Abstract
BACKGROUND:
Parkinson’s disease (PD)-associated psychosis is a well-known non-motor complication, occurring years after diagnosis of PD. Incidence data vary across different studies highlighting a need for long-term observation and clinical definition.
OBJECTIVE:
To determine the incidence of psychosis in patients with PD and to investigate their survival in an incident cohort study from 1991–2010 in Olmsted County, MN.
METHODS:
We used the Rochester Epidemiology Project to define an incident-cohort study of parkinsonism (1991–2010) in Olmsted County, MN. A movement-disorder specialist reviewed the electronic medical records and applied diagnosis criteria to PD. Psychosis was diagnosed using of NINDS/NIMH unified criteria.
RESULTS:
We identified 669 cases of parkinsonism; 297 patients were clinically diagnosed with PD. 114/297 (38.4%) patients had evidence of psychosis (60% male); the median onset age of psychosis was 79.4 years. The incidence of Parkinson’s disease psychosis (PDP) was 4.28/100 person-years. PDP patients had a 71% increased risk of death compared to PD patients. In PD patients without psychosis, men had 73.4% increased risk of death compared to women, whereas no significant sex difference was observed among PDP men vs. women. Of 114 patients diagnosed with psychosis, 59 were treated with antipsychotics. There was no significant difference in survival between treated and untreated patients.
CONCLUSION:
PDP increased the odds of death compared to PD patients. Men with PD without psychosis had greater odds of death compared to women; however, in PD with psychosis the odds of death were comparable among sexes. Lastly, treatment with anti-psychotics did not significantly affect survival.
INTRODUCTION
Parkinson’s disease (PD) is a progressive neurodegenerative disorder that recognizes the loss of dopaminergic neurons within the substantia nigra accompanied by Lewy body inclusions and alpha-synuclein deposit as the pathological hallmark [1]. PD is the second most common neurodegenerative disorder after Alzheimer’s disease, with an incidence of 14 cases per 100,000 person-years and affecting men more often than women [2]. The clinical course of PD is variable and can include motor and non-motor symptoms. Among non-motor symptoms, psychosis has major clinical implications for patients with PD and represents a significant challenge in the treatment of PD [3, 4]. PD-associated psychosis (PDP) significantly hinders quality of life, increases caregiver burden, and increases nursing home placement [5]. The frequency of psychosis symptoms in PD is variable with a frequency between 16% and 75% [6–13]. Unfortunately, the treatment of PDP still represents a challenge for clinicians, given the convoluted relationship between anti-parkinsonian medications, anti-psychotic medications, and clinical course of the disease. The use of antipsychotic medications is the usual initial strategy to treat PDP; however, such treatments reportedly have relatively poor efficacy and a possible risk of cardiovascular mortality, particularly the typical antipsychotics.
Our study aims to determine the incidence and prevalence of PDP, assess the mortality of PDP patients who are receiving antipsychotics versus PDP patients who are not receiving antipsychotics, to assess the efficacy of antipsychotic medications in resolving PDP, and to examine the possible presence of sex differences within PDP.
MATERIALS AND METHODS
All data were collected through the electronic medical records-linkage system of the Rochester Epidemiology Project (REP). The REP encompasses all local medical facilities within Olmsted County, Minnesota, with a catchment rate of over 99% in this population [14, 15]. This also includes all private practitioners and nursing homes. The electronic index of the REP includes all medical records, diagnostic and procedure codes, health services utilization data, outpatient drug prescriptions, demographics, and additional medical data.
Study population
Case ascertainment and exclusion criteria
We used the unique infrastructure of the REP medical-records linkage system [14]. Through the REP, 669 incident cases of parkinsonism were identified within Olmsted County, MN, from 1991 to 2010 [2]. Given that some patients with PD do not report immediately to the physician, the disease onset of the can be retro-dated by a few years, and the cohort may have incomplete incidence cases over the last 5 years (2006–2010). Thus, there is a complete population-based cohort from 1991 to 2005 and an incident cohort from 2005 to 2010. A movement-disorders specialist (R.S.) reviewed the clinical records of the 669 patients with parkinsonism, and then confirmed the diagnosis of the different parkinsonism variants. This methodology has already been used and it is reported in detail elsewhere [2]. A board-certified neuropathologist reviewed available autopsy reports to confirm the clinical diagnoses when possible. Details regarding clinicopathologic characteristics of this cohort have been reported elsewhere [16].
