Abstract
BACKGROUND:
Few studies have reported on long-term harms caused by ADHD drugs but they are known to impair growth.
OBJECTIVE:
To assess whether ADHD drugs impair reproduction in mammals.
METHODS:
Systematic review of reproduction in studies of animals treated with ADHD drugs.
DATA SOURCES:
PubMed, Biosis and EMBASE.
RESULTS:
We included 17 studies. The studies were generally of poor quality or poorly reported. Two studies reported the use of one of three advised randomisation methods. Fifteen studies used placebo which suggested blinding. On clonidine, the ability to produce offspring was reduced for male rats, which approached two females each. In one study, 10 treated rats produced no offspring while all four controls did. In another study, 10 treated rats impregnated nine females while 10 controls impregnated 16. On methylphenidate, vaginal opening was delayed in two studies (in one, the mean difference was 4.0 days, 95% CI 2.5 to 5.6, and number of estrous cycles was halved; in the other, the minimum delay was 6 days), while in two other studies no difference occurred. Generally, the impairments improved after a drug-free period and were less pronounced when treatment started later in life.
CONCLUSION:
ADHD drugs impair the reproduction in animals.
Background
Little is known about long-term outcomes of ADHD drugs but they do not appear to be positive. The largest study, the Multimodal Treatment Study (MTA), randomised 579 children to methylphenidate, behavioural therapy, both, or routine community care [1, 2]. There were no clinically important effects of methylphenidate after 14 months, or after three and eight years of follow-up [3–5]. Similarly, a large cohort study that investigated overall functioning at six years of follow-up found that psychostimulants did not lower ADHD symptom severity [6].
The randomised trials included in a recent Cochrane review underreported the harms of methylphenidate in children [7]. Only 5% of the included trials reported on serious adverse events, while 14% reported non-serious adverse events; these were more common with methylphenidate than with placebo (risk ratio 1.29; 95% confidence interval 1.10 to 1.51).
Methylphenidate is a norepinephrine-dopamine reuptake inhibitor that acts predominantly through the dopamine system. There are concerns that treating children with stimulants may cause irreversible brain damage [8], as well as damages to the neuroendocrine system during development [9], because dopamine acts both as a neurotransmitter and a regulator of hormonal pathways [10].
Exposure to ADHD drugs stunts growth [11] and may possibly also impair the development of the reproductive system. A case has been described where a boy before being put on drugs experienced sexual hyperactivity and was diagnosed with precocious puberty [12]. He was treated off-label with methylphenidate from age five, which caused a decrease in both aggression and hypersexuality. Another case report is about a boy treated with methylphenidate for 17 years since childhood who experienced delayed puberty; at age 20 he had underdeveloped genitalia, lack of libido and also erectile dysfunction [13]. A study that investigated pubertal development in 65 boys on stimulant medication for at least three years found a slower rate of physical development of the genitals during puberty compared to 174 controls [14]. In contrast, some studies and case reports have shown increased sexual activity and priapism with methylphenidate [15, 16].
When methylphenidate was administered to young rats, corresponding in age to prepubertal children, it resulted in altered levels of dopamine transporters that persisted into adulthood, long after termination of treatment [9]. Another study in rats found that methylphenidate impaired spermatogenesis after eleven weeks of treatment, which indicated that the drug might reduce male fertility [17].
A large report from 2005 reviewed the reproductive and developmental effects of methylphenidate [18]. There were no data on humans and for animals, the working group felt it did not have sufficient data to give advice, although they mentioned two studies with rodents demonstrating reduced sperm motility and altered estrous cycling, respectively, and one study reporting no differences.
We decided to do a systematic review of animal studies to assess the impact of ADHD drugs on the development of the reproductive system.
Aim
To assess whether drugs approved for ADHD cause abnormal development of the reproductive system in animals.
Methods
Search strategy
Searches were performed in PubMed, Biosis and Embase, using MeSH terms or equivalents, for the eight most sold ADHD drugs in 2012 as reported by the US Food and Drug Administration: atomoxetine, clonidine, dexmethylphenidate, dextroamphetamine, guanfacine, lisdexamphetamine, methylphenidate and amphetamine [19]. The search strategies are available in Appendix A. We also checked the references in a previous review for eligible studies [18].
