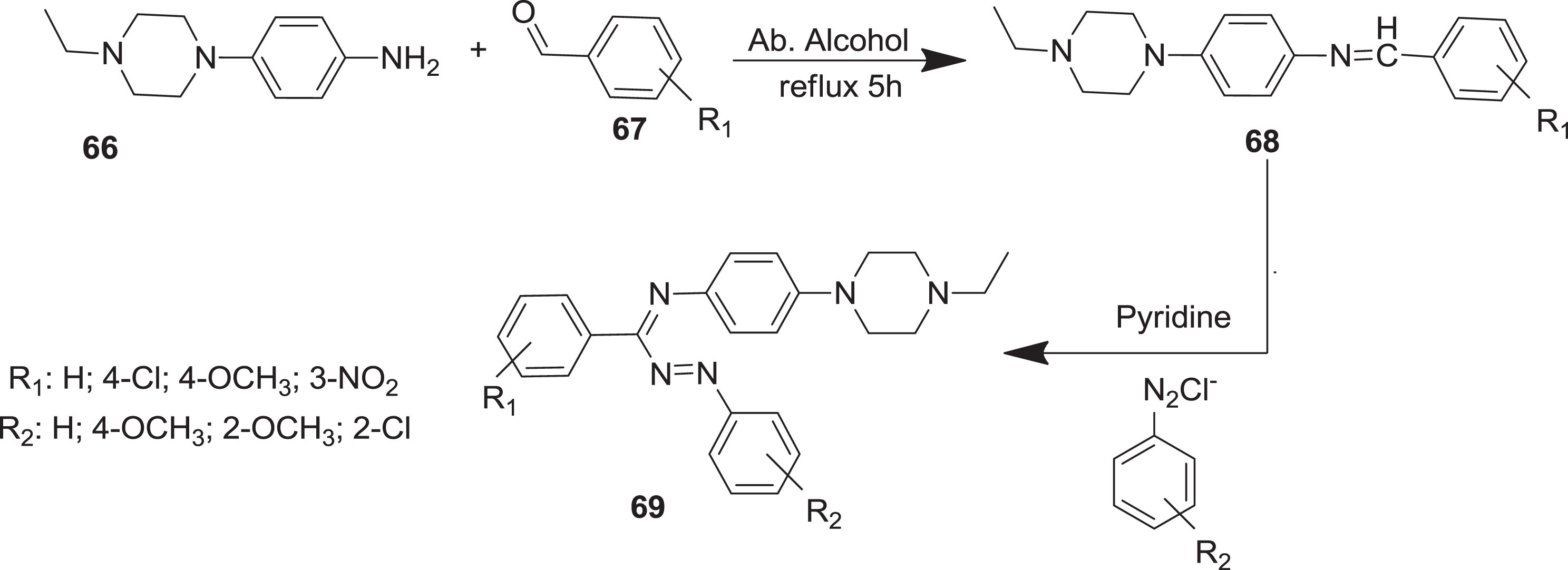
Synthetic route for formazan derivatives.
Recently, Gu et al. [61] originated a mixture of gluconic-meglumine acid as a stimulating vehicle for the reaction of methanal and β-ketosulfones. N-methylglucamine is a potential candidate having physiological inertness and biodegradability. Among other organic compounds, it has recently been investigated as precursors for the synthesis of 1,2-diazoles, pyranopyrazole derivatives, dihydro-pyridines, 2-amino-4H-pyrans, and pyrazolo-pyranopyrimidines [62–65]. Firstly, Khan et al. [66] synthesized the aromatic hydrazone deploying meglumine as a promoter, which then interacted with an aromatic diazonium salt to form formazans. Metal complex formazans were produced using a metallization mechanism with a variety of metal salts and then tested for pharmaceutical applications such as antibacterial, antifungal, and anticancer.
The hydrazine
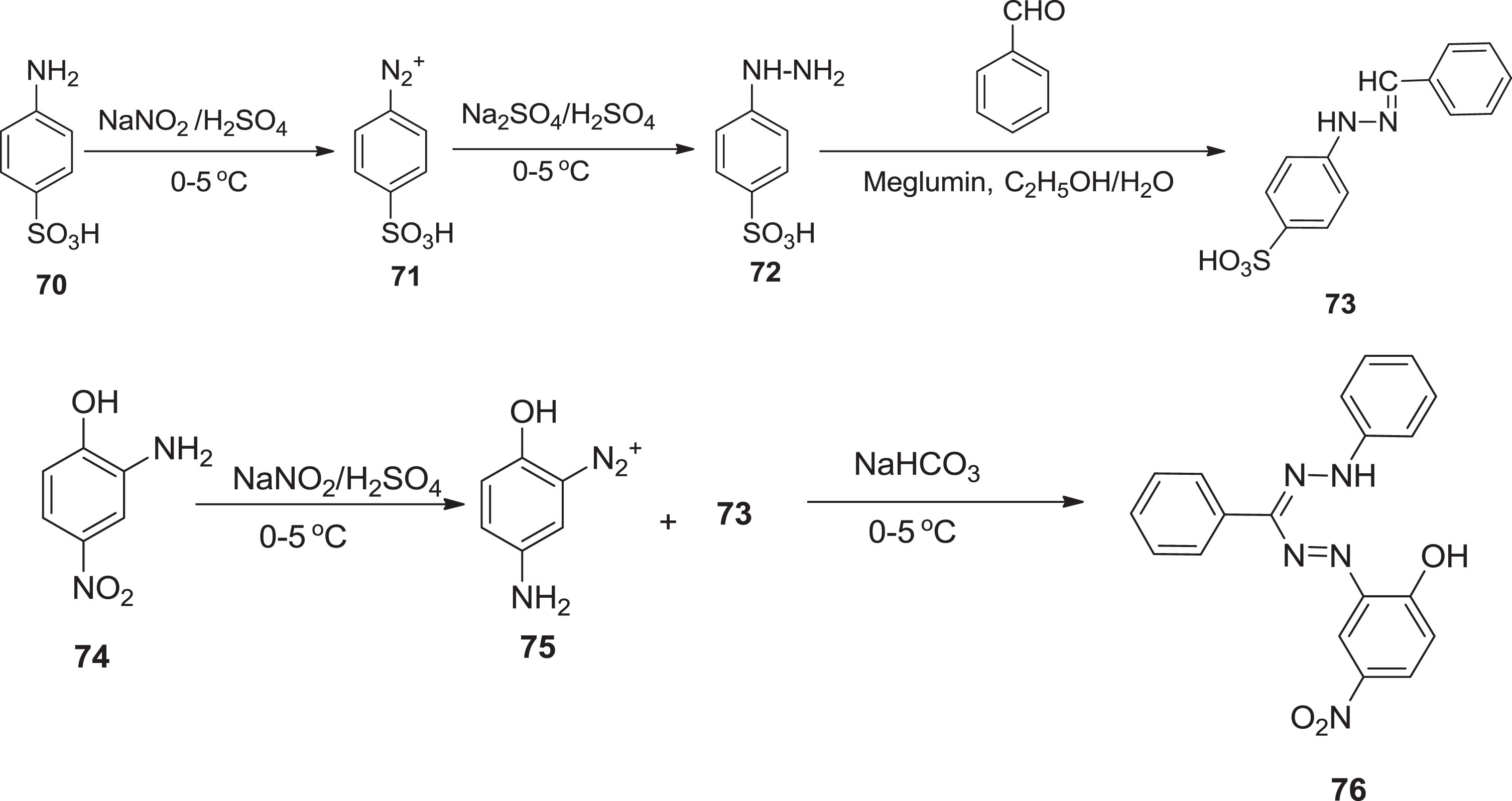
Reactions pathways for preparation of compounds.
The reported results indicate that the synthesized formazan compounds
In continuation, Turkoglu & Berber [9] produced one more series of compound
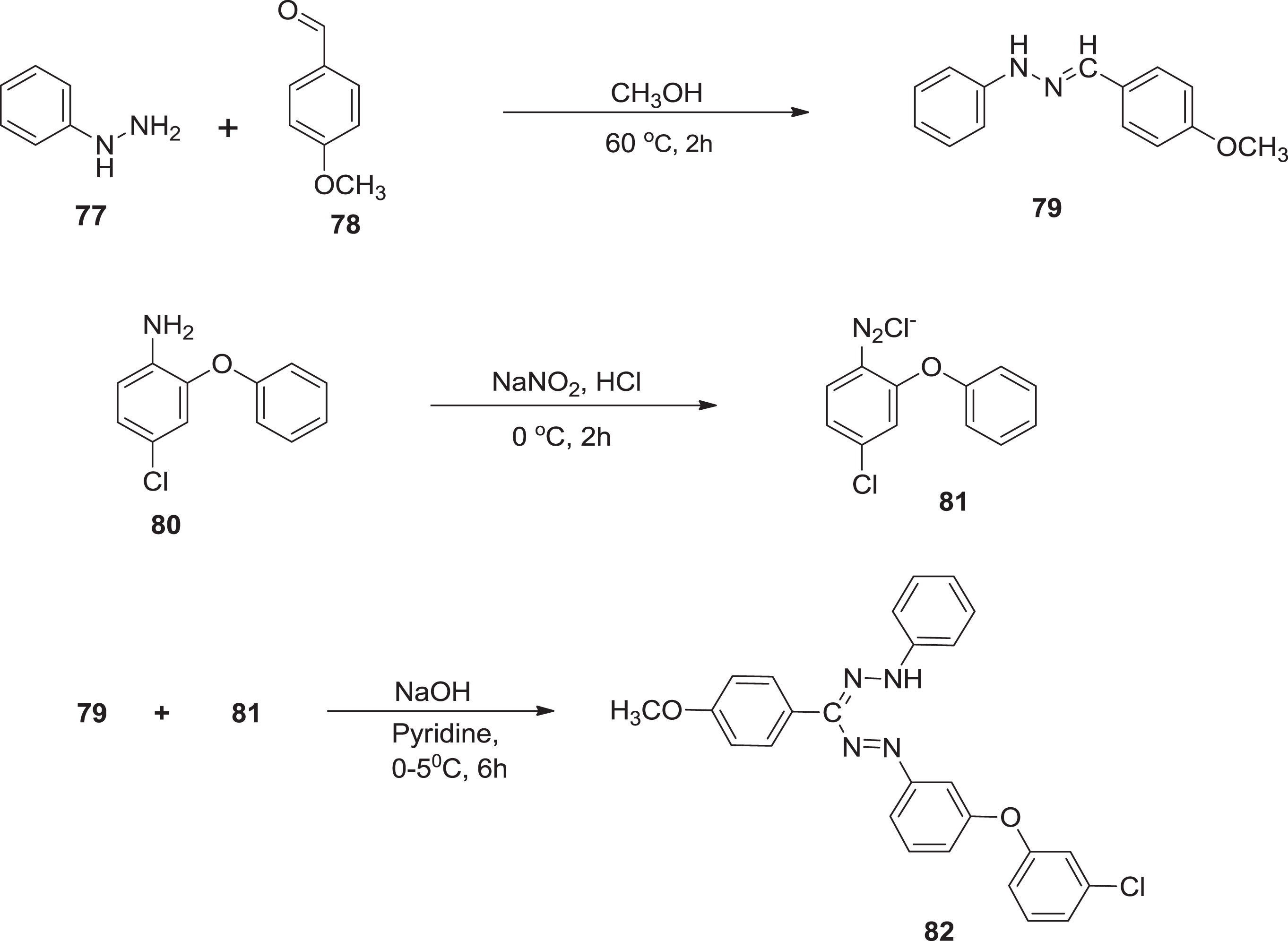
Preparation of formazan compounds.
