Abstract
Pathologies characteristic of Alzheimer’s disease (i.e., hyperphosphorylated tau and amyloid-β (Aβ) plaques), cardiovascular disease, and limbic predominant TDP-43 encephalopathy (LATE) often co-exist in patients with Parkinson’s disease (PD), in addition to Lewy body pathology (α-synuclein). Numerous studies point to a putative synergistic relationship between hyperphosphorylation tau, Aβ, cardiovascular lesions, and TDP-43 with α-synuclein, which may alter the stereotypical pattern of pathological progression and accelerate cognitive decline. Here we discuss the prevalence and relationships between common concomitant pathologies observed in PD. In addition, we highlight shared genetic risk factors and developing biomarkers that may provide better diagnostic accuracy for patients with PD that have co-existing pathologies. The tremendous heterogeneity observed across the PD spectrum is most likely caused by the complex interplay between pathogenic, genetic, and environmental factors, and increasing our understanding of how these relate to idiopathic PD will drive research into finding accurate diagnostic tools and disease modifying therapies.
Parkinson’s disease (PD) is neuropathologically characterized by deposits of α-synuclein (α-syn) as Lewy bodies (LB) in the neuronal somata and as Lewy neurites (LN) in the axons and dendrites. There is evidence that in PD pathology originates in the enteric nervous system and propagates to the brainstem and follows a stereotypical pattern of progression throughout the brain. Limbic structures are affected next, and finally in end stages of the disease, there is widespread distribution of α-syn deposits throughout the cerebral cortex, where LB pathology would represent the neuropathological correlate of clinical diagnoses of Parkinson’s disease dementia (PDD) or dementia with Lewy bodies (DLB) [1]. However, the site of origin of α-syn deposition is of continuous debate as alternative hypotheses propose α-syn originates in the central nervous system rather than the enteric nervous system or independently in the olfactory bulb and progresses to interconnected brain regions [2, 3]. More recently neuropathological evidence has been published representing caudo-rostral and amygdala-based progression, representing the body-first and brain-first hypothesis with differing clinical trajectories and genetically distinct progression patterns [4, 5]. Similar to other neurodegenerative pathologies LB pathology rarely exists in isolation and already in 2008, it could be shown that 53.3% of neurodegenerative disease cases have lesions associated with more than one neurodegenerative condition [6]. This is particularly evident in PD as pathology associated with Alzheimer’s disease (AD), argyrophilic grain disease, vascular pathology, and to a lesser extent Creutzfeldt Jacob disease, and progressive supranuclear palsy, were observed at autopsy as additional pathologies [6]. Moreover, a recent large autopsy study utilizing 1,647 postmortem brains, found up to seven concurrent pathologies, resulting in 161 pathological combinations [7]. Another study focusing on concomitant pathology in Parkinsonian disorders demonstrated 38% of neuropathologically confirmed PD cases had additional pathologies associated with AD, progressive supranuclear palsy, argyrophilic grain disease, cerebral white matter rarefaction, and cerebral amyloid angiopathy (CAA) [8].
Different hallmark lesions of age associated neurodegenerative diseases (i.e., LB/LN, amyloid-β (Aβ), tau, and TDP-43) are initially deposited in different specific regions in the brain and progress though the brain in a stereotypical manner (Fig. 1) [1, 9–12]. There is growing evidence that a synergism exists between pathological proteins, which contributes to an accelerated disease trajectory observed in these cases. Since the observation was made that multiple pathologies can alter the prognosis, investigations into the potential mechanisms underlying this could be crucial in designing disease modifying therapies. This review will highlight the most common concomitant pathologies observed in PD cases, putative interactions between α-syn and tau, Aβ, cerebrovascular pathology, and TDP-43, and the clinical consequences in idiopathic and familial PD.
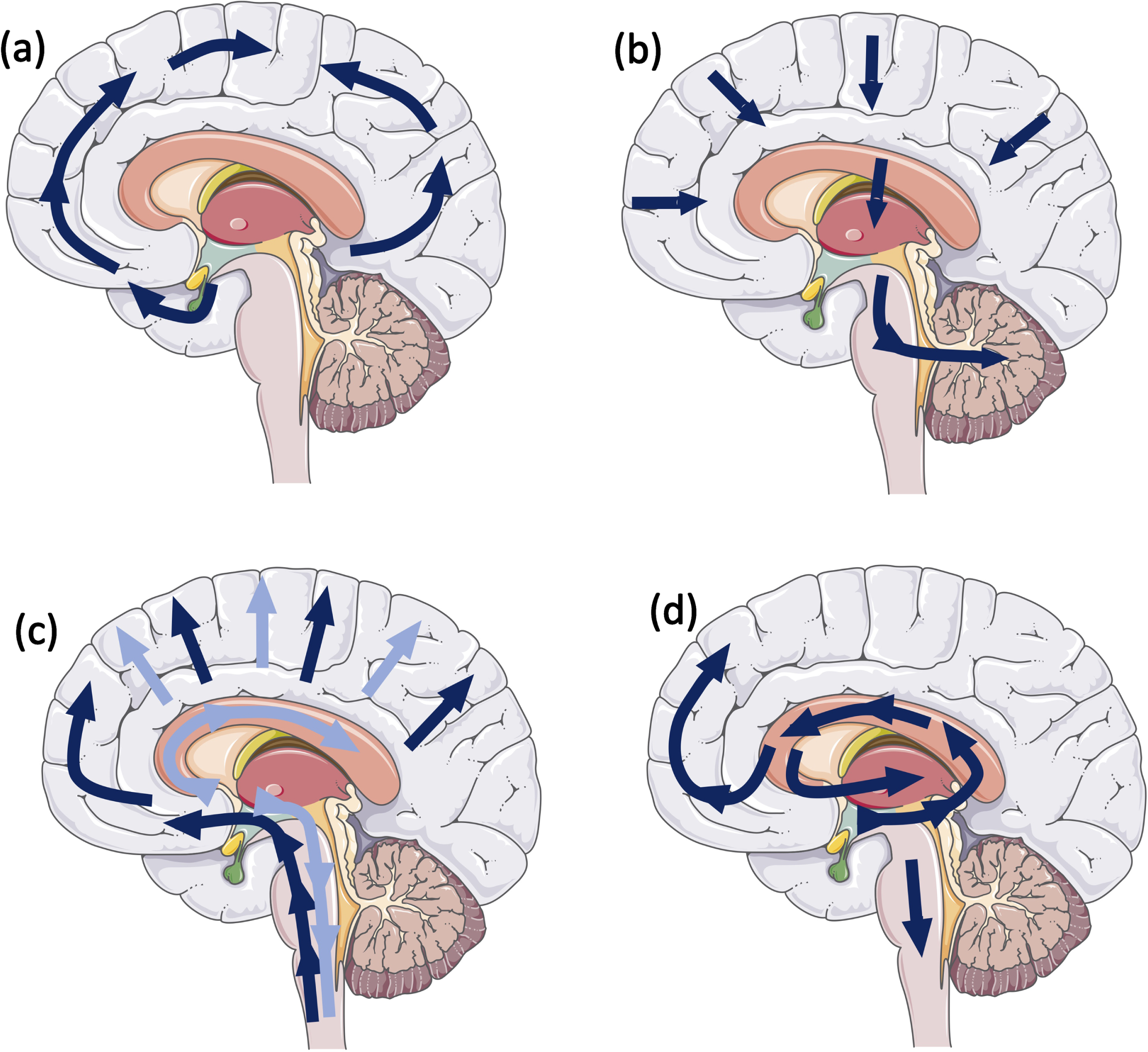
Schematic illustrating stereotypical progression for (a) tau pathology: Neurofibrillary Braak stages (9); (b) amyloid-β pathology: Thal phases (10); (c) α-syn: Braak stages (1), dark blue arrows, Lewy pathology consensus criteria, light blue arrows (12); and (d) Limbic predominant age-related TDP-43 (11).
