Abstract
Background:
Freezing of gait (FOG) in Parkinson’s disease (PD), especially the “L-dopa-unresponsive” subtype, is associated with the dysfunction of non-dopaminergic circuits.
Objective:
We sought to determine whether cortical sensorimotor inhibition evaluated by short-latency afferent inhibition (SAI) related to cholinergic and gamma-aminobutyric acid (GABA)-ergic activities is impaired in PD patients with L-dopa-unresponsive FOG (ONOFF-FOG).
Methods:
SAI protocol was performed in 28 PD patients with ONOFF-FOG, 15 PD patients with “off” FOG (OFF-FOG), and 25 PD patients without FOG during medication “on” state. Additionally, 10 ONOFF-FOG patients underwent SAI testing during both “off” and “on” states. Twenty healthy controls participated in this study. Gait was measured objectively using a portable Inertial Measurement Unit system, and participants performed 5-meter Timed Up and Go single- and dual-task conditions. Spatiotemporal gait characteristics and their variability were determined. FOG manifestations and cognition were assessed with clinical scales.
Results:
Compared to controls, PD patients without FOG and with OFF-FOG, ONOFF-FOG PD patients showed significantly reduced SAI. Further, dopaminergic therapy had no remarkable effect on this SAI alterations in ONOFF-FOG. Meanwhile, OFF-FOG patients presented decreased SAI only relative to controls. PD patients with ONOFF-FOG exhibited decreased gait speed, stride length, and increased gait variability relative to PD patients without FOG and controls under both walking conditions. For ONOFF-FOG patients, significant associations were found between SAI and FOG severity, gait characteristics and variability.
Conclusion:
Reduced SAI was associated with severe FOG manifestations, impaired gait characteristics and variability in PD patients with ONOFF-FOG, suggesting the impaired thalamocortical cholinergic-GABAergic SAI pathways underlying ONOFF-FOG.
Keywords
INTRODUCTION
Freezing of gait (FOG) in patients with advanced Parkinson’s disease (PD) is of increasing clinical and scientific interest given its impact on deterioration of mobility, risk of falls, and patient quality of life [1]. This mirrors the fact that FOG is difficult to treat [2]. A consensus statement defined FOG as the “brief, episodic absence or marked reduction of forward progression of the feet despite the intention to walk” [3]. Based on dopaminergic responsiveness, FOG is generally classified into three subtypes, including “off” FOG (OFF-FOG; freezing episodes are completely relieved by L-dopa), “on” FOG (the blocks are induced or exacerbated by L-dopa), and “L-dopa-unresponsive” FOG (ONOFF-FOG; the effect of dopaminergic stimulation is not enough to prevent FOG even if other parkinsonian motor signs are improved in the “on” state) [4].
While dopaminergic deficiency is a primary contributor to the emergence of FOG in PD, non-dopaminergic degeneration also contributes, particularly the degeneration of central cholinergic neurons [5]. The hypothesis is consistent with the fact that dopaminergic replacement therapy fails to improve gait symptoms in FOG, especially in the ONOFF-FOG subtype. Central cholinergic projection systems originate primarily from the basal forebrain nuclei and the mesopontine tegmental nuclei, most importantly the nucleus basalis of Meynert (NBM) neurons in the substantia innominata and the pedunculopontine nucleus (PPN). The NBM mainly projects to the cortex, which degenerates in Alzheimer’s disease (AD), dementia with Lewy bodies (DLB), and PD [6–8]. The PPN mainly projects to the thalamus, subthalamus, brainstem, and cerebellum. Compelling evidence has pointed to structural and functional abnormalities in the PPN, as demonstrated with damaged white matter, altered structural and functional connectivity with the thalamus and cortex in PD patients with FOG [9–14]. Notably, the thalamocortical cholinergic systems are regulated by the gamma-aminobutyric acid (GABA) signaling [15], whose dysfunction plays a critical role in FOG pathogenesis as well [16]. Therefore, dysfunctions of the thalamocortical cholinergic or GABAergic system, or impaired cholinergic-GABAergic interactions may contribute to FOG in PD patients.
Short-latency afferent inhibition (SAI), traditionally considered as a surrogate for cholinergic activity in the brain, is an electrophysiological technique that noninvasively assesses an inhibitory circuit in the sensorimotor cortex via coupling peripheral nerve stimulation with transcranial magnetic stimulation (TMS) of the contralateral motor cortex [17, 18]. Cholinergic dysfunction leads to a reduction in SAI (i.e., a smaller decrease in motor evoked potentials [MEPs] was recorded when the SAI protocol is applied). Indeed, decreased SAI has been identified in AD and DLB patients [6]. Significantly reduced SAI was also observed in PD patients with subclinical symptoms of cholinergic dysfunction, such as olfactory dysfunction [19, 20], oropharyngeal dysphagia [21], visual hallucinations [22] and REM sleep behavior disorder [23]. Furthermore, SAI deficits were associated with cognitive decline and gait dysfunctions in PD [24, 25]. However, pharmacological studies demonstrated that the thalamocortical cholinergic pathways underlying SAI are heavily modulated by the availability of GABAA alpha1 subtype receptors [26, 27]. Accordingly, in this study, SAI was used to assess cortical sensorimotor inhibition, depending on the integrity of the thalamocortical cholinergic and GABAergic systems as well as the coordination of their interactions.
