Abstract
Background:
Mobile devices and their capabilities (e.g., device sensors and human-device interactions) are increasingly being considered for use in clinical assessments and disease monitoring due to their ability to provide objective, repeatable, and more accurate measures of neurocognitive performance. These mobile-based assessments also provide a foundation for the design of intervention recommendations.
Objective:
The purpose of this work was to assess the benefits of various physical intervention programs as they relate to Parkinson’s disease (PD), its symptoms, and stages (Hoehn and Yahr (H&Y) Stages 1–5).
Methods:
Ninety-five participants (n = 70 PD; n = 25 control) completed 14 tablet-based neurocognitive functional tests (e.g., motor, memory, speech, executive, and multi-function) and standardized health questionnaires. 208 symptom-specific digital features were normalized to assess the benefits of various physical intervention programs (e.g., aerobic activity, non-contact boxing, functional strength, and yoga) for individuals with PD. While previous studies have shown that physical interventions improve both motor and non-motor PD symptoms, this paper expands on previous works by mapping symptom-specific neurocognitive functionalities to specific physical intervention programs across stages of PD.
Results:
For early-stage PD (e.g., H&Y Stages 1 & 2), functional strength activities provided the largest overall significant delta improvement (Δ= 0.1883; p = 0.0265), whereas aerobic activity provided the largest overall significant delta improvement (Δ= 0.2700; p = 0.0364) for advanced stages of PD (e.g., H&Y Stages 3–5).
Conclusions:
As mobile-based digital health technology allows for the collection of larger, labeled, objective datasets, new ways to analyze and interpret patterns in this data emerge which can ultimately lead to new personalized medicine programs.
Keywords
INTRODUCTION
As the prevalence of digital health technology and the subsequent collection and analysis of large amounts of complex health data increases, clinicians are provided with novel tools and methods to increase the quality of care of individuals with neurodegenerative diseases such as Parkinson’s disease (PD) [1–4]. The increased availability of this data and the use of machine learning methods presents major opportunities in the healthcare space for discovery, improving patient safety, and translating information into actionable knowledge for the recommendation of personalized intervention programs [5–7].
Currently, many intervention programs are implemented as a one-size fits-all concept as current systems are not sufficient to develop personalized rehabilitation programs [8]. In the presence of neurodegenerative diseases like PD, both pharmacological and nonpharmacological therapies are advocated for. Nonpharmacological therapies include physical interventions (e.g., functional strength activities, boxing, and yoga) as well as speech, occupational, psychological, and music therapies [9, 10]. These nonpharmacological therapies are continually advocated for as they are shown to provide slower rates of motor and non-motor symptom progression in people with neurodegenerative conditions [11]. However, with the increased amount of collected digital information, individualized, patient-oriented recommendations should be a primary focus [7].
The objective of this work was to assess symptom-specific effects of multiple physical intervention programs as they relate to individuals with PD, across its stages (Hoehn and Yahr Stages 1–5) [12]. This paper focuses on individuals with PD as they demonstrate impaired functionality in both motor and cognitive areas [13]. The analysis in this work was completed through normalization of collected features from digital health technology specifically designed for individuals with PD [14] as they relate to various intervention programs.
Related work
Digital health technology
Digital health technology and its capabilities allow for the collection of far more information and objective metrics in the areas of neurocognition, than can be achieved using traditional pen-and-paper assessments [15, 16]. Previous studies have found that the configured implementation of device-based sensors in addition to user-device interactions (e.g., accelerometry based gait assessments or speech recognition systems for healthcare) enhance the utility and quality of collected data [2]. Mobile devices allow for the implementation of digital versions of standardized assessments (e.g., Montreal Cognitive Assessment (MoCA) [17]; Mini-Mental State Examination (MMSE) [18]); and questionnaires (e.g., PDQ-39 [19]) for the collection of objective digital features in addition to relevant patient reported outcomes (PROs) [20]. The collection of PROs in tandem with digital versions of standardized assessments allows for an increased level of reliability and accuracy by reducing individual variability and/or bias [21–23]. Further, as the volume of relevant health data increases, novel ways to interact with and extract meaning from the data emerge [1, 4]. Specifically, digital health technology has demonstrated the ability to translate large health data sets into actionable knowledge for intervention recommending systems [5, 25]. This process revolves around the utilization of clinically relevant and objective features, as the performance of algorithms is heavily dependent upon the quality of the input features [26].