AD-RELATED PATHOLOGY
Tau
Both tau and Aβ are present at postmortem examination in PD patients, with a study from 2019 classifying 0% PD cases with a neurofibrillary tangle Braak stage of 0, 33.4% of cases with Braak stage I, 33.3% of cases with Braak stage II, and 33.3% of cases with Braak stage III [9, 13]. Neuropathological and experimental studies have suggested a direct synergistic interaction between AD pathology and α-syn (Fig. 2). In vitro studies using confocal microscopy and FRET based techniques found tau co-localizes and interacts with α-syn aggregates, changing the pattern of α-syn aggregation, reducing the size and increasing the number of aggregates [14]. Furthermore, α-syn has been highlighted as a candidate capable of polymerizing tau and initiating fibrilization. Once tau fibrilization is initiated, tau and α-syn synergistically interact to promote the polymerization of each other into amyloid fibrils [15, 16]. In a mouse model of synucleinopathy (TgA53T), tau is required for synaptic and memory deficits, and removal of endogenous mouse tau expression ameliorated cognitive and synaptic dysfunction [17]. However, in contrast to this, several studies have shown cognitive impairment in tau knock out mouse lines [18, 19]. Therefore, as it seems an excess reduction in tau protein results in motor and cognitive impairment, treatments that may restore tau function rather than targeting tau expression directly may be a safer therapeutic strategy. The in vivo relevance of a putative relationship between tau and α-syn was grounded further in animal studies. Tau inclusions are observed in 50% of mice expressing Ala53Thr mutant human α-syn, with the development of α-syn positive inclusions that led to motor dysfunction [15]. Supporting evidence demonstrated hyperphosphorylated tau deposits accumulate in transgenic mice overexpressing human α-syn under the PDGF promoter, which increases in an age-dependent manner [20, 21]. Transgenic animal studies have also demonstrated increased accumulation of tau aggregates following simultaneous inoculation of α-syn mouse pre-formed fibrils and AD lysate-derived tauseeds [22].
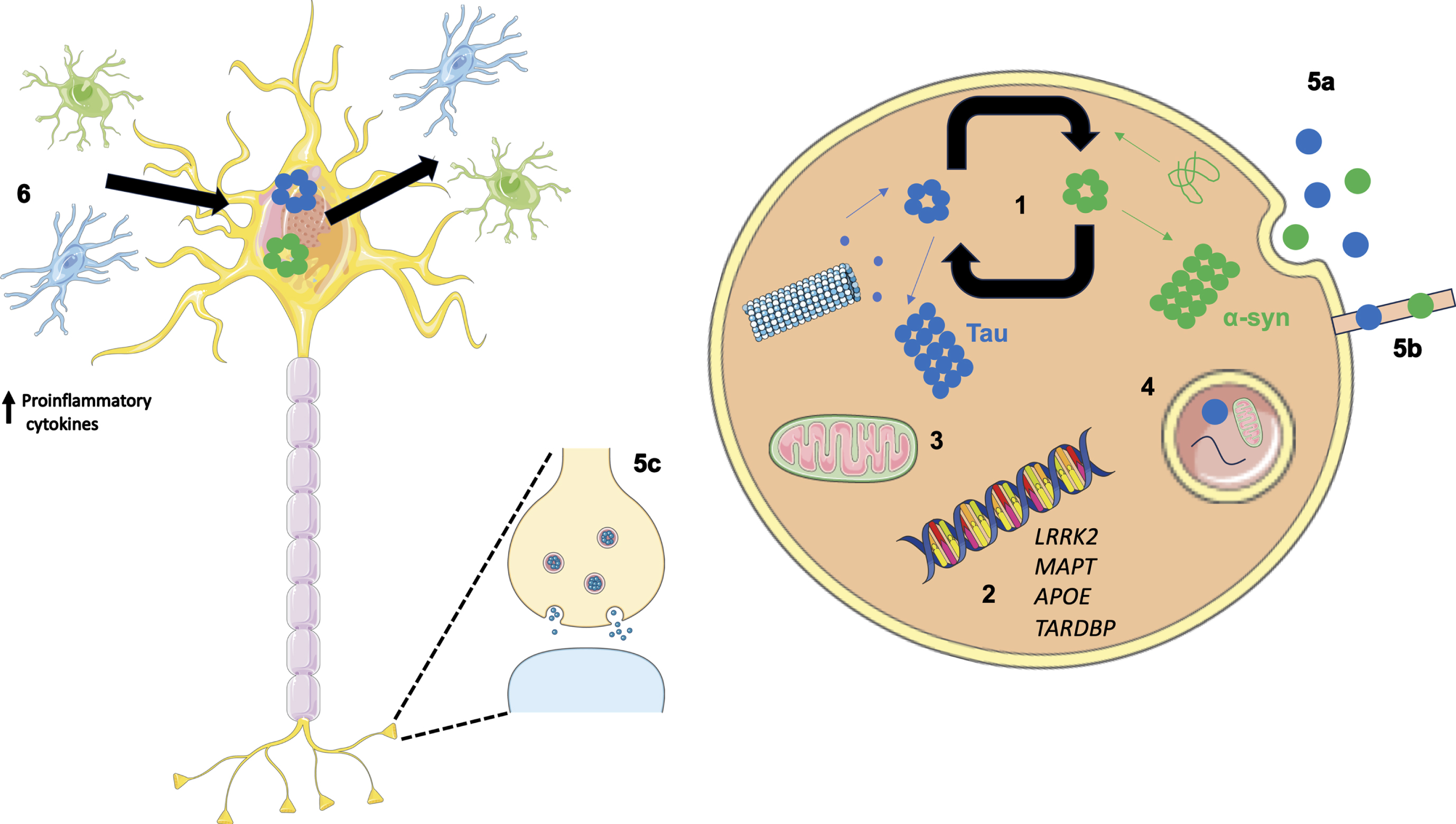
Overview of potential mechanisms illustrating how co-pathologies may arise and interact. (1) Tau and α-syn monomers assemble into oligomers followed by larger aggregates. Intracellular tau and α-syn have been shown to interact and promote the fibrilization of each other. (2) Genetics can play a role in the relationship between tau and α-syn as both protein aggregates have been observed in LRRK2 carriers. In addition, genome wide association studies have revealed the MAPT locus is associated with increased risk in Alzheimer’s disease (AD) and Parkinson’s disease (PD). APOE ɛ4 is a well-known risk factor for AD; however, when APOE ɛ4 is expressed in α-syn mice motor dysfunction and general neurodegeneration is observed. TARDBP mutations are associated with motor neuron disease; however, the presence of a TDP-43 mutation (p.N267S substitution) has been found in a patient clinically diagnosed with PD. (3) Aβ and α-syn exist in different subcellular compartments; however, in pathological states both have been detected in mitochondria, lysosomes, and autophagosomes. (4) Failing cellular mechanisms can also contribute to the increased accumulation of pathologies observed in neurodegenerative diseases such as PD, as disrupted autophagy has been shown to impact α-syn, Aβ, and tau metabolism and clearance from the brain. (5) α-syn and tau are thought to propagate in a manner reminiscent of prions increasing the likelihood of aggregating in the same cell. Methods associated with this include exocytosis (5a), formation of tunnelling nanotubes (5b) and via synapses (5c). (6) Oligomeric species of tau and α-syn are capable of eliciting and inflammatory response from astrocytes and microglia resulting in an increase in proinflammatory cytokines. This in turn has a feed-forward affect where the upregulated immune system can lead to further protein aggregation and neuronal damage.