However, to our best knowledge, few studies have evaluated SAI in PD patients with ONOFF-FOG. Only Picillo and colleagues attempted to compare SAI in PD patients with and without FOG, yet they failed to detect any abnormalities [28]. To further substantiate the cortical sensorimotor inhibition dysfunction and its potential impact on ONOFF-FOG and gait characteristics in PD, we aimed to investigate SAI and gait features under normal walking and dual-task walking in controls, PD patients without FOG (NO-FOG), PD patients with OFF-FOG, and PD patients with ONOFF-FOG.
METHODS
Subjects
Patients with a diagnosis of PD according to the clinical criteria of the Movement Disorder Society (MDS) were consecutively recruited in our study [29]. Inclusion criteria were 1) age 40 or older to rule out early-onset Parkinsonism, 2) disease duration over 3 and less than 10 years, 3) independent walking more than 30 meters, and 4) antiparkinsonian treatments at a stable and optimized daily dosage during the 4 weeks prior to the study entry. Exclusion criteria were 1) contraindications of TMS, such as the presence of intracranial metals, 2) severe tremor and levodopa-induced dyskinesias in medication “on” state interfering with electromyography (EMG) recording, 3) major anxiety and depression according to the Diagnostic and statistical manual of mental disorders fourth edition criteria, 4) dementia according to the criteria of MDS [30], 5) intake of cholinesterase inhibitors, benzodiazepines, neuroleptics, anti-cholinergic or anti-depressant drugs, and 6) other disorders interfering with gait, such as primary progressive freezing gait and normal pressure hydrocephalus. The recruitment lasted more than one year from March 2021 to October 2022 and involved all eligible PD patients in our center.
Among eligible PD patients, we further ascertained FOG subjects following the three steps: 1) identification of “off” and “on” states based on the periodic recurrence of motor and non-motor symptoms of PD and their response to drug intake; 2) confirmation of FOG based on (i) score of 1 or above of new FOG Questionnaire (NFOGQ) [31] and (ii) clinical freezing episodes confirmed by two researchers (KZ Zhang and LN Wang) during 10-m walking, turning or going through a narrow doorway; 3) recognition of FOG subtypes based on the following question “When do you experience FOG?”. Patients with ONOFF-FOG experienced FOG in both medication “on” and “off” states, while OFF-FOG patients experienced FOG only in medication “off” state, that is, freezing episodes could be completely relieved by L-dopa.
Following the above criteria and procedures, a total of 70 PD patients were enrolled, comprising 30 ONOFF-FOG, 15 OFF-FOG, and 25 NO-FOG PD patients. However, two ONOFF-FOG patients were excluded due to severe peak-dose levodopa-induced dyskinesia and a complaint of discomfort during TMS examination. Meanwhile, 20 age-, gender-, and education level-matched healthy control (HC) subjects participated in this study. Our study was approved by the Ethical Committee of the First Affiliated Hospital of Nanjing Medical University (2021-SR-209). All participants gave written informed consent.
Clinical assessments
During medication “on” state, all patients received semi-structural interviews and experienced a battery of clinical evaluations. Motor and FOG-related function was evaluated using Hoehn and Yahr (H&Y) stage, the part III of Unified Parkinson’s Disease Rating Scale (UPDRS-III), Tinetti Mobility Test (TMT), and the NFOGQ. In addition, Mini-Mental State Examination (MMSE), Montreal Cognitive Assessment (MoCA), and Frontal Assessment Battery (FAB) were used to estimate global cognition and executive function. Lastly, the total levodopa equivalent daily dose (LEDD) was calculated to describe the dopaminergic drugs administered.
Electrophysiological evaluations
Recordings
Surface EMGs were recorded from the abductor pollicis brevis (APB) of the symptomatic arm of patients or the dominant hand of controls. The active electrode was placed over the motor point of the muscle and the reference electrode over the metacarpophalangeal joint of the thumb. EMG signals were amplified and bandpass filtered (20 Hz –2000 Hz). Somatosensory evoked potentials (SEPs) were recorded through adhesive electrodes applied to the scalp (contralateral to median nerve stimulation) after appropriate skin preparation, using the active electrode 2 cm posterior to C4/C3 (10–20 System) and reference electrode placed on the forehead.
Median nerve stimulation
SEPs were obtained by stimulating the median nerve on the more affected side in patients and on the dominant side in controls. Stimuli (single pulses, pulse width 200μs) were delivered to the median nerve at the wrist using a constant current stimulator through adhesive electrodes (cathode proximal). Stimulus intensity was adjusted to just above motor threshold, as determined by a visible twitch in the APB muscle.
