Abstract
Background:
Pathological changes in the brain can affect the gastrointestinal tract, whereas there is less evidence regarding the brain-gut axis.
Objective:
To identify whether cerebral endogenous phosphorylated α-synuclein induces gastrointestinal dysfunction via the brain-gut axis, mediated by the vagus nerve.
Methods:
α-syn N103/tau N368 preformed fibrils were injected into the dorsal lateral striatum of rodents, and the cerebral and colonic synucleinopathies and changes in the enteric nervous system were analyzed. Moreover, subdiaphragmatic vagotomy was conducted to confirm the role of the vagus nerve in brain-gut propagation.
Results:
An anterograde propagation of phosphorylated α-synuclein from the brain to the proximal colon mainly via the vagus nerve was observed at one month. The accumulation of phosphorylated α-synuclein was detected in the proximal colon over time, accompanied by infiltration of macrophages and eosinophils in the mucosa and submucosa. Upon injection with lower doses of preformed fibrils, the accumulation of phosphorylated α-synuclein and dopaminergic neuron loss was reduced to levels consistent with control at six months, while the expression levels of GFAP, Iba-1, and IL-6 increased. Under high preformed fibrils dose conditions, fecal traits and gastrointestinal motility were significantly reduced at six months, and aggregations of phosphorylated α-synuclein and an increasing level of IL-1β appeared.
Conclusion:
Induced endogenous α-synuclein can quickly propagate into the proximal colon mainly via the vagus nerve. Injections of low doses of preformed fibrils can elicit recovery of the enteric nervous system and degradation of α-synuclein aggregates whereas high doses cause accumulation of pathological α-synuclein, enteric inflammation, and prominent gastrointestinal dysfunction.
INTRODUCTION
Parkinson’s disease (PD) is a disorder characterized by the neurodegeneration of dopaminergic neurons in the substantia nigra (SN). Its hallmark is the appearance of Lewy bodies and Lewy neurites, which both contain misfolded α-synuclein (α-syn) [1]. Phosphorylated α-syn (α-syn pSer129) is a prominent pathological feature of PD; it can promote the transition of α-syn monomers to oligomers or fibrils and thereby aggregate α-syn. Phosphorylated tau co-aggregates with phosphorylated α-syn in the Lewy bodies [2, 3], and the proteins interact with each other and synergistically promote fibrillization in vitro [4]. Previous studies have found that both tau N368 preformed fibrils (PFFs) and α-syn N103 PFFs are highly cell-permeable and will trigger the occurrence of endogenous α-syn pSer129 formation [5]. α-syn N103/tau N368 PFFs cleaved by δ-secretase are more neurotoxic and compact, and also travel faster along the vagus nerve from the gut to the brain than their full length or individual fragment fibrils [5]. Thus, α-syn N103/tau N368 PFFs might be a promising candidate for PD pathology research, especially regarding the rostrocaudal progression of phosphorylated α-syn.
The enteric nervous system (ENS), the intrinsic nervous system of the gastrointestinal (GI) tract, plays an essential role in regulating GI motility and fluid secretion [6]. GI inflammation is believed to play an essential role in PD pathogenesis and α-syn aggregation [7, 8], shedding new light on potential avenues for the diagnosis and treatment of PD. PD often manifests as non-motor symptoms, including GI, cardiovascular, and genitourinary disorders. These symptoms may precede motor dysfunction and may severely impair the quality of life in patients with PD [9]. GI dysfunction is highly prevalent, ultimately affecting 80–90% of PD patients and includes symptoms such as dysphagia, gastroparesis, and chronic constipation that can deteriorate further with PD progression [10]. Phosphorylated α-syn pathology has been detected in different sites of the ENS (e.g., stomach, duodenum, and colon) and may precede the onset of motor disturbances in PD [11]. According to the Braak hypothesis, the ENS is considered to be the initial site of pathogenesis in some patients with PD [12], but whether PD starts from the ENS is still controversial.
According to reports, α-syn can spread bidirectionally via the vagus nerve or by non-vagal circuits between the brain and the ENS [13–16]. Moreover, rat brains exposed to 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine, 6-hydroxydopamine, and rotenone PD models, which create PD models [17, 18], showed that the brain pathology influenced ENS functions, causing alterations in enteric glial cells (EGCs), gut microbiota, and enteric motility. The results of an in vivo study revealed a route-specific transmission of pathological α-syn from the dorsal motor nucleus of the vagus nerve (DMV) to the stomach via vagal preganglionic projections without the appearance of enteric symptoms and pathology [19]. Injection of α-syn in the substantia nigra showed that the pathology in the brain can elicit subsequent alterations in the ileum [20]. Numerous studies have focused on the transfer of α-syn aggregates in the ENS to the brain eliciting PD pathology, but there is limited evidence supporting the fact that the accumulation of misfolded α-syn in the gut, seeded by brain-derived α-syn toxic aggregates, can lead to enteric inflammation and alterations of GI function. Here, we hypothesized that the injection of α-syn N103/tau N368 PFFs into the brains of wild-type rats would trigger induced stereotypic anterograde progression of α-syn-positive pathology and replicate the features of the neuropathology in the GI tract, which may be associated with enteric inflammation. We tested this hypothesis by stereotactic injection of α-syn N103/tau N368 PFFs into the right dorsolateral striatum to elucidate the role of the vagus nerve in the propagation of α-syn aggregates and the inflammatory response of the proximal colon.
MATERIALS AND METHODS
Study design
This study aimed to establish whether the injection of α-syn N103/tau N368 PFFs into the striatum of rats contributes to α-syn propagation and pathology in the ENS. All experiments were performed in a blinded manner. All rats were divided into two major groups (A–C and S1–S3). Group A–C comprised rats that were subjected to a single intraparenchymal injection of 15μg α-syn N103/tau N368 PFFs (n = 8 per subgroup A1–C1) or 30μg α-syn N103/tau N368 PFFs (n = 8 per subgroup A2–C2), whereas corresponding control rats were injected with the same volume of PBS (n = 8 per subgroup A3–C3). Rats were sacrificed at one month (Group A), three months (Group B), and six months (Group C) after brain stereotactic injections. To study the effects of a disrupted vagal CNS-gut pathway on the colon, rats were divided into Group S1 (sham surgery+30μg α-syn N103/tau N368 PFF injection into the right striatum, n = 5), Group S2 (vagotomy+30μg α-syn N103/tau N368 PFF injection into the right striatum, n = 5), and Group S3 (vagotomy+PBS injection into the right striatum, n = 4) and sacrificed one month after treatments.
For biochemical analysis, animals from groups A–C were decapitated, and both the proximal (ascending) colon and the brain were quickly removed. The proximal colon was cleaned with cold saline, fragmented into 5-mm long sections on an ice-cold surface, and stored at –80°C until further analysis. For histological analysis, half of the animals from Groups A–C and all animals from Group S underwent trans cardiac perfusion, and the brains, vagus nerves, and colons were removed, fixed, and embedded in paraffin until further analysis.
Further experimental details and protocols, including animal care/handling and the number of biological/technical replicates, are presented in the following sections and figure legends.
Animals
Adult male Sprague-Dawley rats (weight: 180–200 g, about 6 weeks old) were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd., China. All rats were housed in a specific-pathogen-free environment under controlled conditions (22±1°C, 55–65% humidity, 12-h light/dark cycle) with adequate access to food and water, and they were allowed to acclimate for one week before study initiation. All experiments were performed in accordance with the guidelines of the Laboratory Animals Ethics of Tongji Medical College, Huazhong University of Science and Technology. The study was approved by the Institutional Animal Care and Use Committee of Tongji Medical College, Huazhong University of Science and Technology, China (IACUC Number: 2593). Rats from each condition were randomly selected and sacrificed at the indicated time points to collect tissue for analysis and comparison. Researchers involved in data collection and analysis were not blinded to the experimental conditions.
Purification and generation of α-syn N103/tau N368 PFFs
Fibrils were prepared as described previously [5]. Briefly, they were generated in 500μL containing 3 mg/mL of α-syn N103 and tau N368 protein monomers in PFFs reaction buffer (50 mM Tris-HCl, pH 7.4, 50 mM NaCl). Determination of concentration was performed by bicinchoninic acid assay (BCA) (wavelength 595 nm). Then, the protein was lyophilized. Before using, the proteins were diluted in PBS and incubated for several days (usually 7–9 days) at 37°C with orbital shaking at 300 rpm until the samples appeared cloudy, and then sonication was applied on the ice for 30 min. PFFs were validated using the Thioflavin-T in vitro assay.
Transmission electron microscopy (TEM)
A 20μL sample was placed on carbon-coated copper grids and left for 3–5 min. Then, 2% phosphotungstic acid was added to the grids for 1–2 min, and excess liquid was moved with filter paper. The grids were allowed to dry at room temperature. A TEM system (Hitachi HT7800/HT7700, Japan) was used to capture negative stained TEM images of α-syn N103/tau N368 PFFs on Day 4 and Day 8 (Supplementary Figure 3).