Uncovering the pathomechanisms that could potentially drive the accelerated decline observed in patients with considerable levels of α-syn pathology and high AD neuropathologic change would greatly benefit the design of disease modifying therapeutics. Indeed, several groups have conducted studies to spatially localize intracellular proteins α-syn and tau in pathological protein aggregates. Arima and colleagues reported tau can be incorporated into filaments in brainstem LBs [23]. Further to this, evidence from electron microscopy experiments demonstrated that α-syn selectively labelled 9- to 13-nm-thick straight filaments, which are analogous to LB filaments and AT8 antibody for hyperphosphorylated tau recognized twisted tubules with 80–100 nm-interval constrictions which correspond to the twisted tubules seen in AD [24]. Further studies in the brainstem structures indicate that, in particular, phosphorylated tau antibodies, preferentially labelled LBs compared to non-phosphorylated antibodies [25]. Colom-Cadena and colleagues also reported co-localizations between tau and α-syn extended to limbic regions [26]. Although α-syn is primarily located at the axonal terminal, during axonal transport it is also present at low levels in the axonal processes. On the occasion of aberrant dissociation of tau from the microtubule in disease states, this may give rise to tau-α-syn interactions and subsequent increase in aggregated forms of both proteins [20]. Increased tau phosphorylation at Ser396 has been reported in synapse-enriched fractions of PD brains in addition to phosphorylation α-syn Ser129. This suggests synapses are a site for putative protein interactions [27]. Further to this, in vitro NMR experiments have demonstrated monomeric α-syn and tau variants Tau23 and K19 synergistically promoted amyloid fibrilization, whereas S129D (a mimic of C-terminal phosphorylation of α-syn phosphorylated at Ser129) significantly enhanced the activity of α-syn in facilitating Tau23 and K19 aggregation [28]. Taken together these studies provide convincing evidence for the mutual promotion and aggregation of tau and α-syn to further advance and aggravate neurodegenerative processes. On the other hand, some studies provide evidence against an interaction between the proteins. Benussi and colleagues reported the P301L tau mutation prevented the interaction between tau and α-syn leading to an increase in unbound α-syn [29]. However this may lead to an increased burden of α-syn as a result of self-polymerization [30]. Furthermore, Esposito and colleagues demonstrated α-syn and its disease-related mutants interact differently with tau as in cell culture the interaction between α-syn and tau is decreased by the A30P mutation unlike the A53T and E46K mutations [31]. Finally Morris and colleagues demonstrated that reduction of tau does not protect mice against motor deficits and pathological alterations in the 6-OHDA mouse model or mice expressing human wildtype α-syn (hSYN) [32]. These studies implicate the tau-α-syn interaction is complicated and further study is required to elucidate the crosstalk between the proteins.
We have shown that the stereotypical patterns of pathology can be altered in Lewy body disease (LBD) cases with concomitant AD pathology [33], potentially sparing the hippocampus in a small number of cases [34]. The mechanisms by which tau and α-syn propagate to different brain regions are under debate, with increasing evidence suggesting they progress from neuron-to-neuron in a manner reminiscent of prions [35–37]. Evidence from case studies involving the transplant of fetal graft tissue into PD patients demonstrated tau and α-syn proteins at much denser concentrations compared to surrounding host tissue [38]. The trauma of the transplantation itself could leave the graft vulnerable to pathogenic insults or create a suitable environment to stimulate intracellular progression after initial seeding. Mechanisms associated with prion like spread include exocytosis, formation of tunnelling nanotubes, and via synapses (Fig. 2).
