Abstract
Background:
The role of peripheral phosphorylated-α-Synuclein (p-α-syn) deposition on nerve degeneration in synucleinopathies is still unknown.
Objective:
To assess the cutaneous neural distribution of p-α-Syn deposits and its correlation with clinical data and with morphology and function of cutaneous sensory and autonomic nerves in early Parkinson’s disease (PD) and multiple system atrophy-parkinson type (MSA-p).
Methods:
We recruited 57 PD (F/M = 21/36; age 63.5±9.4 years) and 43 MSA-p (F/M = 16/27; age 62.3±9.0 years) patients within 2 years from motor symptoms. We applied questionnaires and clinical scales, sensory thresholds, and sudomotor testing to assess severity of motor and non-motor involvement and sensory and autonomic dysfunction. We quantified, in skin biopsy from thigh, leg, and fingertip, epidermal, pilomotor, and sudomotor nerve fibers, Meissner corpuscles and intrapapillary myelinated endings and the neural distribution of p-α-syn deposits.
Results:
Compared to controls, we found a cutaneous denervation paralleling functional and clinical impairment. Sensory and autonomic denervation was more severe in MSA-p than in PD. Deposits of p-α-syn were found in the majority of patients, with no significant differences among sites in both groups. Higher occurrence of p-α-syn deposits in autonomic nerves differentiated (
Conclusion:
Our work demonstrated an early peripheral sensory and autonomic involvement in synucleinopathies, more severe in MSA-p than in PD. Higher p-α-syn deposits in autonomic nerves differentiated PD from MSA-p. p-α-syn deposits were associated with preserved innervation and slower disease progression.
Keywords
INTRODUCTION
Synucleinopathies are a heterogeneous group of neurodegenerative diseases characterized by phosphorylated α-synuclein (p-α-Syn) deposition as a common hallmark [1]. p-α-syn deposits occur in neurons in Parkinson’s disease (PD) and mainly in oligodendroglia in multiple system atrophy (MSA) [2, 3].
In the early stage of the disease, differentiation between PD and MSA-parkinsonian type (MSA-p) may be challenging. The development of a clear clinical picture fulfilling all the diagnostic criteria for MSA-p [4] may require a few years and a definite diagnosis is possible only postmortem with evidence of Lewy body pathology in the brain [5].
In the last few decades, the study of cutaneous innervation through skin biopsy has provided evidence for peripheral involvement in PD [6] as well as in atypical parkinsonism [7–9] with the observation that cutaneous nerve loss parallels motor involvement and the occurrence of postganglionic sudomotor damage in MSA, previously considered a primarily central neurodegenerative disorder. Moreover, the postmortem observation of p-α-Syn in the skin of PD patients with CNS Lewy body pathology [10] prompted the search of cutaneous p-α-Syn as “
In recent years, several reports [12–14] documented differences in the distribution of p-α-Syn cutaneous deposits in PD and MSA, mainly found in autonomic nerves in PD and in sensory nerves in MSA. However, several discrepancies were reported regarding: the percentage of p-α-Syn positivity in the skin, varying from 23 to 100% of PD patients [10, 15] and from 0 to 78% in MSA [12, 16]; the preferential distribution of p-α-Syn in autonomic nerves in PD and in somatic nerves in MSA [16–18]; the correlation of p-syn deposits with cutaneous denervation [12, 17], not confirmed in further study [18]. These discrepancies may be due to different laboratory protocols, site of skin biopsy evaluated, section thickness, number of stained sections, staining technique and type of antibody applied [13, 20].
Finally, it is still unclear if cutaneous denervation starts early in MSA, as reported in PD [8] and if the pattern of cutaneous denervation is able to differentiate the two conditions at an early stage.
The aim of this work was to assess the cutaneous neural distribution of p-α-Syn deposits and its correlation with clinical data and with morphology and function of cutaneous sensory and autonomic nerves in early PD and MSA-p. We aimed to evaluate the early stage of the disorder when there is greater diagnostic uncertainty and before the occurrence of changes related to disease progression or treatment.
MATERIALS AND METHODS
Subjects
One hundred patients affected by parkinsonism (43 MSA-p and 57 PD), within 2 years from motor symptom onset and before starting L-Dopa treatment were recruited from three centers (University College London Queen Square, UK, Skin Biopsy Lab of “ICS Maugeri” IRCCS of Telese Terme, Italy, and Neurology department, University of Naples Federico II) between November 2017 and December 2019 in a prospective longitudinal study (PE-2013-02359028).
We included patients who received the diagnosis of clinically established PD and probable MSA-p according to current diagnostic criteria [4, 21]. The diagnosis was confirmed after 12 months of follow up. Patients who did not fulfill the diagnostic criteria for either of the diseases at follow-up were excluded from the present analysis.
Patients with abnormal neurography, a previous diagnosis of peripheral neuropathy and/or with conditions potentially affecting the peripheral nervous system such as glucose intolerance, dysendocrinopathies, vitamin E, vitamin B12, and folic acid deficiency, hepatic or renal failure, HIV, or connective tissue disorders, as assessed by blood testing, were excluded from the study.
Before recruitment, all patients underwent neurologic examination, dopaminergic responsiveness, and brain MRI.
Clinical evaluation, questionnaires, sensory and autonomic functional assessments, and skin biopsies were performed at the baseline visit, while after 12 months of follow-up only the clinical assessment was repeated.
All skin samples were processed and analyzed at Skin Biopsy Lab of ICS Maugeri – IRCCS of Telese Terme.
Morphologic and functional sensory and autonomic findings were compared with data extracted from our age- and sex-stratified normative dataset of controls that included 200 healthy volunteers [22–24]. Further morphological analyses were performed on sections and slides from controls’ skin biopsies, available at Skin Biopsies laboratory archive of ICS Maugeri - IRCCS of Telese Terme. The study was approved by the Institutional Review Board (Fondazione G. Pascale n. “5/15 Maugeri”) and all patients gave their informed written consent to participate in the study.
Preliminary data on sudomotor morphology and function at distal leg of this cohort of patients have been recently described [25] and are included here to assess potential correlations with synuclein deposits in cutaneous nerves.
Clinical scales
Motor impairment was assessed using the clinician-scored motor evaluation (part III) of the Movement Disorders Society Unified Parkinson Disease rating scale (MDS-UPDRS) [26] and Hoehn and Yahr staging score (H&Y) [27]. For patients with a diagnosis of MSA, the Unified Multiple System Atrophy Rating Scale (UMSARS) [28] was also applied. We calculated the ratio between H&Y stage (I to V) and disease duration (years) to give a measure of disease progression. The non-motor symptoms scale (NMSS) [29], the COMPASS-31 questionnaire [30], and the Small Fiber Neuropathy Symptoms Inventory Questionnaire (SFN-SIQ) [31] were used to assess non-motor symptoms and in particular, autonomic and sensory symptoms.