Furthermore, Samel et al. [12] formed multiple formazan derivatives
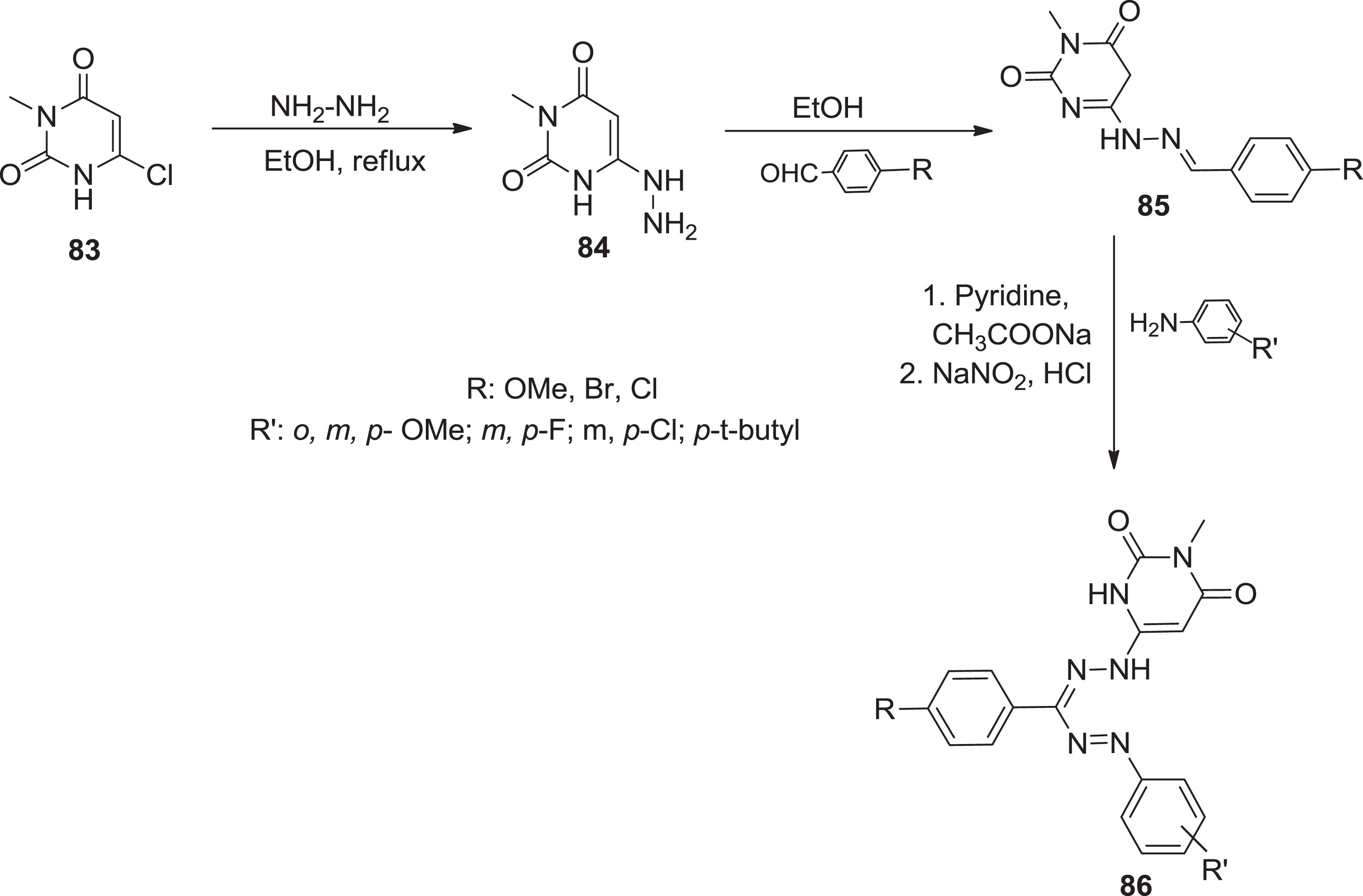
Synthesis of formazans via coupling Schiff bases.
Recently, photodynamic therapy (PDT) against tumor tissues revealed variant promising anticancer activity in formazan derivatives and their conjugates[72]. However, formazans are more practical in terms of determining the efficacy of anti-cancer drugs [73].
All newly formed compounds by Turkoglu & Akkoc [74, 75] have slightly elevated cytotoxic activities in this cell line at various concentrations. Compounds
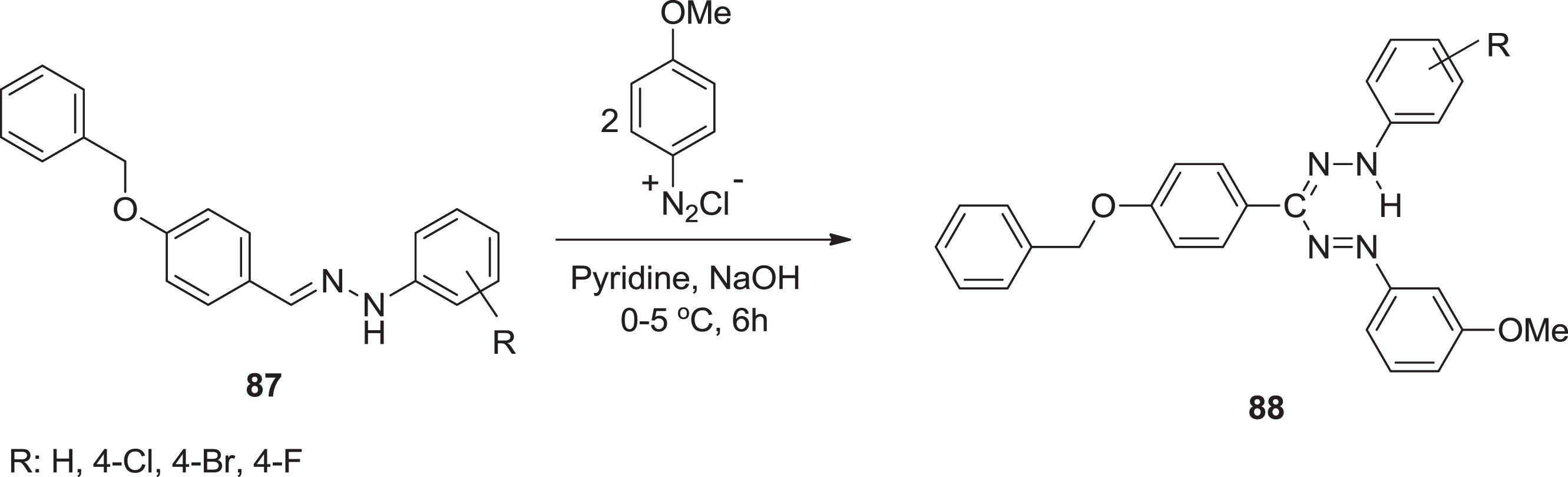
Preparation of formazan compounds from aryl amines.
Several formazans also showed favorable anti-fertility and anti- parkinsonian activity [76]. Isoniazid (isonicotinic acid hydrazide, INH) has been a prominent anti-tubercular agent for decades, but more information about its action against Mycobacterium tuberculosis has recently emerged [77].
For example, Ayed et al. [78] incorporated a set of formazans via a solution of amine derivatives. The amine derivative
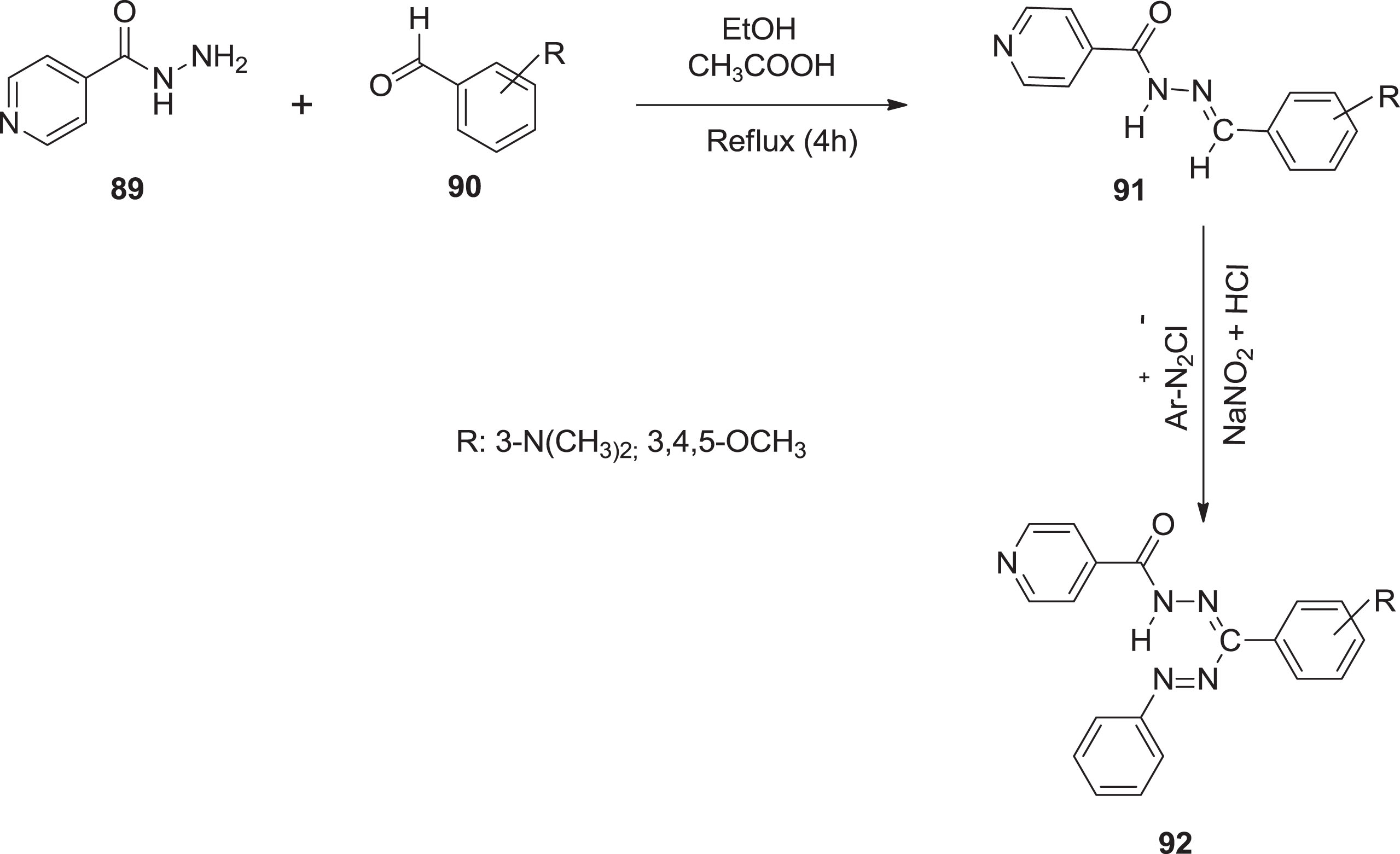
Synthesis of formazan derivatives.