Magnetic stimulation
Electrophysiological evaluations were performed on the more affected side in patients and on the dominant side in controls. TMS of motor cortex was performed with a high-power magnetic stimulation (Neurosoft, Russia). A figure-of-eight coil (with external loop diameters of 10 cm) was positioned at the scalp over the hand motor area to evoke motor responses in the contralateral APB muscle.
Short-latency afferent inhibition
SAI was studied using the technique described by Tokimura et al. [17]. Conditioning stimuli were single electrical pulses applied to the median nerve at the wrist preceded cortical TMS by varying interstimulus intervals (ISIs). The intensity of the conditioning peripheral stimulus was set at just over the motor threshold to evoke a visible twitch of the APB muscle (Fig. 1A). The intensity of the test cortical magnetic shock was adjusted to evoke an MEP in relaxed APB muscle with peak-to-peak amplitude of approximately 1 mV. SAI was tested at different ISIs, which were determined relative to the latency of N20 component of the average SEP (two averages of 500 –2000 responses). ISIs ranged from 0 to 8 ms after N20 latency in 4 ms steps (Fig. 1B). For each ISI, we calculated the amplitude of the basal MEP (average of five consecutive responses obtained after cortical stimulation alone) and the amplitude of the conditioned MEP (average of five consecutive responses obtained after the conditioning peripheral electrical stimulus). The amplitude of conditioned MEP, expressed as a percentage of the basal MEP amplitude at each ISI, was used to evaluate the amount of SAI. All subjects utilized audiovisual feedback of EMG signal at high gain to maintain complete relaxation during experiments.
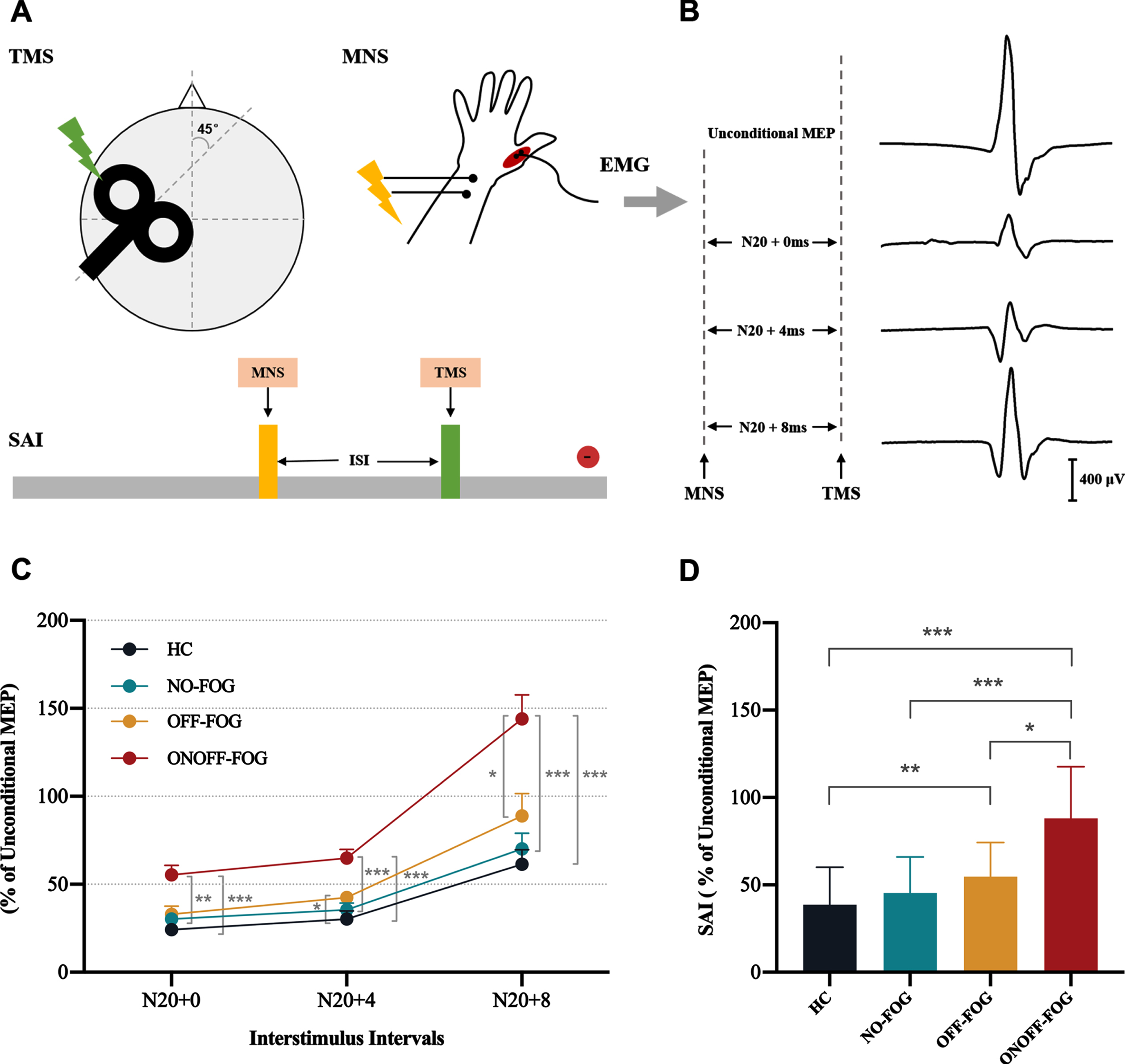
Comparison of short-latency afferent inhibition in the study participants. A) SAI was evaluated by applying MNS stimuli at the wrist before the TMS stimuli. B) Conditional and unconditional MEP was recorded from a control subject (aged 61 years). C) ONOFF-FOG PD exhibited reduced SAI (increased MEP ratio) compared to NO-FOG PD patients and controls at ISIs 0, 4, and 8. ONOFF-FOG patients also showed impaired SAI compared to OFF-FOG patients at ISI 8. Meanwhile, PD patients with OFF-FOG presented decreased SAI relative to controls at ISI 4. D) For grand mean of SAI, ONOFF-FOG patients showed reduced SAI relative to OFF-FOG patients, NO-FOG patients, and HC subjects. OFF-FOG PD patients also presented decreased SAI compared to controls. EMG, electromyography; HC, healthy controls; ISI, interstimulus interval; MEP, motor evoked potential; MNS, median nerve stimulation; NO-FOG, no freezing of gait; OFF-FOG, “off” freezing of gait; ONOFF-FOG, L-dopa-unresponsive freezing of gait; PD, Parkinson’s disease; SAI, short-latency afferent inhibition; TMS, transcranial magnetic stimulation. *p < 0.05; **p < 0.01; ***p < 0.001.