Sensory function
Quantitative sensory testing
All tests were performed at the hand and dorsal foot using a standardized protocol [32] on a total number of 70 patients (50 PD and 20 MSA-p).
Mechanical pain perception was evaluated with a calibrated monofilament connected to a sharp, nonpenetrating probe. The percent of stimuli out of 10 perceived as painful was recorded. Tactile thresholds were assessed with calibrated monofilaments (Semmes-Weinstein). Thermal thresholds (cold, warm, cold pain and heat pain) were evaluated with a thermal sensory analyzer (TSA2001, Medoc, Ramat Yishai, Israel) and the method of limits [32].
Autonomic sudomotor function
Sympathetic skin response (SSR)
SSR was evoked in 56 patients (10 with MSA-p and 46 with PD) by random single 1-ms stimuli of 5 to 20 mA intensity delivered at the volar wrist and recorded simultaneously at the hands and feet. It was used a bandpass of 0.2–100 Hz and skin temperature was maintained between 32° and 34°C.
Dynamic sweat test (DST)
Postganglionic sudomotor function was evaluated in all patients at lateral distal leg bilaterally using the DST [33] and assessing sweat output (nL/min) per gland and per skin area (cm2), and density of activated sweat gland per cm2 after stimulation with 1% pilocarpine iontophoresis.
Morphology study
Skin biopsy
Patients recruited at the London center underwent 3-mm-punch-skin biopsies from the leg bilaterally. All patients recruited at the Naples and Telese centers underwent four 3-mm-punch-biopsies from the leg and thigh bilaterally and 60 of them accepted an additional 2.5-mm-biopsy from the fifth fingertip. Ten patients (5 MSA-p and 5 PD) also underwent biopsies from the cervical site (C8) and 5 patients had an additional biopsy from the leg.
All 3 centers used the same protocol for performing skin biopsies. In particular, for hairy skin, a hair follicle was included in the specimen to maximize the sampling of arrector pili muscles and sweat glands. Samples were fixed in Zamboni solution for 5 h, cryoprotected in a solution of polyvinylpyrrolidone cryoprotectant and kept at –20°C until processed. Fifty microns-thick sections were obtained using a freezing sliding microtome (Leica 2000). Sections were processed using indirect immunofluorescence technique according to standard procedures [6] and a large panel of primary antibodies to mark neural and vascular structures (Table 1). Species-specific secondary antibodies coupled with Cy2, Cy3, and Cy5 fluorophores were used to visualize the marked structures. Digital confocal images were acquired using a non-laser confocal system (Apotome2 Zeiss, Jena, Germany, EU). Z-stacks of at least fifteen 2μm optical sections at 20x magnification were considered for quantification of nerves in dermal annexes. Intraepidermal nerve fiber (IENF) density was obtained according to current guidelines [34]. Quantification of pilomotor and sudomotor nerves using pan-neuronal and selective cholinergic and noradrenergic markers was performed following previously validated procedures [7, 35]. Briefly, for pilomotor nerve fibers (PNF), only arrector pili muscle segments running with their major axis parallel to the focal plane were considered for quantification and then acquired at the confocal microscope. The single optical section having the highest number of fibers running for at least 100μm parallel to the major axis of the muscle was selected from the Z-stack. A line intercepting the highest number of fibers in focus was then traced, perpendicular to the major axis of the muscle. PNF density was calculated as number of intercepts per muscle width in mm (fibers/mm). For each skin biopsy PNF density was expressed as the average of the measurements performed in all muscles suitable for quantification for each staining. For sudomotor nerve quantification, we used Neurolucida 360 software (Microbrightfield Bioscience [MBFB], Williston, VT), that identified and traced nerve trajectories that were morphologically compatible with the nerve structure. Sudomotor nerve length density (SNF) was obtained by dividing the total length of sudomotor nerves over the volume of the acquired sweat glands (nm/μm3). All measurements were performed by the same operator who was blinded to the diagnosis of all subjects.
Name, source, and dilution of immunohistochemical markers
p-α-syn staining
In preliminary experiments, the mouse antibody recognizing synuclein phosphorylated at 129 ser (Wako #015-25191) was tested at increasing dilutions. The dilution that allowed the best signal/background (1:6000) was then applied to all skin samples.
At least five 50μm-thick sections for each sample were double stained with p-α-syn and PGP. In a subgroup of 20 subjects, we applied a staining protocol including consecutive sections double stained with p-α-syn and CGRP, p-α-syn and Sub-P, p-α-syn and Dbh, and p-α-syn and VIP, to look for colocalization of p-α-syn with peptidergic and autonomic fibers. In addition, all sections from an additional skin biopsy from the leg of five subjects were processed with p-α-syn and PGP to assess the presence of p-α-syn deposits in consecutive sections of the entire specimen.
We searched for the presence of p-α-syn in all axons around autonomic structures such as sweat glands, vessels and arrector pili muscles, or running in the dermis as nerve fascicles, or as isolated nerve fibers. Colocalization with PGP-9.5 or other neural markers was required to consider p-α-syn positivity in axons.