We contacted the authors when data were unclear or not reported for papers published in 2000 or later.
Eligibility criteria and data extraction
We included controlled studies of ADHD drugs given postnatally to healthy mammals, which were not primed by drugs or had been operated upon, if they had data on one or more of the following outcomes: morphology, motility of spermatozoa, ejaculation, cell counts, cycles, pregnancies and births (impregnations, resorptions, number of stillborns, number of liveborns with malformations).
We did not assess the reproductive organs of offspring from dams treated during pregnancy.
One observer screened all records and a second observer screened all included records and a random sample of 15 excluded records (no disagreements). Potentially eligible articles were collected and read in full. References of eligible articles were scanned to locate further studies. Data were recorded in Excel. One investigator carried out data extraction and a second investigator checked the extracted data. Disagreements were resolved by consensus.
We extracted data according to published guidelines [20–23]: animals (species, strain, age, sex), study design (randomisation, blinding, drug intervention, group sizes, dose, route of administration, length of treatment and of follow-up), selective reporting within studies and relevant outcome measures.
Quantitative data synthesis
Meta-analyses were performed with Review Manager using data from the end of the intervention and, if available, the last time point after a drug free follow-up period. If the test had been carried out on several consecutive days, we only used the results from the first day.
If more than one drug had been tested in the same trial, we split the control groups accordingly to avoid double counting. For studies with several drug doses, dose groups were pooled [24]. Data from animals kept group-housed or isolated were pooled. Missing standard deviations were calculated or estimated by standard methods [24], e.g. imputation from SEs or p-values.
Risk of bias was assessed according to SYRCLE [20, 24]. For animal studies, the SYRCLE risk of bias tool recommends assessment of three categories of randomisation: randomisation of animals to intervention or control groups; random housing to prevent behavioural differences between groups introduced through differences in light intensity and temperature; and random outcome assessments to prevent influence on results from diurnal variation [20].
Statistical methods
We used a random effects model. For binary data, risk ratios were reported. For meta-analyses of continuous data and ranking scale data, the mean difference or the standardised mean difference were calculated.
The basic assumption was that the healthy control animals showed normal development and behaviour. As there were differences in the direction of the scales and outcomes, we multiplied some of the outcome values by – 1 to ensure that values to the right in the graphs always meant an effect in favour of placebo or no treatment.
Proportions were compared with Fisher’s exact test.
Amendment to the protocol
As our data extraction resulted in a large amount of outcomes, we decided to focus on the outcomes directly related to the ability to reproduce (motility of spermatozoa, ejaculations, cycles, pregnancies and labour) and not the indirect or surrogate ones (cell counts, morphology).
We had planned sensitivity analyses but there was too little data to allow these.
Our protocol is in Appendix B.
Results
We identified 518 unique records and excluded 461 based on title or abstract for one or more of these reasons: not an ADHD drug, primed animals, irrelevant outcomes, in vitro studies or otherwise not an animal study, or polytherapeutic interventions. We also excluded a study in Japanese [25]. Full text screening of the remaining 57 articles led to inclusion of 17 studies (see flow chart in Appendix C).
Study characteristics
All 17 included studies were controlled and they were published between 1975 and 2016 (Table 1). Thirteen studies were conducted in rats [17, 26–37]; two in mice [38, 39]; two in dogs [40, 41]; and one rat study also included rabbits [36]. Median number of animals per study was 30 for the intervention groups (range 4–100) and 17 for the control groups (range 4–41) for the 16 studies where information was given.
Overview of included studies. PND – postnatal day, GD – gestational day
Overview of included studies. PND – postnatal day, GD – gestational day
Methylphenidate was used in nine studies, clonidine in seven and amphetamine in one. The median intervention period was 28 days (range 1–90) and there were follow-up data for eight studies (median 18 days, range 1–63 days) [17, 27, 31, 33, 34, 36, 37, 41].