Aβ
Aβ plaques are not as common in PD; one study found 55.6% showing any Aβ plaques (22.2% Thal phase 2, 22.2%, Thal phase 3, and 11.2% Thal phase 4) and none showed neuritic plaques [10, 13]. Regional prevalence of Aβ pathologies differ in PD cases compared to PDD. Temporal cortex is significantly more affected in PDD and frontal cortex, caudate nucleus, and putamen trending towards significance [39]. Aβ burden as such was significantly increased in parietal, and occipital cortices, striatum and nucleus accumbens in PDD compared to PD [39]. Associations between Aβ and α-syn have been observed in human autopsy tissue. A neuropathological study by Lashley and colleagues found a significant correlation between Aβ plaque load and LB density in postmortem tissue from 40 PD cases and 20 controls [40]. This was further supported by Swirski and colleagues as an association was established between soluble and insoluble Aβ with insoluble levels of α-syn phosphorylated at Serine 129 [41]. Additionally, transgenic mice expressing Aβ and α-syn develop severe cognitive and motor deficits compared to single transgenic animals [42]. At a cellular level exposure of SH-SY5Y cells transfected with wild-type human SNCA cDNA to aggregated Aβ42 significantly increased the phosphorylation of α-syn at the Serine 129 position [41]. While α-syn fibrils are capable of catalyzing the heterogeneous nucleation of Aβ42 aggregates [43]. As the majority of Aβ is visualized as extracellular plaques mechanisms of the putative interaction between Aβ and α-syn is an interesting point for debate. A study conducted by Friedrich and colleagues in 2010 demonstrated internalized Aβ contributes to the formation of extracellular plaques [44]. Aβ becomes sorted to multivesicular bodies and detrimental to cell survival. Ultimately cells die and intracellular structures including intracellular amyloid species are released into the extracellular space [44]. Interestingly mice expressing neuronal Aβ and α-syn develop severe cognitive and motor deficits and showed prominent age-dependent degeneration of cholinergic neurons and presynaptic terminals compared to single transgenic animals [42]. In healthy neurons, Aβ and α-syn do not exist in the same subcellular compartment therefore limiting the potential for direct interaction. However, in pathological states both have been detected in mitochondria, lysosomes and autophagosomes (Fig. 2) [45, 46]. These data provide supporting evidence of a putative synergistic relationship between the proteins.
CEREBRAL AMYLOID ANGIOPATHY
In addition to extracellular plaques, Aβ can be deposited in the cerebral vessels including arteries, arterioles, meninges, capillaries and very rarely veins, a condition which is termed CAA. Although most commonly in AD (20–100%) [47–50], CAA has been observed in LBDs including PD [51, 52]. CAA exists in 2 forms: type 1, involvement of capillaries, with or without arterial involvement, and type 2, where arteries are affected without capillary involvement [53]. Similar to the other AD-related pathologies CAA in PD cases is less common compared to other LBDs [51], and severity scores are lower in PD cases. Postmortem studies have indicated that type 1 CAA, in particular, is associated with the presence of dementia in LBDs which could in part account for PDD and DLB having an increased presence of type 1 CAA compared to PD without dementia [54]. The prevalence of CAA is highest in the occipital lobe [55], and a neuroimaging study has also indicated this region is susceptible to cerebral microbleeds in PD cases that have developed dementia [56], suggesting that CAA could contribute to the vascular impact of cognitive impairment in PD.
TDP-43
TDP-43 inclusions are the pathological hallmark of amyotrophic lateral sclerosis (ALS) and a subtype of fronto-temporal lobar degeneration (FTLD-TDP) that is tau negative with neurons and sometimes glia positive for ubiquitin (FTLD-U). However, TDP-43 positive inclusions have been identified in aging and age-associated neurodegenerative diseases, and this has been referred to as limbic-predominant TDP-43 encephalopathy (LATE) [11]. Although these inclusions are phosphorylated and considered pathological, they do not follow the stereotypical pattern of deposition of TDP-43 observed in ALS and FTLD-TDP [57]. In LATE, TDP-43 first appears in the amygdala then progresses to the limbic areas, followed by the neocortex (Fig. 1) [58, 59]. The prevalence of LATE in PD is up to 7%, while in PDD 19% of cases are affected [60]. Within LBD cases there was a relationship between LATE and more severe symptomatology suggesting TDP-43 may have co-morbid effects in the neurogenerative pathways of LBDs. Although, the precise mechanisms behind this are not yet known as TDP-43 and α-syn are not co-localized in LBs; however, they were occasionally colocalized within dystrophic neurites [60]. Nonetheless, in vivo and in vitro experiments suggest the existence of a synergistic relationship between the pathological protein aggregates. When concomitantly over expressed in mice, TDP-43 and mutant α-syn synergistically induced dopaminergic neurodegeneration [61]. Wild type mice injected with α-syn fibrils resulted in dot-like inclusions of TDP-43 [62]. In addition, SH-SY5Y cells double infected with α-syn and TDP-43 lacking nuclear localization and incubated with synthetic α-syn fibrils resulted in inclusions positive for phosphorylated TDP-43 [63]. Interestingly, TDP-43 and α-syn have been found to be co-localized in cases with a G51D SNCA mutation [64] and in glial cytoplasmic inclusions in multiple system atrophy, another synucleinopathy [65].
INDIRECT INTERACTIONS
Inflammation
Neurodegenerative diseases are multi-factorial disorders with numerous pathological mechanisms contributing to the clinical phenotype. Neuroinflammation has been associated with several neurodegenerative diseases such as PD, AD, and FTLD with growing evidence suggesting the innate and adaptive immune system can contribute to cognitive decline in dementia [66–68].
Oligomeric species of proteins associated with neurodegeneration are capable of activating an inflammatory response as oligomeric α-syn has been shown to be an endogenous agonist for Toll-like receptor 2, which activates an inflammatory response in microglia [69]. Oligomeric tau has been shown to co-localize with astrocytes, microglia, and HMGB1, a pro-inflammatory cytokine [70]. Whereas soluble Aβ oligomers caused an increase in pro-inflammatory cytokines via a sensitized response of Toll-like receptor 4 over a period of time [71]. Ultimately activation of the immune response may initiate a feed-forward cycle. Upregulated inflammatory responses may lead to neuronal damage and protein accumulation, and thus more neuroinflammation (Fig. 2).