Physical interventions
Following diagnosis, both pharmacological and physical therapeutic interventions are advocated for individuals with PD. Previous studies suggests that physical activity during the critical window of early- and mid-stage of the disease is vital to the management of disease progression and PD symptoms [27, 28]. Physical activities encompass both routine activities of daily living (ADLs) (e.g., household activity, walking) and dedicated exercise (e.g., aerobics, strength training) [29]. Further, supervised and structured exercise is noted to be effective at improving functional performance outcomes (e.g., balance and functional ambulation) in individuals with PD [30, 31].
Personalizing physical intervention programs
Presently, the recommendation of physical interventions occurs as a one-size- fits-all concept where current evidence is not sufficient to develop personalized rehabilitation programs [8]. To gain further insights for the creation of personalized rehabilitation programs, it is imperative to administer precise and objective assessments (e.g., by leveraging digital health technology) for the provision of symptom-specific information in relation to physical rehabilitative efforts and further understanding of various intervention approaches [32]. Although previous work has depicted the use of mobile based sensors for the personalization of gait impairment rehabilitations [33, 34], gaps remain with respect to identifying which populations benefit most from intervention programs based on the following: 1) the physiological benefits of exercise with respect to disease modification across disease stages; 2) the best type of exercise; and 3) the optimal intensity of exercise [35].
METHODS
Hoehn and Yahr Scale
PD staging scales are a means of assessing symptoms and monitoring the course of the disease in addition to providing information regarding an individual’s quality of life. The Hoehn and Yahr Scale (H&Y) is an internationally used PD progression rating method for clinical practice for the measurement of how PD symptoms progress and depict the level of clinical disability [12, 37]. This staging scale is depicted below: Stage 1: Symptoms are present on one side only (unilateral). Stage 2: Symptoms are present on both sides but no impairment of balance. Stage 3: Balance impairment and moderate disease progression. Stage 4: Severe disability, but still able to walk or stand unassisted. Stage 5: Needing a wheelchair or bedridden unless assisted.
The H&Yahr scale was used in the supervised classification of individuals into two subgroups: early stage (H&Y Stages 1 & 2) and advanced stage (H&Y Stages 3–5) [38] for the purpose of this study.
Cohort
Ninety-five adults between the ages of 50 and 85, divided into two groups- those with a confirmed diagnosis of PD and age-matched healthy controls participated in this study. The PD population included 70 individuals; with 40 being in confirmed early stages of PD (H&Y Stages 1 & 2), 30 being in confirmed advanced stages of PD (H&Y Stages 3–5). A breakdown of the population is shown in Table 1. All individuals in the PD group who participated in this study had a confirmed diagnosis of PD by a neurologist or movement disorder specialist who was also responsible for assigning the HY staging score. Given the population size, grouped stages of PD (e.g., early (H&Y Stages 1 & 2) or advanced (H&Y Stages 3–5)), was used to maintain a more even group distribution rather than individual observed stages 1–5 [38]. Of the total individuals with a confirmed diagnosis of PD, slightly more than half (n = 36 or 51.4%) were enrolled in physical interventions (e.g., structured exercise programs); 24 in confirmed early stages of PD (H&Y Stages 1 & 2) and 12 were in confirmed advanced stages of PD (H&Y Stages 3–5). Of the total group of individuals diagnosed with PD, slightly less than half were female (n = 33 or 47%). Of the PD population completing structured physical interventions 13 females were in confirmed early stages of PD (H&Y Stages 1 & 2) and 5 were in confirmed advanced stages of PD (H&Y Stages 3–5). The age-matched control population included 25 individuals; with slightly less than half (n = 12 or 48%) being female. Participants were recruited for this work through advertisements, designed rehabilitation programs, physician and clinician referrals, spouses or caretakers of the diagnosed population, and those who participated in prior studies from our laboratory. As the mean onset age for PD in the Western world is early-to-mid 60s [39], recruitment efforts for this study were limited to individuals in the aforementioned groups aged 50 years or older. Participants were excluded from the current study if they were unable to provide informed consent or if they were unable to speak and/or understand English (as all instructions and tests were formatted in English). The work presented in this manuscript is part of an IRB approved study for the Protection of Human Subjects. All methods were performed in accordance with the relevant guidelines and regulations from the IRB. Written informed consent was collected from all participants included in this study.
Cohort Breakdown
The separation of the population into three groups: ‘Age Matched Healthy Control Population’, ‘PD Control Population’ (e.g., those diagnosed with PD but did not participate in any structured physical intervention programs), and ‘PD Population Completing Physical Interventions’ was utilized to understand the benefits of structured physical activity programs for individuals with PD. The PD Control Group (n = 34) and Age Matched Healthy Control Population (n = 25) were included in this work to provide comparative baselines of neurocognitive functionality and did not participate in structured physical intervention programs.