The studies were generally of poor quality or poorly reported. Two studies did not use a placebo [17, 26]. Fifteen studies were placebo-controlled, which suggests that they were also blinded. Only one study [32], however, described in detail the blinding of the observers. According to the SYRCLE guidelines [21], we considered blinding of caregivers and personnel as unclear or at high risk of bias either because blinding was not mentioned or because the control groups did not receive placebo.
Randomisation to either intervention or control was mentioned in two trials [32, 41], but the method was not described. Randomisation of housing and outcome assessments was not described in any of the reports.
Three studies reported the outcomes selectively [26–28] whereas the remaining studies reported all described outcomes. Five studies reported on mortality or adverse events of the drugs [17, 26, 34–36].
Nine reports mentioned that funding was provided by public institutions [17, 28–33, 37, 39] and the authors of seven studies declared that they had no conflicts of interest.
In total, there were 12 studies eligible for meta-analysis; for the five remaining studies there were missing data we could not obtain [26, 28, 34, 40] or the data were not independent [32].
Amphetamine
One study tested amphetamine on 95 mice compared to 24 controls; there were no differences between the groups for number of ejaculations and ejaculation latency [39], see Fig. 1.
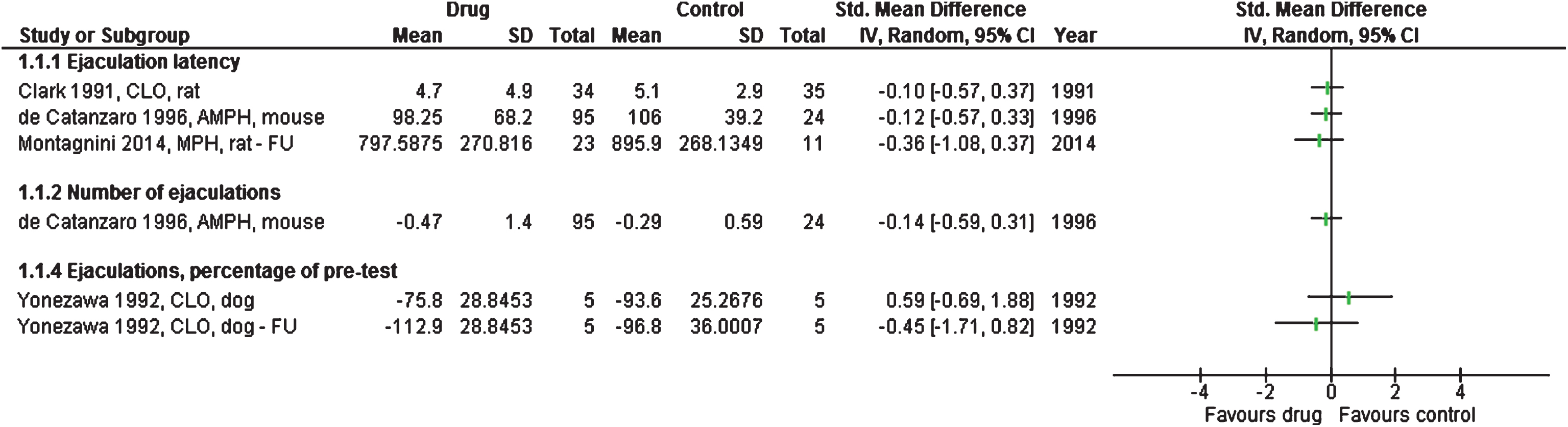
Studies or subgroups – as listed by first author, publication year, short for the drug used, species, and FU if feasible – showing ejaculations as standardised mean difference and 95% confidence intervals. Total is number of animals per group. FU – follow-up, CLO – clonidine, AMPH – amphetamine, MPH – methylphenidate.
