Abstract
Background:
Quality of life (QoL) of persons with Parkinson’s disease (PD) is diminished by (non-)motor symptoms, that require personalized care. Parkinson Nurses (PN) may be pivotal promoting tailored care offerings. This systematic review and meta-analysis investigates PD care models and aims at furnishing current concepts of PN to offer personalized care.
Objective:
The purpose of this study is to identify the various roles and functions that PN may hold for personalized PD care.
Methods:
We performed a systematic literature review, utilizing: PubMed, Web of Science, The Cochrane Library, and PsycINFO. The review qualitatively evaluated articles, which described personalized care models involving PNs and was guided by the personalized care management model. A meta-analysis compared patient-reported QoL (quantified using the 39-item Parkinson’s Disease Questionnaire) between personalized care interventions involving PN versus standard care with.
Results:
Twenty-seven publications were identified, including six randomized, controlled trials ascertaining with health related QoL (n = 1830 PwPs). The qualitative evaluation revealed that PN contribute to all aspects of personalized care. The meta-analysis showed no improved QoL in personalized care models compared to standard care, thought a great heterogeneity among study design and interventions was outlined (Standardized Mean Difference = –0.8935; 95% Confidence Interval, –2.1177 to 0.3307; z = –1.43, p = 0.1526).
Conclusion:
PN fulfil important functions in personalized PD care. For the future, a clear role definition will be necessary to adjust training for PN across healthcare systems and care settings but especially to realize their full potential for PD care.
Keywords
INTRODUCTION
Parkinson’s disease (PD), is an incurable neurodegenerative disorder presenting with a heterogenous phenotype including motor- and non-motor symptoms [1]. With increasing disease duration, the majority of patients experience an increased level of symptom burden [2], and a growing demand for medical and social care services. From a healthcare resources demand perspective, PD already ranks among the top ten most resource-intensive brain disorders in Europe with 1.2 million affected people to date [3]. An increasing incidence among older people and the population development forecasts in mind, it is foreseeable, that persons with Parkinson’s disease (PwPs) will have doubled by 2030 [4].
Traditional physician-centered care models for PwPs may reach their limits when degenerative processes disperse and multiple psychosocial needs require individualized treatment strategies. In addition, the increasing number of PwPs is in conflict with the scarcity of medical resources. Ideal and sustainable care services are nowadays considered to be those that are comprehensive but, above all, that are coined to the patients and their environment [5]. The term “personalized care” embodies such care services and has already found its place in science and practice alike [6]. In the broadest sense, can, “personalized care” be defined as “tailored to the needs and preferences of each individual” [7]. Regarding PD care, van Halteren et al. defined the five core elements of personalized care management for PwPs, which are summarized in Table 1.
Five dimensions of the personalized care management model for people with Parkinson’s disease
cf. (van Halteren et al. [7]), p. S14.
One might argue that an implementation of these elements in PwPs, requires expertise, networking skills and good oversight of the care process. While no doubt exists that PD care improves with multi-professional collaboration, the question arises which profession may drive the implementation of personalized care elements. There is no uniform definition of the sufficient or ideal composition of a multidisciplinary care team, and a strong heterogeneity exists in practice [8]. Nevertheless, when it comes to achieving care personalization, the role of specialized nurses has been recognized as a pivotal facilitator [9, 10].
In research and practice, such nurses are called Parkinson’s Nurses (PN). In a broad sense, PN are specially trained professionals who deal with individual issues related to the care and support of PwPs [11]. However, a precise description of the tasks PN are supposed to master is ill-defined, and wide range of assignments and roles has been described [12–14]. This heterogeneity is problematic because role clarity bolsters effective interprofessional collaboration, adequate training, and the future perpetuation of job profiles. The aim of this paper is therefore to provide an overview of the role of PN in care personalization for PwPs and explore its impact on quality of life (QoL) in PwPs, using a systematic review approach.
MATERIALS AND METHODS
Review question
This present systematic review and meta-analysis aim to examine the following research questions: What are existing models of personalized care for PwPs in which PN are involved? What is the role of involved PN within existing models of personalized care for PwPs? Can complex, personalized care models improve the QoL of PwPs compared to standard care?
Search
The following databases were utilized: PubMed, The Cochrane Library (Cochrane Database of Systematic Reviews, Cochrane Central Register of Controlled Trials (CENTRAL), Web of Science (science and social science citation index), and PsycINFO. The study protocol was registered with PROSPERO on February 11, 2021, and confirmed for registration on March 15, 2021 (CRD42021236755). The search followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) reporting guidelines [15]. The initial search of the databases was performed in April 2021 and a follow-up search was conducted in December 2021 to update the review in a timely manner. Literature search was based on several MeSH terms adapted to the databases to be employed, including: “parkinson’s disease”, “parkinsonian disorders”, “nurse”, “management”, “rehabilitation”, “quality of life”, with individual search terms adapted according to each database (cf. Supplementary Table 1 for PubMed’s exemplary search strategy).