Qualitative assessment questionnaires
All participants were given a set of questions commonly administered in clinical settings for aging populations in addition to questions specifically regarding their diagnosed neurodegenerative disease. These questions included information regarding their diagnosis, disease stage, and symptoms (e.g., how the individual felt in general, their energy levels, their pain level, their sleep quality, and a rating of their cognitive functions of memory, speech, motor, and executive functions). Finally, a specific quality of life questionnaire (e.g., PDQ-39) was used to allow the individual the ability to assesses their mobility, activities of daily living, emotional well-being, stigma, social support, cognition, communication, and bodily discomfort [19].
Mobile application testing
All participants were administered a tablet-based neurocognitive assessment specifically designed for individuals with PD that focused on user-device interactions for the collection of objective metrics [14]. Each participant completed mobile versions of 14 neurocognitive functional tests (e.g., digitally translated versions of clinically implemented tests to assess PD) across the areas of motor, memory, speech, and executive function. Functional tests included single functional tests (e.g., having focus on only one area of neurocognition: motor or memory) and multifunctional tests (e.g., combining two or more single functional tests into one functional test). The 14 administered neurocognitive tests collected 208 objective tablet-based digital features for all participants. All test descriptions are listed below: For a fine-motor tracing test the individual was instructed to use their index finger to trace a depicted shape. For a gross-motor emulation test the individual was instructed to manipulate the device in space to “air-trace” a depicted shape. For a reaction test, the user was to tap on the screen to interact with a set of targets. In a set of card-matching tests the user was to tap on depicted cards, in pairs, until all cards have been matched. In a set of Trail Making Tests the user was instructed to draw a line using their index finger connecting all shapes in increasing numerical order. For a set of speech-based tests, the user was prompted to read both a sentence and passage out loud, and name prompted objects. Examples of multi-functional tests include a motor test (e.g., tracing or emulating an object) paired with a non-automatic speech test (e.g., listing the months of the year, aloud, in reverse order; December to January). For an executive function/multi-functional test a digital version of the Stroop Word Color Test (SWCT) [40] was utilized where the user was required to discern the difference between prompted colors and words and then speak the correct response. In an expanded multi-functional test approach (e.g., Narration Writer), the user was instructed to narrate a sentence (e.g., speech) while also writing (e.g., motor) word by word (e.g., writing the same word being said aloud) in the space provided (e.g., executive function).
Physical intervention
Of those in the PD group, a subset of the individuals (n = 36) participated regularly (e.g., at least twice a week) in structured rehabilitation/intervention programs specifically designed for PD. As this is a long-standing physical exercise intervention program, all participants were enrolled in these structured exercise programs prior to the start of this study. The supervised physical intervention program activities included aerobic activities (e.g., walking or cycling), non-contact boxing, functional strength, and yoga, on a rotational basis (e.g., individuals participated in local versions of the same program and rotated around through each of the aforementioned activities over time). Each participant that was enrolled in these structured intervention programs, designed specifically for PD populations, completed each aforementioned activity at least once during this study. Each intervention training session lasted between 45 and 60 minutes and consisted of guided warm-up, main, and cool-down activities. A further depiction of these structured intervention programs is included in Table 2 where program timing breakdowns and a sample subset of warm up, main (e.g., aerobic activity, boxing, functional strength, or yoga), and cool-down activities are presented. All sessions were led by certified personal trainers. As there were multiple certified personal trainers leading these physical activities, biannual training programs were instructed by a clinical education manager for both guidance and instruction on how to ensure standardization of delivery. The lowest ratio of trainers to diagnosed participants in structured intervention programs was 1:1 whereas the highest ratio was 1:8. Individuals were given a mobile device assessment, consisting of the afore- mentioned functional tasks, both prior to and directly after these supervised physical intervention programs. As the participants were required to take the functional assessment twice within a period of 2 hours, each administered task was internally randomized to aid in the minimization of test-retest phenomena (e.g., for a memory task, the location of matching card pairs; or in the Stroop Word Color Test, the order of colors and word combinations differed between assessments). In addition, patients were asked to give information regarding their medication schedule such that the completion of the assessment/physical intervention protocol occurred at trough pharmacological levels. The rationale for this parameter is that the assessment of the patient near trough levels of pharmacological interventions should depict the most extreme effects of different interventions without the effect of any medication. Although individuals in the PD control population may still be active, they were categorized into a separate group as they did not participate in structured physical intervention programs.