Brian stereotaxic injections
The rats were deeply anesthetized with isoflurane (3% induction and 1.5%–2% maintenance in pure oxygen) and fixed on a stereotaxic apparatus (RWD Lifescience, China). A temperature controller system was used to keep the rats warm during the operation. A dose of α-syn N103/tau N368 PFFs (1.5μg/μL and 3.0μg/μL, 10μL) was injected into two points of the right dorsolateral striatum through a 10-μL microsyringe at 1μL/min. The stereotactic coordinates were as follows: anteroposterior +1.2 mm, mediolateral – 2.4 mm, and dorsoventral 4.0 mm from the dura, and anteroposterior +0.2 mm, mediolateral – 3.5 mm, and dorsoventral 4.0 mm from the dura. All control groups were injected with an equal volume of sterile PBS solution at the same coordinate points.
Subdiaphragmatic vagotomy and vagus nerve isolation
The rats were anesthetized with isoflurane before subdiaphragmatic vagotomy. Briefly, a midline abdominal incision was made, and the liver and stomach were carefully displaced to provide access to the esophagus. Both the dorsal and ventral branches of the vagus nerve were exposed and dissected. Each vagal nerve trunk was isolated from the surrounding connective tissue and vasculature and then ligated with surgical sutures, clearly cut at two levels, and the middle segment was extracted to confirm total vagotomy. Finally, the abdominal muscles and skin were sutured using surgical silk and wound clips, respectively. In addition, we performed sham surgeries in which each of the main vagal nerve trunks was exposed but not ligated or excised. After 7 days, the rats received a single intraparenchymal injection of α-syn N103/tau N368 PFFs or PBS into the right striatum. To study the role of the vagal pathway in the possible effects of GI function, rats were subjected to sham surgery + 30μg α-syn N103/tau N368 PFFs injection into the right striatum, vagotomy + 30μg α-syn N103/tau N368 PFFs injection into the right striatum, or vagotomy + PBS injection into the right striatum, and then sacrificed after one month. As expected, the postmortem size of the stomach increased significantly in vagotomized animals.
To isolate the vagus nerve, we opened the tracheal region after the rats had been euthanized and identified the trachea, carotid artery, and masticatory muscles. The trachea was removed to allow access to the carotid and the adhering vagus nerve, and to cleanly isolate the carotid from the surrounding tissues. Without cutting the vagus nerves, the tissues were immediately fixed in 4% paraformaldehyde.
Gastrointestinal function assessment
Fecal output and body weight
Before the experiments, baseline body weight and fecal output were measured by housing the rats in individual cages. Bodyweight was recorded before PFFs injection and one, three, and six months after PFFs injection. For the assessment of fecal output, rats were placed in a clean plexiglass cylinder for 15 min. After the test, fecal pellets were blotted on paper towels to absorb urine that may have been expelled during the task. The pellets were weighed, dried at room temperature for three days, and then reweighed to determine the total weight and proportion of water weight in the pellets.
Intestinal transit
Thirty minutes before sacrificing the rats, a solution of 2.5% Evans blue (CAS: 314-13-6, Sigma-Aldrich, St. Louis, MO, USA) in 1.5% methylcellulose (1.0 mL per animal) was intragastrically administered to the rats. After euthanasia, intestinal transit was measured as the distance from the pylorus to the most distal point of migration. The total length of the small intestine (distance from the pylorus to the ileocecal valve) and the distance traveled by the marker were measured and expressed as a percentage as follows: intestinal transit (%) = distance traveled by the marker / total length of the small intestine×100.
Tissue preparation
After assessment of the GI tract, three rats from each group were decapitated under isoflurane anesthesia for western blotting. The proximal colon was quickly removed, cut open on ice, and rinsed with ice-cold normal saline. The tissues were minced and homogenized in RIPA buffer containing protease inhibitor cocktail, phosphatase inhibitor cocktail, and PMSF (Servicebio, China), and then centrifuged at 14,000 rpm for 20 min at 4°C to obtain the supernatants.
For histological analyses, rats were first perfused through the ascending aorta with physiological saline solution and then with 4% ice-cold paraformaldehyde in 0.1 M phosphate buffer. The brain, vagus nerves in the rat neck, and proximal colon were collected and fixed in 4% paraformaldehyde solution at 4°C for 48 h. The fixed samples were dehydrated, embedded, and sectioned (i.e., brains and proximal colons were sectioned coronally, and bilateral vagus nerves were sectioned longitudinally) for immunohistochemistry and immunofluorescence staining.
Western blotting
Total protein concentrations in supernatants were measured using a BCA kit (Beyotime Biotechnology, China). Equal amounts of protein (40μg) from each sample were separated by 10% or 12% SDS-PAGE and then transferred to a polyvinylidene 205 difluoride membrane (Millipore, USA), followed by blocking in 5% non-fat milk or 5% bovine serum albumin (Servicebio) for 1.5 h at room temperature. After washing, the membranes were incubated using the following primary antibodies overnight at 4°C: anti-α-syn hosphor S129 rabbit monoclonal antibody (mAb) (1:1,000, Abcam, ab51253), anti-Tau (Ser202, Thr205) mouse mAb (AT8, 1:1000, Thermo, MN1020), anti-α-syn rabbit mAb (1:1,000, Abcam, ab212184), anti-tau rabbit mAb (1:1,000, Abcam, ab32057), anti-tyrosine hydroxylase (TH) rabbit mAb (1:1,000, Abcam, ab75875), anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mouse mAb (1:3,000, Abbkine, A01020), anti-GFAP mouse mAb (1:2,000, Proteintech, 16825-1-AP), anti-ionized calcium-binding adaptor molecule 1 (Iba1) rabbit mAb (1:2,000, Abcam, ab178846), anti-tumor necrosis factor (TNF-α) rabbit mAb (1:6,00, Proteintech, 17590-1-AP), anti-interleukin (IL)-1β rabbit polyclonal antibody (1:1,000, Proteintech, 16806-1-AP), and anti-IL6 mouse mAb (1:1,000, Proteintech, 66146-1-Ig). The day after incubation, the membranes were washed three times and incubated with the appropriate secondary horseradish peroxidase-conjugated antibodies for 1 h at room temperature: goat anti-rabbit IgG (1:10,000, Abbkine, A21020) or goat anti-mouse IgG (1:10,000, Abbkine, A21010). After washing, we used enhanced chemiluminescence kits (Biosharp, BL520A) to dye the membranes, and used a fluorescent chemiluminescence gel imaging system to detect bands (Syngene, United Kingdom). ImageJ software was used to analyze band intensities. Regions of interest were drawn around the bands of interest, and the integrated density was also measured. Relative integrated densities were calculated against the average of the control group samples on each membrane, which were normalized to housekeeping genes (GAPDH) or total protein (for α-syn, tau). All three samples were derived from the same experimental group, and the blots were processed in parallel and repeated three times.
Immunohistochemistry staining
Immunohistochemistry was performed as described previously [21]. Briefly, the paraffin-embedded brains, colons, and vagus nerves were cut in the coronal or longitudinal plane with a 4-μm thickness and mounted on slides. After deparaffinization and rehydration, the sections were treated with Tris-EDTA buffer (pH 9.0) or citrate buffer (pH 6.0) at 95°C for 30 min for antigen retrieval. The tissue samples were then treated with 3% H2O2 for 20 min to block endogenous biotin. After rinsing thrice with phosphate-buffered saline (pH 7.4), the sections were blocked with 5% bovine serum albumin (Sigma-Aldrich) for 30 min at room temperature, then incubated with the following primary antibodies overnight (48 h for vagus nerves) at 4°C: anti-α-syn phospho S129 rabbit mAb (1:1,000, Biolegend, MMS-5091) with and without proteinase K (10μg/mL; CAS: 39450-01-6, MedChemExpress, USA) pretreatment, anti-Tau (Ser202, Thr205) mouse mAb (AT8, 1:1,000, Thermo, MN1020), anti-GFAP mouse mAb (1:1,200, Proteintech, 16825-1-AP), and anti-Iba1 rabbit mAb (1:2,000, Abcam, ab178846). After antigen retrieval, treatment with proteinase K was performed for 30 min at room temperature. Subsequently, the sections were washed and incubated with horseradish peroxidase-labeled secondary antibodies for 1 h at room temperature, followed by visualization with 3,3-diaminobenzidine solution, and counterstained with Harris hematoxylin (without counterstaining for vagus nerves), dehydration, and coverslipping. An Olympus camera (Olympus SV102, Japan) was connected to a microscope to obtain images under the same light intensity, and ImageJ software was used to measure the integrated density. Quantitative analysis of histological and fluorescence staining was performed using ImageJ software as well [22]. Integrated densities were measured in three different sections for each rat, and at least 20 randomly selected microscopic fields were examined in each section (objective, ×20). Integrated densities were calculated per unit area (average optical density (AOD) = integrated optical density (IOD) / area) and normalized to the average of the control group samples.
Histological evaluation of eosinophils
Sections from formalin-fixed full-thickness colonic samples were processed routinely, followed by staining with hematoxylin and eosin to detect eosinophils, which appeared as deep magenta cells. The density of eosinophils in the tunica mucosa/submucosa was evaluated. Cells were counted in three different sections of each rat, and at least 10 randomly selected microscopic fields were examined in each section (objective, ×20). The values obtained from all examined fields for each rat were averaged and expressed as the number of cells per square millimeter of tunica mucosa/submucosa areas, which were estimated using ImageJ software. These values were then used to calculate the mean values of each experimental group.