The flow chart of the study selection process is displayed in Fig. 1.
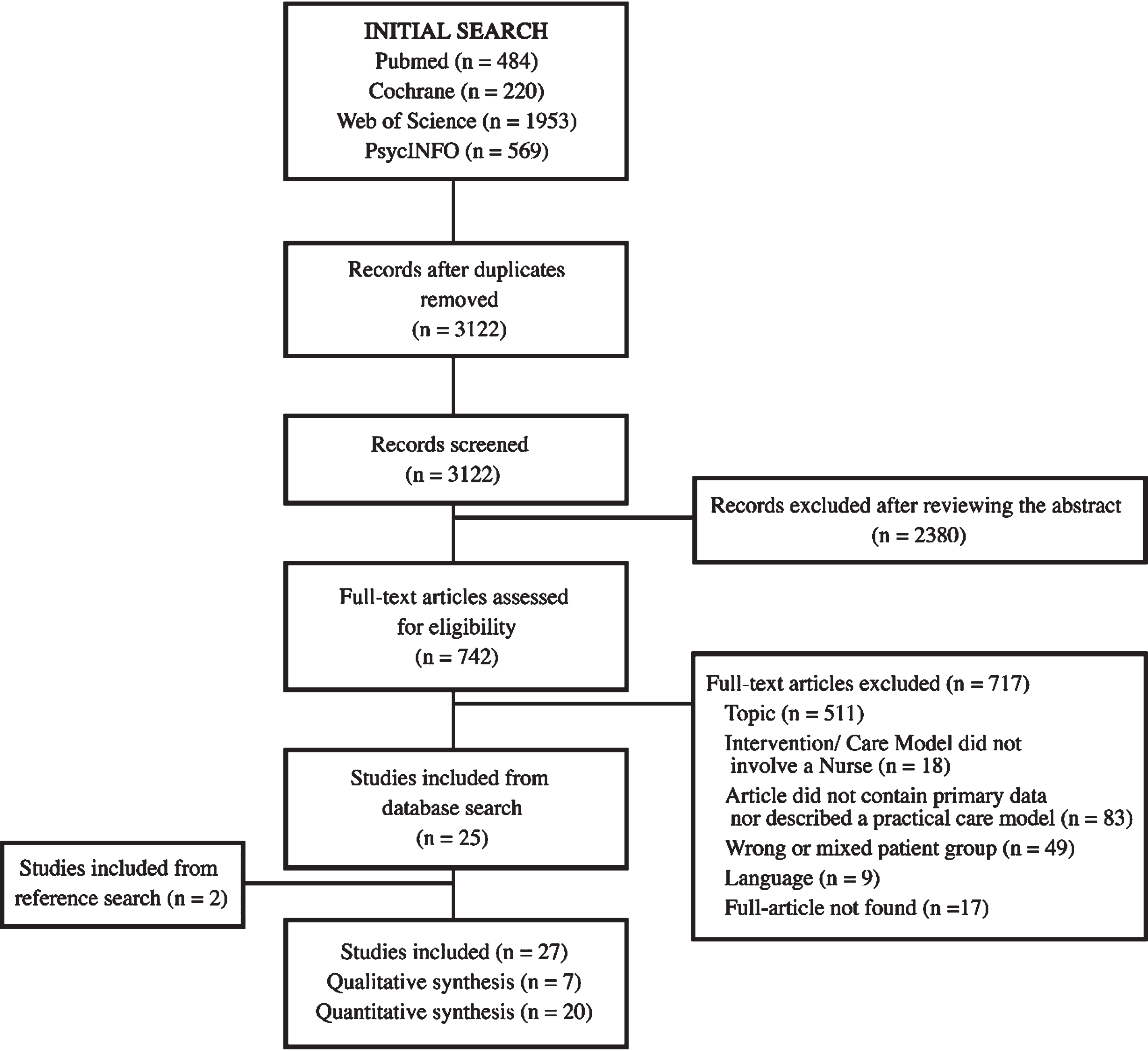
Study flow diagram (ref. [15]).
Studies published in English and German language or with English and German language translation available were included. There were no restrictions for inclusion based on date of publication and/or geographical location.
