Abstract
The alpha-synuclein gene promoter (SNCA-Rep1) is associated with Parkinson’s disease (PD), but its relationship with performance across individual cognitive domains in early PD is unknown. This study aims to investigate Rep1 polymorphism and longitudinal change in cognition in early PD. In this longitudinal study, Rep1 allele lengths (“long” and “short”) were determined in 204 early PD patients. All participants underwent annual neuropsychological assessments and followed up for 3 years. Linear-mixed model was performed to investigate the association of Rep1 status and longitudinal change in individual cognitive domains. At 3 years, significant decline in executive function was observed in long Rep1 allele carriers vs short allele carriers, controlling for potential confounders. This is the first longitudinal study demonstrating that long Rep1 allele carriers are at higher risk for executive dysfunction in early PD.
INTRODUCTION
Parkinson disease (PD) is characterized clinically by motor features of rigidity, tremor, and bradykinesia, and non-motor features including cognitive impairment and PD dementia. Pathologically, degeneration of nigrostriatal dopaminergic neurons with alpha-synuclein-containing Lewy body deposits occur and widespread accumulation of alpha-synuclein in cortical regions associate with cognitive impairment and dementia in up to 50% of PD patients during their disease course [1]. Identifying patients at risk of cognitive decline remains crucial so that early intervention can be implemented. Multiplications of the alpha-synuclein (SNCA) gene are reported in familial PD and result in greater alpha-synuclein pathological burden, earlier disease onset and PD dementia (PDD) [2]. Non-amyloid component Rep1 is a polymorphic microsatellite repeat located ∼10 kb upstream of the SNCA transcription start site and regulates alpha-synuclein expression, with homozygous carriers of the longer Rep1 allele potentially mimicking SNCA locus multiplication [3]. The SNCA Rep1 promoter has not only been shown in multiple cohorts to be a risk factor for sporadic PD [4–8], but also a risk factor for motor [9] and cognitive decline [10] in carriers of the long Rep1 allele. Our group recently reported that PD patients carrying long Rep1 alleles are more impaired on tests of global cognition and motor function independent of APOE4 genotype [11]. The relationship between SNCA Rep1 polymorphism and longitudinal assessment of individual cognitive domains, however, remains unknown. To address this gap, we investigated the association of Rep1 allele length with change in individual cognitive domains in patients with early PD. We hypothesized that longer SNCA Rep1 allele carriers will show greater impairment in cognitive domains of greater relevance in PD, e.g., attention, executive function and visuospatial function over time, compared with shorter allele carriers.
MATERIALS AND METHODS
Clinical recruitment
Participants were recruited from the National Neuroscience Institute, Singapore, between November 2014 and July 2019. All PD patients fulfilled the National Institute of Neurological Disorders and Stroke (NINDS) criteria for the diagnosis of PD [12]. The Early Parkinson’s Disease Longitudinal Singapore (PALS) study is an ongoing prospective cohort study analysing the progression of early PD over a follow-up period of 5 years. Patients were defined as ‘early PD’ using the following inclusion criteria: (i) motor symptoms ≤2 years, and (ii) carrying a diagnosis of PD of ≤1 year using the NINDS criteria as diagnosed by a movement disorders specialist. Ethics approval was obtained from the Singapore Health Services Centralised Institutional Review Board (CIRB) for the use of human participants in this study, and all participants provided informed written consent.
Neuropsychological assessment
Comprehensive neuropsychological assessment comprising 10 tests covering all five cognitive domains (two tests per domain) was used to assess cognition [13]. Verbal and visual memory was assessed using the ADAS-Cog word list learning [14] with delayed recall and Rey–Osterrieth complex figure (ROCF) [15] delayed recall respectively. Visuospatial function was assessed with the ROCF copy test and Benton judgement of line orientation [16]. Attention/working memory was assessed using the WAIS-IV digit span backward [17] and the Weschler Memory Scale (WMS-IV) [18] symbol span. Language was assessed by the Boston Naming Test [19] and WAIS-IV similarities [17]. Executive function was assessed using verbal (fruit) fluency and the Frontal Assessment Battery [20]. Means and standard deviations (SD) for each test appropriate for age, gender, and education level were based on locally validated norms when available, and international norms when local norms were not available. Standardized score for each test was calculated based on means and SDs. Individual cognitive domain scores were calculated as the average of the two standardized scores from the two tests in each domain.