Structured Exercise Program –Sample Activities and Durations
Feature normalization
The standardization of feature values is necessary as many features are of unique type and have varying units. For this normalization, Z-scores were used. The calculation of a Z-score is depicted in Equation 1 where x is an individual’s score, μ is the population mean and σ is the standard deviation of the population.
The Z-score is measured in terms of standard deviations from the mean. If a Z-score is 0, it indicates that the data point’s score is identical to the mean. A Z-score of 1.0 would indicate a value that is one standard deviation from the mean. Z-scores may be positive or negative, with a positive value indicating the score is above the mean and a negative score indicating it is below the mean.
RESULTS
Feature normalization
Objective digital features
For a depiction of neurocognitive functionalities between groups, normalized scores from the objective tablet-based assessments were calculated for the functional areas of motor, memory, speech, executive function, as well as multifunctional tests. Weighted Z-scores for neurocognitive capabilities of healthy controls (e.g., a baseline of ‘normal’ neurocognitive functionality), PD populations before a physical activity, PD populations directly following a physical activity, and PD control populations (e.g., individuals with PD that do not participate in structured physical intervention programs) are shown in Fig. 1. Figures 2 and 3 depict weighted Z-score averages based on grouped disease stages. The neurocognitive functionalities of motor, memory, speech, executive function, and multifunctional tests for individuals in early stages of PD (H&Y Stages 1 & 2) are shown in Fig. 2, whereas advanced stages of PD (H&Y Stages 3–5) are depicted in Fig. 3. It should be