Immunofluorescence staining
Immunofluorescence staining was performed as described previously [21]. Sections were incubated with the primary antibodies α-syn phospho S129 rabbit mAb (1:1,000, Abcam, ab51253), TH rabbit mAb (1:1,000, Abcam, ab75875), ubiquitin rabbit polyclonal antibody (1:100, Proteintech, 10201-2-AP), and SQSTM1/p62 rabbit mAb (1:100, Abclonal, A19700) and the same procedure for immunohistochemistry staining was used. The sections were incubated with primary antibodies and washed three times. Thereafter, they were incubated in the dark with FITC-conjugated and CY3-conjugated secondary antibodies for 50 min, followed by incubation in 4’,6-diamidino-2-phenylindole (DAPI) solution for 10 min. Images were collected using a fluorescence microscope (Nikon Eclipse C1, Japan).
Thioflavin-S staining
Sections were deparaffinized and rehydrated after immunohistochemistry and then incubated at room temperature for 8 min with 0.3% filtered thioflavin-S solution (CAS: 13326-12-1, Shanghai Yuanye Bio-Technology Co., Ltd., China), which was diluted with 50% alcohol. After counterstaining with DAPI solution, images were collected using fluorescence microscopy (Nikon Eclipse C1, Japan).
Assessing monoamines by high-performance liquid chromatography coupled with electrochemical detection (HPLC-ECD)
Dopamine (DA; CAS: 62-31-7, Sigma-Aldrich), its metabolites (3,4-dihydroxyphenylacetic acid [DOPAC; CAS: 102-32-9, Sigma-Aldrich], homovanillic acid [HVA; CAS: 306-08-1, Sigma-Aldrich], and norepinephrine [NE; CAS: 329-56-6, MedChemExpress, USA]), and serotonin (5-HT; CAS: 153-98-0, Sigma-Aldrich) were measured by HPLC analysis. Measurements of catecholamines and 5-HT were performed as previously described [21]. Briefly, standards of monoamine mixtures (DA and HVA; 1 mg/mL, in 0.1 M perchloric acid and 0.1% L-cysteine) were prepared by diluting stock solutions with the mobile phase into 500, 166.7, 55.6, 6.2, and 2.1 ng/mL. The colonic tissue was homogenized and centrifuged at 4°C (14,000× g; 20 min). The remaining solution was filtered (0.22μm) and injected (50μL/injection) into the Waters 510 HPLC system and pumped through a column (Waters Spherisorb 5 mm ODS1, 4.6 mm×25 cm). For monoamine analyses, all samples were injected into the column with the temperature set at 30°C. Detection of compounds was performed using a Waters 2465 Electrochemical Detector. The electrode potential was 800 mV, and the mobile phase (100 mM sodium acetate, 85 mM citric acid, 3 mM sodium heptane sulfonate, 0.2 mM EDTA, and 8% methanol) had a flow rate of 1 mL/min. Data were processed with Empower 3 software and expressed as picograms per milligram of wet tissue.
Statistical analysis
Power analyses were conducted using G*Power (University of Düsseldorf) to estimate the number of animals needed to attain >90% power for each experiment. This was based on expected effect sizes of 0.85 calculated from our preliminary studies.
All data are presented as the mean±standard error of the mean (SEM). All data were analyzed using GraphPad Prism software (version 8.0). Group comparisons were made using one-way analysis of variance with Tukey’s or Dunnett’s T3 multiple comparisons test. Measurements were made from distinct samples, and statistics assumed a Gaussian distribution. The annotation for significance values is: *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001. Data were visualized using GraphPad Prism 8.0.
RESULTS
Accumulation of endogenous pathological α-syn following α-syn N103/tau N368 PFFs injection into the brain
We assessed the distribution and accumulation of endogenous α-syn pSer129 at different levels of the coronal brain sections after the injection of α-synN103/tauN368 PFFs (Fig. 1a). Two different doses of α-syn N103/tau N368 PFFs were unilaterally injected into the right striatum of rats. One month after brain injection, α-syn pSer129 immunostaining was observed in the DMV and nucleus of the solitary tract (NTS), which are associated with gastric motility. More was observed in the anterior olfactory nucleus, locus coeruleus (LC), motor cortex, striatum, and SN as well (Fig. 1b). α-syn pSer129 mainly aggregated in the ipsilateral regions. Proteinase-K-resistance of α-syn pSer129 aggregates indicated their insolubility. In addition, the accumulation of phosphorylated tau detected by the commonly used antibody against phosphorylated tau, AT8, was not as significant as α-syn pSer129 in all the above regions (Fig. 1c). The widespread occurrence of α-syn pSer129 in the brain post-injection indicates rostrocaudal brain spreading of this pathological protein.
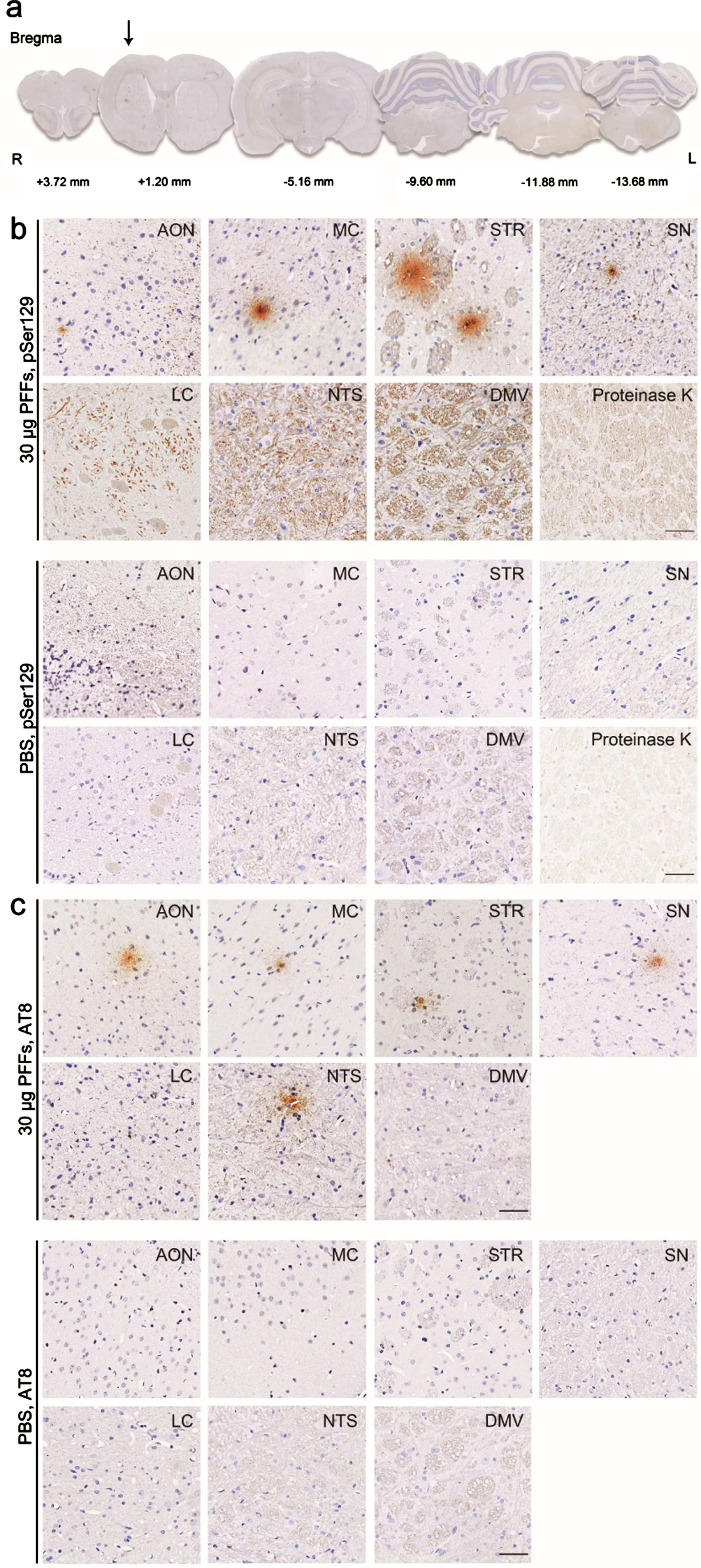
Widespread distribution of α-syn pSer129 in the brain. a) Rats received a single intraparenchymal injection of α-syn N103/tau N368 PFFs into the right striatum. Analyses were performed in eight rats sacrificed after one month. Representative images show brain sections at different bregma levels stained with anti-α-syn pSer129 antibody. For better illustration, different scale factors were used to resize each section. Representative high-magnification photomicrographs show the phosphorylated α-syn (b) and phosphorylated tau pathology (c) in several brain areas, including the anterior olfactory nucleus (AON), motor cortex (MC), striatum (STR), substantia nigra (SN), locus coeruleus (LC), nucleus of the solitary tract (NTS), and dorsal motor nucleus of the vagus nerve (DMV). Proteinase-K-resistant aggregates of α-syn pSer129 in the SN indicate that these α-syn pSer129 aggregates were insoluble. n = 4 each group. Scale bar = 50μm.