Triggering receptor expressed on myeloid cells-2 (TREM2) is one of the most highly expressed receptors in microglia. It suppresses cellular activation and inhibits cytokine production in response to both Toll-like receptor 2 (TLR2) and Toll-like receptor 4 (TLR4) stimulations [72]. In addition, it has been found to inhibit microglia mediated pro-inflammatory responses induced by apoptotic cells [73]. Defects in the TREM2 gene have been implicated in several neurodegenerative diseases, for example the TREM2 p.R47H substitution is a risk factor for both PD and AD [74, 75].
Impaired protein degradation
Failing cellular mechanisms can also contribute to the increased accumulation of pathologies observed in neurodegenerative diseases such as PD. Disrupted autophagy has been shown to impact α-syn, Aβ, and tau metabolism and clearance from the brain (Fig. 2). For example, knock out mice with depletion of Atg7 (an autophagy gene encoding E1-like enzyme in the two ubiquitin-like conjugation systems that are essential for the autophagosome biogenesis) leads to presynaptic accumulation of α-syn [76] and inclusions positive for hyperphosphorylated tau [77]. While knockdown of Atg7 results in an accumulation of Aβ in the Golgi [78]. Conversely abnormal protein accumulation has also been shown to cause disruptions in autophagy, for example α-syn compromises autophagy via Rab1a inhibition [79]. In addition, as microtubule stability and cytoskeletal elements are required for the formation and maturation of autophagic vacuoles [80], dissociation of tau from the microtubules and subsequent disruption of microtubule-dependent transport may result in further impairments autophagy and promotion of tau pathology. It stands to reason that if autophagy is impaired by the presence of one neurodegenerative pathology, this will affect the ability to carry out the fundamental task of degrading other aggregate prone misfolded proteins and will result in accumulation of numerous protein aggregates in the brain parenchyma.
CHALLENGES WHEN INVESTIGATING PROTEIN INTERACTIONS
While human post-mortem tissue provides the perfect cellular environment to study mixed pathologies in neurodegenerative diseases, attempting to understand disease mechanisms particularly early events in end-stage patients is difficult. Therefore, a lot of work in this field has employed animal and cellular models to investigate specific mechanisms and pathways. However, there are caveats with these experiments; firstly age-associated neurodegenerative disease are largely human-specific diseases and naturally animals do not develop the full neuropathological and clinical phenotype. Therefore, transgenic animals have been generated; however, while recapitulating specific neuropathological features of the human disease, transgenic animals cannot recapitulate the full spectrum of pathology seen in human idiopathic disease. Another means of inducing pathology in mice is via intracerebral injection of recombinant pre-formed fibrils and while some studies were able to show successful seeding and aggregation of tau pathology [81–83], others have failed to replicate this [84]. Interestingly in the same study the authors were able to demonstrate the formation of abundant tau inclusions in anatomically connected brain regions in non-transgenic mice when inoculating with AD derived tau fibrils, demonstrating different conformational features between synthetic and AD-derived brain seeds. Additionally recombinant tau fibrils have been shown to differ structurally from brain derived fibrils [85] and are therefore thought to have less translational impact compared to patient derived fibrils. Furthermore, as co-factors are necessary to form and stabilize tau fibrils [86, 87] and as cell-free assays are by design devoid of co-factors the results from these experiments should be interpreted with caution.
CEREBROVASCULAR DISEASE
In addition to proteinopathies, cerebrovascular pathologies (CVP) including lacunes, CAA, white matter lesions, old and recent ischemic infarcts and hemorrhages can present in patients with PD [88, 89]. In a large retrospective pathological study comprising 617 PD and 535 control cases, increased prevalence of CVP was observed in PD cases (44%) compared to age-matched controls (32.8%). However, in PD this included mainly mild to moderately severe lesions (26.8% and 10.6% vs. 20.9% and 6.5%, respectively), and only 6.8% had severe pathology compared to 5.4% in controls [90]. The relationship between motor impairment, vascular burden, and cognition in PD has been explored, and whole brain white matter lesion volume was associated with dementia in PD, freezing of gait and attention deficits [91]. Numerous neuroimaging studies have investigated the link between cognitive decline and CVP in PD, reporting mixed results. Some studies indicate CVP has a negative effect on cognition [92–96], while others have not been able to replicate this [97, 98]. Therefore, the clinical contribution of CVP to cognitive impairment in PD remains controversial. Nevertheless, the mechanisms behind a possible association have been investigated further: in a mouse model of α-synucleinopathy, Elabi and colleagues describe compromised blood-brain barrier integrity at early stages of disease, which is associated with dynamic alterations in vessel density and pathological activation of pericytes reflecting microvascular changes [99]. At 8 months, the density of microvessels is increased in transgenic animals compared to controls, then significantly decreased at 13 months. This could signify initial compensatory angiogenesis. Furthermore, in the human brain, using ultrasonographic examinations of extracranial vessels measuring intima-media thickness, the presence of atherosclerotic plaques, and intracranial vessels was observed. This suggests that comorbid atherosclerosis and subclinical impairment to vessels may contribute to mortality in PD [100].
In addition to studying the contribution of vascular lesions to the pathogenesis and clinical outcome of PD, the influence of vascular risk factors has been investigated. Hypertension, dyslipidemia, diabetes, and obesity are well-established risk factors for cognitive impairment and dementia in older adults. In a clinical study comprising 367 older adults with early PD, hypertension exerted a detrimental effect on memory and verbal fluency suggesting management of blood pressure and cardiovascular health may have implications in reducing cognitive decline [101]. In addition to baseline blood pressure, orthostatic hypertension has also been associated with increased dementia risk in PD [102]. While in the Parkinson Progression Markers Initiative study conducted by Chahine and colleagues a combined vascular risk score, comprising self-reported hypertension, diabetes, body mass index, and measured blood pressure was associated with an annual rate of change in global cognition [103]. Furthermore, in addition to influencing cognitive decline, the presence of at least two vascular risk factors was associated with gait impairment [104]. Diabetes mellitus (DM) has also been linked to cognitive changes in PD, and may induce a more aggressive phenotype as patients with DM demonstrated a faster motor progression and cognitive decline [105]. This is not surprising given evidence that suggests both PD and DM share common molecular pathways [106, 107].