In one study [34], ten male clonidine-treated rats were tested for their ability to impregnate two female rats each, compared to four untreated rats. All control rats impregnated two dams each, but the treated rats failed to impregnate any of the female rats (P = 0.001 for difference between 4 of 4 and 0 of 10, our calculation). The rats were either unable to ejaculate (no sperm present in the vagina); or they were sterile (no embryos present in womb at day 12 after the encounters); or they did not approach the females (four cases). After a drug-free period of seven days, the treated male rats approached one female again; seven of 10 animals were able to impregnate one female rat each compared to all four control rats (P = 0.51, our calculation).
Another study found a similar effect [32]. Ten male rats treated with clonidine impregnated nine of 20 females, and ten control rats impregnated 16 of 20 females (P = 0.05 when disregarding that the observations were not independent, as each male rat approached two females).
For one study with four dogs in each group, the ejaculation score (from 0 to 3), elicited by manual stimulation by the experimenter one hour after a clonidine injection, was lower for treated dogs than for controls [40]. All control dogs scored the maximum of 3 (ejaculation happened), while the mean score for treated dogs was only 0.65 (SD 0.78) (P = 0.004, sign test of 0 vs 8, our calculation), see Fig. 1.
Sperm transit time through the epididymis, which is the time it takes the sperm to become mature, was significantly shorter in clonidine-treated rats than in controls (mean difference 2.1 days; 1.2 to 3.0; 6 treated vs 6 controls) [30], see Fig. 2.
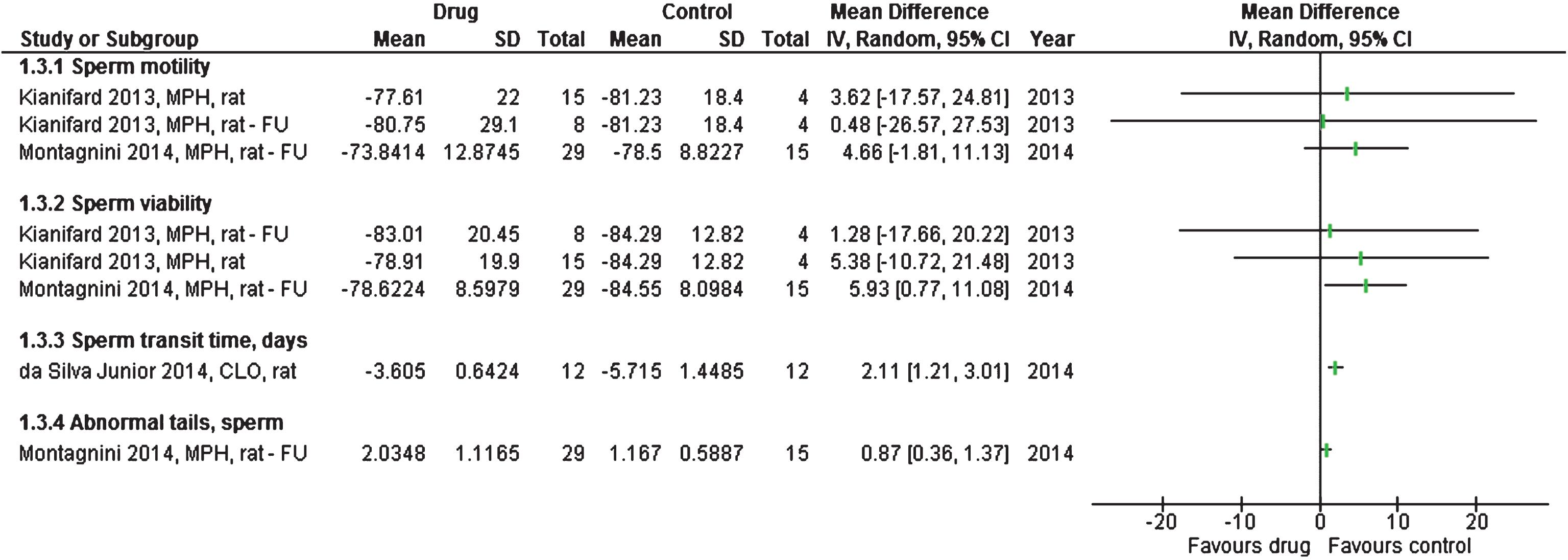
Studies or subgroups – as listed by first author, publication year, short for the drug used, species, and FU if feasible – showing data for spermatozoa as mean difference with 95% confidence intervals. Total is number of animals per group. FU – follow-up, CLO – clonidine, MPH – methylphenidate.