Inclusion/exclusion
For the systematic review, we adopted the following inclusion criteria: 1) Studies qualitatively describing or quantitively evaluating a care model which is tailored towards individual needs and preferences; 2) Studies that addressed the role of PN in the context of personalized care; 3) Setting either inpatient, outpatient or community-based; 4) Sample population comprised adults (≥18 years of age) diagnosed with PD (following United Kingdom Parkinson’s disease Society Brain Bank criteria [16] and the Movement Disorder Society’s clinical diagnostic criteria [17]); any gender included. Exclusion criteria were: 1) Failure meeting the language requirements; 2) Studies not providing primary data (i.e., comments, case series, case reports); 3) full text was unavailable; 4) not relevant to the subject of the review. The following inclusion criteria applied additionally to the meta-analysis: 1) Eligible studies had to be controlled randomized or quasi-randomized; 2) The intervention had to be tailored towards patients’ individual needs and preferences; 3) Use of the PD validated QoL scale (8-/ or 39-item Parkinson’s Disease Questionnaire –PDQ-8/-39).
Outcomes
For the systematic review, we collected the following information: identification of care settings, team composition, descriptions of the care model, intervention and the PN roles/tasks. The dimensions of personalized care delivered by the PN were described based on the five dimensions of the model of personalized care management for PwPs (cf. Table 1) [7, 18]. The identification of dimensions is qualitative in nature [18], which is why there was no quantitative data analysis. For our analysis, models and interventions that documented at least one dimension of personalized care were considered for the review. We added a sixth dimension for tasks that do not match one of the five dimensions but still aims to adapt care to the needs and preferences of PwPs, termed “other”.
The main outcome of the meta-analysis was patient-reported health-related (HR)-QoL. The PDQ-39 and the shortened version (PDQ-8) are widely utilized and validated tools ascertaining QoL in PwPs [19]. Eligible studies had to report PDQ-39/-8 results at 6–24 weeks after randomization. This period was chosen because the power of randomized controlled trials is related to the observation period [20] and the defined period is a common observation period in PD health care studies [8, 21–23]. Since none of the included studies reported the PDQ-8 as an outcome, we will only address the PDQ-39 as an outcome in the following.
Data extraction
Every step of the data extraction process was conducted by two authors independently (M.vM., J.S.). Discrepancies during each step were resolved through discussion with a third researcher (F.T.) and mutual agreement was reached. First, the two authors (M.vM., J.S.) screened independently titles and/or abstracts of studies retrieved using the search strategy. Based on the first screening, full texts of studies meeting inclusion criteria were retrieved and independently assessed for eligibility.
After removal of duplicates, the authors (M.vM.; J.S.) extracted data from every eligible article to a standardized prespecified template. Missing data was requested from study authors (this was once the case; the requested data could be provided by the authors). The template captured study information (authors’ names, year of publication, location), study population (sample size), the intervention (setting, involved professionals, duration (if applicable), and content) and the roles of PN according to the five dimensions of personalized care. For studies eligible for the meta-analysis, we collected additional information on the study population (baseline characteristics, in- and exclusion criteria) and the intervention (outcomes assessed, comparator). Since the review involved a systematic review and meta-analysis of previously published studies, approval by the institutional ethics committee and written informed consent from patients were not required.
Risk of bias assessment
The articles included in the meta-analysis were independently examined for internal validity utilizing the Cochrane risk-of-bias tool [24]. To ensure interrater reliability, the tool was applied independently by the two researchers (M.vM.; J.S.). Subsequently, the results were compared and discrepancies were discussed with a third researcher (F.T.) until a consensus was reached.
Data synthesis and publication bias assessment
For the systematic review, a narrative synthesis of the included studies was conducted. For the meta-analysis, we pooled the results from (quasi-)randomized control trials reporting the outcome measure (PD quality of life; PDQ-39) with a random-effects model of the standardized mean differences [25]. We calculated 95% CI and two-sided p-values for the outcome measure [25] and included studies were weighted according to the inverse-variance. Heterogeneity between studies were assessed using the χ2 test and the corresponding I2 statistic [25]. Substantial heterogeneity was defined as I2 statistic greater than 50% [25]. Additionally, we assessed for publication bias with the Egger’s regression intercept as per the small number of studies [26]. The meta-analysis was conducted in R 4.1.2 using the “meta” package version 5.1-1 [27].
RESULTS
Study characteristics
The combined searches retrieved 3,122 records (cf. Fig. 1 for details on the search strategy). After removal of duplicates and screening for titles/abstracts, 742 records remained for full-text review. In the next step, 511 of the 742 articles were excluded because the topic of the article did not address our primary research interest. The remaining 231 articles were then considered for a full-text review in which176 articles had to be excluded on the basis of poor-fit to the inclusion criteria and to the research question. Thus, 25 articles could be included into the presented systematic review. The excluded articles were related to care models disregarding PN (n = 18), neither reported primary data nor described an implemented care model (n = 83), reported findings for non-PD patients or involved a mixed study population in which the results for PD-patients were not disclosed separately (n 49), unavailable (n = 17) or published in another language as defined in the inclusion criteria (n = 17). Two additional articles were included after hand searching the reference lists of those 25 selected articles.