noted that all executive function tests are inherently multi-functional in nature (e.g., an individual needs to move or speak to carry out the executive function) and therefore are a subset of the multi-functional test digital feature set (e.g., denoted by an * in Fig. 1–3). These figures depict that on average, age-matched healthy control populations have higher neurocognitive functionality scores (e.g., higher Z-scores) compared to all other groups, whereas PD control populations (e.g., individuals with PD that do not participate in structured physical intervention programs) have lower neurocognitive functionality scores (e.g., lower Z-scores). Individuals in the PD group that do participate in structured physical intervention programs, on average, have higher neurocognitive functionality scores when compared to those who do not participate. In addition, overall functionality for the active population increases following various intervention programs.
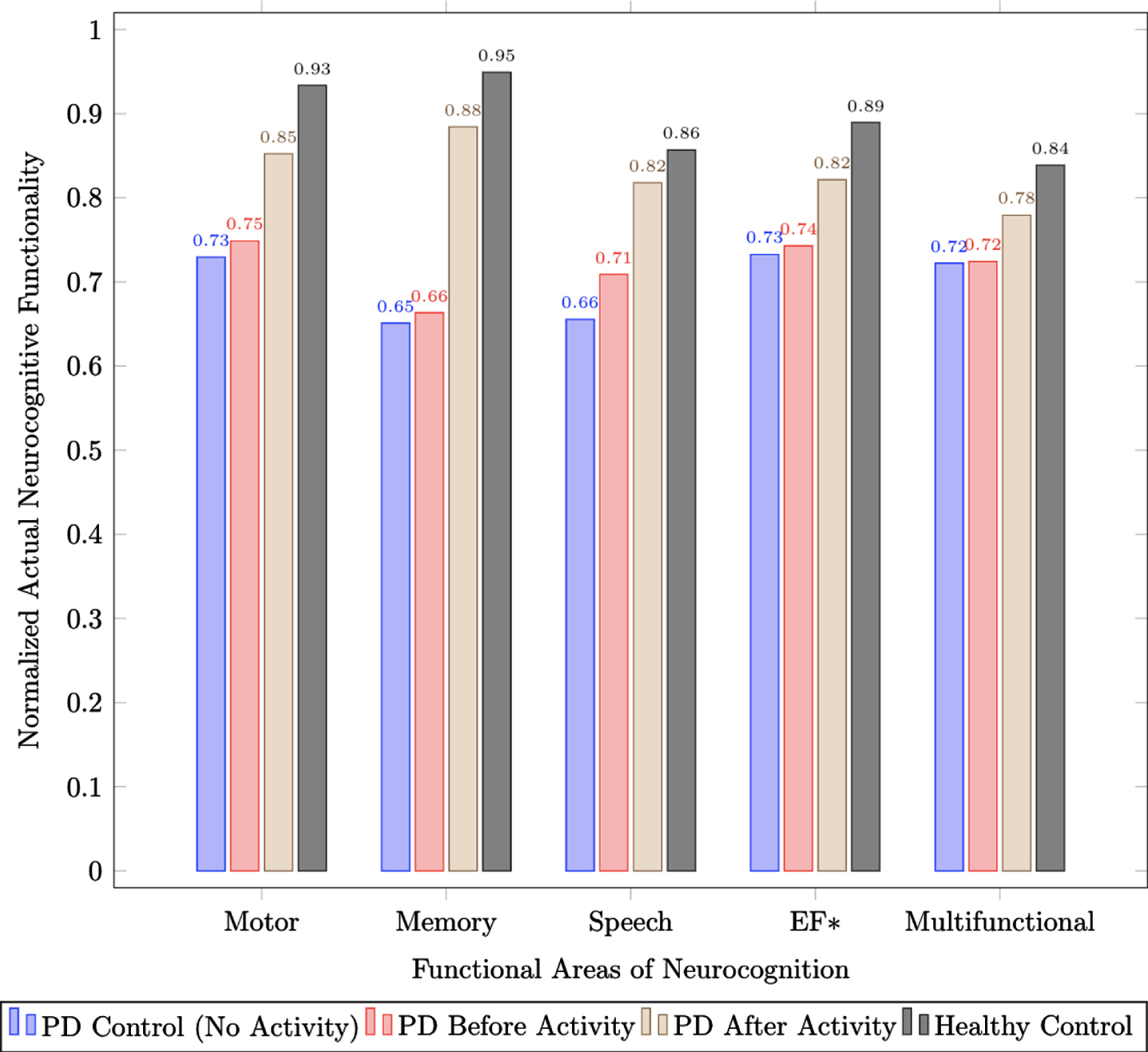
Actual functionality scores across motor, memory, speech, executive function, and multi-functional neurocognition for healthy controls, individuals with PD both before and after activity, and PD controls who did not participate in structured physical intervention programs.
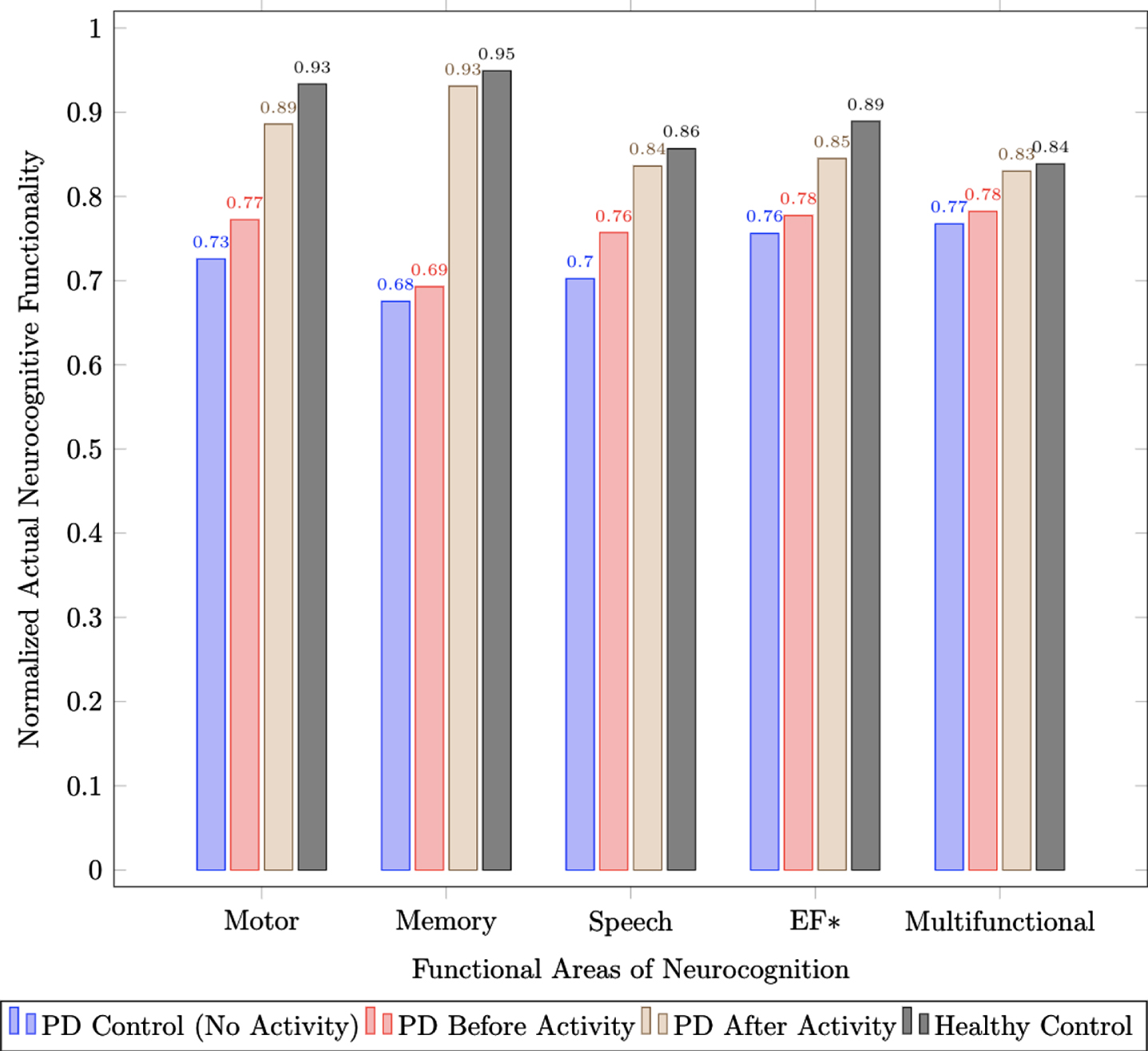
Actual functionality scores across motor, memory, speech, executive function, and multi-functional neurocognition for healthy controls, individuals in early stages of PD (H&Y Stages 1 & 2) both before and after activity, and early stage PD controls (H&Y Stages 1 & 2) that do not participate in structured physical intervention programs).
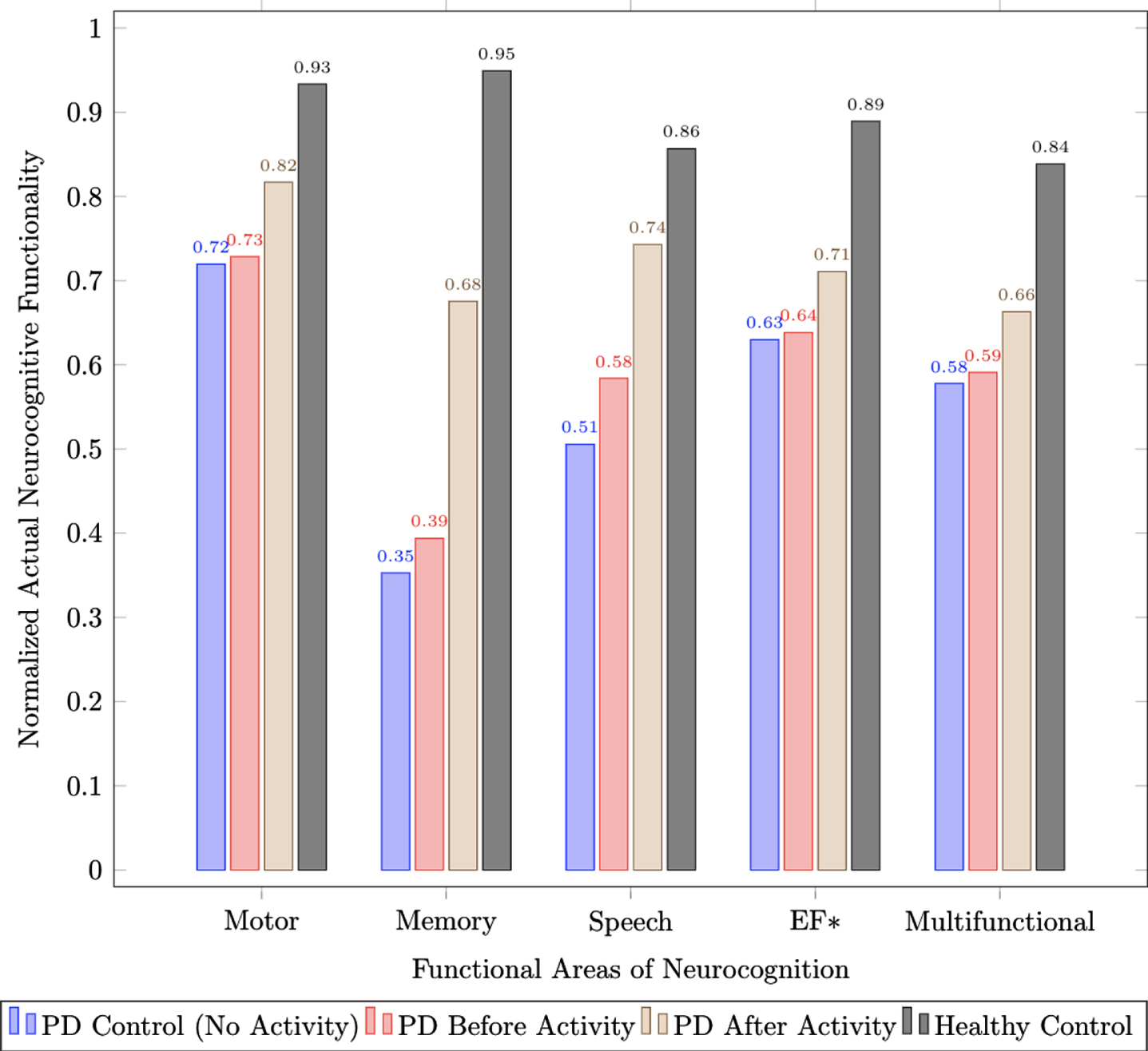