To characterize the pattern of cutaneous p-α-syn distribution, we recorded whether p-α-syn deposits were absent (0) or present (1) in nerve fibers in the following cutaneous structures (Fig. 1):
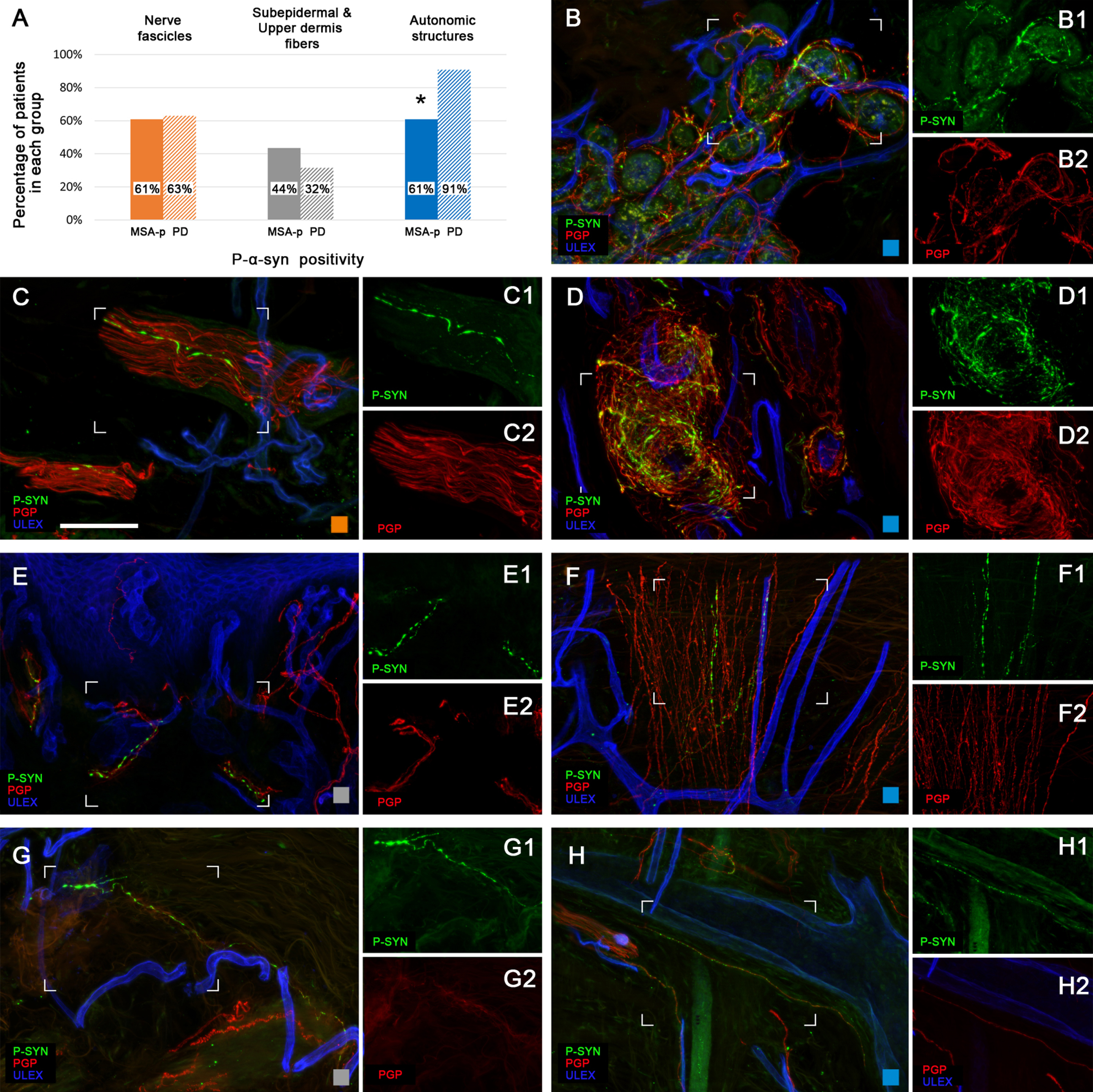
p-α-syn deposits in cutaneous nerves. The graphic in the upper left corner (A) shows the percentage of p-α-syn in MSA-p and PD in dermal structures; *
Blood vessels;
Sweat glands;
Arrector pili muscles (only in hairy skin) or Arteriovenous anastomoses (only in glabrous skin);
Dermal nerve bundles;
Subepidermal neural plexus (within 200μm below the basement membrane);
Isolated fibers in the upper dermis (between 200 and 500μm below the basement membrane);
Categories one to three are related to fibers that, based on the identification of their target, can be identified as certainly “autonomic”.
For each skin biopsy, we obtained a p-α-syn total score that ranged from 0 (no p-α-syn deposits present) to 6 (at least one p-α-syn deposit found in all six categories) and a p-α-syn autonomic subscore that ranged from 0 to 3. For each subject, we calculated a total score and an autonomic subscore as the average from all sites.
We also searched for the presence of p-α-syn in skin biopsies from 30 healthy subjects in the same age range as patients. Two blinded operators (MN and GC) performed the p-α-syn assessment in all skin samples. Scores obtained from two operators (MN, GC) showed an excellent inter-rater reliability (coefficient = 0.9;
For morphological and functional tests, when both sides were tested we considered the average value from the two sides as representative.
Statistical analysis
All data are presented as mean±SD. The Kolmogorov-Smirnov test was used to assess normality of distribution for all parameters. The differences between MSA-p, PD, and healthy control groups were evaluated by the analysis of variance with Bonferroni correction.
Differences in p-α-syn positivity between PD and MSA-p were compared using the Chi-square test.
Intraclass correlation coefficient (ICC) was used to assess inter-rater reliability between different operators measuring presence/absence of p-α-syn.
Pearson and Spearman tests were used as appropriate to assess the correlation between demographic, clinical and morphologic data.
A receiver operating characteristic (ROC) curve analysis was used to verify the ability of the ratio autonomic p-α-syn subscore/total p-α-syn score to discriminate PD from MSA-p. We considered a value of
Data availability
Anonymized data will be shared upon request by a qualified investigator.
RESULTS
Clinical and demographic data are summarized in Table 2.
Clinical and demographic data
All data are expressed as mean±standard deviation.
There were no differences in age, sex and, disease duration in patients with PD and MSA-p. Patients with MSA-p had a more rapid disease progression, a higher score on MDS-UPDRS total and part III (motor score), H&Y, NMSS, and COMPASS-31 compared with PD patients. Non-motor symptoms paralleled motor involvement (
Morphological findings
We examined a total of 676 skin biopsies, including 416 biopsies from patients (200 samples from the leg, 146 from the thigh, 60 from the fingertip, and 10 from the neck) and 260 biopsies (100 from the thigh, 100 from the leg, and 60 from the fingertip) from 100 age and sex comparable healthy subjects for the assessment of sensory and autonomic nerve densities. In addition, we processed and analysed further 90 biopsies from 30 healthy subjects for assessing the presence of p-α-syn.
Cutaneous innervation analysis
Overall, the dermal and epidermal innervation was poor and irregular in both groups of patients compared to controls with the presence of degenerative phenomena such as axonal swelling, nerve fiber fragmentation, varicosities, and irregularities in the distribution of IENF (Supplementary Figure 1). The subepidermal plexus was generally poorer in the MSA-p. In PD patients, we frequently observed nerve fiber clusters as well as aspects of hyperinnervation in the upper dermis and increased IENF branching, features that we rarely observed in patients with MSA-p (Fig. 2).