Ultimately, we could include 27 records in the qualitative review. Details of the included studies are displayed in Table 3. The study characterizes of 7 randomized, controlled trials (RCT) included in the quantitative meta-analysis and 14 other experimental studies included in the systematic review. The remaining 6 articles were nonexperimental and included description of practical care models or qualitative research.
Detailed study characteristics
The included articles were published between 1998 and 2021 (cf. Table 2), with most articles (n = 18) published after 2010. Of the 27 articles, 21 articles stemmed from quantitative research. Studies were conducted more frequently in Europe (n = 11).
Characteristics of the articles (total n = 27)
Systematic review of personalized care models
We identified 25 individual reported models from 27 articles (cf. Table 3) that met our definition of personalized care. In total, 28 tasks performed by PN and covering all five domains of personalized care were identified. All actions and domains are summarized in Fig. 2.
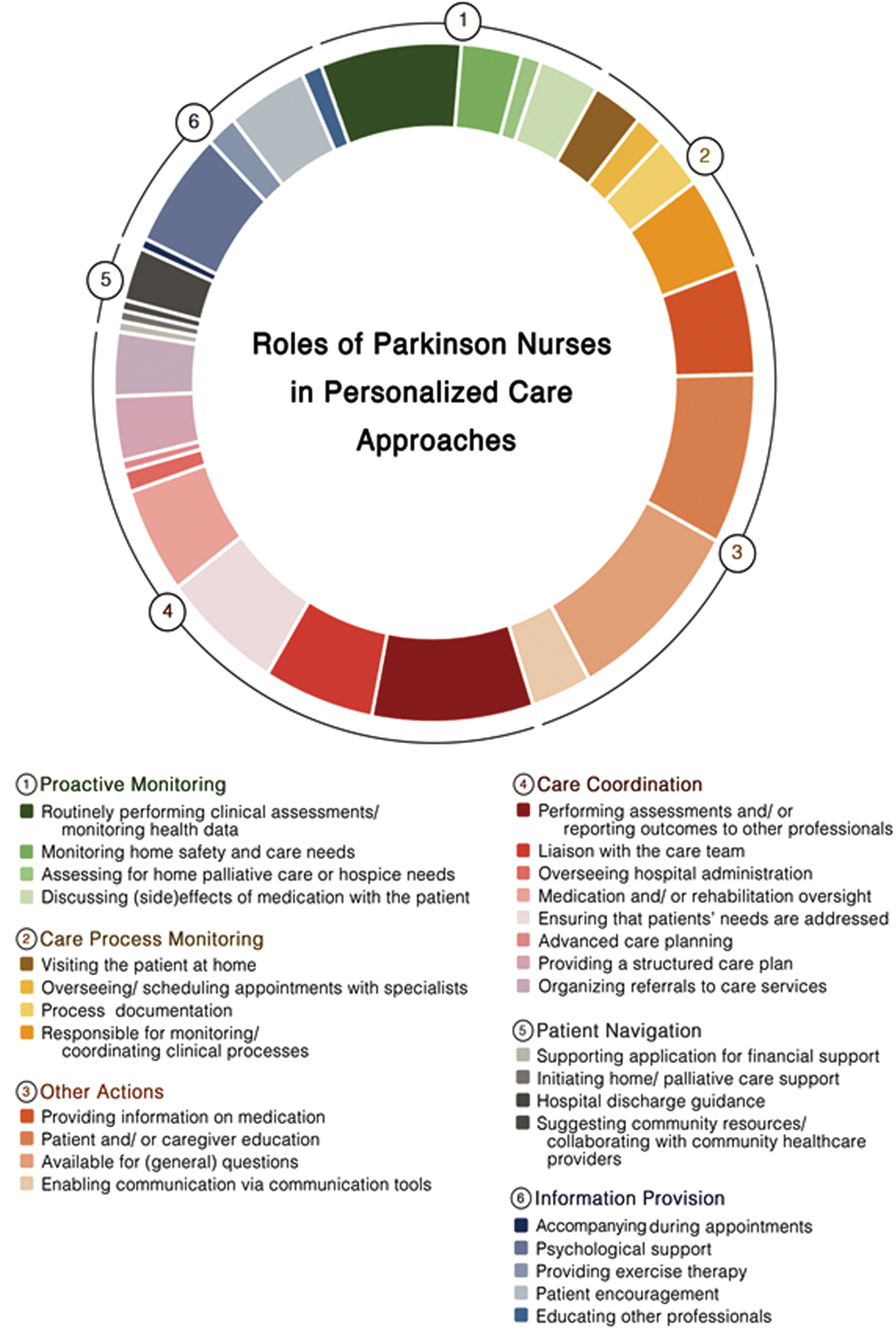
Tasks performed by Parkinson Nurses which relate to the model of personalized care management for people with Parkinson’s disease.