Actual functionality scores across motor, memory, speech, executive function, and multi-functional neurocognition for healthy controls, individuals in advanced stages of PD (H&Y Stages 3–5) both before and after activity, and advanced stage PD controls (H&Y Stages 3–5) that do not participate in structured physical intervention programs.
Physical interventions
A depiction of symptom-specific neurocognitive functional improvement for individuals with PD, following a physical intervention (e.g., aerobic activity, boxing, functional strength, and boxing), is shown in Table 3 where results are given as delta (Δ) values with relation to the normalized score to depict overall functional improvements. This heat map shows the relational improvement of symptom-specific functions given a physical intervention as a delta value of the normalized digital feature score. Subsequently, Tables 4 and 5 show weighted Z-score delta values based on grouped disease stages. Delta value mapping between intervention type and symptom-specific functionality for individuals in early stages of PD (H&Y Stages 1 & 2) are presented in Table 4, whereas advanced stages of PD (H&Y Stages 3–5) are presented in Table 5. These figures depict that physical activities have direct symptom-specific benefits with regard to different intervention programs (e.g., regions of darker green depict a higher delta (Δ) of improvement). Functional strength and aerobic activities are shown to yield the highest overall functional increase to the assessed neurocognitive functions regardless of stage.
Z-Score delta relationship between intervention protocol and neurocognitive functions of interest for individuals with PD
Z-Score delta relationship between intervention protocol and neurocognitive functions of interest for individuals in early-stage PD (H&Y Stages 1 & 2)
Z-Score delta relationship between intervention protocol and neurocognitive functions of interest for individuals in advanced-stage PD (H&Y Stages 3–5)