Diagnostic criteria
Our diagnostic criteria included two steps: the definition of parkinsonism as a syndrome and the definition of types of parkinsonism within the syndrome. Parkinsonism was defined as the presence of at least two of four cardinal signs: rest tremor, bradykinesia, rigidity, and impaired postural reflexes. Among the persons who fulfilled the criteria for parkinsonism, we applied the diagnostic criteria to classify individuals with PD [2]. Additional details regarding the methods have been reported elsewhere [2, 17].
Identification of PDP patients
The most recent NINDS/NIMH Work Group clinical criteria for psychosis were used to classify PDP [18]. Psychosis was defined as the presence of one or more of the following: illusions, false sense of presence, hallucinations, or delusions within the medical records. We could not further define whether the hallucinatory phenomena were visual or auditory because of lack of consistent details in the medical records to distinguish the hallucinations accurately. Symptoms of psychosis had to be continuous or recurrent for a minimum of one month and could not be explained by a neuropsychiatric condition or other medical condition. For the psychosis definition, we excluded single instances of acute changes in mental status such as acute confusion and delirium due to medical conditions. Patients were considered to have unresolved psychosis if the symptoms persisted for at least six months. We abstracted all the data on medications used for psychosis in cases and controls. PDP patients were considered as treated if they were prescribed antipsychotic medications any time after their diagnosis of PDP for psychosis indication. All cases of psychoses were abstracted from the patient’s date of birth until 12/31/2019 or to their death. The list of antipsychotic medications investigated were: quetiapine, clozapine, olanzapine, and risperidone. If psychosis was not reported in medical records, the patient was considered free of psychosis.
Statistical analysis
Continuous variables were summarized with medians and interquartile ranges (IQRs); categorical variables were summarized with frequency counts and percentages. Incidence rates of psychosis per 100 person-years were calculated using the number of incident cases of psychosis divided by the number of years at risk for the cohort during the specified period. Patients with partial follow-up within a period were counted proportionally. Sex-specific incidences were also calculated.
The risk of mortality for PD patients was assessed using a Cox proportional-hazards model [19]. The outcome of interest was follow-up time in years from the onset of PD to date of last follow-up, with an indicator as to whether the patient was deceased (1; event) or alive (0; censor) at last follow-up. The model included time-dependent covariates (TDC) as predictors for psychosis and unresolved psychosis. The TDC of psychosis was created using time from the onset of PD to first instance of psychosis (1; event) or last follow-up if no psychosis occurred (0; censor). Similarly, the TDC for unresolved psychosis were created using the time from the onset of PD to the presence of psychosis lasting longer than 6 months (1; event) or last follow-up, if psychosis resolved before 6 months or no psychosis occurred (0; censor). This model was adjusted for patient sex and age at onset of PD, as well as the presence of each of tremors and bradykinesia at PD onset. Age at onset was centered and scaled to 5-year increments.
In a secondary analysis, the relationship between mortality and the treatment of psychosis with antipsychotic medications was assessed using a Cox proportional hazards model. Only patients in the PDP cohort were included in this analysis. The outcome of interest was follow-up time in years from the onset of psychosis to date of last follow-up, with indication as to whether the patient was deceased (1; event) or alive (0; censor). In this secondary model we again included the same adjustments of sex, age, and symptoms as in the primary model.
All statistical analyses were conducted using R software version 3.6.1 (The R Foundation for Statistical Computing, Vienna, Austria) [20], and a p value of <0.05 was considered statistically significant.
Standard protocol approvals, registrations, and patient consents
The Mayo Clinic and Olmsted Medical Center Institutional Review Boards approved this study (ID reference number 07-008807), and participating patients (or their legally authorized representatives) provided informed written consent for the use of their medical information for research [2]. More detailed information about the Olmsted County population is reported elsewhere [14, 22]. Information about the different types of parkinsonism and the clinical and pathological characteristics of this cohort is also reported elsewhere [16, 23].