In most care models, PN performed tasks covering at least two domains of personalized care. The domain most frequently covered by PN was the provision of personalized information, with PN being especially available for general questions [14, 41–53] and educated patients and/ or care partners [14, 48–53]. In 10 studies, PN provided personalized information to patient and care partners by providing information on medication [14, 53]. Besides, PN enabled communication via tools such as virtual chats [29, 44]. In several studies, PN coordinated care processes by reporting outcomes of clinical assessments to other professionals [28–30, 47–49], liaising with the care team [14, 53], implementing structured care plan [31, 48], overseeing patients’ hospital admissions [48, 53] or their medication and rehabilitation schemes [31, 42–44]. PN additionally ensured that all care providers were aware of the patients’ needs [34, 42–48]. Moreover PN, facilitated advanced care planning [42] or organized referrals to specific care services [14, 48]. In terms of monitoring the care process, PN performed home visits [29–32, 36–38], oversaw and scheduled appointments with specialists for patients [31, 40], documented the care process [38–40, 48] and coordinated these processed within clinical evaluations [31, 53]. However, PN also performed proactive monitoring actions by routinely performing clinical assessments [14, 46–49], monitoring patients home safety and care needs [39, 49], evaluation the demand for home palliative care or admissions to hospice care [42, 43] and by discussing (side-)effects of medication with patients [31, 46]. Actions attributed to patient navigation were only limitedly observable across the studies. In one trial PN’s supported patients in applying for financial support [31, 32], whereas another study mentioned the PN being responsible for initiating home/ palliative care support [42]. In another study the PN navigated the patient through the hospital discharge process [43]. Five studies reported that PN were responsible for suggesting community resources to patients and for collaborating with community healthcare providers [14, 49]. Finally, six themes were identified across the studies which did not fit into one of the proposed dimensions of personalized care but also facilitate the personalization of care processes. Several studies reported that PN provided individual psychological support [34, 51–53] and encouraged patients throughout the care process [14, 49]. In three studies, PN implemented personalized exercise programs for patients [34, 43], while in two studies PNs educated other healthcare professionals about patient needs [14, 50]. Finally, in one care model, PN accompanied patients during medical appointments [31, 32].
Impact of personalized care in PD-related quality of life (meta-analysis)
Description of studies
Study details are displayed in Tables 3 and 4.
Study characteristics of randomized controlled trials
IG, intervention group/; CG, control group; SD, standard deviation; PDQ 39, 39-item Parkinson’s Disease Questionnaire; EuroQol, EuroQol Questionnaire; UPDRS, Unified Parkinson’s Disease Rating Scale; MoCA, Montreal Cognitive Assessment; BDI, Beck’s-Depressions-Inventory; NMS-Score, Non-motor Symptoms; PANDA, Parkinson Neuropsychometric Dementia Assessment; PDNS, Parkinson’s Disease Nurse Specialists; L- dopa, Levodopa; PD, Parkinson’s disease; MADRS, Montgomery–Åsberg Depression Rating Scale; SCOPA-PS, Scales for Outcomes in Parkinson’s disease- Psychosocial Functioning; CSI, Caregiver Strain Index; SF, Short Form 36 item health survey; NHPT, Nine-hole-peg-Test; HADS, Hospital Anxiety and Depression Scale; SGA, Subjective Global Assessment; SAS, Supervisory Attentional System; SDS, Self-Rating Depression Scale; PDSS, Parkinson Disease Sleep Scale.
In this subgroup of RCTs, included studies were conducted between 2002 and 2020 [28, 52]. There were 1830 participants overall, who were all community-dwelling adults with PD. Sample sizes of the individual studies ranged from 10 to 1254 PwP.
The settings for the interventions were: fully inpatient in one study [34] and delivered in an outpatient setting in the remaining ones [28, 52]. While most studies [28, 52] involved a multidisciplinary team for intervention delivery, one study solely involved PNs as care professionals [34]. The duration of the intervention program varied from three to twenty-four months, with two studies implementing weekly patient contact [34, 52], three studies implementing monthly patient contact or when desired [28, 33] and one study disclosing no further information in this regard [31, 32].
Risk of bias assessment
The risk of bias assessment for included studies is presented in Supplementary Figure 1. The trials covered a wide range of methodological quality, but no study achieved low risk in all the seven criteria assessed. The Egger’s regression intercept test was not significant (p = 0.204), suggesting asymmetry and therefore publication bias in these results.
Effects of interventions on patient’s quality of life
QoL scores were available for 1830 participants, as measured by the PDQ-39. Pooling data from all the available RCTs (n = 6) resulted in substantial heterogeneity (I2 = 96.6%, χ2 = 146.31, Fig. 3) and suggested no significant effects of personalized care delivery on QoL in PwP if compared with standard care (Standardized Mean Difference [SMD] = –0.8935; 95% Confidence Interval [CI], –2.1177 to 0.3307; z = –1.43, p = 0.1526).