Also, Al-Faham & Aljamali [79] synthesized formazans as depicted in the following scheme. Compound
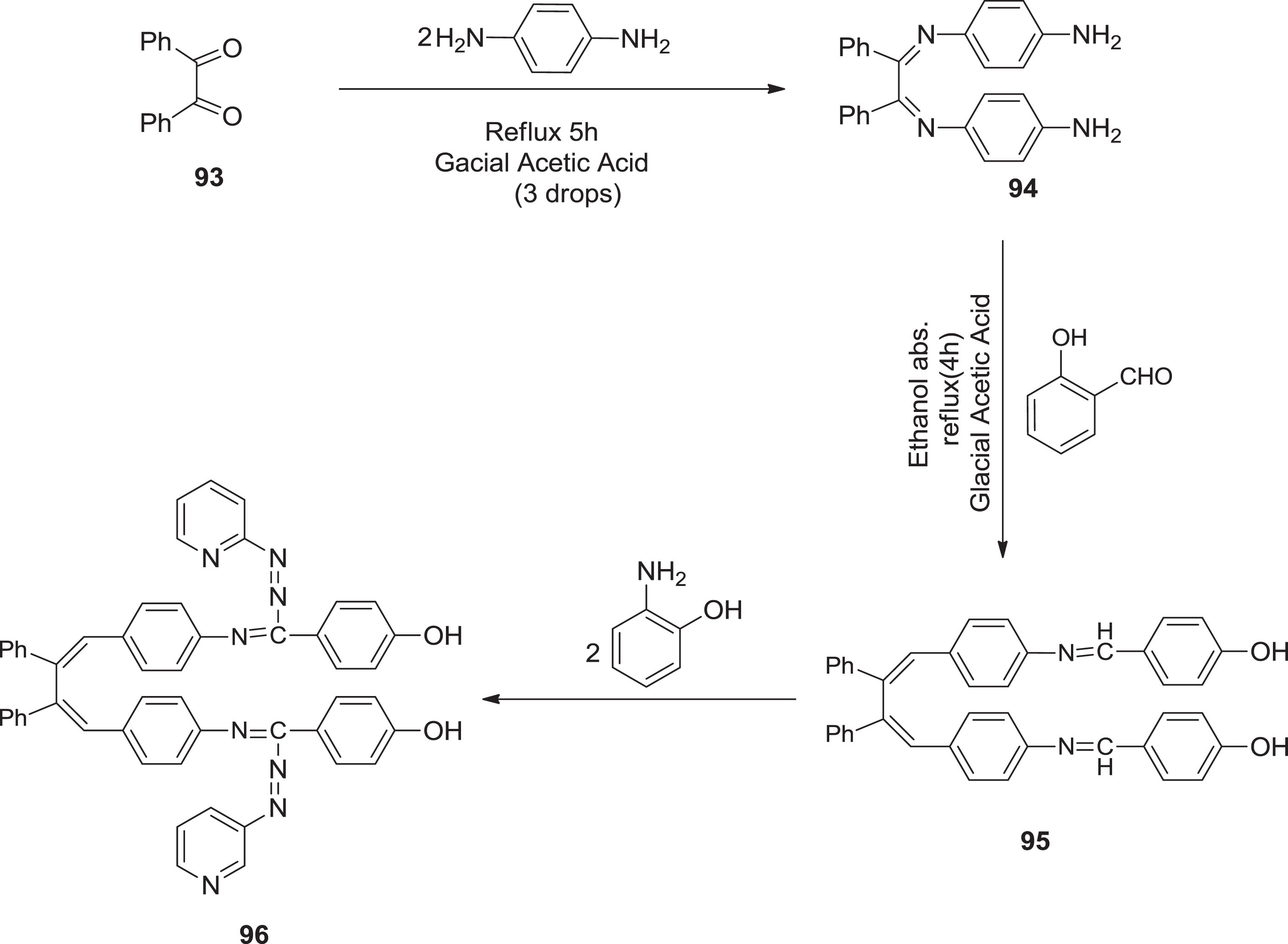
Preparation of bis-formazans.
Analyses based on the metabolic lowering of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide has been used to measure virus-induced mutagenic effects [85], and cell viability [86]. MTT reduction in cells results in the generation of a colored, non-soluble formazan compound
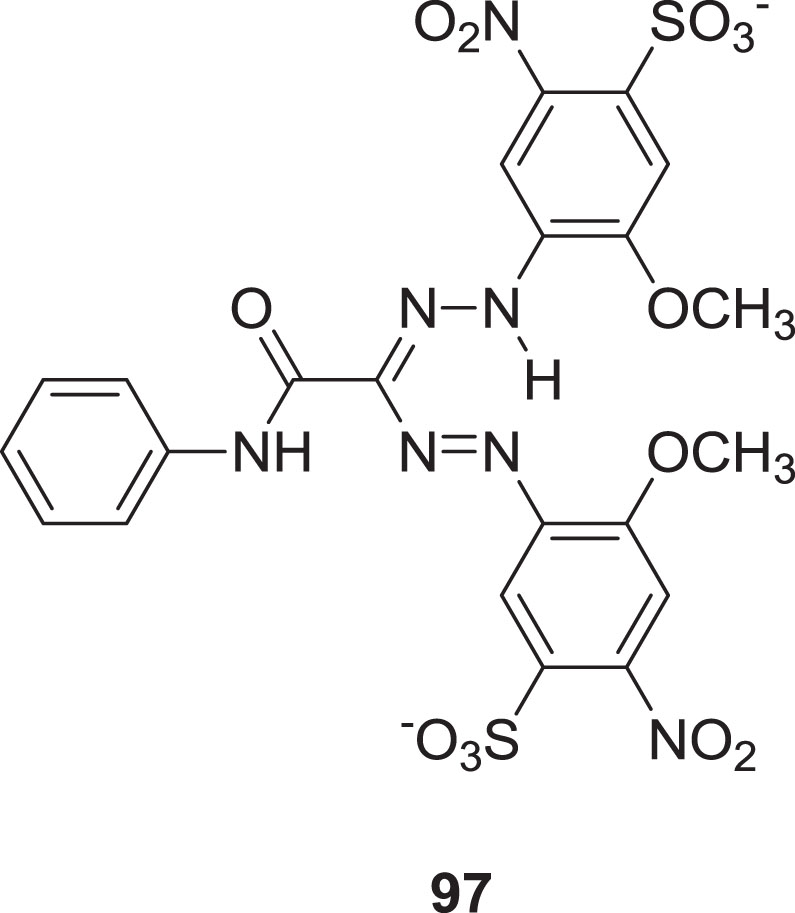
Structure of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT).
Over a broad extent of drug concentrations, the standard anti-HTV-1 agents 2’,3’-Dideoxy-ATP (wells B11, C11-B4, and C4) and AZT (wells D11, E11-D4, and E4) promoted cell survival and thus substantial production of XTT formazan in this compound [87].
Recently, Shinde et al. [88] were obtained a series of fluoro substituted formazan derivatives
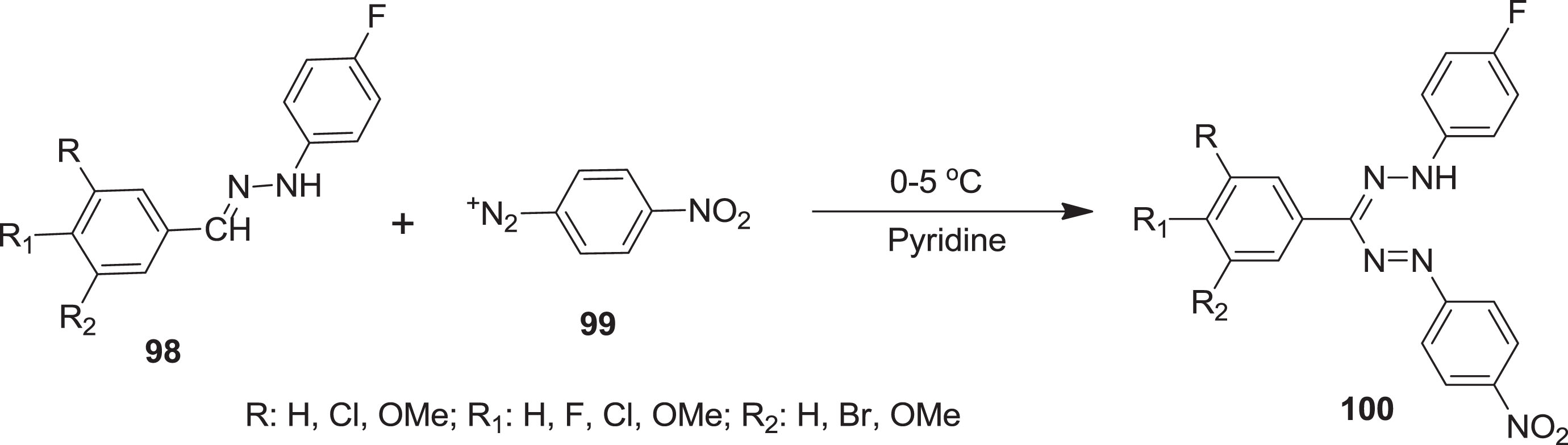
Synthesis of fluoro substituted formazan derivatives.