We could not extract any meaningful data from another clonidine study because the number of rats was not mentioned in the results section [26]. Half of 40 female rats treated with clonidine before and during pregnancy were sterile but the results for the control group were not mentioned [26].
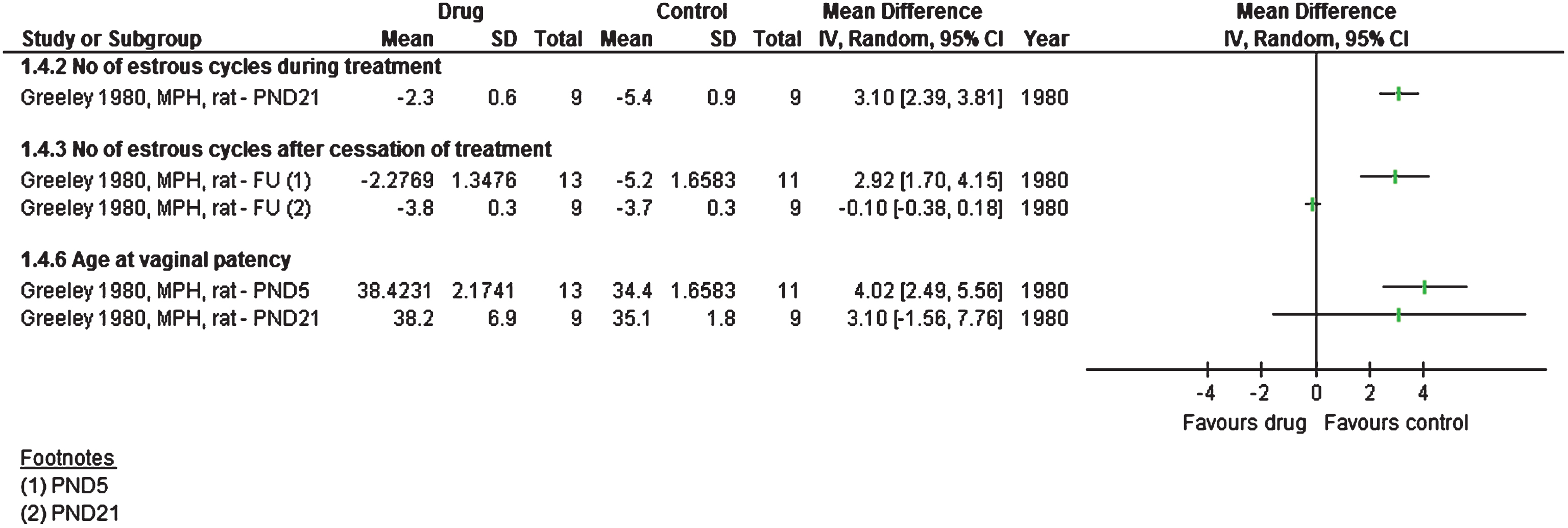
A rat study found that vaginal patency was delayed when treatment started five days after birth (mean difference 4.0 days; 2.5 to 5.6; 13 treated vs 11 controls) [31]. When treatment started 21 days after birth, there was an insignificant delay (mean difference 3.1; – 1.6 to 7.8; 9 treated animals vs 9 controls). Another study with ten rats in each group found vaginal opening to be delayed for a minimum of six days; vaginal opening remained poor throughout the entire 30-day study for six of the treated rats [28]. In contrast, two other studies, with 42 vs 28 [37] and 30 vs 10 rats [27], found no difference between groups for age at vaginal opening.
Female rats showed abnormal estrous cycling (counted as number of days per cycle) when the treatment started at postnatal day 21; the number of cycles was halved for treated rats (mean difference 3.1 days; 2.4 to 3.8; 9 treated rats vs 9 controls) [31]. After a 30-day drug-free period, estrous cycling did not normalise for rats starting treatment early, at postnatal day 5 (mean difference 2.9; 1.7 to 4.2; 13 treated vs 11 controls), but it did normalise for rats that started treatment at postnatal day 21 (9 rats per group).