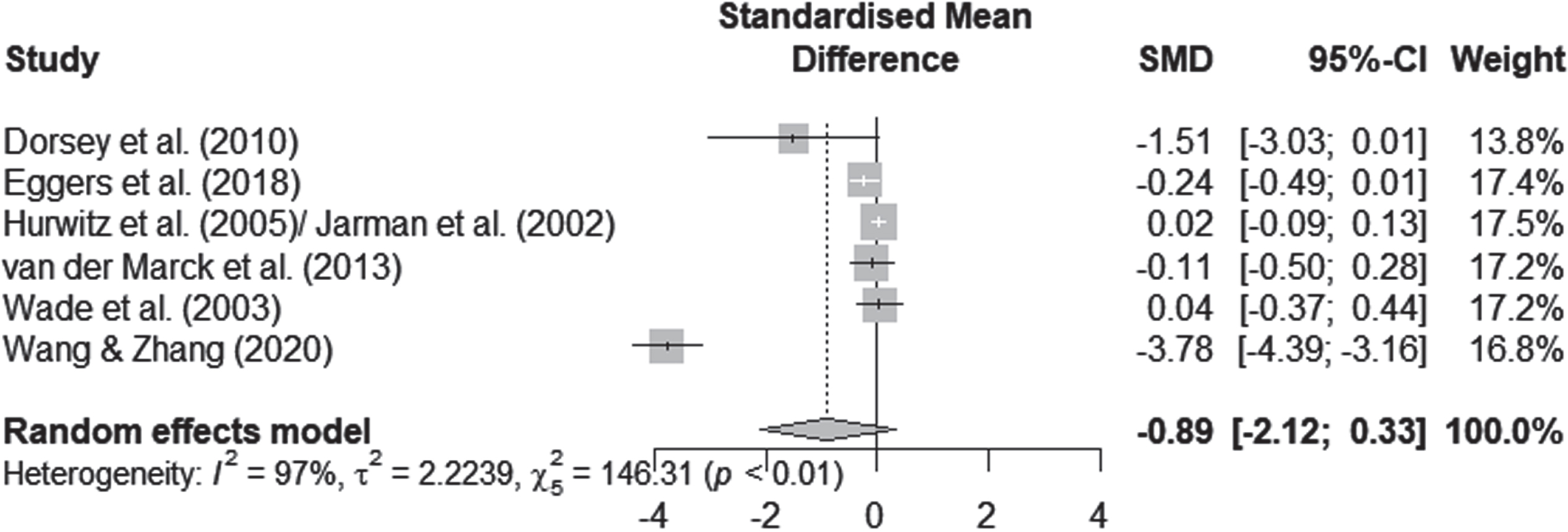
Forest plot of meta-analysis.
Various factors such as the nature of the intervention, the duration of follow-up and the overall quality of the studies may explain the heterogeneity of our results. One study reported the outcome of interest after a considerably shorter period of time (assessment after three months of intervention) [34]. The intensity and frequency of the intervention in the inpatient study differed considerably from the outpatient studies. In addition, it was the only study that showed a significant effect (cf. Fig. 3). We therefore performed an exploratory post-hoc subgroup analysis. In the subgroup analysis, we pooled data from the remaining five care models that reported the outcome of interest over a period of more than six months [28, 52]. According to the I2 statistic heterogeneity was 47% for this subgroup, whereas concerning the outcome of interest no significant effects for QoL improvements after personalized care versus standard care delivery were traceable (SMD, –0.076; 95% CI = [–0.235 to 0.084]; p = 0.353).
DISCUSSION
In this systematic review, we identified models of personalized PD care in which PN play primordial role. These models covered the entire spectrum of healthcare settings (inpatient, outpatient, and community-based) and originated from a broad gamut of healthcare systems. A trend can be seen in the meta-analysis which indicates that personalized care models may impact PwPs’ physical, mental, and social well-being (cf. Fig. 3). Nevertheless, the meta-analysis of pooled data from randomized, controlled trials identified no significant favorable effects of personalized care on QoL compared with usual care. Possibly, this reflects the significant differences in populations included, in the care settings scrutinized, and the timings of outcome assessments. In what follows, we will try to dissect why results of the RCTs do not necessarily imply that the models are ineffective or lack important practical implications but why more high-quality data is imperative.
Heterogeneity instead of a clear line: What do the studies tell us?