Also, the poison plate technique was being operated to evaluate the antifungal activity of synthesized compounds
Furthermore, Ostrovskaya et al. [90] incorporated tetrazolium betaines having sulfo groups and their precursors. The synthesis of
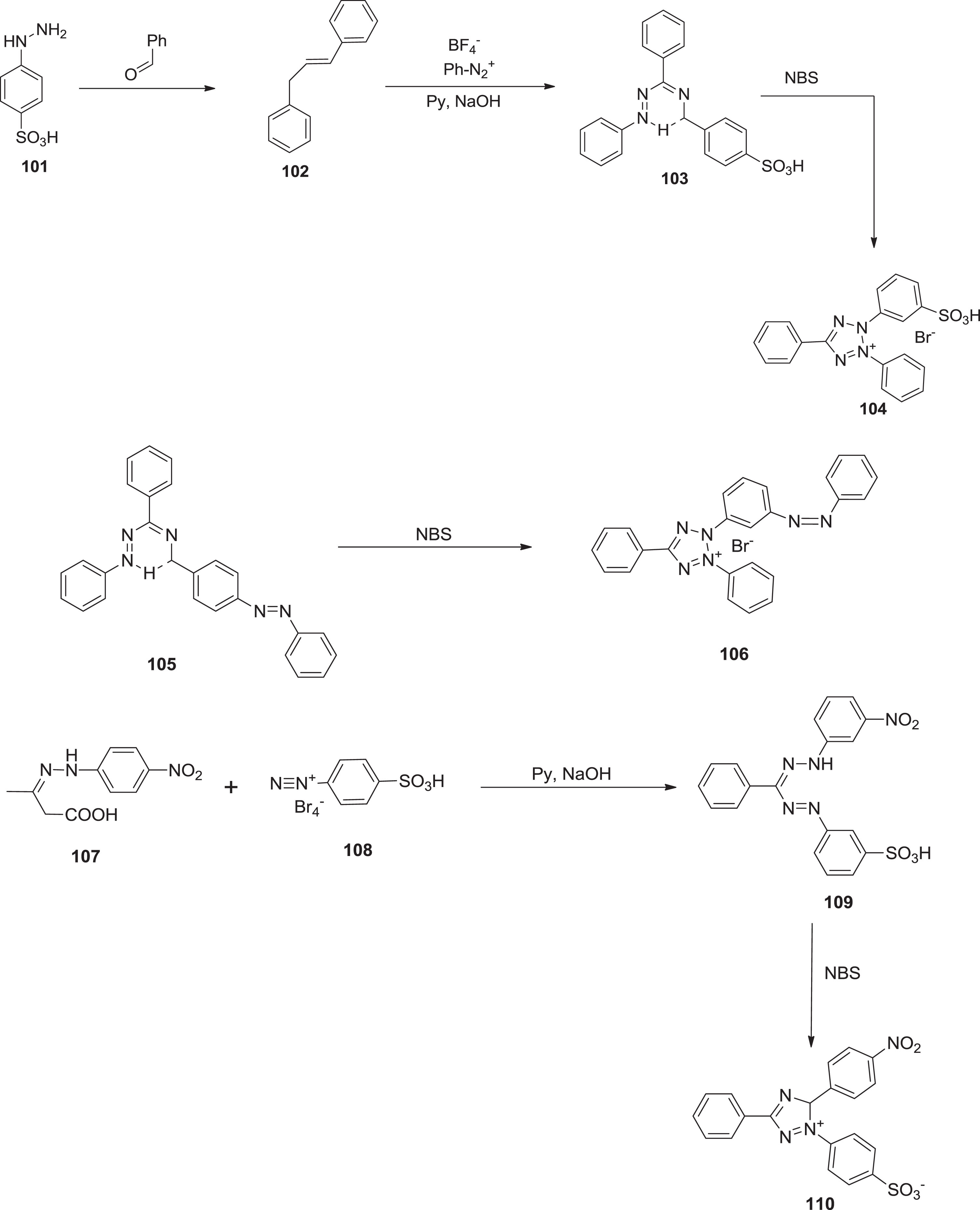
Chemical reactions for 5-(4-nitrophenyl)-3-phenyl-1- (4-sulfophenyl) formazan via cyclization.
The synthesis of formazan dyes necessitates specific circumstances, such as modest temperatures, use of polar solvents, saturated liquids, fraught with equipment degradation and environmental conflicts Metal formazanates are either preferable as environmentally friendly dyes or as elements of digital recording media, photo and thermo sensitive materials, extremely active catalysts [73, 91].
In 1947, Ciba was the first to make use of metal chelates of tetradentate formazan as wool dyes. Since then, various metal complexes of tetradentate formazans have been identified, and become a widespread variety of colors, particularly in volatile and reactive dyes; although they have also been operated for different purposes [92]. Yuzhen et al. [93] formed a set of five Cu(II) chelates of tetradentate formazans
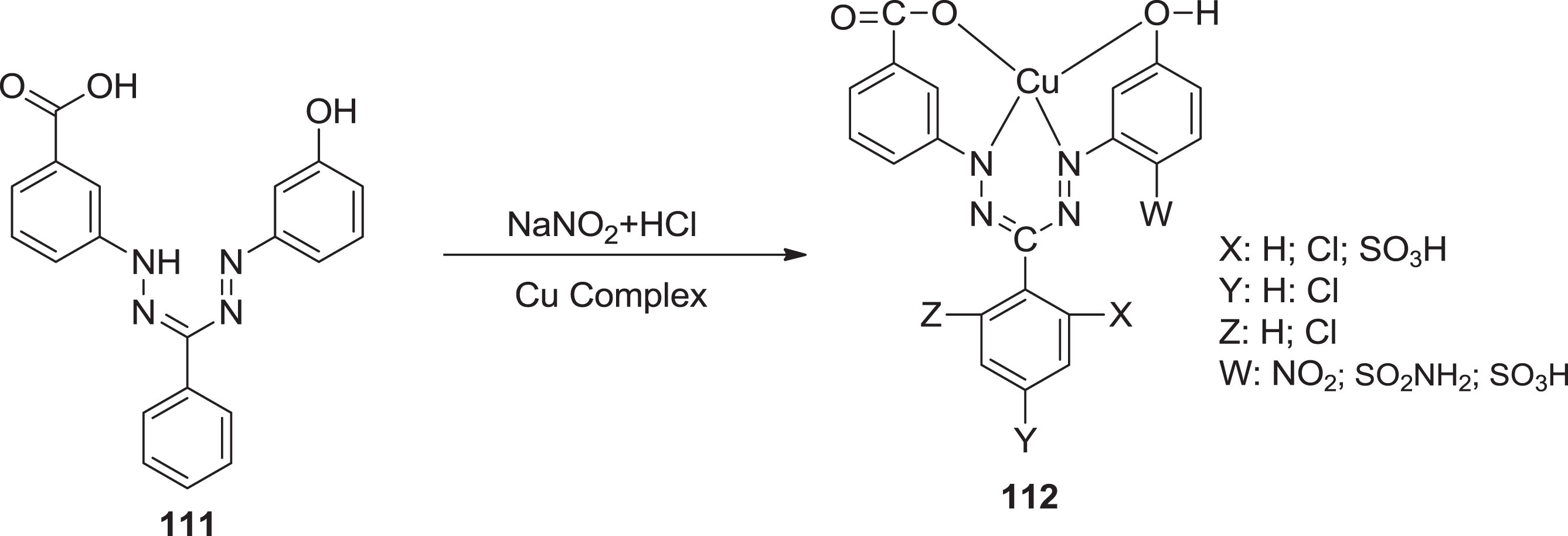
Structure of formazan dye-containing Cu(II) complexes.
Formazan has historically been employed in different application domains. It can, for example, induce very rapid and intense color reactions with a large number of metal ions. They are frequently used as analytic intermediates. Color photography also tends to make use of the reduction of salts containing tetrazolium into metallized formazan pigments. Water-soluble colorless tetrazolium salts have been found to be reducible to water-insoluble deeply colored formazans. Water-soluble complexes with -SO3H groups have a strong tendency for peptide and polyamide fibers and can be utilized to dye and manufacture make synthetic materials. Acid Black 180 (1) is an example (Scheme 28). The most beneficial dyes of formazan are the 1:1 Co chelated formazan organic dyes used in cellulose fiber coloring and printing [94].
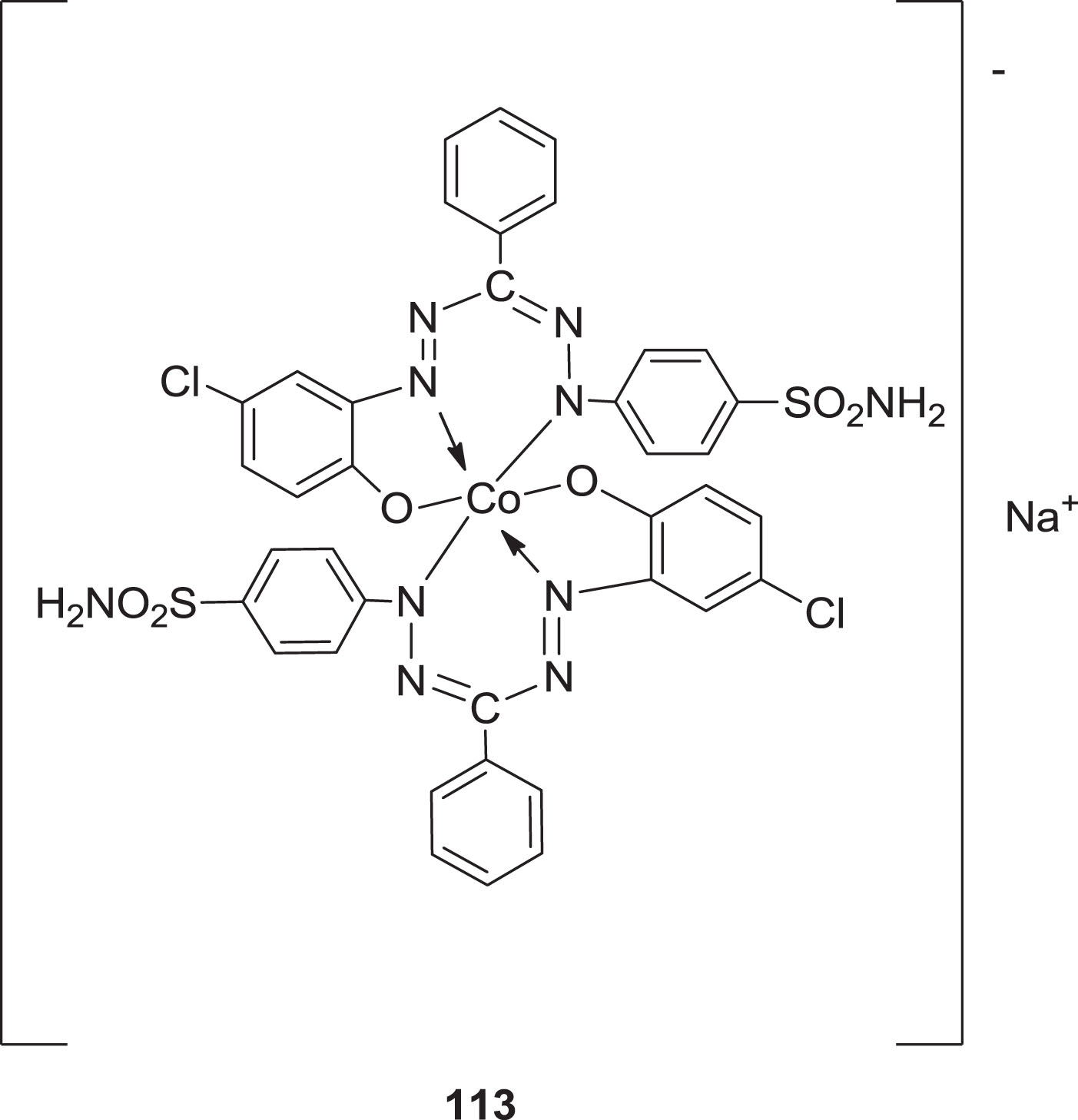
Structure of formazan dye.