SNCA Rep1 polymorphism genotyping
Genomic DNA was extracted from peripheral blood with QIAamp® DNA Blood Maxi Kit (Qiagen) according to the manufacturer’s protocol. Fragment length analysis of SNCA Rep1 and its length determination were performed as described previously [11, 21]. Patients with 259 bp and 261 bp were grouped as “short”, while 263 bp and 265 bp were grouped as “long” carriers [11].
Statistical analysis
Data were analyzed using SAS software version 9.4 for Windows (SAS, Inc. Cary, NC, USA). Demographics and clinical characteristics of PD patients were compared between short and long carriers of SNCA Rep1 allele using two-sided two-sample t test or Mann-Whitney U test (depends whether normality assumption was tenable) and Chi-squared test for continuous and categorical variables, respectively. Associations between Rep1 polymorphism and change in cognitive domains over time were investigated by linear mixed models, adjusted for age, sex, disease duration, education years, and APOE4 status. Un-structured variance-covariance structure method was used to account for repeated measures outcomes. Maximum likelihood approach was used as the estimation method for covariance parameters. Statistical significance was set at p < 0.05, and all tests were two-sided.
RESULTS
Two hundred and four early PD patients were included in the study. Patients’ characteristics are shown in Table 1. There was no significant difference in age, sex, disease duration and education between the two groups. At baseline, both short and long Rep1 allele carriers showed no significant difference in performance on global cognition (MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment) and on tests of individual cognitive domains (Table 1).
Baseline demographics and clinical characteristics of PD patients
*Continuous variables reported as mean±standard deviation; Categorical variable reported as frequency (%). **Two-sided two-sample t test or Mann-Whitney U test (depends if normality assumption is tenable) for continuous variables; Chi-squared test for categorical variable. PD, Parkinson’s disease; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; H&Y stage, Hoehn and Yahr stage; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment.