Data availability statement
All the relevant data have been shared and published in this article; data regarding case ascertainment of parkinsonism and methodology on case identification have been previously published [2].
RESULTS
Demographics, incidence, and prevalence
Among the 669 incident cases of parkinsonism in Olmsted County, MN, from 1991 to 2010, there were 311 cases of PD. Eleven cases were excluded for not having record of the cardinal signs of PD. Three additional cases were diagnosed with psychosis prior to the onset of PD and were excluded, resulting in a dataset of 297 incident cases of PD (178 (59.9%) male, 119 (40.1%) female). The median age at PD onset was 74 years of age (74 years for males, 75 years for females, IQR = 66–81). There was pathological confirmation in 24 (3.4%) patients within our cohort. The clinicopathologic correspondence positive predictive value was 83.3% (20/24) and the sensitivity for the patients with clinical diagnosis of PD was 100%. Of the clinicopathologic discrepancies two were pathologically consistent with Alzheimer’s disease, one with progressive supranuclear palsy, and one with vascular parkinsonism.
Of the 297 cases of PD, 114 (38.4%) patients developed psychosis. The median age of psychosis onset was 79 years of age (80 for males, 79 for females, IQR = 74–86). Among the 114 individuals with PDP, 93 (80%) had died at the time of abstraction with a median age at death of 84 years (84 for males, 85 for females, IQR = 79–90). The median age at death in the 183 individuals without psychosis was 86 years (86 for males, 88 for females, IQR = 80–91). Further details of patient demographics and characteristics can be found in Table 1.
Patient Characteristics by Sex