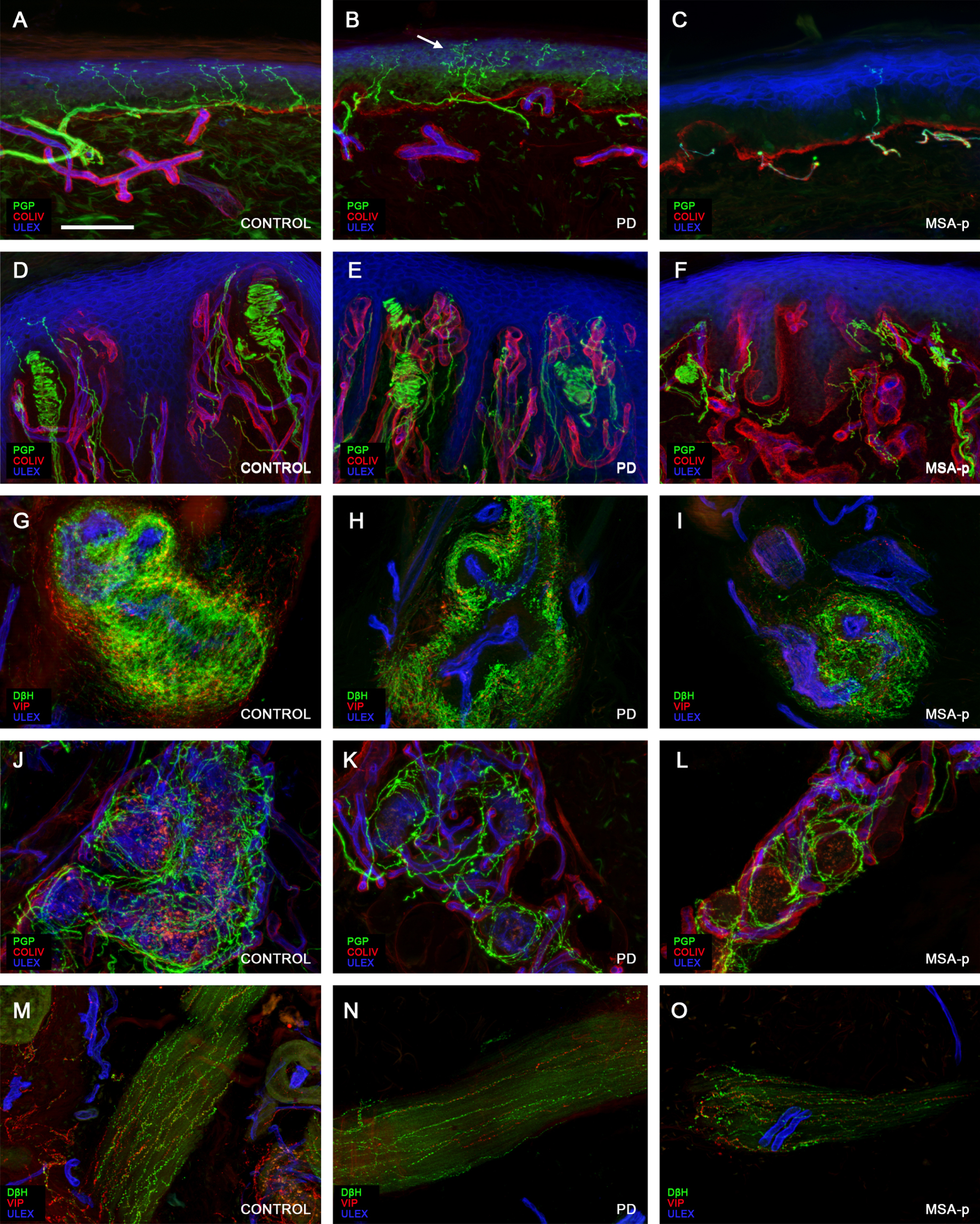
Sensory and autonomic innervation. Confocal images showing somatic and autonomic nerve fiber loss in PD and MSA-p patients. Compared to the control (A) in the skin of a PD patient (B) IENF loss is associated with aspects of nerve regeneration such as subepidermal sprouting (arrow in B) and increased branching. In a MSA-p patient (C) the loss of IENF is even more evident with lack of regenerative signs. Also the loss of Meissner corpuscles in glabrous skin is moderate in PD (E) compared to control (D) and more severe in MSA-p (F). Compared to the complex and rich innervation of arteriovenous anastomosis in the fingertip of the control (G) there is a moderate nerve loss in PD and a more severe loss in a MSA-p patient. The loss of sudomotor nerves is moderate in PD (K compared to J) and severe in MSA-p patients (L compared to J and K). Also pilomotor nerve loss was more severe in MSA-p (O) compared to PD (N) patients and to the control (M). Scale bar is 100μm.
Quantitative data of cutaneous sensory and autonomic nerve fibers density for each site in patients and controls are reported in Table 3.
Cutaneous sensory and autonomic nerve fiber density in MSA-p, PD, and controls
All data are expressed as mean±standard deviation;
Sensory nerves
Quantitative nerve analysis revealed a significant loss of IENF in the thigh, leg, and fingertip in patients compared to controls. The loss of IENFs was more severe in MSA-p than in PD at the leg. A moderate reduction of MC and IME was observed in glabrous skin of patients compared to controls. The loss of MC was higher in MSA-p compared to PD.
Autonomic nerves
Arrector pili muscles were detected in samples from all patients except in the leg of 2 MSA-p and 2 PD. Sweat glands were detected in the skin of all patients except in the leg of two patients with MSA-p. Overall, in patients, we quantified the innervation of 1,220 sweat glands and 1,980 arrector pili muscles in the leg, 949 sweat glands and 1,491 arrector pili muscles in the thigh, and 422 sweat glands in the fingertip.
There was a significant reduction in cholinergic VIP-immunoreactive (-ir) sudomotor and pilomotor nerves in patient skin compared to controls at all sites. There was a loss of noradrenergic Dbh-ir pilomotor nerves in patients compared to controls only at the distal leg. Sudomotor (PGP-ir and VIP-ir) and pilomotor (PGP-ir and Dbh-ir) nerve densities at distal leg and pilomotor nerves (PGP-ir) at the thigh were lower in MSA-p compared to PD.
Neural p-α-syn analysis
In the patient samples, we observed different patterns of p-α-syn deposits, from a fine granular bright staining often around sweat glands, to heavy deposits in axonal swellings. Very often we observed such deposits running for long tracts in nerve fascicles (Fig. 3). Interestingly we frequently observed such p-α-syn positive nerve fibers in consecutive sections of the same sample as well as in samples from multiple sites of the same subject, suggesting that the distribution in cutaneous nerves is less patchy than previously described [36].