Spreading of endogenous pathological α-syn from the brain to the gut and loss of dopamine (DA) neurons in the gut
GI dysfunction, highly prevalent in patients with PD, is controlled by ENS [23]. Various studies focusing on the ENS have indicated that almost all PD patients have aggregated phosphorylated α-syn in the GI tract [10, 25]. To experimentally confirm this observation and test whether phosphorylated α-syn aggregates in a time-dependent manner, α-syn N103/tau N368 PFFs were injected into the right striatum, which has abundant direct or indirect projections with other nuclei, including the SN, thalamus, cortex, amygdaloid nucleus and pedunculopontine tegmental nucleus [26]. At one month after injection, immunofluorescence co-staining with antibodies against both α-syn pSer129 and Ubiquitin or p62 revealed that there were numerous Lewy body-like inclusions in the proximal colon of rats compared to vehicle control (Fig. 2, top and middle panels). To confirm whether the inclusions observed with α-syn pSer129 antibody in the proximal colon were mature amyloid structures, we performed thioflavin-S staining (Fig. 2, bottom panels). To investigate whether loss of DA neurons had occurred in the GI tract, immunofluorescence staining for α-syn pSer129 and TH was performed in the proximal colon at one, three, and six months. The results demonstrated that some of α-syn pSer129 co-localized with TH-positive dopaminergic submucosal neurons (Fig. 3a). Semi-quantitative immunoblotting analysis was performed on these markers to investigate the expression of α-syn pSer129 and TH (Fig. 3e–g). Notably, the intracranial injection of PFFs contributed to an increase in α-syn pSer129 in a time-dependent manner, especially at three months post-injection (Fig. 3c). Different PFFs doses altered this time-dependent accumulation in the colon; at a dose of 15μg, α-syn pSer129 began to aggregate at three months, whereas at a dose of 30μg it was already highly expressed after one month (Fig. 3b, c). Similarly, TH expression was reduced at one and three months, although this decrease changed to levels of vehicle control at 6 months. In addition, AT8 antibody staining against phosphorylated tau showed a significant increase at one month (Fig. 3b) and then gradually decreased to baseline levels (Fig. 3c, d). Taken together, these data suggest that intracranial PFFs injection contributes to the aggregation of α-syn pSer129 in the proximal colon in a time-dependent manner, accompanied by a loss of TH-positive neurons (dopaminergic neurons).
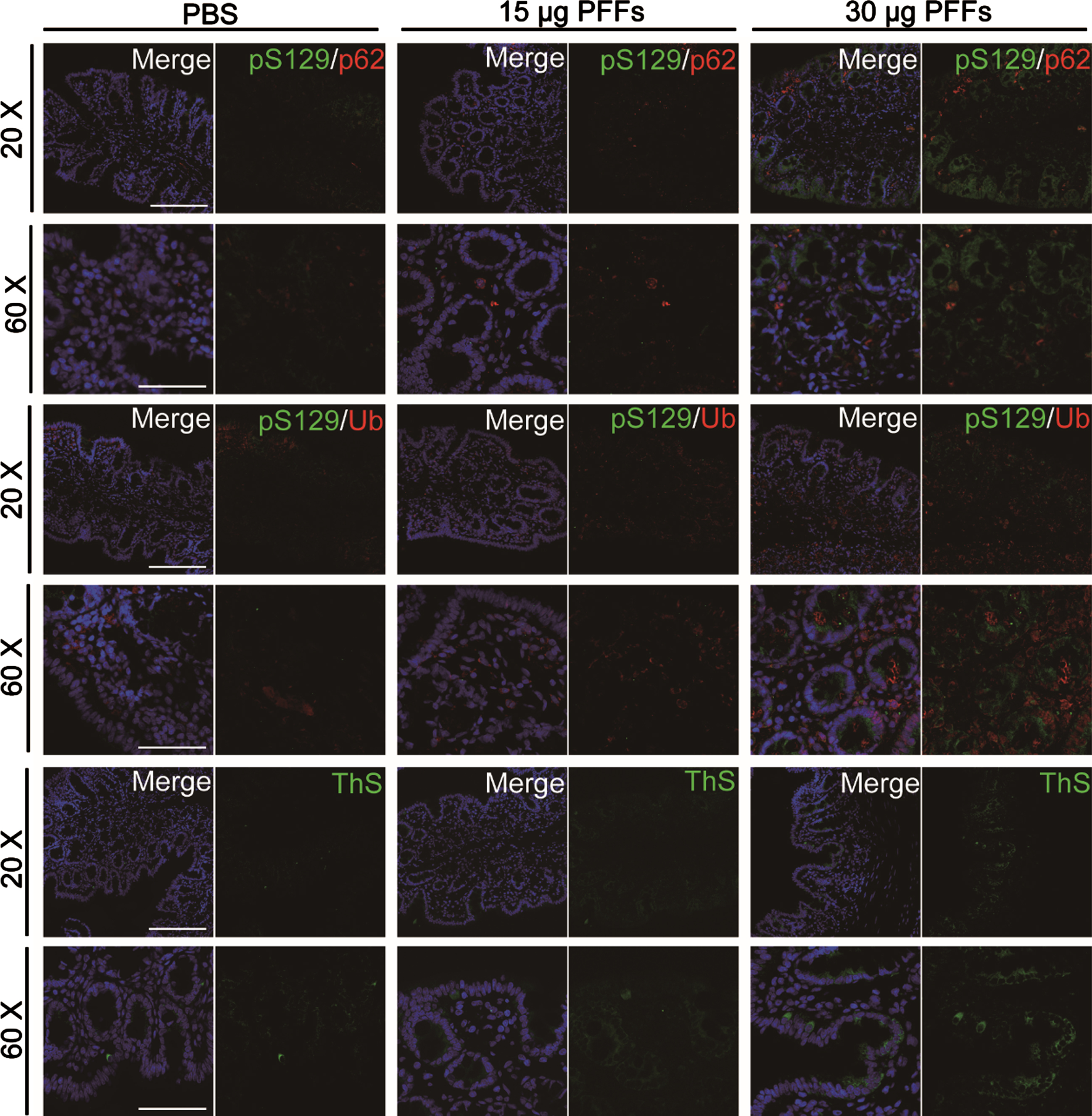
Characterization of phosphorylated α-syn in the proximal colon. One month after injection, phosphorylated α-syn-positive accumulations co-localize with the cargo protein p62 and Ubiquitin, indicating that they share properties with Lewy bodies found in Parkinson’s disease in humans. Thioflavin-S (ThS) positivity indicates mature amyloid structures. The figure shows representative 20× magnification (Scale bar = 50μm) and 60× magnification (Scale bar = 20μm) photomicrographs. n = 4 each group.
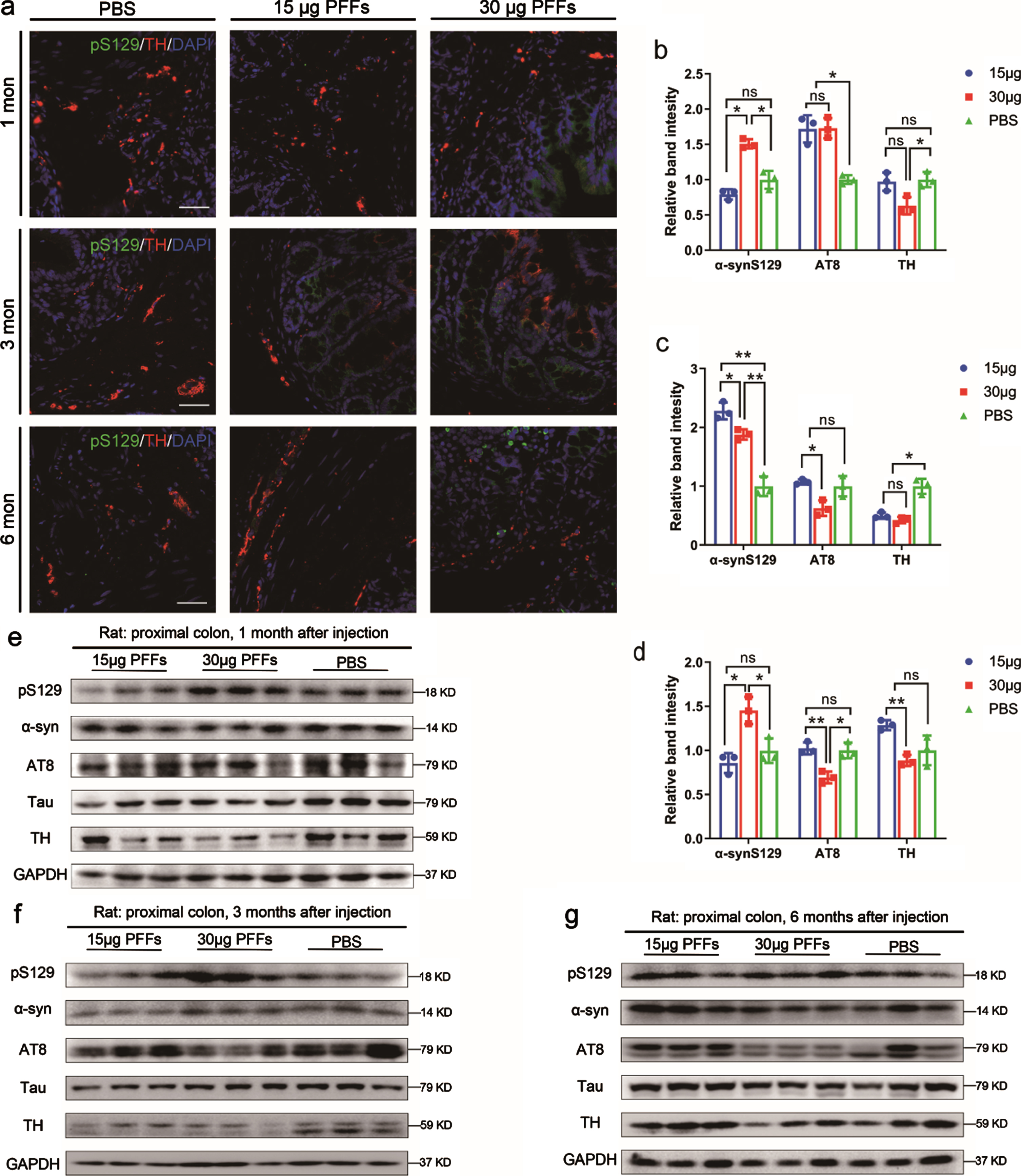
The change of enteric phosphorylated α-syn pathology and dopaminergic loss over time. a) Phosphorylated α-syn-positive accumulations co-localize with TH at one, three, and six months after injection. n = 4 each group. e–g) Representative immunoblotting images showing the expression of α-syn pSer129, α-syn, AT8, tau, and TH in the colon. GAPDH was used as the loading control. All experiments were performed three times independently. Data are shown as the mean±SEM (b–d). n = 3 each group. *p < 0.05, **p < 0.01, p-values were determined by one-way ANOVA.