In our study, all PD patients underwent SAI protocol during medication “on” state. However, a previous study showed that SAI would be decreased after taking dopaminergic drugs on the more affected side in PD patients [32]. In order to determine whether dopaminergic treatment has a similar influence on SAI in PD patients with L-dopa-unresponsive FOG, 10 ONOFF-FOG patients underwent SAI testing during drug “off” and “on” periods. More specifically, these patients were first studied at least 12 h off medication in the “practically defined off state” (drug “off” state). Then, patients taking dopaminergic medication were also studied approximatively 1–2 h after they took their usual doses of medication (drug “on” state).
Gait assessments
A portable Inertial Measurement Unit (IMU) system (GYENNO Science, China) was applied to obtain gait features. Gait was characterized by single-task (ST) and dual-task (DT) 5-meter timed up and go (TUG) tests (Supplementary Figure 1) [33]. In ST TUG test, the participant was instructed to stand up, walk 5 meters, turn 180°, walk back, and sit down. During the DT walking, subjects held a full cup of water while performing the TUG task. Participants were instructed to walk at a comfortable, self-selected pace. Each task was performed only once, but each subject should practice twice before the formal test. Gait variables of interest included spatiotemporal gait characteristics (gait speed [m/s] and stride length [cm]) and their variability (coefficient of variation [CV] of gait speed and stride length).
Statistical analyses
Gender differences were analyzed using Chi-square test. We checked the data normality using histograms and Shapiro-Wilk tests. Age, education level, disease duration, H&Y stage, LEDD, MMSE, MoCA, FAB, and TMT scores were analyzed by Kruskal-Wallis tests due to the non-normality of data. The UPDRS-III of the 3 PD groups were compared with one-way ANOVA. The NFOGQ of the 2 FOG groups were analyzed via two-sample t test. The non-normally distributed SAI variables and gait characteristics were log base-10 transformed to improve normality. A repeated ANCOVA was applied to compare SAI variables obtained at different TIME (within factor: ISIs 0, 4, and 8) and across different GROUP (between factor: HC, NO-FOG, OFF-FOG, and ONOFF-FOG) during medication “on” state, adjusted for MOCA and FAB scores. Besides, SAI obtained at the ISIs 0, 4, and 8 were averaged to obtain a grand mean of SAI in order to reduce the data variation [22]. Grand mean of SAI was analyzed using a one-way ANCOVA, after controlling for MOCA and FAB scores as well. In addition, we also compared SAI between drug “off” and “on” sates in 10 ONOFF-FOG patients with paired-sample t test or Wilcoxon matched-pairs signed rank test. Spatiotemporal gait characteristics and variability under ST and DT conditions were entered in repeated ANCOVAs with the factor GROUP (HC, NO-FOG, OFF-FOG, and ONOFF-FOG) as between factor and the factor TASK (ST and DT walking) as the within factor, adjusted for MOCA and FAB scores. Dual-task cost (DTC; %) was calculated ([DT-ST]/ST * 100) for each gait parameter. The level of significance was set at p < 0.05, and post hoc analyses were performed, applying the Bonferroni correction for multiple comparisons where appropriate. Disease duration and LEDD were also entered as covariates when comparing PD subgroups. To explore the clinical relevance of SAI, partial correlation analyses were performed adjusted for disease duration, LEDD, MoCA and FAB scores. False discovery rate (FDR) correction was subsequently applied due to multiple testing.