Hallucinations were the most common symptom of psychosis, occurring in 99 of the 114 (87%) PDP patients. The median time to the onset of psychosis was 8.1 years (8.3 for females, 8.1 for males, IQR 5.7–10.6) after the onset of PD. The median time of follow up for the entire cohort was 18 years (95% CI: 17–20). 59 out of 114 (52%) PDP patients were treated with antipsychotics (25/49 [51% ] females, 34/65 [52% ] males). Table 2 provides details regarding the characteristics of psychosis for the PDP cohort.
Psychosis Summary for PDP Patients
1Other indicates antipsychotic medication apart from those listed above.
The incidence of psychosis within the first five years of PD was 1.97 cases per 100 person-years (2.22 males, 1.68 females). The overall incidence of PDP was 4.28 cases per 100 person-years (4.47 per 100 person years for females and 4.14 per 100 person years for males). Figure 1 shows the incidence of psychosis by disease year in males and females.
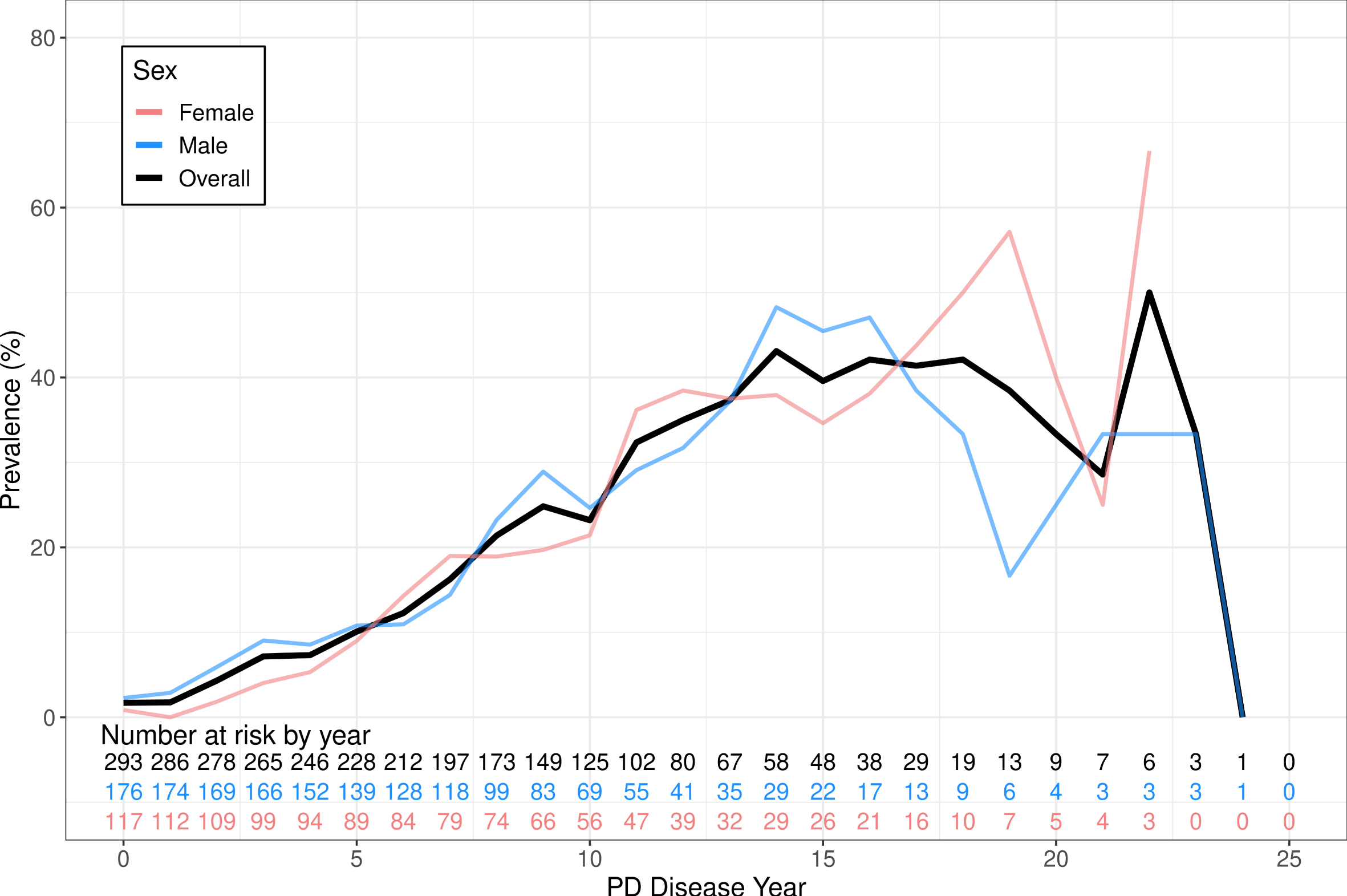
Shows the incidence of PDP comparing male and female sex as well as both sexes combined. Incidence is calculated per 100 patient years. The number at risk table represents the number of PD patients who are still at risk of developing psychosis comparing the overall cohort.
The prevalence of psychosis at disease year 5, 10, and 15 years was 10.1%, 23.2%, and 39.6% respectively. The median duration of the first instance of psychosis was 2.45 years, with 63.2% of initial instances of psychoses lasting longer than 6 months.
Mortality and survival of PDP
Using the Cox proportional-regression model, we observed that PDP patients have an increased risk of death compared to PD patients (HR = 1.71, p = 0.027, 95% CI = [1.06, 2.76]). Figure 2 shows the expected survival of PDP patients compared with PD patients. Individuals with unresolved psychosis had no significant difference in mortality compared to patients with resolved psychosis (HR = 1.41, p = 0.19).