The vagus nerves are required for the brain-to-gut transmission of pathological phosphorylated α-syn
The visceral vagus nerve is long, thin, and unmyelinated. It can be damaged in α-syn pathologies [27]. To determine whether phosphorylated α-syn can travel long distances along this nerve from the brain to peripheral tissues, including the proximal colon, longitudinal sections of the bilateral vagus nerve were stained to obtain α-syn pSer129. These specimens demonstrated the presence of bilateral labeled nerve fibers at one and six months, as selectively shown in the right vagus nerve in Fig. 4a. To directly test the hypothesis that α-syn pSer129 in the proximal colon mainly propagates via the vagus fibers, we performed complete subdiaphragmatic vagotomies on rats of Group S2 (vagotomy + 30μg α-syn N103/tau N368 PFFs injection into right striatum, n = 5) and S3 (vagotomy + phosphate-buffered saline [PBS] injection into right striatum, n = 4), whereas rats of Group S1 (sham surgery + 30μg α-syn N103/tau N368 PFFs injection into right striatum, n = 5) received sham surgery. All rats received a single intraparenchymal injection of α-syn N103/tau N368 PFFs or PBS into the right striatum after 7 days post-surgery (Fig. 4b). Upon sacrifice, the stomachs of the rats in Group S2 and S3 were severely distended, confirming a complete vagotomy. In the three groups, vagotomy resulted in a significant but not thorough elimination of α-syn pSer129 in the appropriate distribution of Group S2 (Fig. 4c, d), considering other pathways (such as sympathetic nerve and blood) may also be involved in the propagation of pathological α-syn. These observations demonstrate that the vagus nerve is the main medium of the observed rostrocaudal spread of α-syn pSer129.
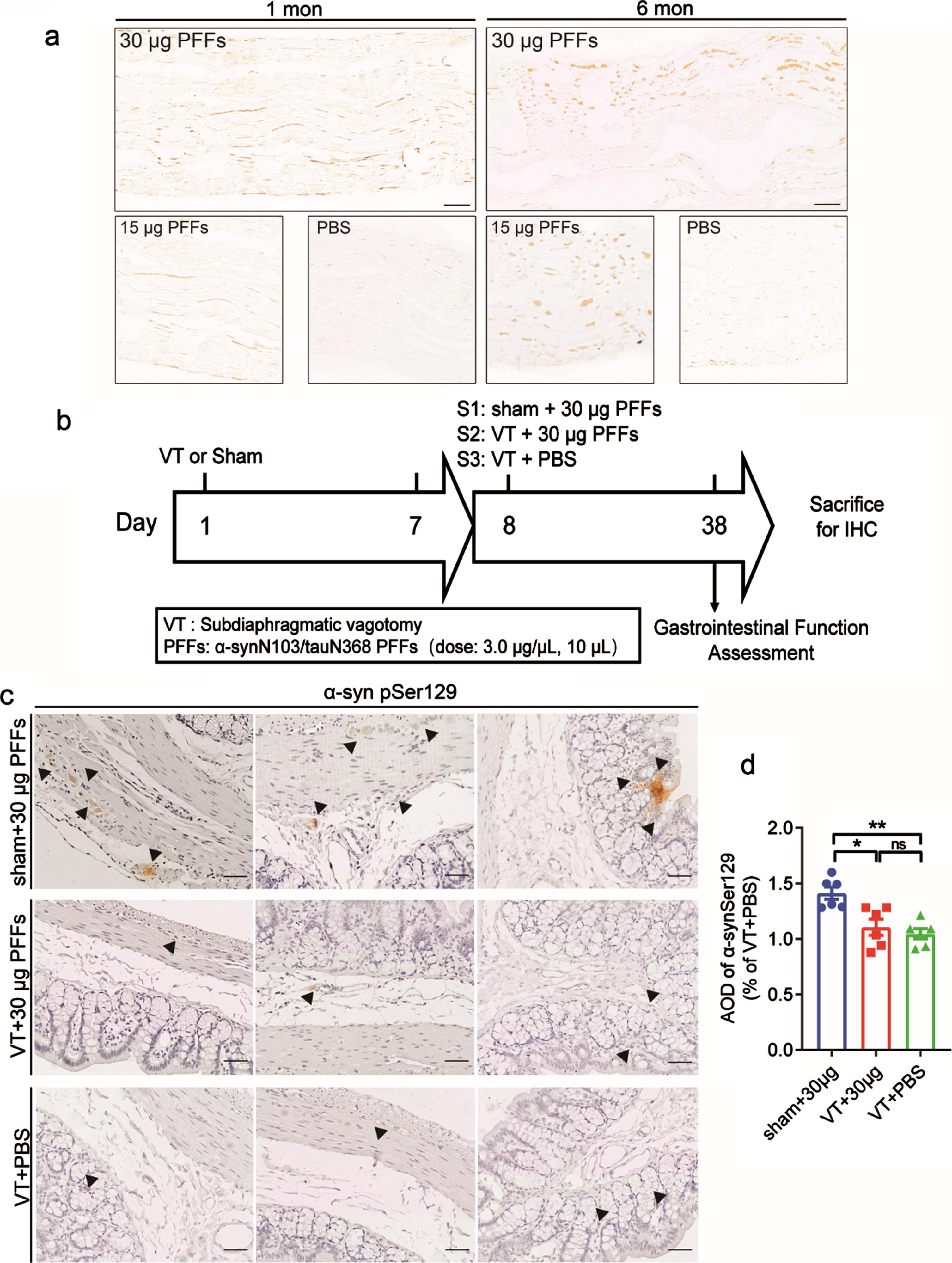
The role of the vagus nerve in the brain-gut transmission of pathological α-syn. a) Fibers of the vagus nerve containing α-syn pSer129. Longitudinal sections of bilateral vagus nerves collected from the rat neck at one and six months after preformed fibrils (PFFs) injection were stained with anti-α-syn pSer129 antibody. n = 4 each group. b) Rats were divided into three groups: sham + 30μg PFFs (n = 5), vagotomy (VT) + 30μg PFFs (n = 5), and VT + phosphate-buffered saline (PBS; n = 4). VT or sham operation was performed before the single intraparenchymal injection of 30μg α-syn N103/tau N368 PFFs or PBS. c) Representative high-magnification photomicrographs of α-syn pSer129 aggregates in the mucosa, submucosa, and muscular layers (arrowheads). Scale bar = 50μm. d) Quantification of the average optical density (AOD) of α-syn pSer129 aggregates. Data are shown as the percentage of the VT + PBS group. **p < 0.01, p-values were determined by one-way ANOVA.
Transmission of pathological α-syn from the brain to gut leads to GI dysfunction
Intracranial injection of PFFs or PBS did not affect the body weight, fecal pellet weight, proportion of water weight, or the intestinal transit of rats after one month (Fig. 5a, d, g, j). The bodyweight of rats injected with a dose of 15μg PFFs began to increase at three months post-injection, whereas rats injected with a dose of 30μg PFFs had weights identical to that of the control group (Fig. 5a–c). No significant difference in fecal pellet weight was found among the three groups (Fig. 5d–f). In the 30-μg group, the proportion of water weight was decreased at three months (30μg vs. Con = 56.13±3.27% vs. 67.50±5.37%, p = 0.034; Fig. 5h) and six months (30μg vs. Con = 53.25±3.81% vs. 61.13±3.72%, p = 0.003; Fig. 5i) post-injection. Up to six months, the intestinal transit of pathological α-syn in rats who received a dose of 30μg PFFs was decreased (30μg vs. Con = 59.70±6.23% vs. 72.15±6.34%, p = 0.018; Fig. 5l). We also compared the colon lengths of rats among the groups but found no difference (Supplementary Figure 1). Although we did not perform detailed motor testing, considering that there were no significant differences in bodyweight and fecal pellet output among groups, these gastrointestinal findings were not the result of severe debilitation.