CLINICAL AND BEHAVIORAL OBSERVATIONS
Limitations of studies investigating isolated pathologies are that they may not reflect the complex cellular environment and pathology loads evident in neurodegenerative diseases. To address this, Clinton and colleagues generated a mouse model that expressed Aβ, tau, and α-syn by crossing 3xTg-AD with mice the A53T mutation in α-syn. The subsequent DLB-AD mice developed all three pathological lesions at a significantly increased load compared to the single transgenic animals and demonstrated an accelerated cognitive decline [108]. In a large autopsy study comprising 670 cases spanning multiple neurodegenerative diseases, 63% of cases exhibited at least one additional pathology, which increased the odds of transitioning from mild cognitive impairment to mild dementia by 20-fold [109]. This is particularly evident in PD, as summated pathology scores of tau, Aβ, and α-syn were the best predictor of cognitive decline rather than one single pathological lesion [110, 111]. There is strong evidence that the burden of Aβ is a contributor to conversion to dementia in patients with PD, with total Aβ plaque burden being an independent predictor or shorter latency to dementia from onset of motor symptoms [112–116]. However, in a large multi-center cohort of patients diagnosed with synucleinopathies, both the progression of tau pathology and quantitative tau burden were the strongest predictors of a decreased interval between the onset of motor symptoms, the onset of dementia, and overall survival time [117]. In addition, shorter duration of parkinsonism prior to dementia is associated with higher CERAD scores [115]. Together these data suggest AD related pathologies are tightly linked to the accelerated clinical disease progression experienced by patients with PD. However, there have been mixed results when examining the relationship between concomitant LB pathology and AD clinical phenotype. Stern and colleagues did not find a relationship between the presence or absence of LB pathology and any clinical features in a cohort of fifty-one clinically and neuropathologically diagnosed AD cases [118]. In addition, Holtzer and colleagues found no relationship between quantitative indexes of LB abnormalities and clinical outcome [119]. To fully understand how the relationship between multiple pathologies contribute to the clinical picture in patients with dementia, it is important to understand which brain regions are affected by multiple pathologies; for example in PD, the hippocampus can be affected by numerous co-existing protein aggregates such as hyperphosphorylated tau, Aβ, α-syn, and TDP-43 (Fig. 3). Furthermore, identifying which pathologies are important in driving cognitive decline particularly in the early stages of disease or before the development of neuropathologically defined neurodegenerative diseases will inform drug discovery into disease modifying therapeutics. In a group of cognitively normal controls 63% of cases reported mixed pathologies, with tau and vascular lesions associated with poorer memory scores, tau with poorer general cognition and amyloid neuritic plaques with higher depression scores [120].
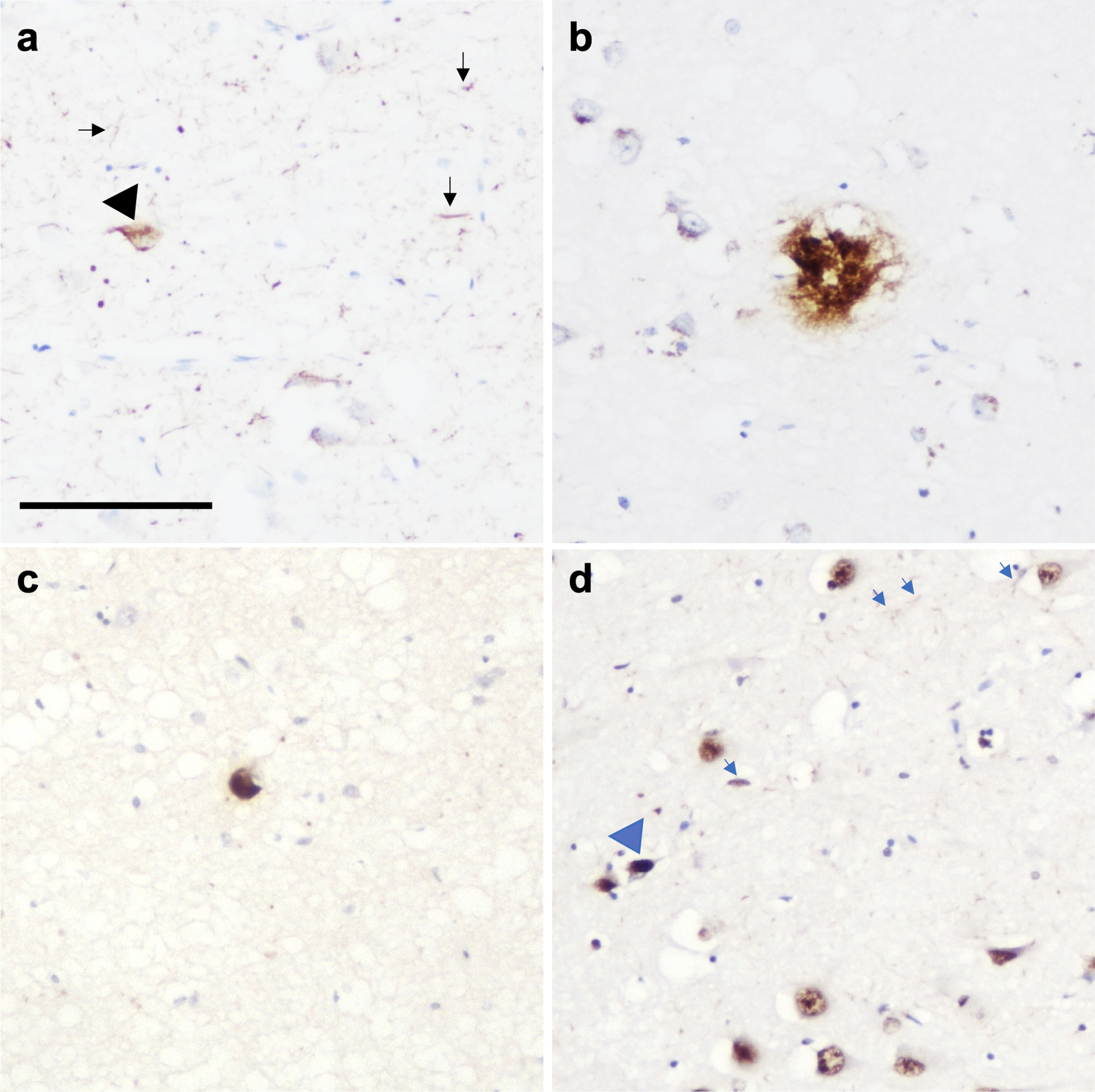
Photomicrograph demonstrating the presence of multiple pathologies in CA1 region of the hippocampus in a PD case. (a) Neurofibrillary tangles (black arrowhead) and neuropil threads (black arrows), (b) amyloid-β plaque, (c) Lewy body, and (d) TDP-43 neuronal intranuclear inclusion (blue arrowhead) and dystrophic neurites (blue arrows). Scale bar represents 100μm.