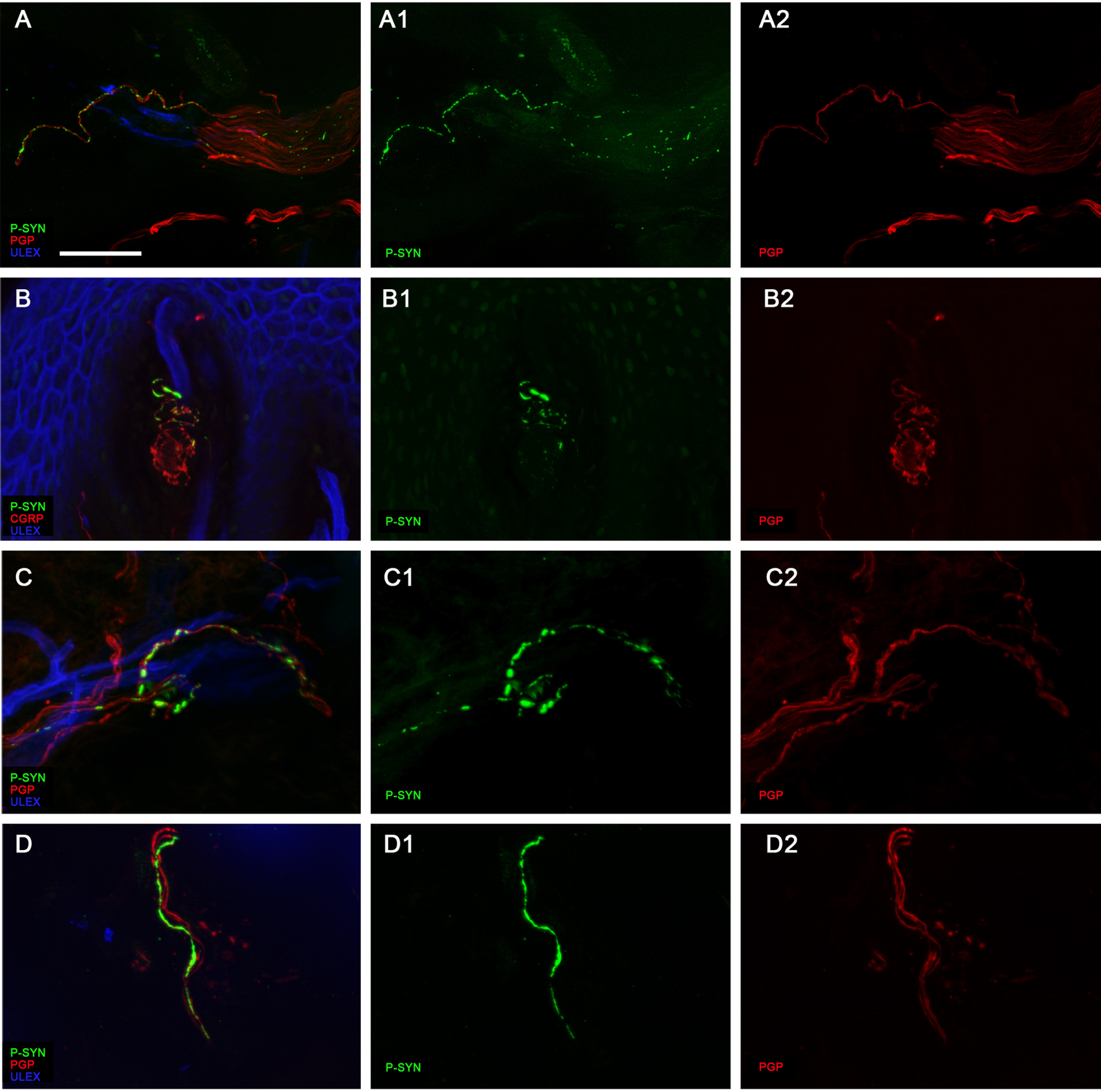
Patterns of neural p-α-syn deposits. Confocal images showing the different patterns of p-α-syn deposits within nerve fibers. In “A” tiny deposits are scattered along fibers in a nerve fascicle. In “B” larger deposits are located within the small fiber component (CGRP+) of a Meissner corpuscle. In “C” p-α-syn deposits appear as multiple adjacent large agglomerates along the fibers that distort its regular shape. In “D” thick p-α-syn deposits completely engulf the nerve fiber along its course. Scale bar is 100μm.
In dedicated experiments we found that p-α-syn deposits could colocalize with Sub-P, CGRP, D
In the fingertip, most of the deposits were localized around arteriovenous anastomoses (Fig. 1E). Sometimes p-α-syn deposits were found in dermal papillae in fibers directed to Meissner corpuscles and double marked with p-α-syn/CGRP (Fig. 3B).
No p-α-syn deposits were found in cutaneous nerves of controls. p-α-syn deposits were present in 95% of our total cohort of patients (91% of MSA-p and 96% of PD).
p-α-syn deposits were observed in the leg of 91/100 patients (91%), in the fingertip of 56/60 (93%), and in the thigh of 61/73 (83%) patients. Percentage of patients with p-α-syn deposits in the cervical site (90%) was similar to that observed at leg (91%).
The total p-α-syn score did not differ (
Considering all skin samples from the entire cohort of patients, p-α-syn deposits were present in perivascular fibers (69%), sudomotor fibers (62%), nerve fascicles (62%), pilomotor fibers (39%), subepidermal fibers (23%), and isolated nerve fibers (22%).
Neural p-α-syn deposits in autonomic structures (blood vessels, sweat glands, and arrector pili muscles) were more frequently observed in PD compared to MSA-p patients (91% vs. 61%;