Another study, in which treatment started at postnatal day 21, found no differences in estrous cycling patterns (42 vs 28 rats) after 40 days of treatment and a 15-day drug-free follow-up [37], whereas a study, in which treatment started at postnatal day 28 and continued for four weeks, with 10 rats per group, reported that all the treated rats showed impaired estrous cycling [28].
Fertility rates were 42% for six methylphenidate-treated mice approaching 3 female mice each and 95% for three untreated animals (counted as number of embryos per corpora lutea) [38]. On average, the gestation period was significantly longer in 100 treated rats compared to 24 control rats, but the difference was only half a day [35]. The results are shown in Figs. 3 and 4.
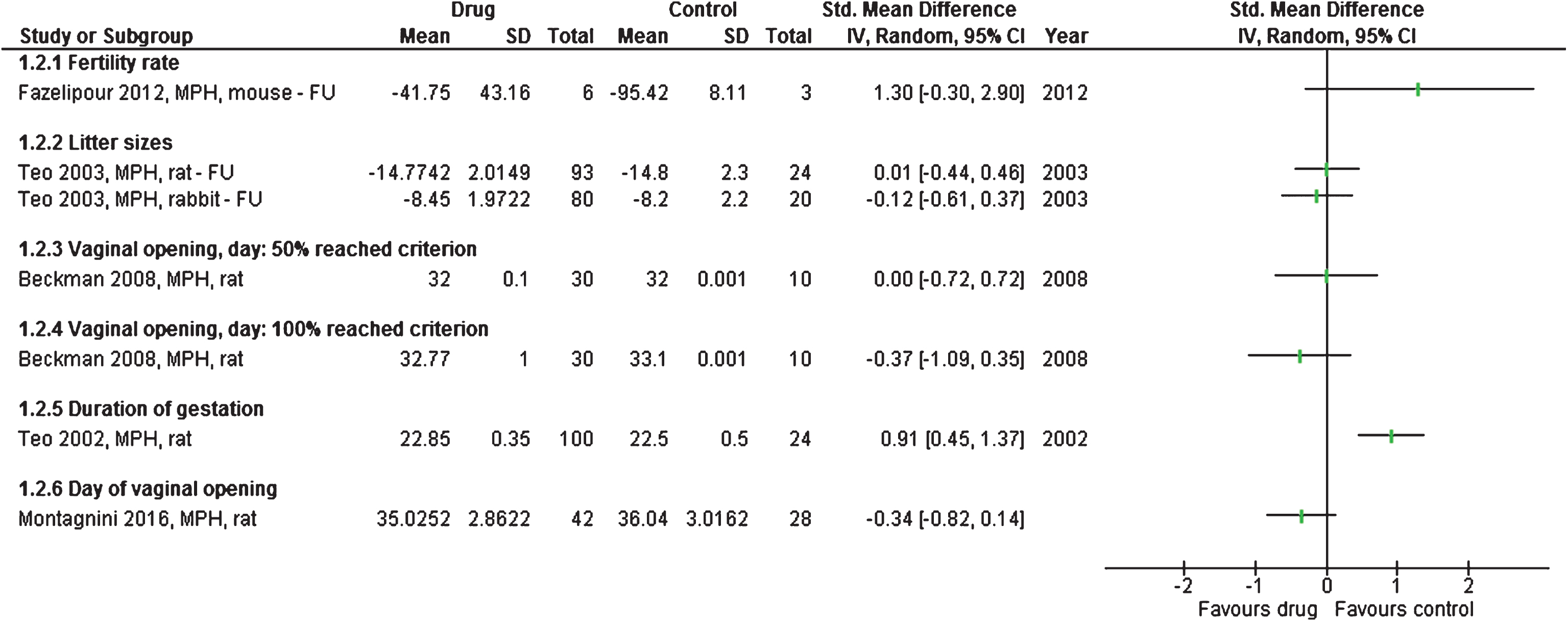
Studies or subgroups – as listed by first author, publication year, short for the drug used, species, and FU if feasible – showing pregnancy and labour outcomes as standardised mean difference with 95% confidence intervals. Total is number of animals per group. FU – follow-up, MPH – methylphenidate.