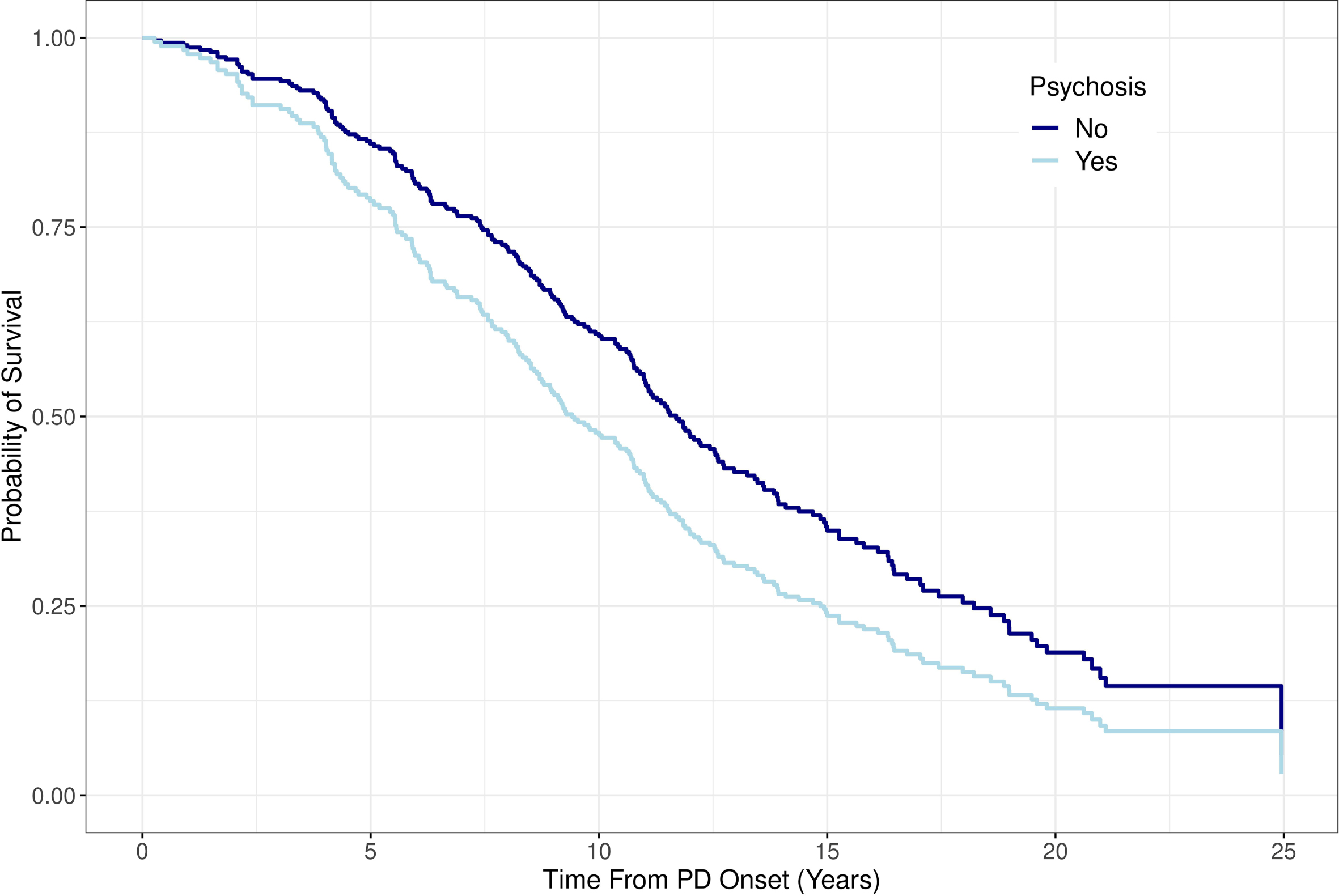
Shows the estimated survival comparing PD patients to PDP patients.
Among the patients who have PD without psychosis, males had a 73.4% increase in the risk of death compared to females (HR = 1.73, p < 0.001, 95% CI = [1.29, 2.33]). However, within the PDP cohort there was no sex difference in risk of death (HR = 1.11, p = 0.70, 95% CI = [0.71, 1.73]). Model estimates regarding the risk of mortality in PD and PDP patients can be found in Tables 34 respectively.
Risk of Mortality in PD Patients
Risk of Mortality in PDP Patients
Antipsychotic treatment in PDP
Out of the 114 PDP patients, 59 (52%) were treated with antipsychotics. There was no significant difference in the risk of death following the onset of psychosis among treated and untreated PDP patients after accounting for age, sex, and the presence of tremors or bradykinesia at PD onset (HR = 1.11, p = 0.60). Similarly, there was no difference in the risk of death following the treatment with antipsychotics after the onset of psychosis between males and females (HR = 1.11, p = 0.70).