EFFECTS OF PATHOLOGICAL LOAD
The amount of pathological load has to be considered since control cases lacking neurological deficits can exhibit low levels of pathology, which are mainly restricted to smaller amounts of pathological load and are generally considered to be age associated. This includes mainly tau and Aβ; however, incidental LBs have been found in a subset of cases, in particular neocortical LBs in PD cases without dementia [121]. TDP-43 pathology has also been observed in control cases [122–124]. It seems unlikely that the presence of a small amount of concomitant pathology will elicit a significant decline in cognition when restricted to specific areas; therefore, when assessing the presence of multiple pathologies in the context of neurodegenerative diseases we must pay close attention to the quantitative pathological load of each protein within each region. This has been demonstrated in studies where the increased burden of α-syn in the parahippocampal gyrus and cingulate cortex allowed to distinguish PDD from PD [125, 126]. Similar results are seen with AD-related pathology as in a longitudinal population-based study of aging and dementia, quantitative measures of both tau and Aβ in the neocortex and hippocampus were strongly associated with the presence of dementia, whereas using semi-quantitative values this association was not observed [127]. It seems there may also be a similar relationship with concomitant pathologies in PD as both imaging and fluid biomarker studies demonstrated Aβ at baseline was not able to distinguish PD patients from normal controls. However, longitudinal data suggested Aβ was associated with a more rapid cognitive decline in patients therefore acting as a future predictor of increased amyloid burden rather than a current marker of cognitive decline when pathological burden becomes high enough to be detected [128]. Tau neuroimaging studies in LBD have reported variable findings as some studies report an increase in tau PET binding in PDD compared to PD without cognitive impairment [129, 130]. Additionally other studies that have found an increased tau PET signal in DLB compared to control participants [131–133]. However, a conflicting study conducted by Mak and colleagues were unable to replicate these findings, reporting [18F]-AV1451 binding is not elevated in DLB relative to normal aging [134]. Interestingly, it has been noted that the amount of [18F]AV-1451 binding in DLB and PDD is substantially lower than that observed in AD [131, 136]. This is in line with our neuropathological study which quantified tau pathology in cases that fulfilled neuropathological criteria for both AD and DLB, i.e., Braak stage VI, Thal phase 5, and neocortical LB disease. Even though all cases were Braak stage VI, those cases that present clinically with AD had significantly more tau pathology in cortical, subcortical, and brain stem regions compared to DLB, which was not observed using semi-quantitative staging criteria [34]. In the future implementation of quantitative methodologies will help disentangle the contribution of multiple concomitant pathologies to clinical and pathological phenotypes.
GENETIC CONSIDERATIONS
While the majority of PD cases are idiopathic, we have gained tremendous insights into disease pathogenesis from genetic mutations that cause PD and others that increase the risk of PD. The SNCA gene was the first recognized to cause autosomal dominant PD and subsequently there have been several missense mutations identified including A30P, E46K, H50Q, G51D, A53T, A53E, A53V, and A30G [137]. Several other genes have been convincingly associated with PD risk such as LRRK2, VPS35, PRKN, PINK1, GBA, and DJ-1. Interestingly, in mice overexpressing A30P, phosphorylated tau also develops in parallel with aggregates α-syn [138]. Furthermore in LRRK2 carriers, while α-syn pathology can been seen in 63.6% of individuals, tau pathology can be found in 100% of carriers, and is abundant in 91% of carriers in a small cohort [139].
Significant efforts have been focused on investigating the contribution of common variants to PD risk and age-at-onset. Several of these mutations, most commonly associated with other neurodegenerative diseases, have been observed in patients with a clinical diagnosis of PD such as APOE and MAPT. It is well known that APOE ɛ4 carriers have an increased risk of AD; however, two recent studies indicate that APOE4 can directly regulate levels of α-syn. Mice with different APOE genotypes were either genetically altered to express mutant α-syn or inoculated with α-syn preformed fibrils. The animals that expressed the ɛ4 allele exhibited the most severe α-syn pathology [140]. In a parallel study by Zhao and colleagues an adeno-associated virus gene delivery of α-syn was performed in human APOE-targeted replacement mice expressing APOE ɛ2, APOE ɛ3, or APOE ɛ4. In addition to expressing abundant α-syn, the mice expressing APOE ɛ4 had the most motor dysfunction and general neurodegeneration [141].
Genome wide association studies (GWAS) of 1,713 patients with PD and 3,978 controls in a cohort consisting of individuals from European ancestry, demonstrated a strong association at the MAPT locus [142], and this agreed with a study by Edwards and colleagues that combined data from two previous GWAS [143], confirming the MAPT region is a major gene whose common variants are influencing risk of PD. However, this failed to be replicated in a Japanese cohort [144] highlighting a population specific heterogeneity in PD.
Mutations in the TARDBP gene have been shown to cause motor neuron disease/ALS; however, in addition to TDP-43 aggregations in the brains of patients with PD mutations, Rayaprolu and colleagues report the presence of a TDP-43 mutation (p.N267S substitution), in a patient with clinically diagnosed PD [145]. Subsequently three additional missense mutations, p. G294A, p.G295S, and p.S393L, in patients with typical and atypical parkinsonism have been identified [146].
Taken together these data suggest that common pathomechanisms are present across the neurodegenerative spectrum, and particularly in patients with PD (Fig. 2). Harmonizing genetic and pathological substrates in terms of mixed pathology and disease pathogenesis will accelerate our upstanding of PD and drive design of disease modifyingtherapies.
BIOMARKERS
To aid the development of disease modifying therapies, it is critical that we are able to classify patients correctly during enrolment into clinical trials. Numerous biomarker studies have focused on the detection of α-syn in patients during life including in biofluids and peripheral tissue [147–152]. However, the presence of mixed pathologies in PD can be exploited in the biomarker field to aid diagnosis, prognosis, and the enrolment of patients into clinical trials.