DISCUSSION
Physical intervention
As PD is often described as a “designer disease”, meaning individuals with PD manifest different symptoms across the spectrum of disease characteristics, personalized medicine should be the goal and is required to optimize care [41, 42]. However, to reach personalized medicine, relevant objective features (e.g., collected from configured mobile devices) cross-referenced with intervention programs are necessary to map the benefits of different therapeutic activities on symptom-specific functions. This work included the feature normalization of 208 device-based metrics collected from digital health technology specifically designed for individuals with PD [14] for use in depicting the relation between symptom-specific areas (e.g., motor, memory, speech, executive function, and multifunctional tasks) as they relate to physical interventions. In accordance to previous studies in literature [27, 31], physical interventions were shown to improve both motor and non-motor PD symptoms as overall normalized scores of neurocognitive functionality were significantly different following physical activity (p = 0.0188). This is further depicted in Figs. 1–3 where the average normalized scores for individuals in physical intervention programs are 1.16x higher following physical interventions as compared to PD control groups (e.g., individuals with PD that do not participate in structured physical interventions). Additionally in individuals who participated in physical intervention regularly (e.g., at least twice a week) had a 1.06x higher average normalized score for symptom-specific functionalities prior to physical interventions as compared to PD control groups depicting the longer-term benefits of structured physical activity programs.