DISCUSSION
Incidence and prevalence of PDP
Few epidemiological studies have explored the frequency of PDP due to the rarity and the lack of a universally accepted diagnostic criterion. Using the REP medical records-linkage system, we report an overall incidence of 4.3 cases per 100 person-years. Prevalence of psychotic symptoms was 38.4% in our population of PD patients. In a previous study, which used a prospective longitudinal design in Rogaland County, Western Norway, the observed incidence was 7.97 per 100 person-years after a 12-year follow-up [24]. Despite supporting similar results, the Norwegian results differ from ours as their study explored only hallucinations or delusions, whereas our study used the NINDS/NIMH Work Group clinical criteria [17]. Furthermore, the longer follow up of about 25 years of clinical observation, in-depth review of medical records, and the approach of our study may have provided a more accurate estimation of PDP. However, the higher incidence of PDP found in the prospective study may indicate an underreporting of psychotic symptoms within patient populations. This may be of importance to bring awareness to the significancy of educating PD patients about psychosis. This may also encourage healthcare providers to examine for psychotic symptoms when working with PD patients.
The prevalence of psychotic symptoms in PD has been reported varying from 16–75% [6, 25]. However, there is limited knowledge of the epidemiology of PDP using the most recent diagnostic criteria, which includes multiple psychotic symptoms [18]. The population-based study from Norway also found a prevalence of 41.9% [26]. A study from the UK also observed a prevalence of 26% of PDP, while 90.8% of those patients met the NINDS/NIMH Work Group clinical criteria for PDP [27]. However, a different cross-sectional study using this criterion reported a prevalence of 60% [25]. On the other hand, methodological differences and differences in the clinical definition should be considered while comparing this study to others. Importantly, the NINDS/NIMH Workgroup has recently proposed new criteria that we adopted but could not be used in previous studies, thus the discrepancies in the clinical definitions. In addition, using hospital-based or community-based cohorts, can lead to an overestimation in the frequency of PDP.
Mortality and survival of PDP
Although several studies have shown that PDP increases caregiver burden and risk of admission to nursing home care [28], few studies have explored the survival of patients with PDP compared to PD. A population-based study from Norway reports that patients with PDP have a 98% increased risk of death compared to PD patients [26]. Our results show that PDP patients have a 71.4% increase in the risk of death; importantly, individuals with a psychosis lasting longer than 6 months (hence defined as “unresolved”) did not have a higher rate of mortality as compared to those with a resolved psychosis. Thus, duration of psychosis does not seem to be associated to an increased risk of mortality in our study. Furthermore, nearly 70% of PDP cases in our population had a psychosis that lasted longer than six months, showing that the past treatment within this population was not effective. Although a number of studies have reported ambiguous results about the efficacy of antipsychotics on PDP symptoms, few have explored their impact on mortality [29–34]. A recent study showed that the off-label use of antipsychotics to treat patients with PDP led to an increased rate of mortality [35]. Our results show that treatment with antipsychotics does not have a statistically significant effect on mortality compared to PDP patients who went untreated. Although we do not observe an association, our results may support that the use of antipsychotics may potentially improve symptoms without decreasing the survival of these patients [29–34]. Notably, we observed that none of the patients experiencing psychosis were prescribed clozapine within our cohort. While clozapine is a drug that can be effective on the treatment of psychosis, the need of a diligent compliance on the management of possible drug-induced neutropenia or agranulocytosis limits the use in the clinical practice, especially in non-neurological/psychiatric providers.
Our study also supports that the clinical phenotype upon presentation of PD symptoms impacts the survival of PD patients. We report that patients who present with tremor predominant (TD) phenotype of PD have a decreased risk of death compared to patients who present with postural instability/gait difficulty (PIGD) phenotype. It has been shown that individuals with PIGD phenotype experience greater subjective intellectual, motor, and occupational impairment than people with TD [36]. These individuals also have a greater incidence of neuropsychiatric disorders compared to individuals presenting with TD phenotype [37, 38]. However, when examining the effect of phenotype on mortality of PDP, we find that there is no significant difference between TD and PIGD. To our knowledge, the clinical phenotype of PD on the mortality related to PDP is relatively unexplored. These results may continue to support that these differing phenotypes may have pathological and molecular differences, which impact disease course and survival. Our findings further highlight the relevance of an individualized treatment of these differing phenotypes. Although these phenotypes have differing impacts on mortality in PD, they did not have a significant difference on mortality in PDP.
Another relevant observation was that the treatment with antipsychotic medications did not further impact the mortality of patients with TD or PIGD phenotypes in PDP. These results further suggest that the current standards of care for patients with PDP may not be effective at treating PDP or decreasing the mortality of PDP.