BF2 complexes of easily available formazan binders are an emanating type of dye with tunable optoelectronic and redox properties. As near-infrared colorants [95, 96], narrow band-gap polymers [97], and cell cytotoxic agents [98], BF2 formazanates have shown potential. Benzosulfonazole-substituted formazans [99] and their coordination compounds [100, 101] have previously been explored, but derivatives of trifluoroborane were previously unknown.
Further. Chang et al. [102] synthesized boron containing formazan complexes via the condensation reaction of
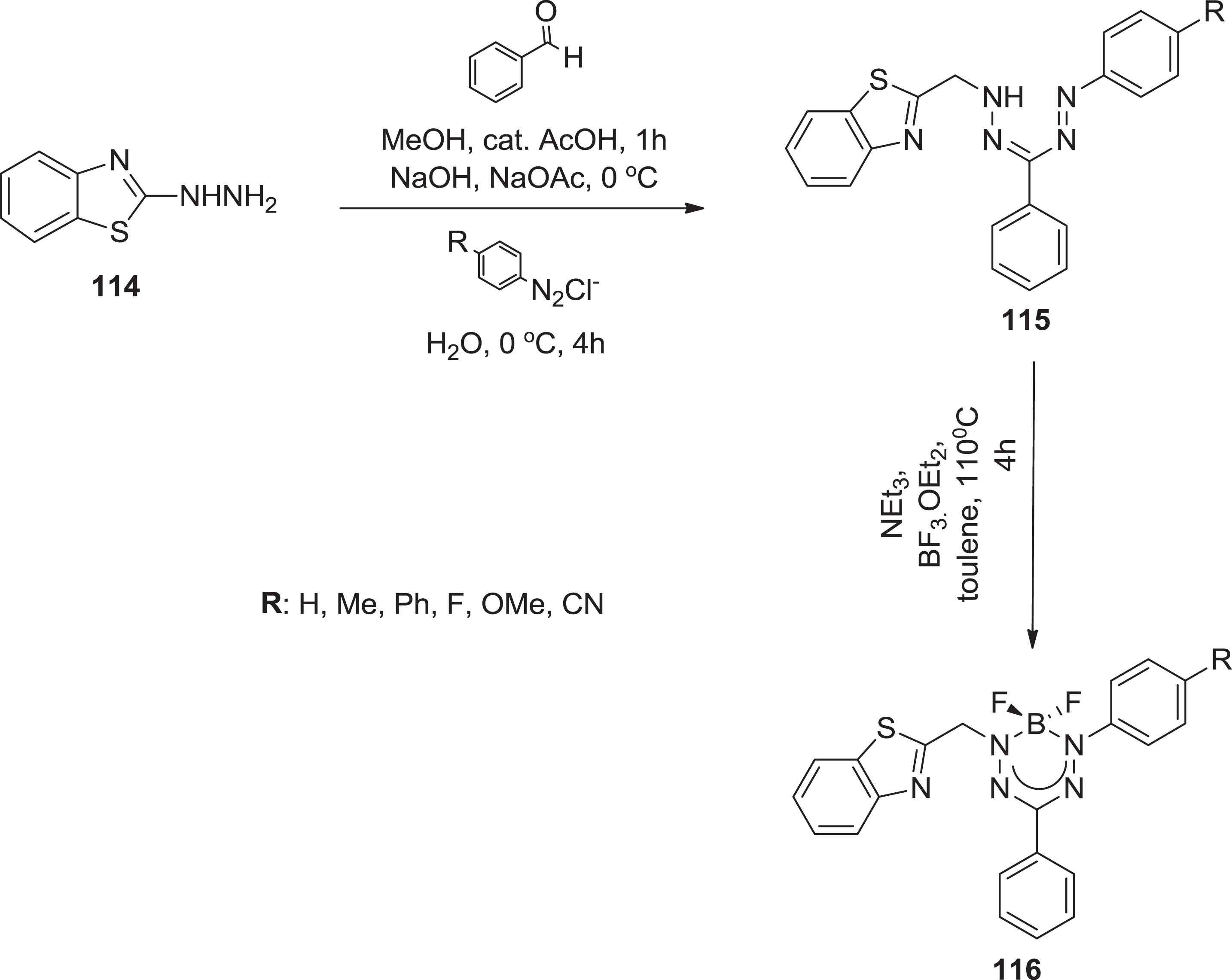
Synthesis of boron trifluoride formazanate complexes.
When metallized with different metal salts, e.g., hydrated form of ferrous sulphate to generate a complex of ferric ions, chromium chloride to produce Cr(III) ion complex, and copper sulphate to form copper coordination complex, the formazan dyes tend to form metal complexes. The dyes of formazan are also more advantageous in terms of determining the efficacy of anti-cancer medications [103].
Khan et al. [104] prepared this series of metal complexes which were screened for antibacterial potential against various strains such as gram-positive and gram-negative bacteria. The
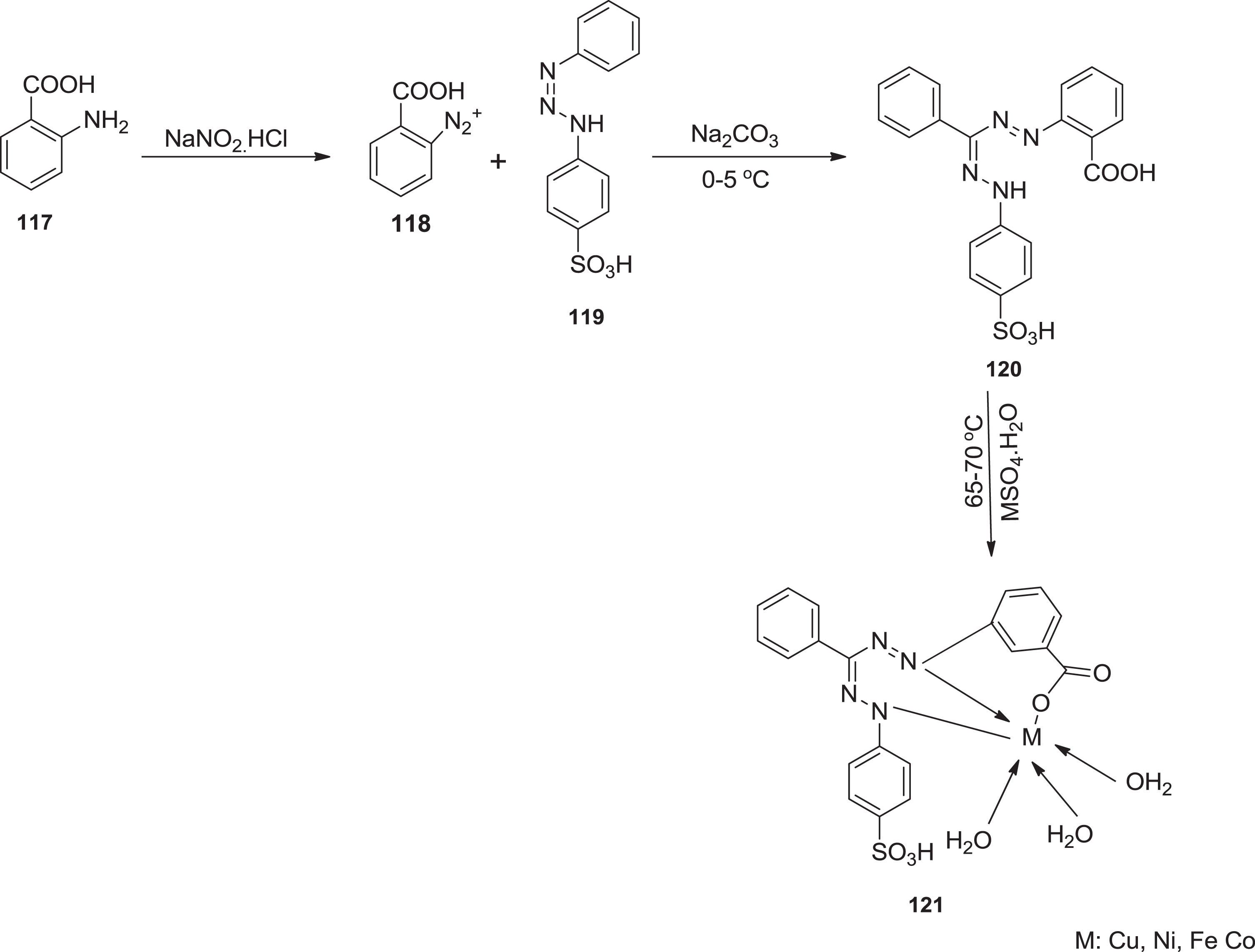
Preparation of formazan metal complexes.
Over the past two decades, interest in the coordination chemistry of formazans and formazanates has been revived due to the simple synthesis and distinctive properties of these complexes. Here, we give an overview of recent research with an emphasis on the reactivity and applications made possible by this special class of ligands. We have opted to arrange our data by periodic table group, starting with alkali metal complexes.
Alkali metals
It has been demonstrated that deprotonation of formazans with highly alkaline metal bases e.g., sodium hydride or potassium hydride generates the corresponding alkali metal formazanate salts. As a result, dimeric, hexameric, and polymeric solid phase systems are formed [105] (Scheme 31).