The process of identifying the neurocognitive benefits of physical activity, as a prescribed therapeutic intervention program, should ultimately occur with increased rigor (e.g., similar to pharmacological intervention optimization) for the purpose of yielding symptom-specific information [43, 44]. This work expands on previously presented studies through the mapping of improvements across multiple symptom-specific functionalities (e.g., motor, memory, speech, and executive function) [45–48] in relation to specific advocated physical intervention programs (e.g., aerobic activity, non-contact boxing, functional strength, and yoga) [11, 49] across grouped stages of PD (e.g., H&Yahr Stages 1–5) [38] as seen in Tables 3–5. The physical interventions of functional strength and aerobic activity yield the highest deltas for improving overall neurocognitive functionality for PD (0.1670 and 0.1574; respectively). However, functional strength activities provided the largest significant delta improvement (Δ= 0.1883; p = 0.0265) for early-stage (H&Y Stages 1 & 2) PD populations, whereas aerobic activity provided the largest significant delta improvement (Δ= 0.2700; p = 0.0364) for advanced stages of PD (e.g., H&Y Stages 3 –5).
Significant symptom-specific effects of physical interventions to note for early-stage (H&Y Stages 1 & 2) PD populations include yoga on motor function (Δ= 0.1809; p = 0.029), functional strength on memory (Δ= 0.5334; p = 0.008) and executive functions (Δ= 0.1913; p = 0.046), and boxing on speech (Δ= 0.1612; p = 0.050). Multifunctional tasks were most affected by the physical intervention boxing; however, the changes (Δ= 0.1155) were not significant (p = 0.076). Uniquely, the symptom-specific effects of physical interventions to note for advanced-stage (H&Y Stages 3–5) PD populations, include aerobic activity on motor (Δ= 0.1624) (p =0.046), memory (Δ= 0.6848; p = 0.014), and speech (Δ= 0.4189; p = 0.041), and functional strength on executive function (Δ= 0.3287; p = 0.043). Yoga was most beneficial for the improvement of multifunctional tasks; however, the changes (Δ= 0.1533) were not significant (p = 0.110).
Future work
The ability to map the improvement of objective, symptom-specific digital features to specific physical interventions, yields a more personalized approach to intervention programs. With the collection of patient reported outcomes clinician observations, and objective digital features from mobile-based assessment tools, clinicians can accurately assess symptom-specific functional deficits across different areas of neurocognition and subsequently provide data-derived intervention protocol recommendations. Future work should focus on the relation between interventional therapies and additional neurocognitive functionalities (e.g., sleep, language, behavior, sensory, and autonomic functions) and their subfunctionalities (e.g., fine motor vs. gross motor vs. balance) [16]. Additionally, a focus on the relation between interventional therapies on neuocognitive functions (e.g., executive function) on a test level (e.g., Trail Making [11] vs. Stroop Word Color Test [46]) should be included as part of future work. Further, an extension of this work should be applied to standardized protocol versions of other physical (e.g., dancing or swimming), pharmacological (e.g., novel drug therapies and/or the monitoring of different pharmacological cycles), medical (e.g., deep brain stimulation), occupational, psychological, dietary, speech, and music therapies [9, 50, 51]. Future work should also assess the differences in disease presentation and progression between males and females and across different demographic breakdowns (e.g., race and ethnicity). Finally, an extension of this work should include the use of machine learning methods (e.g., k-nearest neighbors, decision trees, and random forests) [52, 53] for the automation of intervention recommendations, given collected user data.
Limitations
A limitation of this work arises given the population size, where stage had to be grouped (e.g., early-stage PD (H&Y Stages 1 & 2) or advanced-stage PD (H&Y Stages 3–5)), rather than individual observed stages 1–5. Future work in this domain should include the collection of much larger sample sizes allowing for the separation of individuals into individual stages for a more fine-grained classification approach.
Finally, this work only shows a subset of commonly utilized clinical assessments. Future work should also include further testing and metric collection for the provision of revised digital health system classification models. This is necessary for the classification of individuals with different neurodegenerative conditions (e.g., Alzheimer’s disease, amyotrophic lateral sclerosis, multiple sclerosis, Huntington’s disease, and other types of dementia) and their respective stages.
CONCLUSIONS
Using digital health systems for monitoring individuals with neurodegenerative diseases allows for more comprehensive insights of these conditions and their progression. As digital health technology allows for the collection of large amounts of complex health data, new ways to analyze and interpret this data emerge. In addition, these mobile-based assessment tools may bring to light new features that expand the way digital versions of functional assessments should be administered. Utilizing this information to reevaluate and update standardized techniques and scales can ultimately allow for novel digital health systems and disease monitoring. In turn, this can aid clinicians, diagnosed populations, and caretakers in monitoring all neurocognitive functions while also allowing for increased efficacy for both diagnostic and rehabilitative purposes.
CONFLICT OF INTEREST
All authors of this work declare that there are no conflicts of interest in the authorship or publication of this contribution.