In PD compared to MSA-p, the autonomic p-α-syn subscore was higher (1.0±0.6 vs. 0.5±0.6;
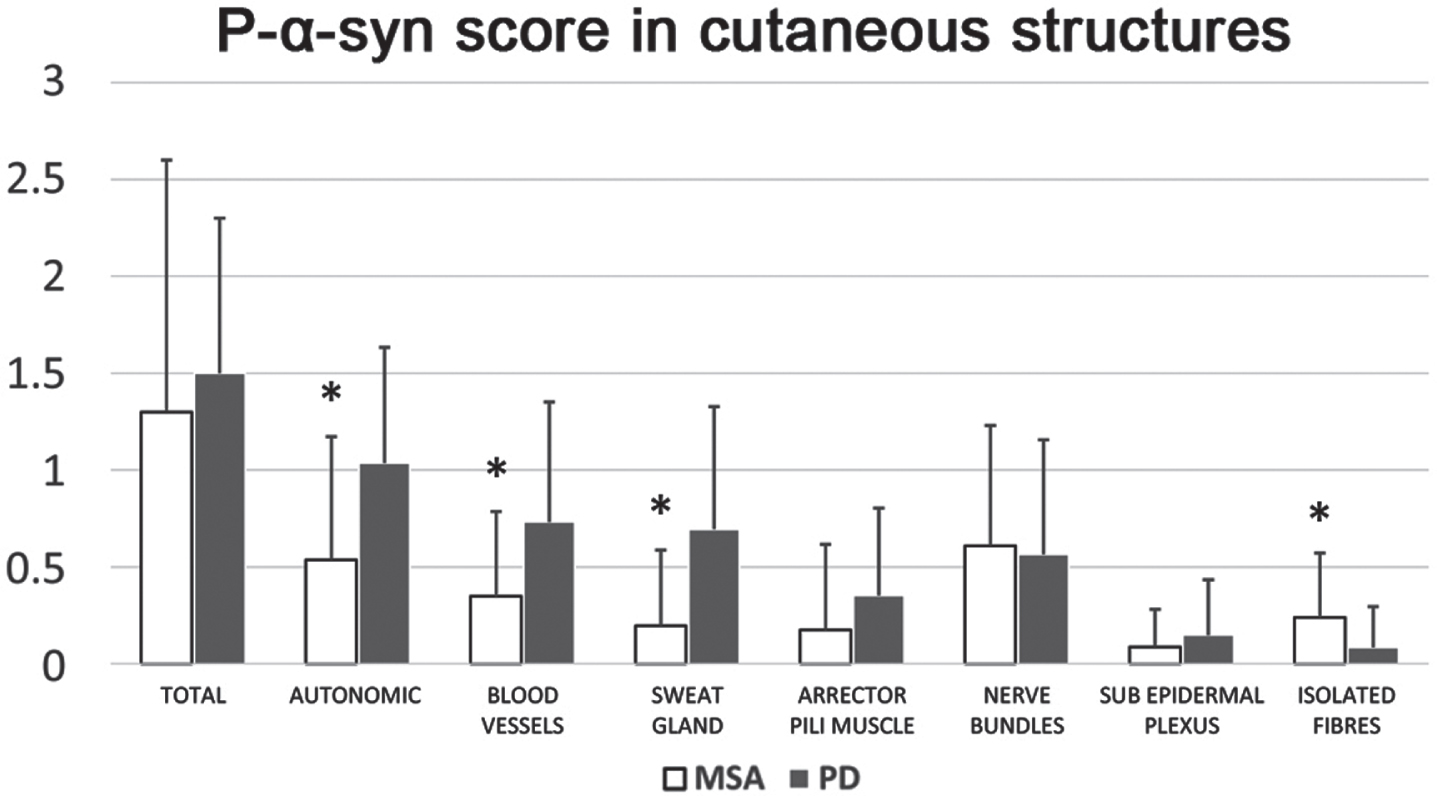
p-α-syn score in cutaneous structures. The graphic summarizes p-α-syn positivity scores in the different cutaneous structures, the total score and the Autonomic sub-score that includes blood vessels, sweat glands and arrector pili muscles. *
Sensory and autonomic findings
A similar proportion of MSA-p and PD patients reported frequent sensory symptoms on the SFN-SIQ questionnaire (26% MSA-p, 25% PD). Also a similar percentage (10%) in both groups reported restless leg symptoms. Burning feet and/or bed sheet intolerance were reported by 13% of MSA-p and 8% of PD patients.
Tactile thresholds and mechanical pain perception were impaired in MSA-p and PD compared to controls. Moreover, we found in both groups of patients increased thermal thresholds for warm and heat pain in the hand and for warm, heat pain, cold, and cold pain in the foot. Although mean thermal thresholds were generally higher in MSA-p compared to PD, a significant difference was found only for cold at the hand dorsum (Fig. 5).
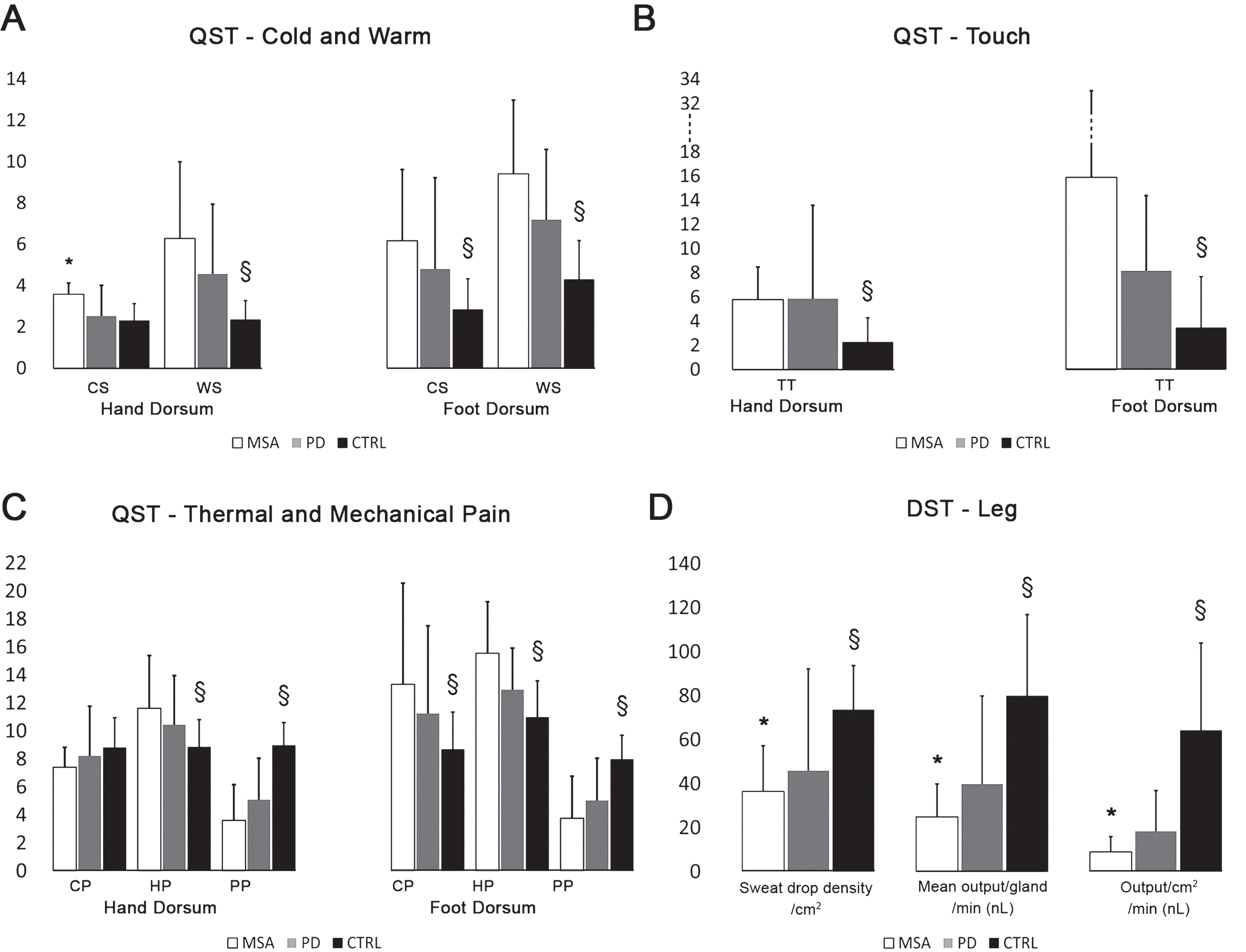
Quantitative sensory testing and dynamic sweat test in MSA, PD, and controls. Thermal threshold values for CS, WS, CP, and HP are expressed in degree Celsius; TT values are expressed in millinewtons and PP values are expressed as number of stimuli perceived as painful out of 10. CS = cold threshold; WS = warm threshold; CP = cold pain; HP = heat pain; PP = mechanical pain perception; TT = tactile threshold. Data are expressed as average±standard deviation. *
Among the autonomic symptoms assessed using the COMPASS-31 questionnaire, 100% of MSA-p patients reported urinary symptoms compared to 30% of PD. Gastrointestinal symptoms were frequent in both groups (95% of PD and about 90% ofMSA-p).