RESULTS
Demographic and clinical characteristics
There were no significant differences in gender, age, education level and MMSE score among the 4 groups. PD patients with NO-FOG, OFF-FOG, and ONOFF-FOG showed matched H&Y stage and UPDRS-III (Table 1). As excepted, ONOFF-FOG and OFF-FOG patients exhibited lower Tinetti balance, gait and total scores compared to NO-FOG patients. However, PD patients with NO-FOG, OFF-FOG, and ONOFF-FOG presented lower MoCA and FAB scores relative to controls. Considering the role of cognition in gait and to reduce potential confounding effects on SAI, the MoCA and FAB scores were entered as regressors in the four-group statistical model. Meanwhile, PD patients with OFF-FOG had longer disease duration than ONOFF-FOG and NO-FOG patients, and higher LEDD than ONOFF-FOG PD patients. Therefore, disease duration and LEDD were also included as covariates when comparing PD subgroups.
Demographic and clinical characteristics of all participants
Data are expressed as mean±SD. FAB, Frontal Assessment Battery; F, Female; HC, healthy controls; H&Y, Hoehn and Yahr; LEDD, Levodopa equivalent daily dose; M, Male; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; NA, not applicable; NFOGQ, New Freezing of Gait Questionnaire; NO-FOG, no freezing of gait; OFF-FOG, “off” freezing of gait; ONOFF-FOG, L-dopa-unresponsive freezing of gait; TMT, Tinetti Mobility Test; TUG, Timed Up and Go; UPDRS, Unified Parkinson’s disease rating scale; y, year. *P < 0.05 was set as significant. a Chi square test. b Kruskal-Wallis test. c One-way ANOVA. d Two-sample t test.
Short-latency afferent inhibition
For SAI variables across different ISIs, a repeated ANCOVA showed a significant main effect of GROUP (F (3,82) = 19.376, p < 0.001), adjusted for MoCA and FAB scores; however, no significant effect of TIME and TIME × GROUP interaction were identified. Specifically, ONOFF-FOG PD patients exhibited reduced SAI compared to NO-FOG PD patients and controls at ISIs 0, 4, and 8 (all p < 0.01, Bonferroni-corrected) (Fig. 1 C). ONOFF-FOG patients also showed impaired SAI compared to OFF-FOG patients at ISI 8 (p = 0.042, Bonferroni-corrected). Meanwhile, OFF-FOG PD patients presented decreased SAI relative to controls at ISI 4 (p = 0.017, Bonferroni-corrected). For grand mean of SAI, ONOFF-FOG patients showed reduced SAI relative to OFF-FOG patients (p = 0.048, Bonferroni-corrected), NO-FOG patients (p < 0.001, Bonferroni-corrected) and HC subjects (p < 0.001, Bonferroni-corrected) (Fig. 1D). However, PD patients with OFF-FOG presented decreased SAI compared to controls (p = 0.008, Bonferroni-corrected) as well.
Additionally, we compared SAI between medication “off” and “on” states in 10 ONOFF-FOG PD patients, however, no significant differences were identified (Supplementary Table 1), indicating no effect of dopaminergic drug on SAI reduction in PD patients with L-dopa-unresponsive FOG.
Gait characteristics
For gait speed and stride length, repeated ANCOVAs revealed significant main effects of GROUP (F (3,82) = 9.310, p < 0.001; F (3,82) = 13.842, p < 0.001) and TASK × GROUP interactions (F (3,82) = 3.914, p = 0.012; F (3,82) = 4.219, p = 0.008) after controlling for MoCA and FAB scores; however, no significant effects of TASK were observed. For gait speed CV and stride length CV, repeated ANCOVAs disclosed significant main effects of TASK (F (1,82) = 6.090, p = 0.016; F (1,82) = 10.924, p = 0.001) and GROUP (F (3,82) = 9.924, p < 0.001; F (3,82) = 8.939, p < 0.001); however, no significant interactions were detected (Table 2). During ST and DT conditions, PD patients with ONOFF-FOG exhibited decreased gait speed and stride length, as well as increased gait speed CV and stride length CV compared to PD patients with NO-FOG and HC subjects (all p < 0.05, Bonferroni-corrected). Relative to controls, OFF-FOG patients also performed slower gait speed, shorter stride length, and higher gait speed CV under both walking conditions (all p < 0.05, Bonferroni-corrected). However, for stride length CV, OFF-FOG patients showed a significant increase in ST condition (p = 0.005, Bonferroni-corrected) and an increase trend in DT condition (p = 0.055, Bonferroni-corrected) compared to HC individuals. When comparing gait characteristics under ST and DT conditions, decline performance in gait speed and stride length was observed in PD patients with and without FOG (all p < 0.01, Bonferroni-corrected). Both ONOFF-FOG and OFF-FOG patients showed an increase in gait speed CV and stride length CV under DT condition compared to ST condition (all p < 0.05, Bonferroni-corrected).