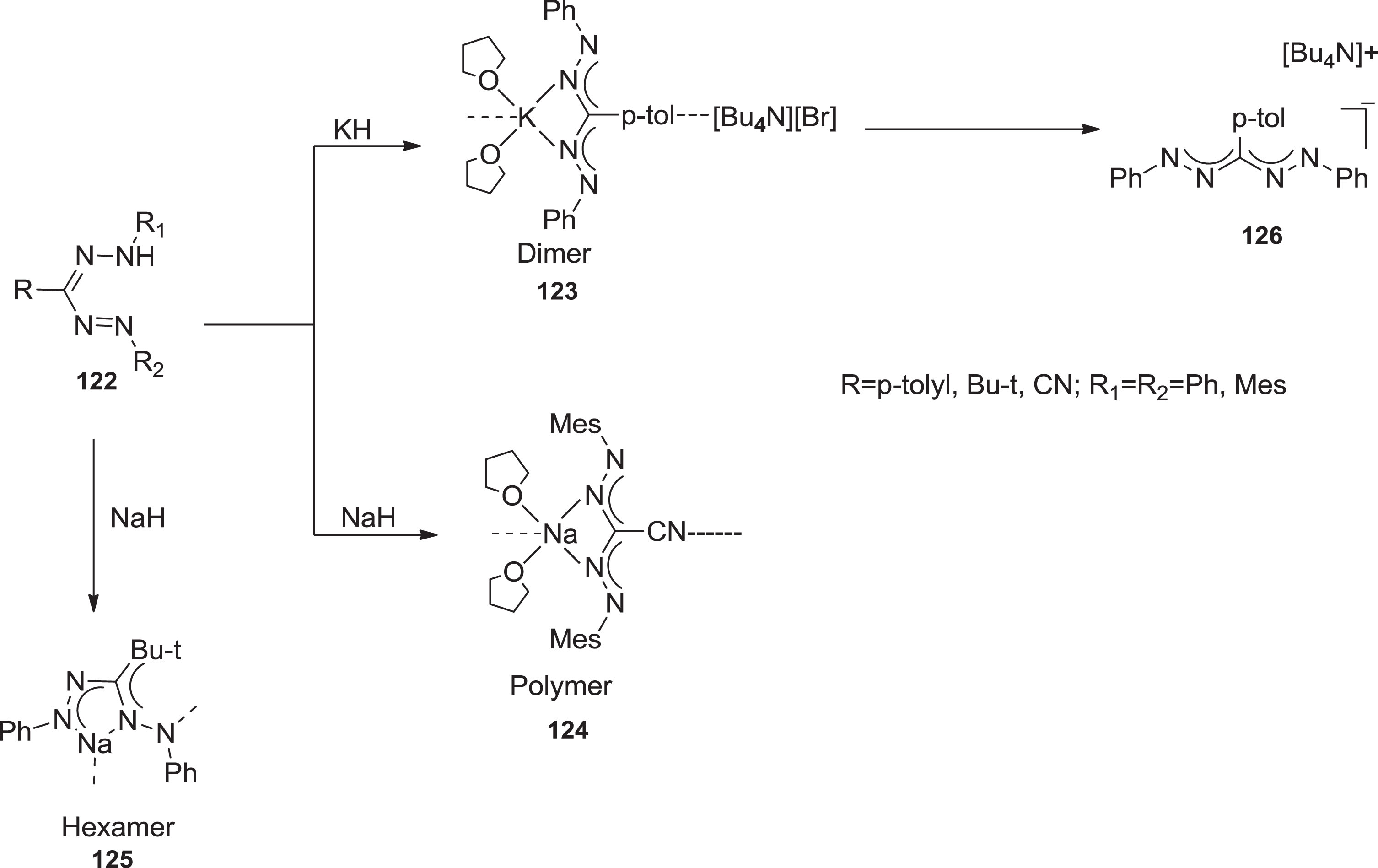
Synthesis of Na and K metal salts of formazanate ligands.
Hicks and associates recently reported an iron composite with a trianionic nitrous oxide linker relying on the moiety of formazanate, in which the N-aryl substituents have an auxiliary phenoxide o-donor component. The low-spin Fe(III) cluster [106]was synthesized by salt hydrolysis reaction from the in situ tetradentate dioxyhydrazine generated Na-salt with FeCl3 (Scheme 32).
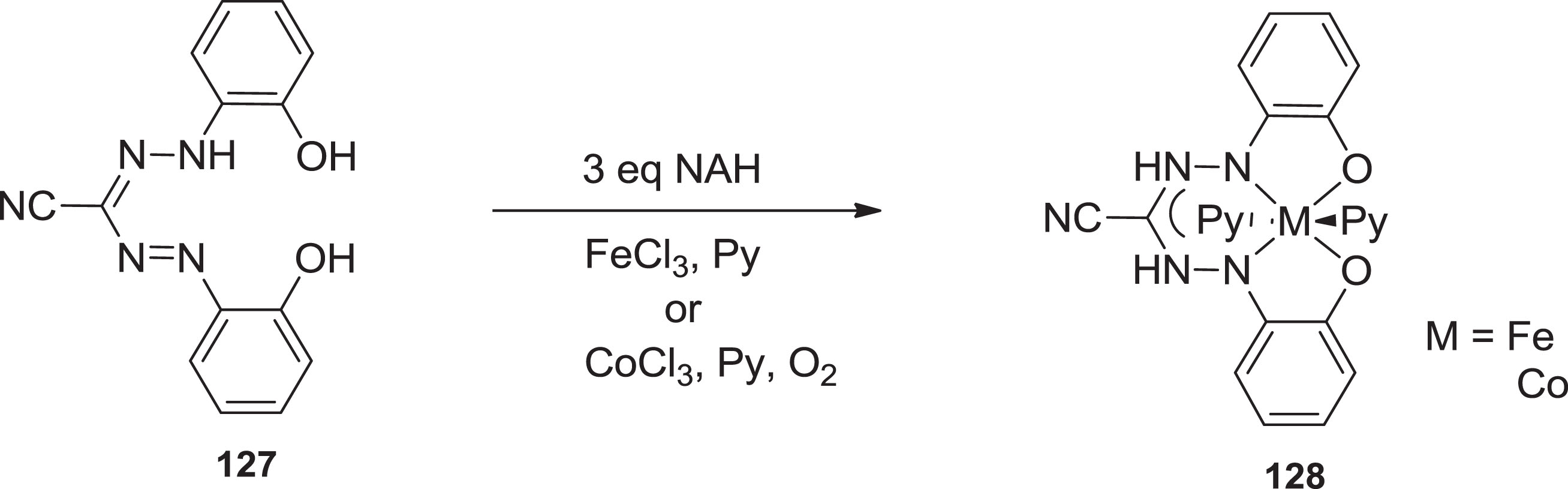
Formation of formazanate scaffold containing iron and cobalt complexes.
The formation of the ferric ion complex with two monoanionic binding sites of triarylformazanate was using salt metathesis and potassium formazanate was reported [107] (Scheme 33).
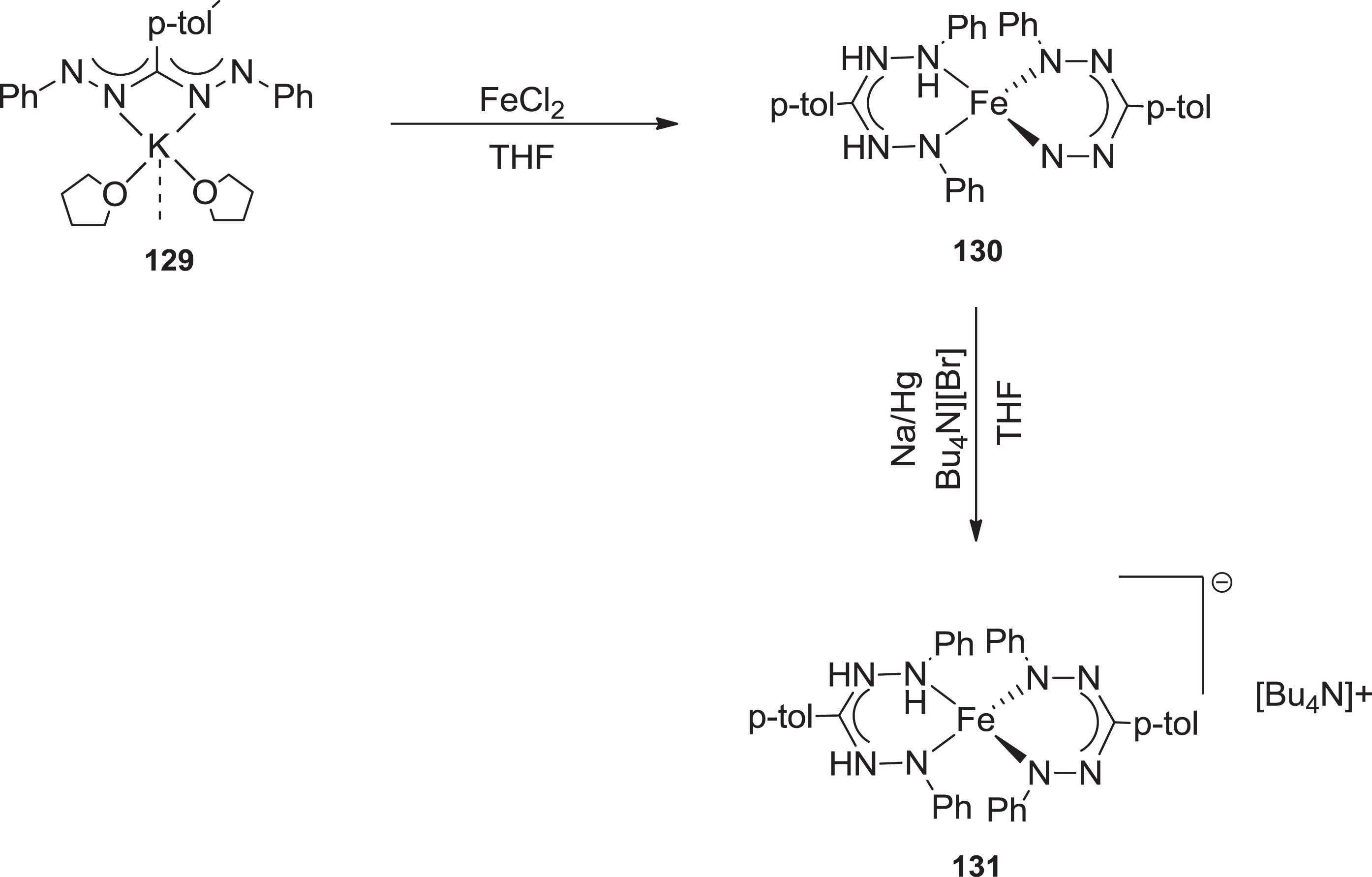
Synthesis of formazanate complex 128 and the corresponding by-product 131.