Studies or subgroups – as listed by first author, publication year, short for the drug used, species, and FU if feasible – showing reproductive ability as mean difference with 95% confidence intervals. Total is number of animals per group. FU – follow-up, MPH – methylphenidate.
For the pregnancies that led to successful deliveries, no differences were seen in number of stillborns, offspring or exterior malformations [27, 35, 36], see Fig. 5.
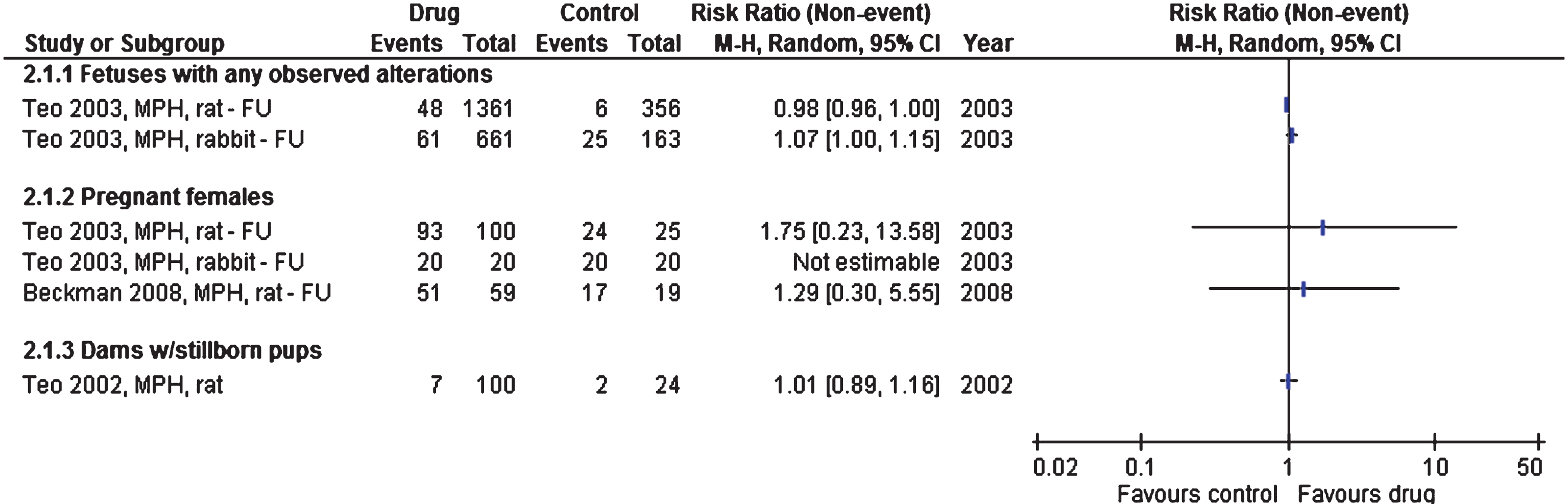
Studies or subgroups – as listed by first author, publication year, short for the drug used, species, and FU if feasible – showing binary outcomes as the risk ratio with 95% confidence intervals. Total is number of animals per group. FU – follow-up, MPH – methylphenidate.
Abnormal tail development occurred in 2.0% of sperms in 29 methylphenidate-treated rats and in 1.2% of 15 controls (mean difference 0.9%; 0.4 to 1.4) [33]. Forty days after end of treatment, 79% vs 85% of the sperms were viable (mean difference 6%; 1 to 11), see Fig. 2. There was no difference in number of ejaculations [33], see Fig. 1.
In a study of 15 vs 8 rats receiving methylphenidate or placebo for 11 weeks, there were only trivial differences between motility and viability of the sperms [17], see Fig. 2.