Descriptive gait characteristics for all subjects under two walking tasks
Data are showed as mean±SD. CV, coefficient of variation; DT, dual task; HC, healthy control; NO-FOG, no freezing of gait; OFF-FOG, “off” freezing of gait; ONOFF-FOG, L-dopa-unresponsive freezing of gait; ST, single task. *p < 0.05 was set significant.
Correlation analyses
Using partial correlation analyses, SAI was significantly correlated not only with the NFOGQ scores (r = 0.507, p = 0.015, FDR-corrected) but also with gait speed (r = –0.520, p = 0.014, FDR-corrected; r = –0.564, p = 0.009, FDR-corrected) and stride length (r = –0.528, p = 0.014, FDR-corrected; r = –0.572, p = 0.010, FDR-corrected) during both ST and DT walking, respectively (Fig. 2A–E). Besides, SAI was also significantly associated with gait speed CV (r = 0.414, p = 0.044, FDR-corrected; r = 0.717, p = 0.001, FDR-corrected) and stride length CV (r = 0.431, p = 0.040, FDR-corrected; r = 0.688, p = 0.001, FDR-corrected) in PD patients with ONOFF-FOG during both ST and DT walking, respectively (Fig. 2F–I). These partial correlation analyses were adjusted for disease duration, LEDD, MoCA and FAB.
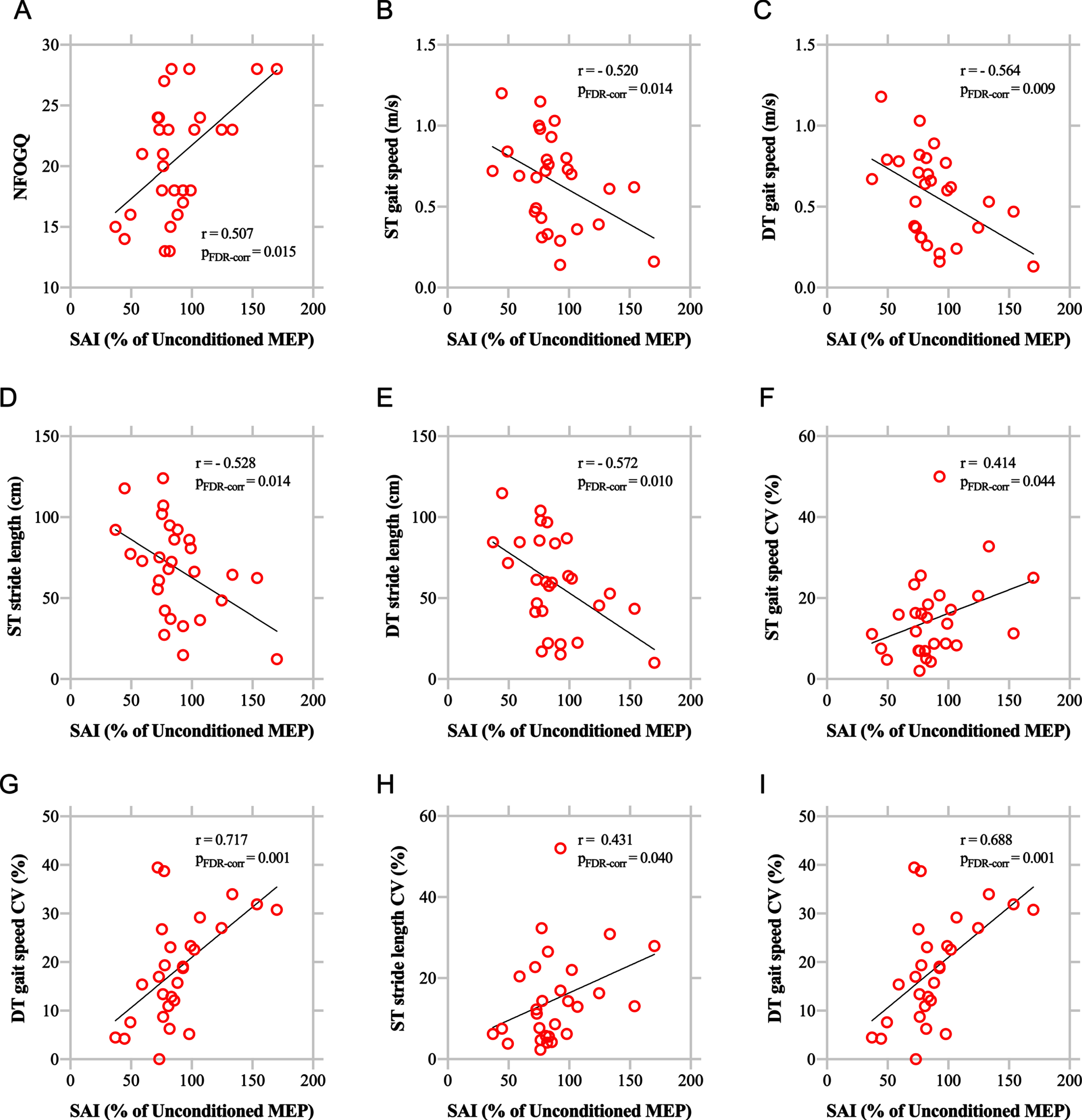
Correlations between SAI and NFOGQ as well as objective gait characteristics. SAI was significantly correlated not only with NFOGQ scores but also with spatiotemporal gait characteristics and gait variability under ST and DT conditions in Parkinson disease patients with L-dopa-unresponsive freezing of gait. CV, coefficient of variation; DT, dual task; FDR, False discovery rate; MEP, motor evoked potential; NFOGQ, New Freezing of Gait Questionnaire; ST, single task.