Gilroy and Otten [108] reported the formation of the Co(III) multiplex with a trianionic and tetradentate N2O2 cyano-formazanate ligand. Complex
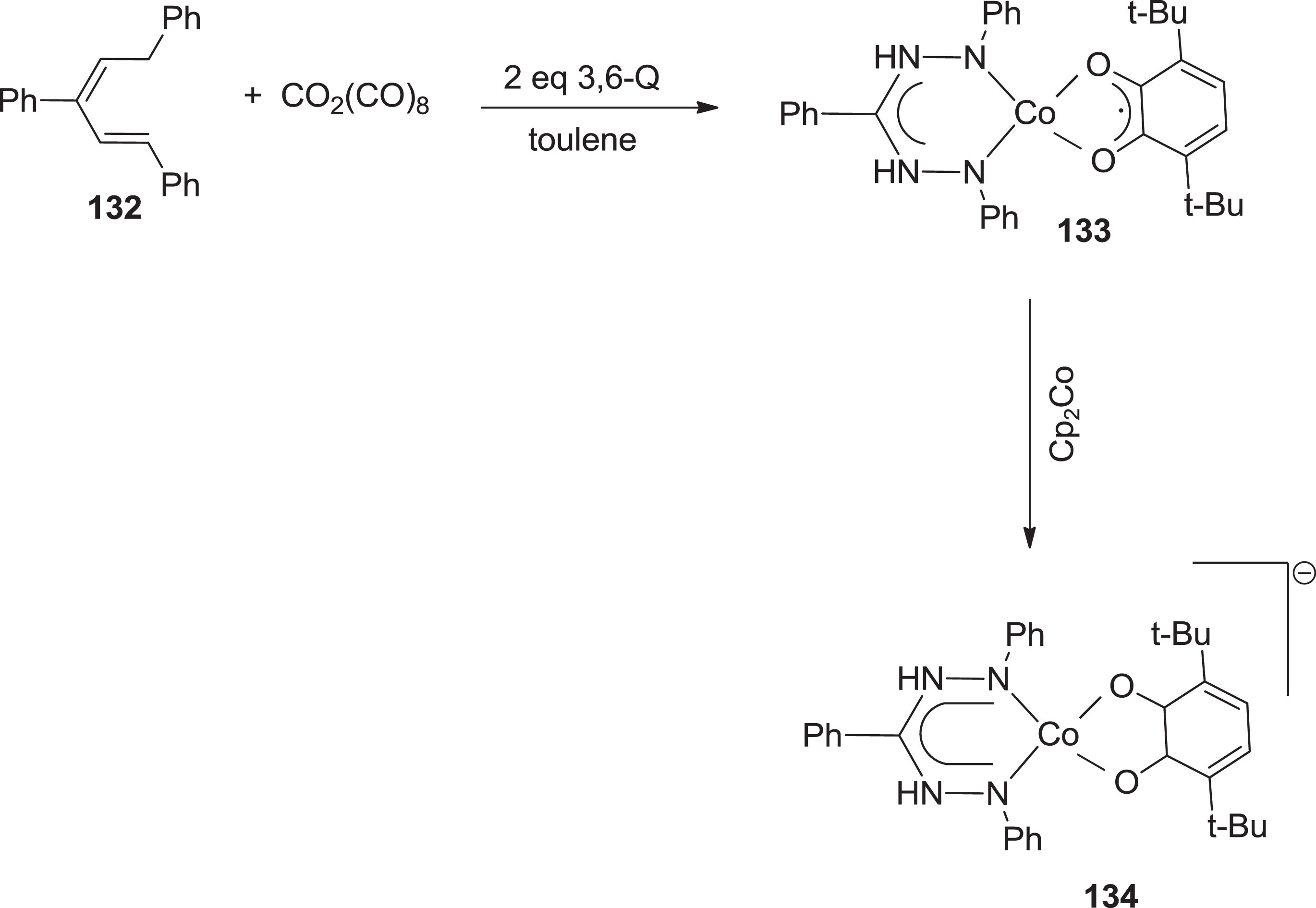
Synthesis of formazanate cobalt multiplex with o-quinone based polydentate.
Palladium complexes
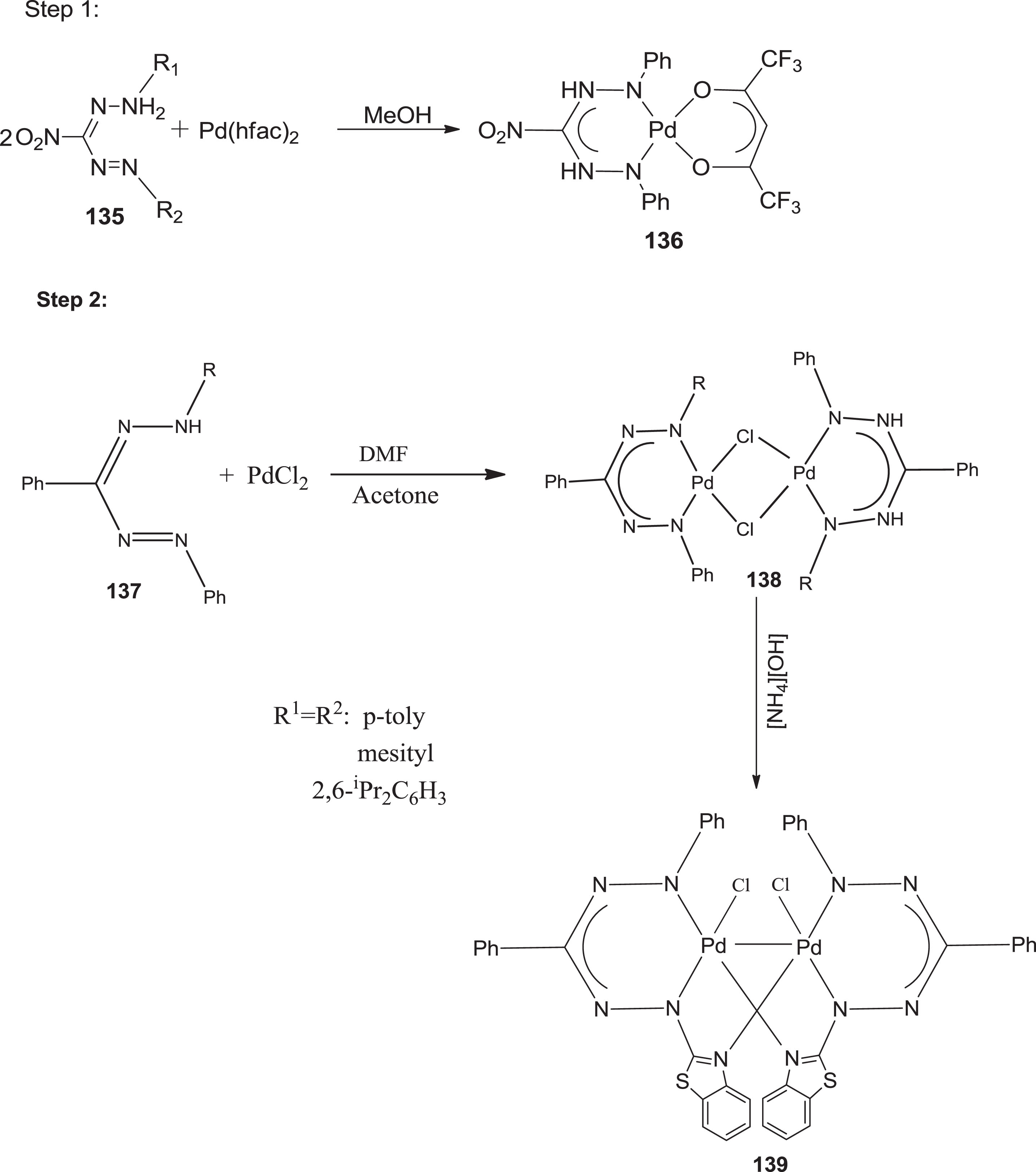
Synthetic route for preparation of Pd–Pd bonded complex 139 via the chloride-bridged dimer 138.
The Hicks group outlined [106] the copper-containing formazanate moiety
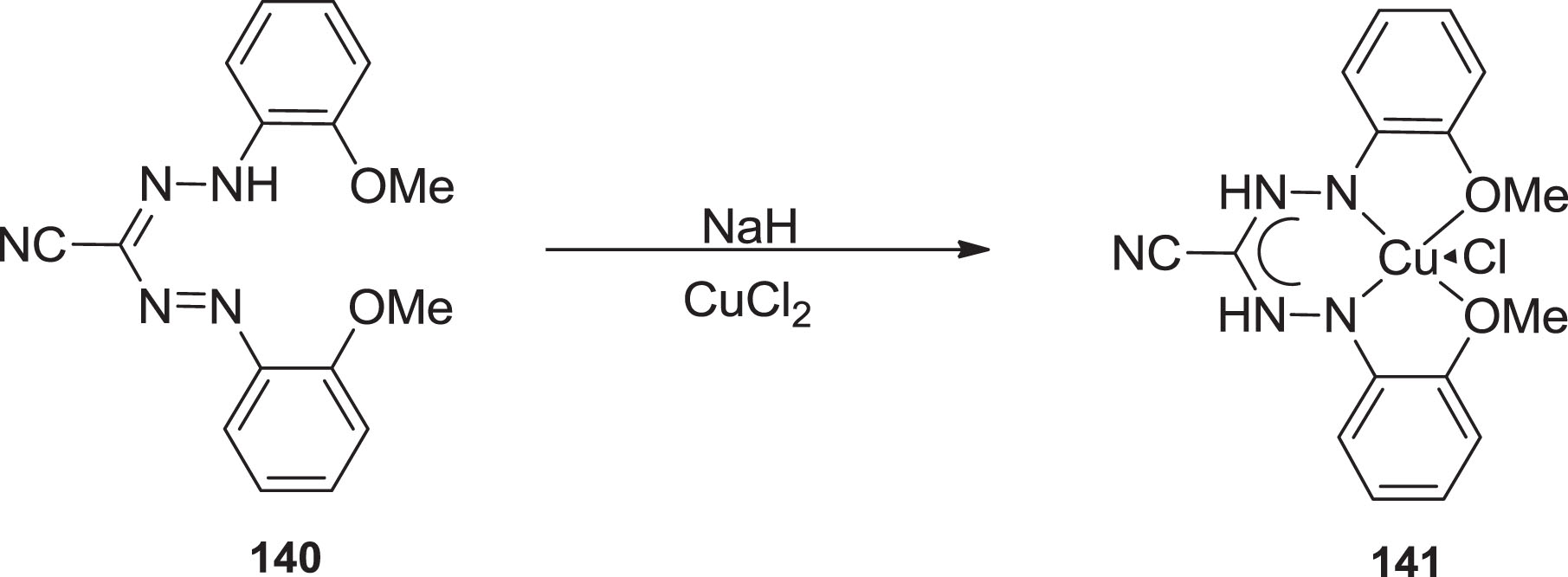
Synthesis of copper containing formazanate complex 141.