We found that clonidine and methylphenidate impaired reproduction in animals. These harms might be reversible if the treatment is not started early in life. We did not find an effect on number of stillborns, number of offspring or visible malformations. However, one of our included studies assessed the newborns’ skeletons and found that the pelvis, pubis and sternum were incompletely ossified compared to controls [36].
Strengths and limitations
It was a strength that nine of the 17 studies were publicly funded and that we only included studies where the animals had not been primed in any way, as priming might make the results difficult to interpret.
Fifteen of the 17 studies were placebo-controlled. This usually implies blinding of the observers, but this information was only given in detail for one study. No studies mentioned whether the caregivers and other personnel involved in the study were blinded, but in animal studies, the caregiver and the observer are sometimes the same person. This may mean that use of a placebo has not necessarily led to blinding since there could be differences in, for example, the colour of the food.
No studies adequately described that the animals were randomised in the three ways advised by SYRCLE [20]. For our study, the possible lack of random outcome assessments may have had little influence, as few of our outcomes were subjective or addressed behavior, which could vary over the day. The possible lack of random housing could have influenced the neuroendocrine system and thereby the results.
Eleven studies were published before the introduction of the ARRIVE guidelines in 2010 [42]. The guidelines have not yet been sufficiently implemented, as researchers, journal editors and ethical committees do not focus enough on the importance of blinding and randomisation for minimizing bias [43]. Animal research is often the basis for studies in humans and they therefore need to be performed with the same rigour as randomised trials in patients in order to reduce research waste because of false leads.
The studies were rather small and highly heterogeneous, using different species, strains, age at start of intervention, treatment periods, route of administration, test settings and outcome measures. In the two studies of methylphenidate that did not find differences between treated groups for age at vaginal opening, the drug was given as oral gavage [27, 37], whereas it was administered subcutaneously in the two studies that did find a difference [28, 31]. The significant findings might be due to the animals receiving a higher dose. Delayed vaginal opening is similar to a late entering into puberty in humans [44].
It can be discussed to which extent the studies reflected clinical use of ADHD drugs [45]. In some studies, the treatment was started shortly after birth, which does not happen in humans. On the other hand, the length of treatment was short compared to the clinical setting. The results we found might therefore have both over- and underestimated the problems with these drugs.
Comparison with data from humans
Data on reproductive parameters in humans when treated with ADHD drugs is sparse [12–16] and we have not found any long-term data that could elucidate whether ADHD drugs irreversibly impair the reproductive system. Children are being treated with ADHD drugs at an ever earlier age and sometimes continue with the drugs for decades although these long-term treatments are based on short-term studies. In a large Cochrane review of methylphenidate in children, the median length of treatment for parallel group trials was only 49 days [7]. This review found that all trials were of high risk of bias [7], and only 5% of trials reported on serious adverse events while 14% reported on non-serious adverse events. In addition to harms being seriously underreported, there is a lack of long-term trials with a drug-free follow-up in both animal and human research.
It is very worrying that the large Multimodal Treatment Study (MTA) in children after 16 years of follow-up reported that people who continued to take methylphenidate lost 5 cm in height compared to those who only took a negligible amount of drug after the 14-months randomised phase was over [46].
Clonidine is an antiadrenergic agent approved for treatment of hypertension in 1974 [47]. It is well known that hypotensive agents can cause sexual dysfunction [48, 49]; to prescribe clonidine to children and adolescents may have a similar negative impact.
Implications
We consider it likely that the harms to the reproductive system seen in animals can also occur in humans. This possibility should be studied in observational studies and also in the large MTA study.
Conflict of interest
None to report.
Funding
Faculty of Health and Medical Sciences, University of Copenhagen, Denmark.
Footnotes
Appendix A:
Appendix B:
Appendix C: Literature search – flow chart
*Additional records identified through “related papers” function and references of screened papers.
Acknowledgments
We thank Marie Solitander Bohlbro and Anders Klokmose Sørensen, PhD students at the Nordic Cochrane Centre, for comments on the manuscript.