DISCUSSION
This is the first study to our knowledge to demonstrate impaired SAI in PD patients with ONOFF-FOG. SAI was reduced in PD patients with ONOFF-FOG compared to controls, NO-FOG and OFF-FOG PD patients; however, there was no significant difference in SAI between drug “off” and “on” states in ONOFF-FOG patients. PD patients with ONOFF-FOG walked with worse spatiotemporal gait characteristics and gait variability in comparison with PD patients with NO-FOG and HC individuals under ST and DT conditions. Furthermore, SAI was significantly correlated not only with NFOGQ scores but also with gait speed, stride length and their variability under ST and DT conditions in ONOFF-FOG PD patients.
The mechanisms underlying SAI are complex. Although the neural pathway traversed by the afferent volley leading to SAI remains obscure, SAI may result from direct thalamocortical projects to the primary motor cortex via cholinergic thalamic nuclei [34, 35]. Support for this pathway emerges from studies demonstrating the abolishment of SAI in individuals with thalamic stroke ipsilateral to the hemisphere receiving the TMS pulse while N20 remained intact [36, 37]. Moreover, scopolamine (a muscarinic antagonist) reduced SAI [34], while acetylcholinesterase inhibitors increased SAI [38], suggesting the key role of acetylcholine in the generation of SAI. Thus, SAI was traditionally viewed as a surrogate for cholinergic activity in the brain. SAI deficits were indeed demonstrated in populations with cholinergic dysfunction, such as AD and PD with mild cognitive impairment [7, 39]. This is in line with postmortem studies revealing a loss of cholinergic neurons in AD and PD [40, 41]. However, SAI circuit is not a sheer cholinergic pathway. It was suggested that GABAA receptor agonists (benzodiazepines) could reduce SAI [26, 43]. More specifically, diazepam has no effect while lorazepam and zolpidem reduce SAI. Diazepam non-selectively binds to the alpha1, alpha2, alpha3, or alpha5 subunit of the GABA receptor, zolpidem has a high affinity for the alpha1 subunit, and the affinity profile of lorazepam is unknown [44, 45]. Thus, SAI may be mediated by the GABAA receptor subtype bearing the alpha1 subunit. Given the known involvement that GABA controls the release of acetylcholine within the brainstem and cortex [15], it is likely that SAI involves a cholinergic pathway whose activity is modulated by GABA. Therefore, normal SAI suppression depends on the integrity of the thalamocortical cholinergic and GABAergic systems, and their coordinated interactions.
However, in our study, SAI was reduced in ONOFF-FOG PD patients compared to controls, NO-FOG and OFF-FOG PD patients, suggesting the impaired thalamocortical cholinergic-GABAergic SAI pathways in ONOFF-FOG. Actually, PD is associated with degeneration of the cholinergic basal forebrain (cholinergic nuclei 1–4) and cholinergic neurons in the upper brainstem (cholinergic nuclei 5 and 6) as disease progresses [40]. Reduced gray matter density of the cholinergic nucleus 4 (Ch4) or NBM, which has extensive cholinergic projections to the cerebral cortex [5], was associated with slower gait speed and a greater precent TUG test deterioration in PD patients when performing a cognitive task [46]. The volume of NBM could predict the longitudinal gait changes unique to PD [47]. Also, a clear relationship between the measures of Ch4 degeneration and cortical cholinergic deficiency, attention and other cognitive impairments has been announced [48, 49]. A molecular imaging study indicated that NBM-cortical cholinergic denervation was related to increased FOG risk, especially in the presence of concomitant cortical amyloid deposition [50]. Thus, NBM-cortical cholinergic dysfunction may contribute to FOG and other gait deficits, especially during highly cognitive tasks. In addition, degeneration of cholinergic neurons in the PPN is also implicated in the pathogenesis of FOG in PD [51]. The PPN supplies the bulk of the cholinergic input to the thalamus, with projection to the striatum, cerebellum, and brainstem, which are critical for motor planning, gait initiation, and control [52, 53]. Compelling MRI studies have evidenced that PD patients with FOG exhibited abnormal PPN functional connectivity in corticopontine-cerebellar pathways, and diffuse white matter deficits extending to motor, sensory and cognitive regions [13]. Moreover, a 72-month follow-up study showed that white matter microstructural damage to PPN could predict gait symptoms in PD [54]. Thus, NBM-cortical and PPN-thalamic cholinergic dysfunctions may be involved in the complex pathophysiological mechanisms underpinning FOG in PD through affecting cognitive or gait control. Additionally, GABAergic neurons in the thalamic nucleus receive collaterals from thalamocortical fibers, and modulate information transfer between the thalamus and cortex [55]. The thalamic nucleus receives inputs from a large cholinergic projection from the brainstem and basal forebrain [56]. Furthermore, abnormal GABAergic neurotransmission contributed to some of axial symptoms in PD, including FOG [16]. Therefore, dysfunctions of the thalamocortical cholinergic or GABAergic system, or the uncoordinated cholinergic-GABAergic interactions might contribute to SAI degeneration in ONOFF-FOG patients. Moreover, FOG symptoms were significantly correlated with reduced SAI, indicating that the impaired cholinergic-GABAergic SAI pathways are involved in the pathophysiology mechanisms underpinning ONOFF-FOG in PD patients.