The bis(formazanate) zinc complexes
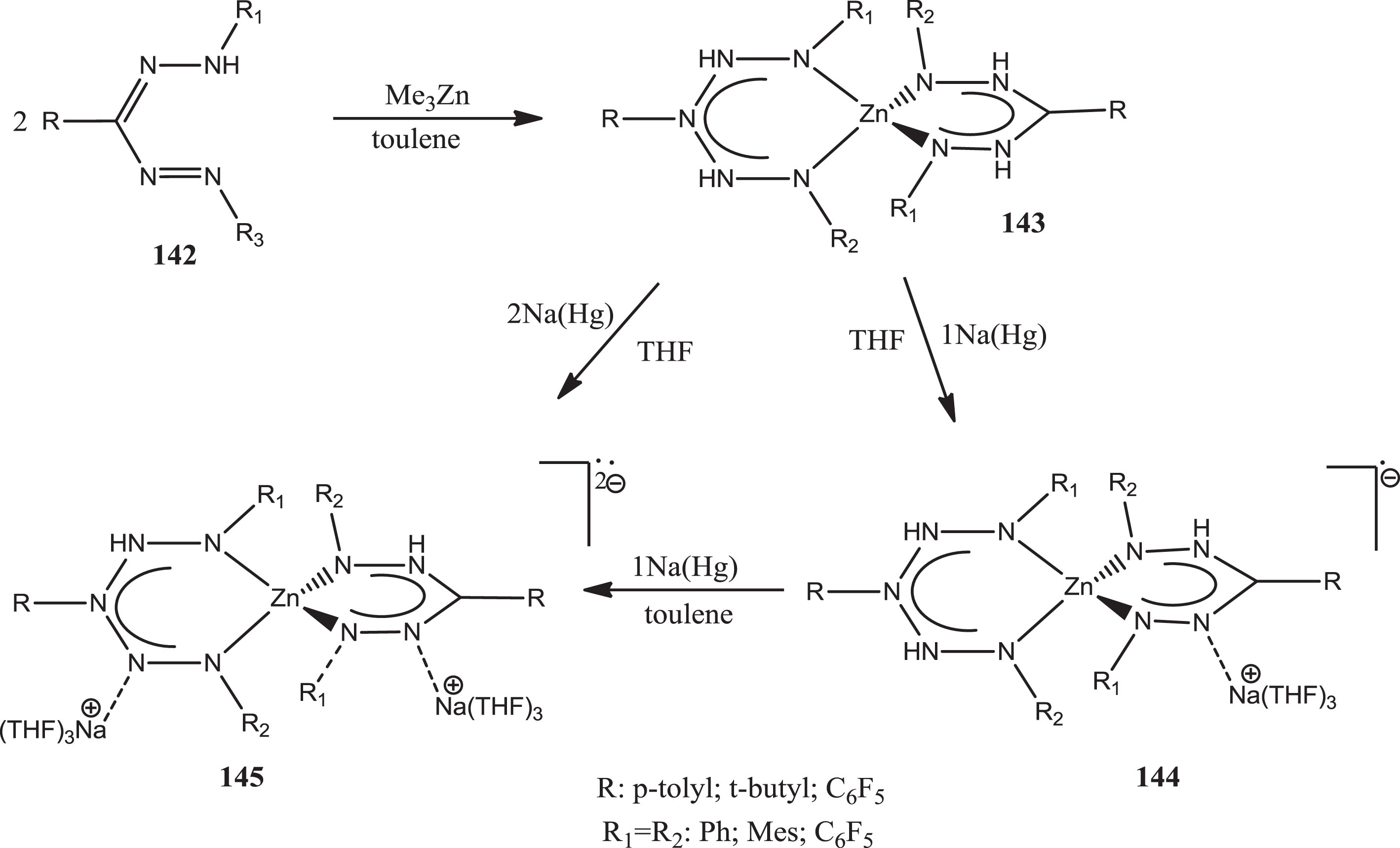
Synthesis of complex 132 and corresponding radical anions 144 and dianions 145.
Hicks and colleagues [114] piqued interest in this family of chemical compounds by converting triphenyl formazans into complexes of tetraacetyl diborate formazanate ligands and demonstrating conversion into verdazyl-type radical anions (Scheme 38).
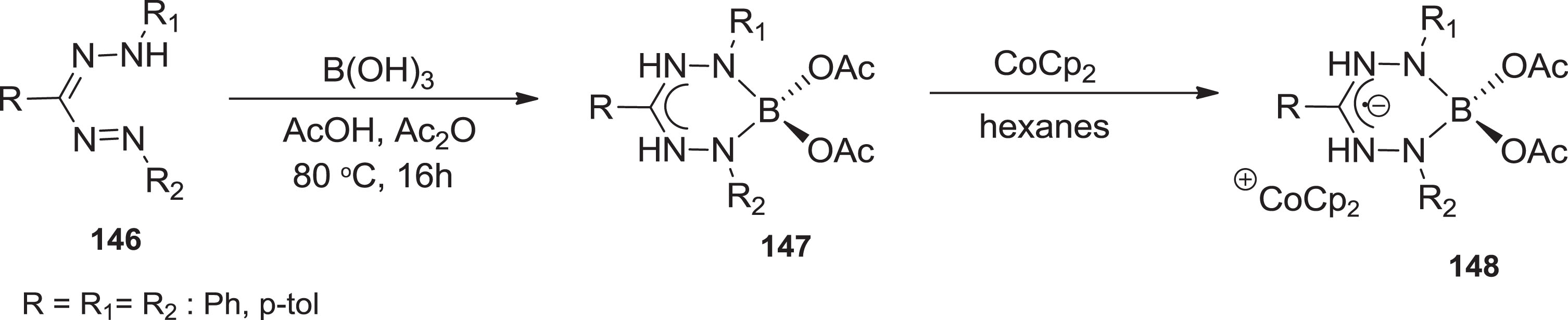
Formation of boron complexes and their corresponding radical anions.
An alternative synthesis involves heating toluene solutions containing excessive triethylamine and borane for conversion of 3-cyanoformazan to BF2 complexes. BF3.OEt2 [115] and was later elongated to formazans containing nitro and phenyl groups to yield BF2 ion complexes [116] (Scheme 39).
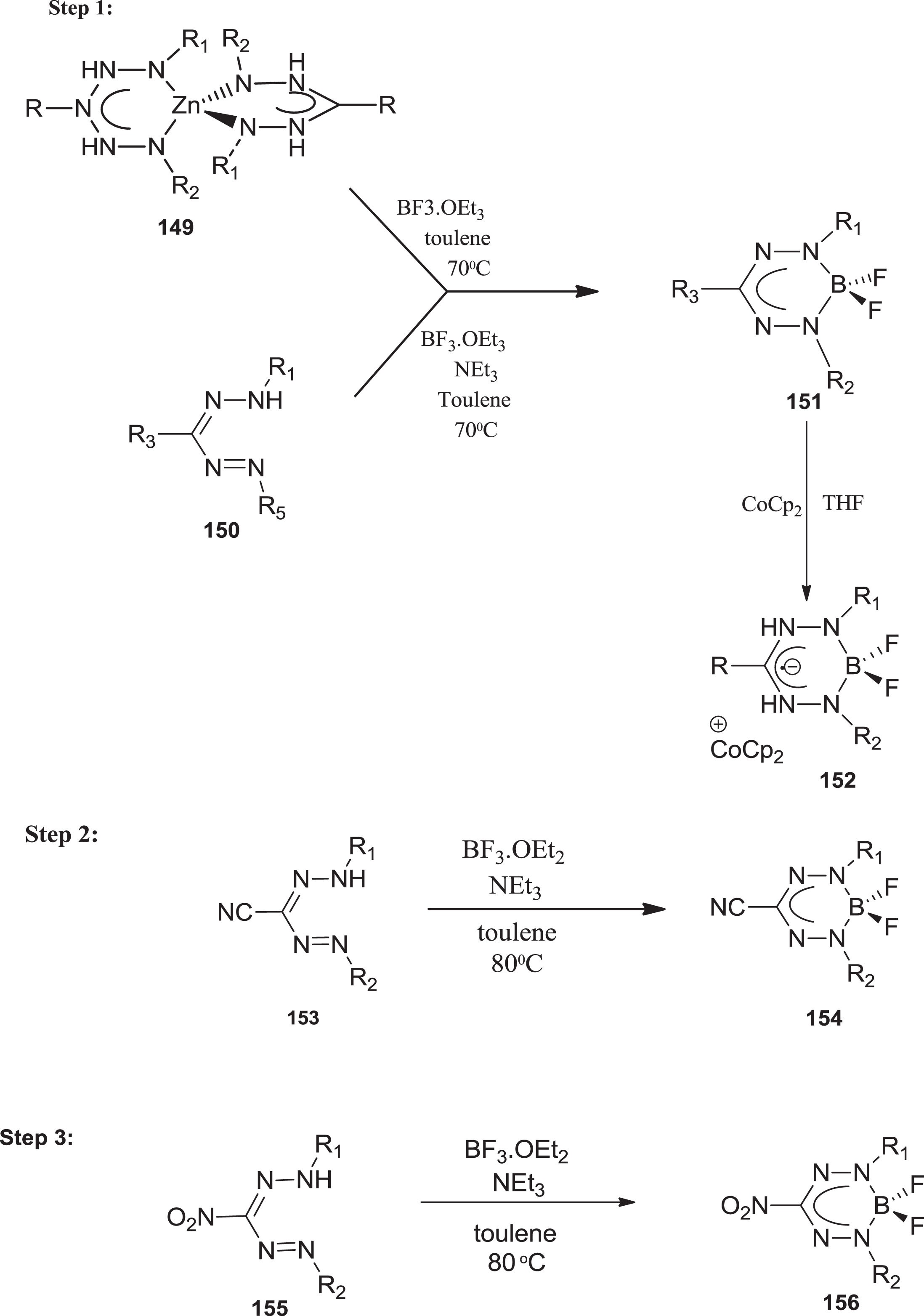
Synthetic route for preparation of boron-containing formazanate ligands.
From the above table, we observed that group
If
If
If
Conclusion
After a thorough review of the literature and critical analysis, it was investigated that the effect of a definite type of substitution on a particular position is specific, for example, the electron withdrawing groups (EWGs) on the ortho and para-positions of phenyl rings (R, R1 & R2) attached to the formazan group can increase activity to a greater extent. Aside from that, as per the study the electron donating groups (EDGs) in the different positions (R, R1 & R2) of the substituted moieties showed no efficient activity. From SAR studies, the substitution of various substituents on –NH moiety decreases the efficiency of the biological activities. It is hoped that this review would pique the interest of other research groups, encouraging them to develop additional formazans that are more efficacious.
Footnotes
Acknowledgments
The authors are thankful to Chandigarh University for providing financial support for the research.
Conflict of interest
No potential conflict of interest was reported by the authors.
ORCID
Neetu 0000-0003-2334-3631
Harvinder Singh Sohal 0000-0001-7190-8508
Meenakshi Verma 0000-0002-2407-0818
Manvinder Kaur 0000-0002-4173-7194