At 3-year follow-up the long Rep1 carrier group showed significant decline in tests of executive function compared to the short allele group (Fig. 1), adjusted for age, gender, disease duration, education and APOE4 carrier status. There was no significant decline in other cognitive domains in both groups at 3 years (Supplementary Table 1). After additional adjustment for multiple testing for the 5 cognitive domains, the association between Rep1 and decline in executive function was not significant.
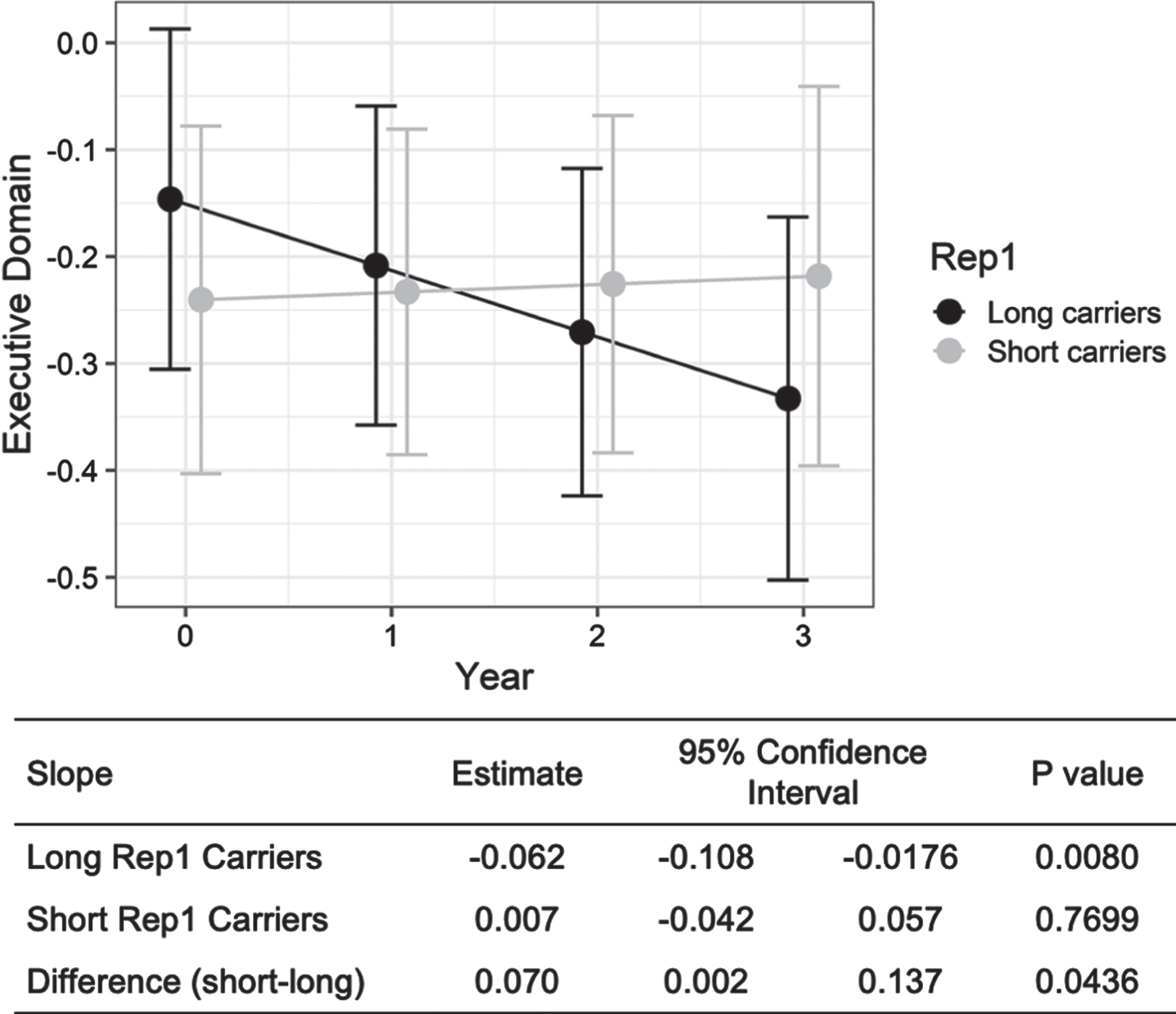
Long Rep1 carriers showed significant cognitive decline in executive function over 3 years. Results are generated using linear mixed model analysis, controlling for age, sex, disease duration, education and APOE4 status. The estimate (and 95% confidence interval) for “Long Rep1 Carriers” and “Short Rep1 Carriers” represent the slope of the regression line on the outcome over time and the corresponding p value indicates whether such slopes are significantly different from zero. Difference (short-long) shows the difference in slopes between “Long Rep1 Carriers” and “Short Rep1 Carriers” over time, and the corresponding p value indicates whether such difference is significantly different between the long and short carriers.
DISCUSSION
Overall, we report that in our study of patients with early PD, long Rep1 allele carriers demonstrated greater decline in executive function approximately 4 years after disease onset compared to short allele carriers. There was no significant difference in performance on memory, attention or visuospatial tests between both groups at baseline and at 3 years. Although the result in executive function decline did not survive multiple testing correction, it remains a clinically important and meaningful finding, given that cognitive impairment is common in newly diagnosed PD, with deficits being most prominent in the executive function domain [22]. Both groups had a mean disease duration of 1 year and had been followed up for a 3-year period after study recruitment.
PD patients with mild cognitive impairment (PD-MCI) show predominant impairment in executive function and attention, and executive dysfunction has been identified as a risk factor for PD [23, 24], particularly in the prodromal stage [25]. ‘Executive function’ encompasses several cognitive abilities, including problem-solving, planning/sequencing, rule-shifting/maintenance, task-switching, manipulation in working memory and response inhibition [26, 27] and worsens with disease progression [28], resulting in difficulties with concentration, retaining information, planning and organizational skills that interfere with social and occupational function [29].