Exciting developments in the AD biomarker field has proven tau (particularly detection in biofluids) is an accurate and reliable biomarker and has the potential to identify PD patients that are more likely to experience an accelerated disease trajectory. In 2009, Compta and colleagues found high total tau and phosphorylated tau levels in cerebrospinal fluid (CSF) in PD cases were associated with cognitive impairment and worsening motor symptoms [153, 154]. However, longitudinal evaluation of CSF total and phosphorylated tau measured 6 and 12 months from baseline demonstrated no significant changes over this time period. This suggests CSF tau and phosphorylated tau does not mirror disease progression, in particular progressive striatonigral degeneration as evaluated by clinical motor ratings (MDS-UPDRS III) and DaTscan measures [155]. More recently advances in plasma biomarkers (which provides a more accessible and less invasive option) have indicated plasma ptau181 is significantly increased in PD compared to controls. However no association was observed between ptau181 and longitudinal cognitive performance, supporting findings in CSF studies [156]. Reduced Aβ42 is associated with reduced cognitive function in PD compared to healthy controls (with cut-off levels being lower than those for AD) [157–159], with lower CSF Aβ42 associated with higher rate of Mini-Mental State Examination and Montreal Cognitive Assessment score decline [160].
It seems diagnostic panels incorporating α−syn and concomitant pathology may provide the most accurate diagnostic accuracy, perhaps highlighting the underlying pathological burden. For example, in a study comprising 45 participants, a diagnostic panel incorporating plasma α-syn and ptau181 combined with age and sex showed good performance in discriminating de novo PD cases from healthy controls [161]. Furthermore, the combination of CSF oligomeric and total α-syn, and Aβ42/tau ratios increase the diagnostic accuracy of PD [157, 160]. In addition to fluid biomarkers, neuroimaging has been trialed in PD biomarker studies. In 2016, Gomperts and colleagues investigated AV-1451 PET in PDD and PD cases, indicating higher standardized uptake value ratios in the inferior temporal gyrus than in healthy controls [131]; however, this difference was not recapitulated in a subsequent study [162]. Further work is required to elucidate the pathological signature of tau ligands in LBDs, particularly including a more quantitative approach to assessing pathological protein loads (see section ‘Effects of pathological load’).
RACIAL AND ETHNIC VARIATION IN PD
To date, racial and ethnic minorities are underrepresented in PD research. This was highlighted by GWAS studies where the MAPT locus was strongly associated with PD in a European population but not in a Japanese population [144]. Examination of diverse cohorts will allow us to investigate the enormous heterogeneity observed in neurodegenerative disease including PD. In a recent study investigating health-related quality of life in PD patients, differences were observed between racial and ethnic groups. After adjusting for co-factors, non-White patients scored lower than White counterparts, with further mediation analysis demonstrating lower cognition partly contributing to this association [163]. In a seminal study by Barnes and colleagues, Black decedents with AD dementia were more likely to have mixed pathologies compared to matched White decedents with AD [164]. A more recent neuropathological study investigating mixed LBD and AD pathologies across different ethnicities demonstrated prevalence of combined LBD and AD pathologies in 18% of non-Hispanic White individuals, 11% of Black individuals, and 35% of Hispanic individuals [165]. It is well documented that racial and ethnic groups are under-represented in brain donation programs for research into neurodegenerative diseases. Studies involving African Americans have identified religious beliefs [166], the effect of the removal process on physical appearance [167], and the lack of understanding regarding the rationale for brain donation in dementia research [167, 168] as contributing factors to lower participation rates in brain tissue donation initiatives. A study conducted by Boise and colleagues investigated this further conducting a survey of research volunteers from four racial/ethnic groups for their willingness to assent to brain donation including African American, Caucasian, Asian, and Latino research volunteers. Positive predictors included older age, Latino ethnicity, understanding how the brain is used by researchers, and an understanding of the process that participants need to follow to ensure the tissue is donated. Negative predictors included African/African American race, belief that the body should remain whole at burial, and concerns that researchers may not be respectful of the tissue following donation [169]. While the research community should be respectful of different religious beliefs and cultures regarding opinions on brain tissue donation, there are aspects that may be amendable to increase participation in such programs. Taking opportunities to familiarize under-represented minority groups with the enormous impact post-mortem tissue has on the research into neurodegenerative diseases should be prioritized. In addition to this, researchers should be transparent with research practices involving the use of donated tissues. Future studies focused on PD should include a more representative population including diverse ethnicities and also social, economic, and cultural backgrounds, and strive to make this data accessible to researchers, to fully appreciate the disease heterogeneity across the PD spectrum.
CONCLUSION
Aging and the accumulation of age-associated pathologies is the biggest risk factor for developing dementia in later life. Common protein aggregations such as hyperphosphorylation tau, Aβ, α-syn, and TDP-43 are assumed to lead to one specific neurodegenerative disease; however, it is rare for pathologies exist in isolation. Multiple pathologies can be observed in the PD brain at post-mortem examination, and while this is less frequent and severe compared to PDD and indeed even less compared DLB, a growing body of evidence suggests concomitant pathologies, even at low levels, can contribute to cognitive impairment in PD patients. It is assumed concomitant pathologies have a synergistic relationship that drives cognitive decline and research is ongoing to resolve the pathomechanisms underpinning this to inform the design of disease modifying therapies. Biomarker research (in particular fluid biomarkers) are demonstrating tremendous promise in identifying PD patients during life and will aid the stratification of patients for clinical trials. Finally, to fully appreciate the prevalence of concomitant pathologies and identify disease pathways in PD, it is crucial to include diverse populations of patients in research studies and clinical trials.
Footnotes
ACKNOWLEDGMENTS
This paper presents independent research funded and supported by the NIHR Newcastle Biomedical Research Centre (BRC). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. Figures 1 and ![]() were partly generated using Servier Medical Art, provided by Servier, licensed under a Creative Commons Attribution 3.0 unported license.
were partly generated using Servier Medical Art, provided by Servier, licensed under a Creative Commons Attribution 3.0 unported license.
FUNDING
LW is funded by an Alzheimer’s Research UK fellowship (ARUK-RF2020A-010). Brain tissue provided for the images in this manuscript was provided by Newcastle Brain Tissue Resource, which is funded in part by a grant from the UK Medical Research Council (G0400074), by NIHR Newcastle Biomedical Research Centre awarded to the Newcastle upon Tyne NHS Foundation Trust and Newcastle University, and by a grant from the Alzheimer’s Society and Alzheimer’s Research UK as part of the Brains for Dementia Research initiative. The NIHR Newcastle Biomedical Research Centre (BRC) is a partnership between Newcastle Hospitals NHS Foundation Trust, Newcastle University, and Cumbria, Northumberland and Tyne and Wear NHS Foundation Trust and is funded by the National Institute for Health and Care Research (NIHR).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