Recent studies have been exploring the effect of sex on the clinical features, disease course, response to treatment, mortality, and pathophysiology in patients with PD. It has been shown that male sex is associated with higher incidence, earlier disease onset, more severe motor symptoms, hastened disease progression, and more frequent cognitive symptoms [39]. Our study finds that both men and women have a similar incidence of PDP as well as a similar frequency of presenting psychotic symptom and no significant difference in mortality when comparing men and women with PDP.
Our study has several strengths, including its long follow-up period (25 years) and having access to all medical history of each patient, which allows us to minimize the effect of selection bias on our results. However, we also acknowledge several limitations; additionally, confounders and reverse causality were considered in the interpretation of the data, but our study design and statistical adjustments minimized this risk. First, using a retrospective study design may overlook under-reporting of psychotic symptoms. In fact, it is possible that the provider writing the clinical notes could have omitted psychosis symptoms; therefore, there is the risk that our percentage of PDP (38%) could be underestimation. However, to minimize this effect, a study abstractor reviewed all cases of PD using the most recent criteria to classify PDP. Secondly, due to PDP’s rarity, in 114 cases, we only had 59 cases treated with antipsychotics, limiting more granular analyses. Thus, the accuracy of the documented clinical parameters may be affected. Using a retrospective study design, we could not confirm that patients who were prescribed anti-psychotic medications used them; also, it was not possible to have detailed report of visual symptoms in many cases; also, the clinicians used the term “hallucinations” to include “illusions” (hence, the absence of illusions in Table 2); moreover, we could not distinguish between visual and auditory hallucinations, since they were both reported as “hallucinations”. Hence, we were not able to break down our analysis between these two symptoms. Additionally, the study period used may no longer reflect the current incidence or treatment efficacy. Importantly, our study focused only on incident cases of PD that developed psychosis; we excluded all the cases of PD that later developed dementia; indeed, exploring the epidemiological figures of psychosis in PD-AD, PDD and DLB was beyond the scope of the study. We could not analyze PD patients with dementia (PDD) because we did not have detailed, consistently collected data on cognitive decline and dementia. Therefore, we excluded PDD cases from our study. Finally, our cohort lacked detailed pathological confirmation in most cases and, thus, is a clinically defined PD cohort.
Conclusion
Our results indicate that the overall incidence of PDP was 4.28 per 100 person-years and the prevalence was 38.4%. PDP patients had an increased risk of death compared to PD patients. Men with PD without psychosis had greater odds of death compared to women; on the other hand, in PD with psychosis the odds of death were comparable among men and women. However, there was not an increased risk of mortality in patients with unresolved psychosis compared to those who had resolution of their psychosis, and there was no difference in risk between treated and untreated PDP patients. Our results, if confirmed, highlight the importance of an early treatment of PDP and the need for more effective medications/treatment for PDP. The role of PDP in the physiopathology of PD needs to be further explored in additional studies that can investigate the current era and different medications that may change the progression of PD and PDP.
Footnotes
ACKNOWLEDGMENTS
This study was sponsored by ACADIA Pharmaceuticals, Inc.
CONFLICT OF INTEREST
MMM reports consulting for Brain Protection and receiving research funding from the NIH and Biogen. KAJ reports research support from NIA and NINDS. JHB reports research support from AbbVie, Inc. BFB serves on the Advisory Board of the Tauopathy Consortium; has contracts with Axovant and GE Healthcare and grants from NIA, NINDS, the Mangurian Foundation, and the Little Family Foundation. RS receives research support from the National Institute on Aging, the National Institute of Neurological Disorders and Stroke, and the Mayo Clinic Small Grants Program National Center for Advancing Translational Sciences (NCATS) and Acadia Pharmaceuticals Inc. The remaining authors have no conflicts of interest to report.
This study used the resources of the Rochester Epidemiology Project (REP) medical records-linkage system, which is supported by the National Institute on Aging (NIA; AG 058738), by the Mayo Clinic Research Committee, and by fees paid annually by REP users. The content of this article is solely the responsibility of the authors and does not represent the official views of the National Institutes of Health (NIH) or the Mayo Clinic.