Personalized PD care describes a comparatively new approach and has therefore not yet been finally and uniformly established throughout literature. In the included studies, it remained at times unclear whether concepts of personalized care were actively pursued, not only as care models differed significantly, but also due to the applied terminology. These discrepancies are paralleled beyond the scope of this review, insofar as that some aspects of personalized PD care can be met in patient-centered or integrated care models with somehow differing nomenclature [54, 55]. In agreement with this blurred definition, we could identify tasks corresponding to but not matching exactly one of van Halteren’s five elements of personalized PD care which might nonetheless be attributable to their idea of care according to the individuals’ needs and preferences [7]. On the one hand, divergency of services may reflect strengths of care models incorporating specific, e.g., regional elements. It is hard to imagine anyone assuming personalized care approaches being a universal concept in the sense of a one-fits-all approach. Otherwise, great heterogeneity in implementation or nomenclature hinders scientific assessment and the development of binding and specific descriptions of professions like PN. In the future theoretical work, a balance between precision and reflection of theories and concepts in the international comparisons is desirable.
Against the background of distinct concepts of personalized care, peculiarities of healthcare systems are only comprehensible. The differences in the adopted approaches revolved around the implementation of personalized care and the role of PN being responsible for at least single elements, but at times also for two or more. This divergence is also found in everyday life: for example, Prell et al. observed that in practice there is a wide range of training, tasks and areas of work exists [56]. These results are likely to be related to the organization of healthcare systems with regard to professional collaboration. For example, while the German system is not geared towards cooperation [57], other healthcare systems are deemed collaborative [58–60]. Besides, the traditional values and training of staff might also play a role, as in countries where PN do not yet exist and no funding mechanisms are implemented [61], versus those in which these specialists are already an integral part of care approaches [8, 63]. Cross-country analyses may help differentiate the roles of PN in personalized care delivery.
Regardless of the structure of healthcare systems, multispecialty care models with multiple healthcare professional working in a team are intuitive for PwPs’ personalized care. The relevance and effectiveness of a multi-professional collaboration is gaining momentum, as reflected in clinical recommendations, practical observations and scientific studies [11–13, 64–68]. So, what should the role of PN be in the care process of PwPs?
In practice, PN represent an important member of multispecialty care teams, as they fulfill several functions. They may enable the integrated flow of care between patients and care teams, and between professionals in the care team [56]. Moreover, they may facilitate the implementation of PD-specific care in previously unspecialized healthcare facilities. Possible other roles of PN have been highlighted in recent clinical recommendations [13, 66]. Thus, Radder et al. consider them a core team member and explicitly mention them as putative team coordinator, a role to which they attach central importance for its success [66]. MacMahon broaden the range of roles of PN to include assessments of patients’ concerns and challenges, the provision of educational and emotional support, and the facilitation of health or social services [64]. Finally, Lennaerts et al. bring up the facilitation of self-management in PwPs, the establishment of multidisciplinary collaboration among care professionals and the execution of specific nursing-technical interventions as tasks for PN [13]. All these recommendations nourish in particular the view that the profession of PN is not yet clearly defined.
Strength and weaknesses of the method
Thus far, our results have rendered evidence on existing models of personalized care for PwPs involving PN. We could elaborate their distinct roles within existing models of care and highlight, in which roles within healthcare teams the PN was favored. At the same time, some limitations must be acknowledged. Firstly, even though, full texts were requested from the authors, it was not possible to access all publications that were found during the systematic search process. The number of unavailable studies, as well as the language restrictions might have biased the results of this review.
Contrary to the expectations, no evidence was traceable that personalized care models improve the QoL of PwPs compared to standardized care. It would be tempting to restrict this to the relatively few studies eligible for our meta-analysis. Yet, some other aspects may also explain this apparent contradiction. Another source of variance is the considerable difference in sample sizes, modes and content of delivery for multidisciplinary programs for PwPs but also the intervention length. The occurrence of reporting bias may have led to a deceptively positive perception of the interventions’ efficacy and should therefore be addressed. Information was lacking in some studies, particularly about to the in-depth characterization of the concept of PN, so that future research should address this gap with detailed descriptions of each professional’s role within care teams.
Nevertheless, some important strengths of this work are to be highlighted. It is well-known that PD impacts severely the lives of PwPs and their care partners [69, 70] and that the PDQ-39 is a validated and widely-used tool assessing QoL [71]. Along with a rather large sample size, the use of the PDQ-39, a validated and widely-used tool assessing QoL [71], as primary outcome enabled greater statistical power and generalizability. The pre-specified analysis in this systematic review and the few exclusion criteria but especially the meticulous methods, underline our results as the current state of knowledge. Yet, a question to be addressed is whether a holistic view of QoL is possible [5, 72]. Further research should also investigate whether this primary outcome is adequate when examining the impact of personalized care approaches for the manifold limitations PD entails over its course.
Considerations for future research in the field of Parkinson’s care
Notwithstanding the heterogeneity and diversity of the study results to date, some recommendations for the future are possible. First, scientists should consider the complexity of (PD-)care research when designing studies and critically appraise which elements should be addressed. Focusing on single theoretical concepts may be helpful in planning studies, but limit the evaluation of results. Care models may be multidisciplinary, patient-centered, integrated, and personalized.