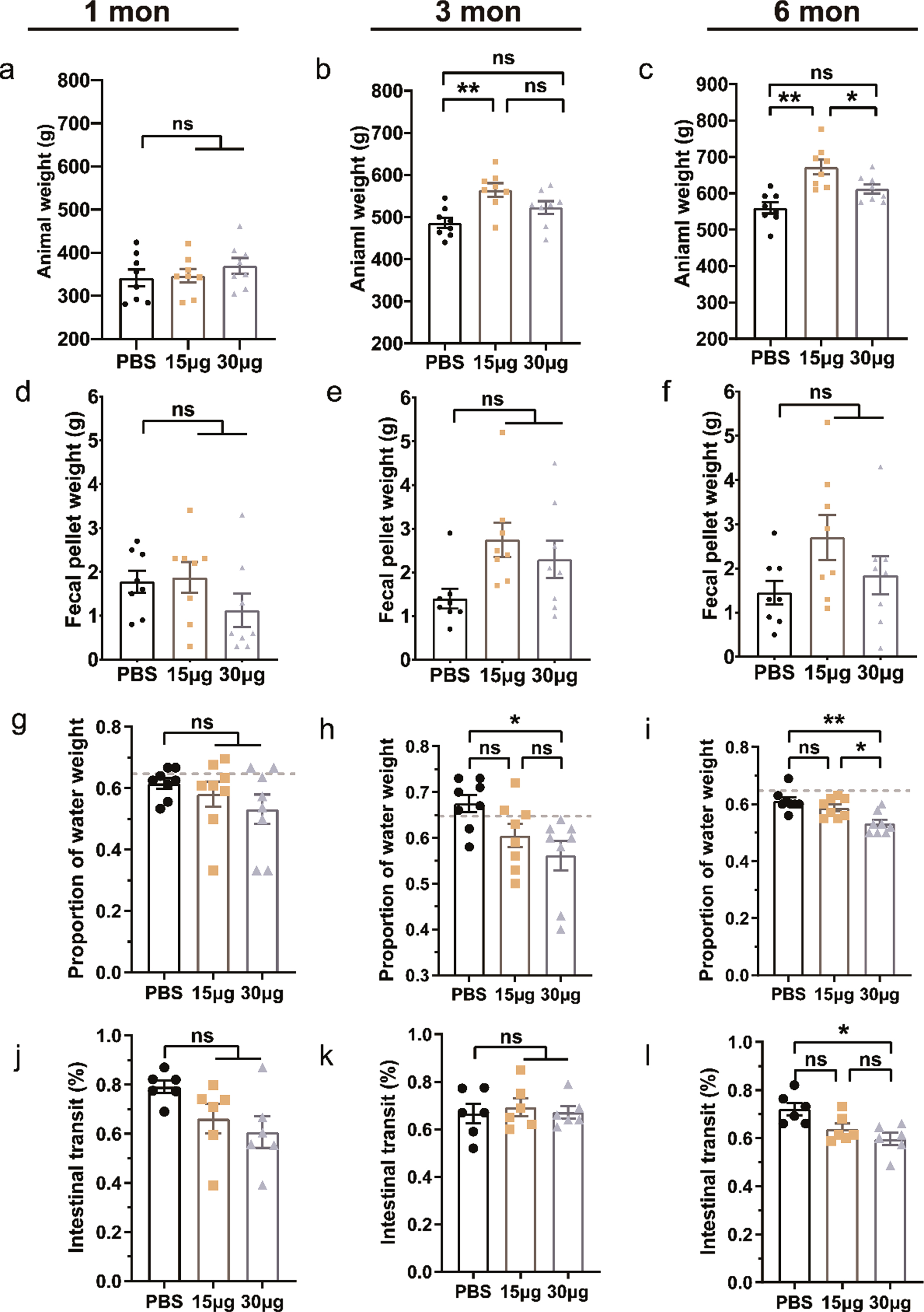
Effects of α-syn pathology on GI function over time. a–c) Animal weights at one, three, and six months after intraparenchymal injection of 15 or 30μg preformed fibrils (PFFs) or phosphate-buffered saline (PBS) were obtained. GI function is characterized by fecal pellet weight (d–f), the proportion of water weight (g–i), and intestinal transit (j–l) in different groups. The grey dashed lines in (g–i) are baselines without interventions. Data are shown as the mean±SEM. n = 8 each group. *p < 0.05, **p < 0.01, p-values were determined by one-way ANOVA.
Transmission of pathological α-syn from the brain to the gut accompanied by inflammation
Eosinophils, frequently found in the mucosa and submucosa of the normal colon, were significantly increased in number in the proximal colon after PFFs injection, and this effect was less prominent at six months (Fig. 6a, b). EGCs, the glial cells in the colonic myenteric ganglia, were GFAP-immunopositive. Iba1 acted as a marker of macrophages in peripheral tissues. Both GFAP and Iba1 showed lower expression at one month than under control conditions (Fig. 7a). At a dose of 15μg PFFs, the expression of GFAP and Iba1 sharply increased at three and six months, while there was no high expression with a PFFs dose of 30μg (Fig. 7a, e, g).
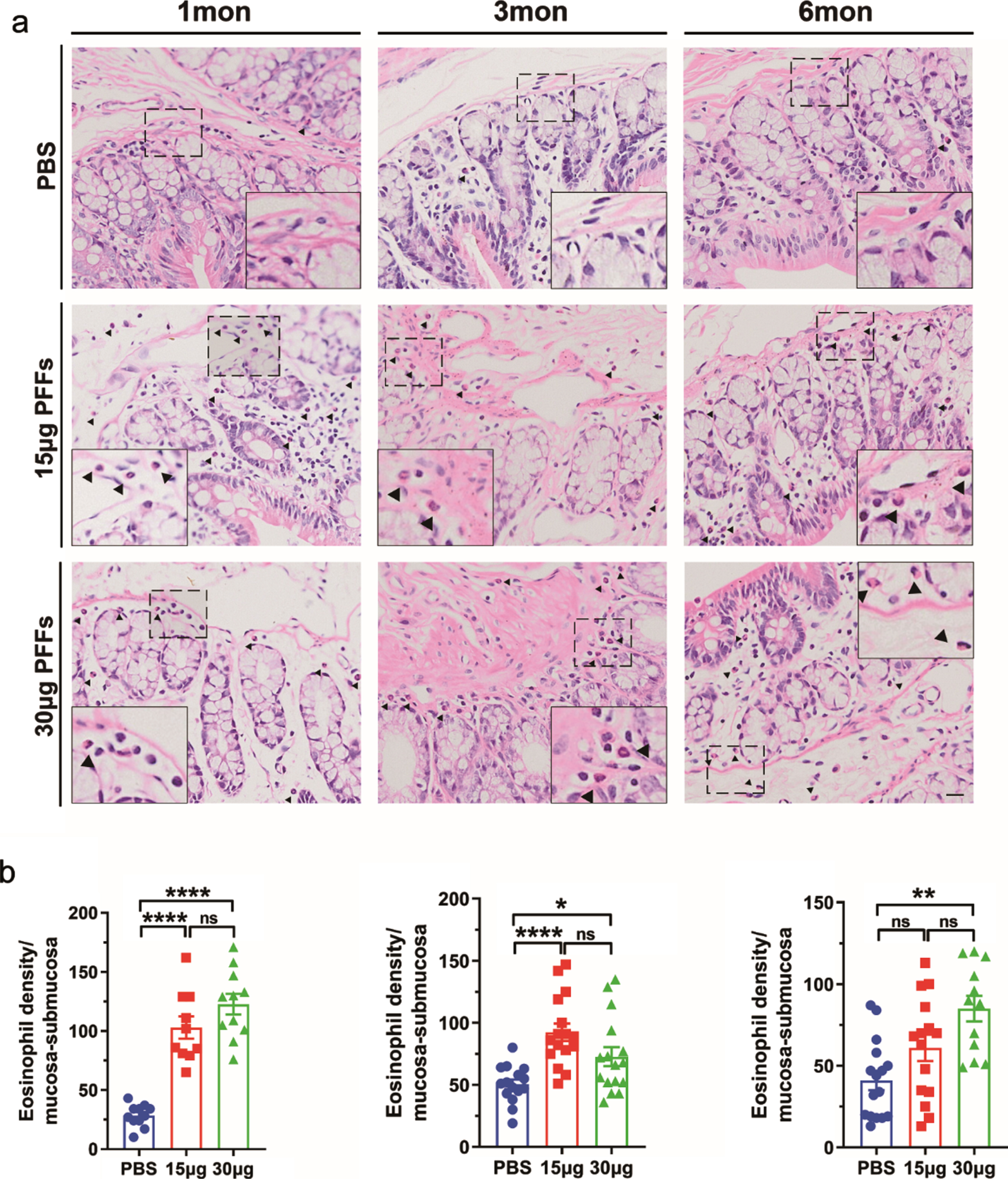
Effects of α-syn pathology on the enteric inflammatory response. a) Representative images of hematoxylin/eosin-stained colonic sections were obtained at one, three, and six months. Dashed lines and arrowheads indicate eosinophils. b) Bars indicate the mean eosinophil cell density per square millimeter of mucosa-submucosa areas (cells/mm2)±SEM value. n = 4 each group. Scale bar = 100μm. *p < 0.05, **p < 0.01, p-values were determined by one-way ANOVA.