Executive dysfunction in PD has been linked to disruption of the fronto-striatal dopamine network, degeneration in the mesocortical dopamine network, and disruptions in non-dopaminergic brain networks [30]. The prefrontal cortices are implicated in executive function and distinct areas of the prefrontal cortex have strong functional connections with the striatum via dopamine-dependent cortico-striatal loops [31]. Functional MRI imaging in PD has linked impairments in set shifting and working memory to hypo-activation within fronto-striatal loops connecting dorsolateral and ventrolateral prefrontal cortices, striatum and thalamus [32, 33], with striatal dopaminergic depletion disrupting transmission in the fronto-striatal network and contributing to executive dysfunction [34]. Additionally, dopamine release from the mesocortical dopamine network modulates prefrontal D2 receptors and facilitates cognitive flexibility, a core feature of executive processing [35], with evidence for a concurrent dysfunction in the mesocortical dopamine network and disruption of projections to the insular cortex contributing to executive impairment and PDD [30]. Apart from dopaminergic networks, the noradrenergic network projecting from the locus coeruleus to the thalamus, amygdala, and cortex is also affected in PD, with the extent of neuronal loss correlating with development of PDD [36].
Neurodegeneration in PD is characterized by the neuronal accumulation of alpha-synuclein, a protein encoded by the SNCA gene, in Lewy bodies that remain the pathological hallmark of PD. The SNCA-Rep1 promoter region regulates gene expression by acting as a negative modulator of SNCA transcription [37]. Alpha-synuclein levels in blood and human post-mortem brain tissue have been found to be lower in shorter Rep1 allele carriers (259/259 bp) compared to longer genotypes of 261/261, 259/261, and 259/263 [38, 39], whilst mice transgenic for the entire human SNCA locus showed increased SNCA mRNA and protein levels in homozygotes for the longer Rep1 allele compared with homozygotes for the shorter allele [3], with deletion of Rep1 significantly reducing human SNCA mRNA and protein levels [3]. Long SNCA-Rep1 alleles were also shown to associate with hypomethylation of the CpG-sites in intron 1 of the SNCA gene [40], suggesting associated increased transcription of the appropriate mRNA and enhanced gene expression with hypomethylation, thereby contributing to neuronal alpha-synuclein accumulation. Together, these studies suggest that longer Rep1 alleles may enhance SNCA transcription and that our findings of greater executive function decline in long Rep1 allele carriers can be explained by evidence for the strong biological effect of longer Rep1 alleles increasing SNCA gene expression, contributing to neurodegeneration in neuroanatomical regions related to dopaminergic, mesocortical, and non-dopaminergic networks subserving executive function as described above.
A limitation of this study is the lack of biomarker and neuroimaging correlates, which will be included in our future work. This study, however, has significant strengths including comprehensive neuropsychological testing in a good-sized cohort of early PD patients, and is the first study reporting the relationship between Rep1 polymorphism and longitudinal performance on individual cognitive domains. Although the results of executive function did not survive multiple testing correction, the significant decline in executive function in this cohort of early PD with long Rep1 carriers is important and should not be ignored, given that executive dysfunction is known to be one of the earliest cognitive domains impaired in PD. It is important to detect patients at risk for cognitive decline, to allow for earlier intervention and closer monitoring, which will help in the development of strategies to delay progression. These findings suggest SNCA-Rep1 as a potential marker identifying patients at risk of cognitive decline and warrant further study in larger longitudinal cohorts in combination with other biomarkers.
Footnotes
ACKNOWLEDGMENTS
The authors thank our patients and families for their valuable contribution to the study. This study was funded by Singapore’s National Medical Research Council (ASLN by the Clinician-Scientist Transition Award (MOH-TA18may-0003), EKT and LCST by the Open Fund Large Collaborative Grant (MOH-OFLCG18May-0002)).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