The planning of problem-orientated research is of central importance. From a methodological perspective, we follow previously made recommendations [8] advocating for a head-to-head design, comparing the standard of care with a complex, personalized care intervention or and for implementing cluster-randomized designs, as blinding participants may be hardly achievable. While randomized controlled study designs are considered the gold standard, more pragmatic research approaches also appear as relevant research approaches [73, 74]. Finally, explicit descriptions of the settings, the objectives, the mechanisms of cooperation, the responsibilities and the preliminary theoretical considerations might promote a universal definition of the roles of PN.
Conclusion
In summary, we provide evidence on existing models of personalized care for PwPs involving PN. The results of the present studies demonstrate a diverse set of tasks a PN can perform and confirm the picture of a manifold understanding of roles, but also show that scientific concepts such as personalized care can help to sort out and make tangible these tasks. For example, van Halteren’s concept of personalized PD care served as a structural basis for the qualitative analysis in this review. The review revealed that, these highly specialized professionals already assume various roles within the care team, some of which go beyond defined elements of personalized PD care, such as providing psychological support. Additionally, we could highlight the roles within healthcare teams that might become more prevalent in the future because they provide extraordinary benefits, such as including patients’ preferences in medical decisions. Overall, it seems important to further harmonize concepts of the PN across healthcare systems and to emphasize their role as team members of multidisciplinary care approaches and, perhaps, in leading the implementation of personalized care. Thus, further scientific evidence of the PN role in care models is desirable to generate a larger data pool supported by clearly stated theoretical concepts and well-designed evaluative studies.
Footnotes
ACKNOWLEDGMENTS
We would like to thank Beate Weber-Schicker and Elena Keuchel for their assistance in the collection of data. Special appreciation to Annika Dörrhöfer for her support in the preparation of charts and tables.
This review and meta-analysis were conducted as part of the research project “iCARE-PD”. This is an EU Joint Programme –Neurodegenerative Disease Research (JPND) project. The project is supported through the following funding organisations under the aegis of JPND –![]() (Canada –Canadian Institutes of Health Research; Czech Republik –Ministry of Education, Youth and Sport of the Czech Republic; France –Agence National de la Recherche; Germany –Bundesministerium für Bildung und Forschung; Spain –National Institute of Health Carlos III; United Kingdom –Medical Research Council).
(Canada –Canadian Institutes of Health Research; Czech Republik –Ministry of Education, Youth and Sport of the Czech Republic; France –Agence National de la Recherche; Germany –Bundesministerium für Bildung und Forschung; Spain –National Institute of Health Carlos III; United Kingdom –Medical Research Council).
M.vM. and J.S. are funded by the aforementioned research project “iCARE-PD”.
F.R. is supported by the Helmholtz Association under the joint research school “Munich School for Data Science –MUDS"
CONFLICT OF INTEREST
M.v.M. declares no COI; J.S. declares no COI; F.T. declares no COI; F.R. declares no COI; T.C. declared no COI; K.C. declared no COI; D.P. received honoraria as a speaker on symposia sponsored by Boston Scientific and Desitin Pharma. The institution of D.P., not D.P. personally received funding by Boston Scientific, the German Research Foundation, the German Ministry of Education and Research and the Deutsche Parkinson Vereinigung.
T.M. has received personal compensation for serving as a Consultant for CHDI, Sunovion, Valeo Pharma, Roche, Biogen and nQ, received personal compensation serving on a Speakers Bureau for Abbvie, Valeo Pharma, and has received research support from the Canadian Institutes of Health Research, EU Joint Programme— Neurodegenerative Disease Research, the Ontario Research Fund, Michael J Fox Foundation, Parkinson Canada, uOBMRI/Parkinson Research Consortium, Parkinson Canada, Brain Canada, Ontario Brain Institute, and PSI Foundation.
O.R. has received personal compensation for serving as a Consultant for AbbVie, Adamas, Acorda, Addex, Aguettant, Alkahest, AlzProtect, Apopharma, Astrazeneca, Axovant, Bial, Biogen, Britannia, Buckwang, Cerevel, Clevexel, Contera, Handltherapeutic, Irlab, Lilly, Lundbeck, Lupin, Merck, MundiPharma, Neuratris, Neuroderm, Novartis, ONO Pharma, Orion Pharma, Osmotica, Oxford Biomedica, Parexel, Pfizer, Prexton Roche,Therapeutics, Quintiles, Sanofi, Servier, Sunovion, Théranexus, Takeda, Teva, UCB, Watermark Research, XenoPort, XO and Zambon and has received research support from Agence Nationale de la Recherche (ANR), CHU de Toulouse, France-Parkinson, INSERM-DHOS Recherche Clinique Translationnelle, MJFox Foundation, Programme Hospitalier de Recherche Clinique, European Commission (FP7, H2020).