SSR amplitude at foot was lower (
The DST, after pilocarpine stimulation, showed a lower sweat output/cm2 (Fig. 5D) in all patients compared to controls and in MSA-p compared to PD.
Correlations between nerve loss and functional impairment
Considering the total cohort of patients, the IENF density from leg strongly correlated with IENF density from thigh (
A correlation between autonomic nerve loss and disease severity was also observed (PNF PGP-ir and VIP-ir at thigh inversely correlated with UPDRS III;
Finally IENF density at leg correlated with disease progression rate (
Correlation between nerve loss and p-α-syn deposits
When considering all patients with alpha-synucleinopathies, we found a positive correlation between p-α-syn total score and IENF at leg (
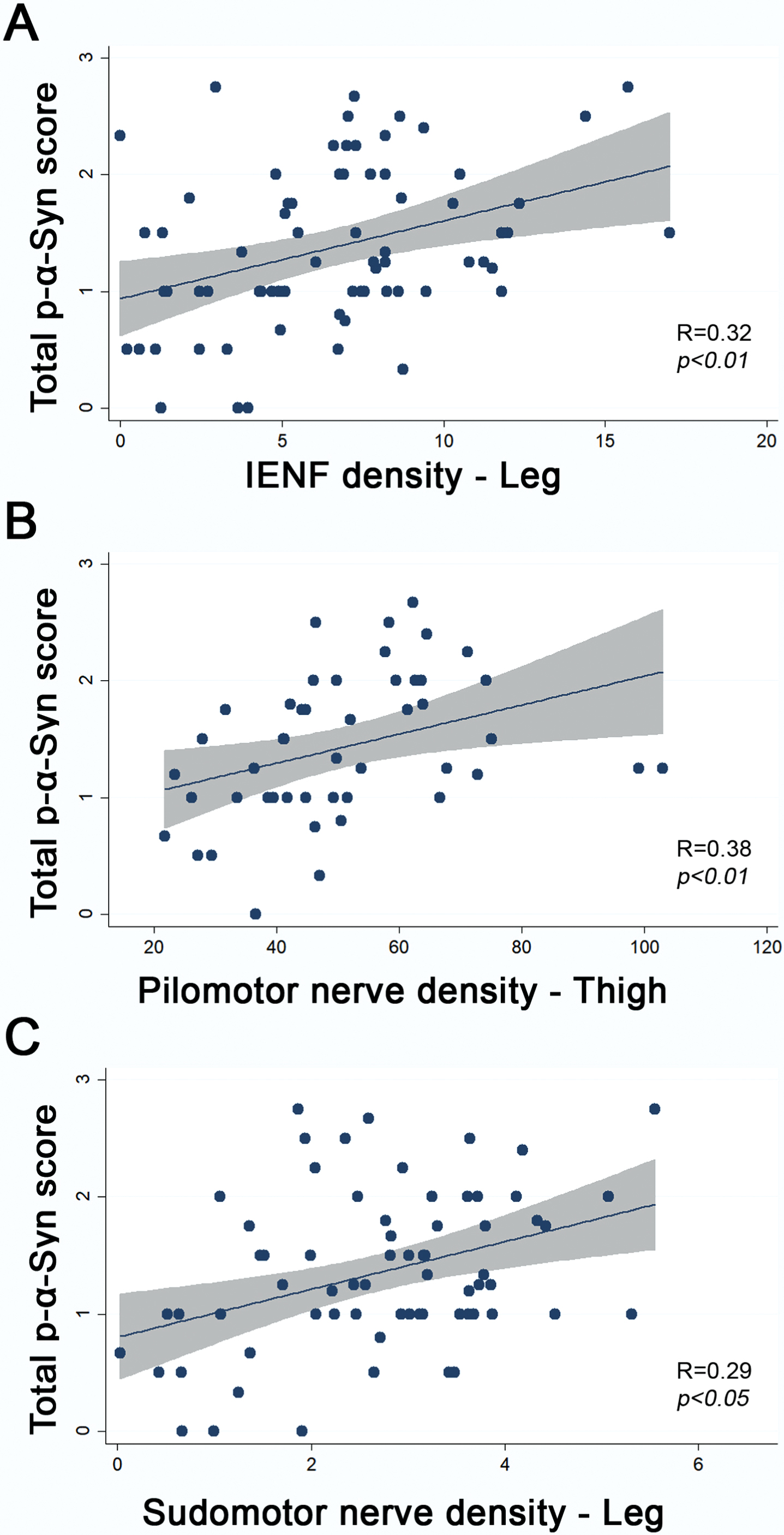
Correlations between p-α-syn score with sensory and autonomic innervation. R, regression coefficient; p, significance with Spearman Test.
DISCUSSION
In our study we found evidence of early peripheral nervous system involvement in a population of 57 PD and 43 MSA-p patients, demonstrating a loss of sensory and autonomic nerves that correlated with functional and clinical impairment with a more severe picture in MSA-p than in PD. Deposits of p-α-syn were found in the majority of patients, without significant differences among sites in both groups, and an higher occurrence in autonomic nerves in PD.
p-α-syn deposits were associated with preserved innervation.
Cutaneous sensory and autonomic nerve loss
The loss of peripheral sensory and autonomic endings in our PD patients is in line with previous works [6] and further supports the hypothesis that cutaneous denervation is an intrinsic feature of the disease [37], unrelated to L-Dopa treatment [38] and occurring in the early stage of the disorder. Instead, in MSA, studies on cutaneous innervation are more scanty and discordant [17, 40] reporting mostly preserved innervation. Therefore, in our study, the loss of sensory unmyelinated (IENF) and myelinated endings (MC, IME) as well as the loss of autonomic sudomotor and pilomotor nerves in all the examined body sites of MSA-p patients, compared to controls, is a relatively new finding. Moreover, the nerve fiber loss was overall more severe in MSA-p compared to PD with lower densities of IENF at leg, MC at fingertip, autonomic pilomotor nerves at thigh and leg, and sudomotor nerves at leg. Moreover the features of regeneration such as nerve fiber clusters, hyperinnervation, and increased IENF branching were often found in PD patients and not in MSA-p. This finding suggests that the peripheral involvement occurs early also in MSA-p and should be considered an intrinsic aspect of this multisystemic degenerative pathology and the lack of overt cutaneous regeneration is in line with the overall higher disease aggressiveness compared to PD.