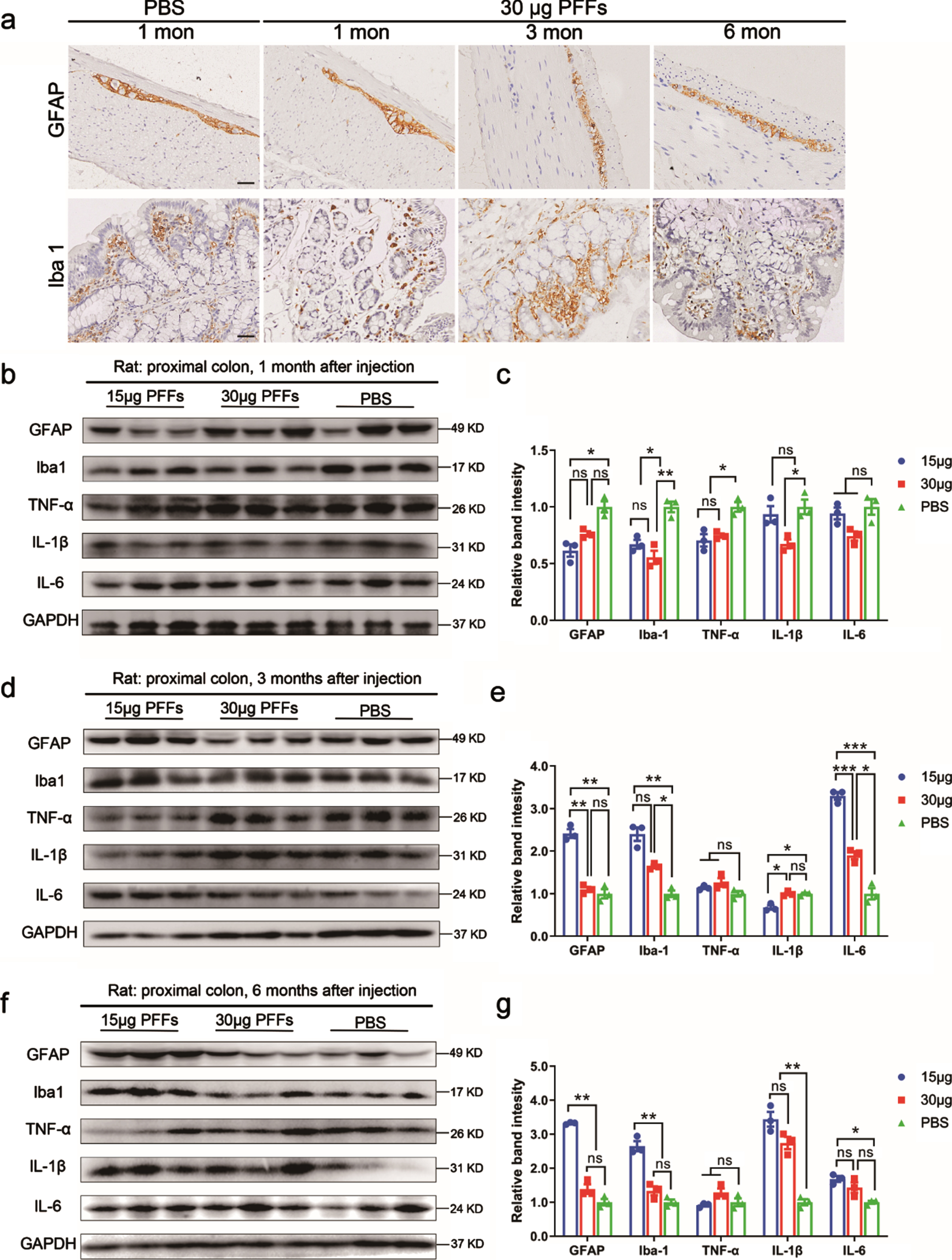
Effects of α-syn pathology on the enteric nervous system and cytokine expression. a) Representative images of glial fibrillary acidic protein (GFAP)- and ionized calcium-binding adaptor molecule 1 (Iba1)-positive immunostaining of the colon at one, three, and six months. n = 4 each group. Scale bar = 30μm. b, d, f) Representative immunoblotting images showing the expression of GFAP, Iba1, tumor necrosis factor (TNF)-α, interleukin (IL)-1β, and IL-6 in the colon. GAPDH was used as the loading control. All experiments were performed independently three times. c, e, g) Data in the right panel are shown as the mean±SEM. n = 3 each group. *p < 0.05, **p < 0.01, p-values were determined by one-way ANOVA.
Based on the occurrence of activated microglia and infiltrating T lymphocytes, as well as the accumulation of pro-inflammatory cytokines, including TNF-α, IL-1β, IL-6, and interferon γ, the analysis of postmortem brains of PD patients supports the involvement of inflammation in PD pathogenesis [28, 29]. In the present study, we evaluated the expression levels of the pro-inflammatory cytokines TNF-α, IL-1β, and IL-6 (Fig. 7b, d, f). The expression of TNF-α showed a decrease at 1month after PFFs injection (Fig. 7c), and there was no difference in the expression of TNF-α among the groups at three, and six months (Fig. 7e, g), even when there was a high expression of IL-1β and IL-6. IL-1β levels in colonic tissues were significantly higher at six months than those of the control group. IL-6 expression was increased starting from three months and returned to the baseline levels at 6 months (Fig. 7e, g). Taken together, the pathological α-syn pSer129 aggregation in the proximal colon is associated with inflammation, and mainly manifested by increased eosinophils and macrophages, as well as high levels of pro-inflammatory cytokines, especially IL-6 and IL-1β.
To test the influence of phosphorylated α-syn aggregates on neurotransmitters in the colon, the levels of monoamine neurotransmitters (NE, DOPAC, DA, HVA, and 5-HT) in the proximal colon at six months post-injection were tested (Supplementary Figure 2). Significant increases in DA and DOPAC levels were observed for both doses of PFFs, whereas 5-HT showed only high levels with PFFs dose of 30μg. NE and HVA levels in the proximal colon remained unaffected.
DISCUSSION
At present, there are two hypotheses about the pathological origin of PD: the Body-first or the CNS-first [30]. Braak staging is mainly applicable to PD patients with early-onset and slow progression (the disease starts from the peripheral nervous system, such as GI tract), and is partially applicable to patients with dementia with Lewy bodies who exhibit late-onset and more rapid disease progression [31]. Though Braak staging is still controversial, from the brain to the gut or from the gut to the brain, which may both be applicable to different types of PD.
In our previous study, we observed that α-syn N103/tau N368 PFFs were much more neurotoxic and triggered phosphorylation of the endogenous α-syn and tau. Furthermore, α-syn N103/tau N368 PFFs injected into the colon progressed to the brainstem along the vagus nerve [5]. In this study, we first identified that the top-down propagation of phosphorylated α-syn aggregates under the controlled microinjection of α-syn N103/tau N368 PFFs into the right dorsolateral striatum of wild-type rats, and revealed the changes of enteric inflammation and GI dysfunction at different time points based on the degrees of α-syn aggregation in the proximal colon whole-mounts (summarized in Table 1).
Summary of indicators expression in the colon at one, three, and six months
The aggregated syn propagates in a brain-vagus nerve-gut pathway after PFFs injection
α-syn and tau proteins have prion-like properties, and they can self-duplicate, misfold, and promote fibrilization of each other mutually [32, 33]. Although previous studies have suggested that α-syn PFFs have cross-seeding tau aggregation abilities, the accumulation of tau is relatively low compared with α-syn inclusions [34]. In the current study, the expression of phosphorylated tau (AT8) in different brain regions at one month was significantly lower than that of α-syn Ser129. Moreover, AT8 expression increase preceded that of α-syn Ser129 in the proximal colon, and then returned to the baseline levels after three months. This suggests that in this model, tau plays an essential role in promoting fibrilization of α-syn. Notably, AT8 expression increased at a dose of 15μg PFFs while the expression of GFAP, Iba1, and TNF-α decreased without the accumulation of α-syn Ser129. A cross-sectional study in breast cancer survivors revealed negative correlations among peripheral tau levels and IL-10, IL-12, IL-7, IL-4, and IL-1β levels [35]. This suggests that AT8 might suppress the expression of some cytokines or alter its neurotoxicity to the ENS. The relationship between tau and cytokines has not yet been defined and needs further research.