OFF-FOG PD patients also exhibited SAI reduction in drug “on” state compared to controls in our study. Some suggest that FOG may progress through a continuum for responsive to unresponsive, indicating OFF-FOG is a mild intermediate state prior to ONOFF-FOG [57]. Thus, this reduction in SAI may be attributed to mild thalamocortical cholinergic and GABAergic dysfunctions in OFF-FOG. Alternatively, a previous study showed that SAI was normal during “off” medication but reduced in the “on” medication state in PD patients [32]. The authors argued that dopaminergic drugs could worsen proprioception and sway in PD, which might contribute to dyskinesia, possibly related to SAI alterations in medication “on” state. Anyway, it is insufficient to conclude that reduced SAI is a characteristic of OFF-FOG, given that OFF-FOG only showed SAI decline relative to controls. Nevertheless, our observation that dopaminergic therapy failed to alter SAI in ONOFF-FOG patients reinforces that SAI reduction is a predominant feature of ONOFF-FOG, independent of dopaminergic drugs.
However, using the same method, Picillo et al. failed to investigate aberrant cholinergic activity in PD patients with FOG [28]. The reason for this contradiction may be twofold. First, their sample size is small. Their study included only 14 PD patients with FOG and 10 PD patients without FOG, whereas we enrolled twice as many patients. Second, as the authors stated, biased recall might influence self-report of recall in their patients. Therefore, it is possible that they tested patients with mild FOG (only referred and objectified) and failed to see a difference in SAI for this reason. In contrast, only PD patients with clinically confirmed FOG were included and objectively evaluated in our study, which ensured the reliability of our results.
In addition to FOG manifestations, other gait domains including spatiotemporal gait characteristics and their variability under normal walking and dual-task walking were also described in the current study. Consistent with previous studies, decreased gait speed and stride length were discovered in PD patients with ONOFF-FOG in both ST and DT conditions compared to PD patients with NO-FOG and HC participants [58, 59]. Meanwhile, the increased CV of gait speed and stride length in ONOFF-FOG patients could indirectly reflect the inconsistency in stepping patterns and the impairment in postural control during both ST and DT walking [60]. Furthermore, impaired spatiotemporal gait characteristics and their variability were strongly associated with SAI reduction, supporting the hypothesis for the cholinergic and GABAergic role in gait [24, 52].
There are some limitations to the study that should be acknowledged. First, only five trials for each condition were collected and they were not delivered randomly as commonly performed in TMS studies. However, using the same technical procedure, previous studies have succeeded in finding a reduced SAI in patients with visual hallucinations [22]. Second, SAI indirectly reflects the function of thalamocortical cholinergic and GABAergic systems, rather than directly assessing the integrity of cholinergic-GABAergic pathway. Moreover, a positron emission tomography study suggested that FOG is linked with reduced vesicular acetylcholine transporter expression in the striatal cholinergic interneurons in PD [61]. This is another limitation of SAI, which does not reflect alterations in striatal cholinergic interneurons. Lastly, SAI is also modulated by movement, but we only performed our experiments at resting state. It would be interesting to evaluate if we are able to capture some differences in the amount of sensorimotor inhibition between PD patients with and without FOG during finger movements or repetitive stepping in place.
Conclusion
Reduced SAI was identified in PD patients with ONOFF-FOG, suggesting the cholinergic or GABAergic dysfunction underlying the pathophysiology mechanisms of L-dopa-unresponsive FOG in PD patients. In addition, SAI was demonstrated to be associated with FOG and other gait symptoms in PD patients with L-dopa-unresponsive FOG.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the National Natural Science Foundation of China (No. 81671258) and Jiangsu Social Development Project (No. BE2022808).
CONFLICT OF INTEREST
The authors have no conflicts of interest to declare.