Cutaneous denervation may contribute to the occurrence of pain and dysautonomia in alpha-synucleinopathies. Pain has been described in both PD and MSA, and abnormal pain sensitivity has been demonstrated at early stages in both conditions [41, 42].
In our PD and MSA-p patient population, about 1 out of 4 reported sensory disturbances and among them burning feet, bed sheet intolerance and restless leg syndrome. We found also an impaired perception of innocuous and noxious thermal stimuli, as well as of mechanical pain and touch in both groups of patients compared to controls. Sensory thresholds abnormalities were overall more severe in MSA-p compared to PD although a significant difference between the two patient populations was found only for cold thresholds at hand dorsum.
Autonomic dysfunction is common in the synucleinopathies [43] and it may manifest at any stage of the disease course and often preceding the onset of motor impairment. Dysautonomia is due to the involvement of both central and peripheral autonomic networks, with the first considered more affected in MSA and the second in PD [43]. In our study population, as expected, we observed higher autonomic symptoms score (COMPASS 31) in MSA-p compared to PD. We found higher postganglionic sudomotor dysfunction and higher cutaneous autonomic denervation in MSA-p compared to PD at both proximal site and distal leg as recently described [25].
Our finding is not in line with previous works reporting a more severe denervation in PD compared to MSA. However we enrolled subjects in early stage of disease and naïve to L-Dopa treatment and only MSA-p patients while previous studies included older PD patients, with longer disease duration, and with orthostatic hypotension [17, 44] and mixed cohorts of cerebellar and parkinsonian-type MSA patients.
p-α-syn deposits in cutaneous nerves
The physiological role of α-synuclein is still debated. α-synuclein is predominantly located in the presynaptic terminals of neurons and is thought to play an important role in synaptic plasticity [45]. Post-translational modifications of α-synuclein, like phosphorylation, favor oligomerization [46] and α-synuclein inclusions
In the last decades, the identification of p-α-syn deposits in peripheral nervous system rendered it a potential pre-mortem biomarker of the disease. Several skin biopsy studies demonstrated the occurrence of p-α-syn in dermal nerves in alpha synucleinopathies and differentiated these from tauopathies with high sensitivity and specificity [11, 15]. p-α-syn deposits are described to be mostly found in autonomic nerves in PD and in subepidermal plexus in MSA. However recent comprehensive reviews of the literature on p-α-syn in skin biopsy addressed the methodological problems and the lack of standardized procedures that may be the major cause of discordant results in previous studies [20, 52].
We aimed at improving signal/noise ratio and we were able to visualize p-α-syn deposits in the majority of skin samples (95%). We found p-α-syn deposits mostly in perivascular nerve fibers, in sweat glands and nerve fascicles and less frequently in pilomotor nerves, subepidermal fibers and isolated fibers.
The total p-α-syn score (indicating the spread of p-α-syn distribution among different cutaneous structures, and not the quantity of p-α-syn) was not different in the two groups of patients indicating a widespread involvement of all skin structure in our patient cohort. We found higher occurrence of p-α-syn deposits in autonomic nerves (91% vs. 61%) in PD compared to MSA-p. The ratio between autonomic and total p-α-syn score was higher in PD compared to MSA-p and efficiently differentiated the two conditions with a sensitivity of 71.2% and specificity of 85.7%.
Conversely, p-α-syn deposits in the subepidermal neural plexus and isolated nerve fibers, although more frequently observed in MSA-p, could not differentiate the two conditions. We did not find significant differences in p-α-syn positivity among sites (leg, thigh, and fingertip) and although only a limited number of skin biopsy was performed on the neck, we observed a similar occurrence of p-α-syn deposits in the cervical site as in the leg. However leg and fingertip revealed almost always p-α-syn deposits and the amount of p-α-syn appeared relatively higher in the fingertip, around arterio-venous anastomoses.
The occurrence of p-α-syn in the majority of our patients irrespective of the diagnosis, imply that PD and MSA are multisystemic diseases sharing, at least in part, common pathogenetic mechanisms with an early involvement of peripheral cutaneous nerves.
This finding seems apparently in contrast with the normal cardiac innervation observed with the MIBG in MSA but not in PD [18, 44]. However the study of skin innervation provides a different point of view with the analysis of nerve fiber populations that may be preferentially affected compared to cardiac innervation.
We found a positive correlation between the p-α-syn deposits and the density of epidermal, pilomotor and sudomotor nerve fibers and the sudomotor function. Therefore p-α-syn deposits are greater in patients with more preserved fibers and function and may decrease and be more difficult to visualize in the skin with the progression of the disease and severe denervation. The latter is confirmed by the inverse correlation between the p-α-syn autonomic deposits and the disease progression and NMSS indicating that p-α-syn deposits are associated with slower disease progression, relatively preserved innervation and less severe symptoms score. The overall findings support the hypothesis that p-α-syn deposition is as an event that precedes nerve degeneration and is affected by the severity and rate of progression of the disorder. Further progression of the degenerative process, together with the loss of nerve fibers, might be associated with loss of p-α-syn aggregates likely removed by the macrophage mediated inflammatory response, as observed in the gut [53].
Of relevance is also the widespread patterns of p-α-syn deposits abnormalities we have observed in this patients cohort and ranging from a fine granular bright staining to tiny segments along the fiber, to heavy deposits in axonal swellings. This might indicate different stages of the degenerative process resulting in progressive accumulation of aggregates paralleling more evident degenerative aspects such as reduced PGP immunoreactivity (with a faint staining), axonal swelling and fragmentation.
Limitations
We present detailed assessment of the distribution of p-α-syn deposits in different body sites and in different nerve fiber populations but we did not perform a quantification of the total amount of such deposits in cutaneous nerves. Moreover, our findings are based on a cross-sectional assessment of cutaneous sensory and autonomic innervation and p-α-syn deposits that should be confirmed in a longitudinal study.
Conclusive remarks
Cutaneous sensory and autonomic dysfunction and denervation occurs early in both PD and MSA-p and should be considered an intrinsic feature of these multisystemic disorders contributing to clinical sensory and autonomic features.
Cutaneous sensory and autonomic impairment is more severe in MSA-p than in PD in contrast with current knowledge.
The presence of p-α-syn deposits in autonomic, but not in somatic nerves appears able to differentiate PD from MSA-p.
p-α-syn deposits visualization in the skin is associated with preserved innervation and slower disease progression.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This study was financed by Italian Ministry of Health “Ricerca Finalizzata 2013” – project code PE-2013-02359028. Dr Valeria Iodice is supported by National Institute for Health Research University College London Hospitals Biomedical Research Centre. Dr Shiwen Koay was supported by the Guarantors of Brain Entry Fellowship.