The DMV and NTS are considered to be the main sensory and motor centers of the vagus nerve, and they are involved in GI function [36]. A newly discovered nigrovagal pathway demonstrated evidence of direct projections among the DMV, NTS, and SN [37]. Projections between the LC and SN are also known, and the LC receives the projections from the NTS [38]. In this study, we injected the PFFs to the right dorsolateral striatum and found that the occurrence of α-syn Ser129 was more evident ipsilaterally, because the nigrostriatal pathway is more strongly connected on the ipsilateral side of the nerve fibers. Coronal brain sections showed substantial α-syn Ser129-positive aggregates in the SN, DMV, and NTS, indicating anterograde propagation. Notably, the anterior olfactory nucleus and motor cortex also developed inclusions at one month, suggesting retrograde rostral spreading. Different PFFs exhibited different speeds along the vagus nerve, and α-syn N103/tau N368 PFFs can reach the brainstem from the colon after 9 days [5]. Thus, intrastriatal PFFs injection in rats might be a PD model which mimics rapid propagation. In recent studies, we mainly focused on the participation of the vagus nerve in rostrocaudal propagation, while neglecting the other pathways, such as sympathetic nerve and blood or the spinal cord. These pathways have all been implicated in α-syn spreading [16, 40]. The α-syn pSer129 staining of the vagus nerve revealed the presence of labeled nerve fibers, and further subdiaphragmatic vagotomies studies also support the important role of the vagus nerve in the propagation of α-syn pSer129 aggregates.
The degree of α-syn aggregation mainly influences the emergence and severity of GI symptoms
Although PFFs showed rapid propagation in the models, the abnormalities of fecal traits did not emerge until three months after injection of 30μg PFFs, indicating that the number of α-syn pSer129 aggregates in the GI tract at one month was insufficient to cause symptoms. Neighboring neurons and glial cells can mediate the degradation of α-syn inclusions via the ubiquitin-proteasome system and autophagy-lysosome pathway to regulate α-syn homeostasis [41, 42]. Pathological α-syn overload and impairments of the degradation systems can disrupt the balance [43, 44]. After injection of 30μg PFFs, the immunofluorescence co-staining of α-syn pSer129 and Ubiquitin or p62 in the proximal colon was more prominent than in the control, demonstrating that these aggregates are Lewy body-like and may be closely related to the pathways mentioned above. Since the overload of α-syn aggregates can further damage these degradation systems, in our findings, the rats exposed to high doses of PFFs showed significant α-syn pSer129 aggregation and the occurrence of GI dysfunction at six months, while the aggregates of α-syn pSer129 returned to the baseline levels after injection with lower doses of PFFs. Besides, GFAP elevation was not evident at any time point in animals receiving a higher dose of PFFs, and IL-6 elevations were only mild at 3 months, which also suggested that the protective degradation systems in the ENS were overwhelmed, with the consequence of less reactive gliosis and IL-6 production and resultant ongoing damage within the ENS. It is worth noting that at the PFFs dose of 30μg, the proportion of water weight in the fecal pellet decreased at three months, and at this dose, ENS bore a heavy α-syn pathology.
Recovery of the ENS is a limited compensatory response
EGCs play an important role in PD-related GI dysfunction and shift toward a pro-inflammatory phenotype in response to stimulation, which triggers the release of pro-inflammatory cytokines and mediates cytokine signaling in the gut to recruit immune cells [45, 46]. Macrophages recruited during these inflammatory processes play an essential phagocytic role in the removal of α-syn inclusions [47, 48]. GFAP is a molecular marker for EGCs and is often overexpressed in patients with PD [49]. Therefore, in our study, GFAP was expressed at a high level in the proximal colon with lower PFFs dose at three, and six months. This high expression of GFAP indicates reactive gliosis in response to phosphorylated α-syn aggregates. At a lower dose of PFFs, similar proliferation was found in macrophages at three and six months. The glial response under pathological conditions, which is called “reactive gliosis,” is special in PD because it is not found in patients with atypical parkinsonism [49]. This reactive proliferation of EGCs and macrophages may help clear the overload of phosphorylated α-syn aggregates.
It has been reported that IL-1β and TNF-α stimulation can make α-syn expression decrease in the enteric primary culture of rats [50]. Further studies on mice with acute dextran sulfate sodium-induced colitis also confirmed that acute enteric inflammation can elicit a downregulation of pathological α-syn expression [50]. According to the levels of IL-1β and α-syn pSer129 examined, we discovered a negative correlation. These data also support that enteric inflammation in response to phosphorylated α-syn aggregation can downregulate the expression of α-syn pSer129. Besides, IL-6 promotes the survival of enteric neurons and the morphologic differentiation of EGCs [51], as IL-1β, and TNF-α also have been shown to increase GFAP expression in cultured EGCs [52]. Observations of PD patients revealed a positive correlation between pro-inflammatory cytokines and glial markers [8]. In the present study, IL-6 was the first of the three examined pro-inflammatory cytokines to show higher levels at three months, together with elevated GFAP and Iba1. At low doses of PFFs, we speculate that IL-1β and IL-6 together promoted the expression of GFAP and Iba1 at six months.
Some studies have suggested a time-related disruption of efferent vagal terminals in the GI tracts, which has already been investigated in older transgenic PD mice models [53]. In the present study, TH-immunopositivity in the proximal colon was decreased over time, indicating PFFs’ neurotoxicity. Nevertheless, we found that the TH expression levels in the gut were reversed at six months, indicating that at the PFFs dose of 30μg, the restoration of the function or number of DA neurons is a transit compensatory response, given that phosphorylated α-syn still aggregated and GI symptoms emerged.
Thus, the proliferation of EGCs and macrophages and high expressions of IL-1β and IL-6 are compensatory responses to the stimulation of enteric α-syn aggregates after PFFs injection. A study of patients diagnosed with PD also suggested that elevated pro-inflammatory cytokines and glial markers in the colon were negatively correlated with disease duration and not disease severity [8], indicating that the upregulated pro-inflammatory cytokines in the early stage of PD are a compensatory response to defense. However, this compensatory function is limited, as heavy enteric pathological α-syn aggregates can worsen GI symptoms.
Neurotransmitters also can alter the function of the ENS [46]. The vagus nerve has dual anti-inflammatory properties, and it can inhibit the release of TNF-α by releasing acetylcholine (Ach) at terminals [54], which may have prevented the upregulation of TNF-α in our study. However, we lacked an examination of Ach levels in the colon. It is known that dopamine inhibits GI motility, and both Ach and 5-HT increase GI motility, but the influence of 5-HT on GI motility is now controversial [55, 56]. In our study, we tested the production of monoamine neurotransmitters at six months in the proximal colon. The changes in NA and HVA levels were not significant, whereas DA, DOPAC and 5-HT levels in the colon increased. A previous study has demonstrated that EGCs can promote the generation of enteric neurons, including dopaminergic and serotonergic neurons, in response to injury [57]. Although the presence of TH does not unequivocally reflect DA involvement [58], we found that DA levels were upregulated with the restoration of TH expression. 5-HT levels only were elevated at high PFFs dose, together with higher expression of DA and DOPAC. The balance between DA and 5-HT levels and GI symptom development is difficult to elucidate, because the amounts of cell receptors and expression of other neurotransmitters should be considered, and it needs to be assayed at longer time points to verify.
Enteric inflammation associated with α-syn aggregation also contributes to GI dysfunction
Cytokines such as TNF-α, IL-6, IFN-γ, and IL-1β are upregulated in PD [8]. It has been reported that the stimulation of lipopolysaccharide can increase the expression of IL-1β from EGCs, and TNF-α and IL-6 from the ENS [59]. Interestingly, in our study, the pro-inflammatory cytokines in the gut did not always increase, and some even decreased at one month. TNF-α expression had not changed since injection after three months, which is inconsistent with the previous findings of high levels of TNF-α and IL-1β in PD patients [8]. Only the level of IL-1β was still upregulated at six months, but there was no high expression of GFAP and Iba1. Cytokines have dichotomous effects on the ENS [46], and the anti-inflammation effect was detected at one, and three months after high PFFs dose injection. Peripheral administration of lipopolysaccharides in the transgenic A53T mouse model aggravated the neurotoxicity of α-syn oligomers [60]. IL-1β expression increased at two different doses of PFFs, and the IL-1β effect in the gut differed according to the aggregations of α-syn and GI symptoms. It is reasonable to believe that the high expression of IL-1β at six months with prominent α-syn pSer129 aggregation and GI symptoms is considered to be an effect of pro-inflammatory factors. Moreover, the eosinophil density in the mucosa and submucosa was also significantly high. All these features suggest that gut inflammation plays an essential role in the observed GI symptoms, and the underlying mechanism needs further research.
Conclusion
Our results suggest that intrastriatal injection of α-syn N103/tau N368 PFFs triggers α-syn pathology in the ENS, supporting the brain-to-gut hypothesis of synucleinopathy. We developed a prodromal PD model with quick anterograde propagation of α-syn pSer129 and GI dysfunction. Furthermore, the restored α-syn pSer129, AT8, and TH expression levels at low doses of PFFs highlighted the restoration of the ENS. At present, the expression of EGCs and their role in the clearance of pathological α-syn aggregates and the pathogenesis of PD are still undefined. Understanding the mechanism of gut recovery in synucleinopathy will promote the development of therapeutic options for patients with PD. Our work expands the knowledge of the mechanism of anterograde propagation in PD synucleinopathy and the potential role of the ENS in prodromal synucleinopathies.
Footnotes
ACKNOWLEDGMENTS
We would like to thank Editage and Ziqin Cao for the English language help. We thank the Medical Subcenter of HUST Analytical & Testing Center in data acquisition.
This study was supported by grant from the National key R&D Program of China (2017YFC1310300), National Natural Science Foundation of China (81873734, and 81974200).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
