Abstract
Parkinson’s disease (PD) is in some cases predisposed-or-caused by genetic variants, contributing to the expression of different phenotypes. Regardless of etiology, as the disease progresses, motor fluctuations and/or levodopa-induced dyskinesias limit the benefit of pharmacotherapy. Device-aided therapies are good alternatives in advanced disease, including deep brain stimulation (DBS), levodopa-carbidopa intestinal gel, and continuous subcutaneous infusion of apomorphine. Candidate selection and timing are critical for the success of such therapies. Genetic screening in DBS cohorts has shown a higher proportion of mutation carriers than in general cohorts, suggesting that genetic factors may influence candidacy for advanced therapies. The response of monogenic PD to device therapies is not well established, and the contribution of genetic information to decision-making is still a matter of debate. The limited evidence regarding gene-dependent response to device-aided therapies is reviewed here. An accurate understanding of the adequacy and responses of different mutation carriers to device-aided therapies requires the development of specific studies with long-term monitoring.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative condition after Alzheimer’s disease and is the fastest-growing neurodegenerative disorder, with a projected prevalence of 12 million by 2040 [1]. The incidence ranges from 5 to 25 annual cases per 100,000, with the mean age of onset in the seventh decade [2].
There is still a knowledge gap in our understanding of the molecular basis for neurodegeneration in PD. Several environmental and genetic risk factors have been identified, including rare monogenic disorders [3].
A broad genotype-phenotype correlation can be recognized for certain variants [4]. Besides, monogenic PD may benefit from gene-specific treatment strategies (i.e.,
Surprisingly, genetic screening has demonstrated an overrepresentation of specific genetic variants in DBS cohorts (up to 29%) compared to the overall PD population (estimated at 5–10%).
This association raises whether individuals with certain genetic variants represent better candidates for specific device-aided therapies and whether these genetic factors affect the response to such therapies. We suggest reading a previous publication addressing the phenotype-genotype relationship in decision-making on device-aided therapies [10]. The latter issue, how genetic factors affect treatment response, is addressed below.
DEVICE-AIDED THERAPIES
The success of device-aided therapies depends on selecting the suitable device for the right patient. Key eligibility features include: (I) ≥1 hour of troublesome dyskinesia per day, or≥2 hours “off” symptoms per day and need to take levodopa≥5-times per day; (II) no more than mild dementia and absence of troublesome hallucinations; and (III) significant difficulty with activities of daily living. Patients demonstrating good levodopa response who are emotionally stable, physically healthy, cognitively intact, and younger (preferably < 70 years of age) are ideal candidates for CSAI, LCIG, or DBS [11]. While these therapies seem to provide a similar improvement in reducing “off” time by around 40–60%, their effects on dyskinesia and non-motor symptoms are heterogeneous, and their side effects and complications can be quite different [12]. Table 1 summarizes their main indications, advantages, disadvantages, and contraindications.
Comparison between different device-aided therapies
DBS, deep brain stimulation; LCIG, Levodopa-carbidopa intestinal gel; CASI, continual apomorphine subcutaneous infusion; iPD, idiopathic Parkinson’s disease; NMS, non motor symptoms; PEJ, percutaneous endoscopic Jejunostomy; AE, adverse effects; ICD, impulse control disorders; LEDD, Levodopa equivalent daily dose.
In terms of selecting which device therapy is most appropriate, DBS is favored with younger patients and minimal non-levodopa-responsive motor symptoms (except for tremor) [13, 14]. DBS may be contraindicated if there is dementia, hallucinations, uncontrolled depression, marked postural and gait problems, severe brain atrophy, or suspected atypical parkinsonism.
LCIG can still be considered for patients with mild-to-moderate dementia or age > 70 years, even with severe depression [15]. However, patients with dopamine dysregulation, punding, or pre-existent peripheral neuropathies may be less favorable candidates.
CSAI can be considered if there are mild hallucinations or moderate cognitive impairment. Moreover, it might improve neuropsychological performance [16, 17]. In addition, this device may ameliorate depression, apathy, “off” pain, and slowness of thinking [18]. However, it seems less favorable in patients with impulse control disorders (ICD), marked psychosis, daytime somnolence, and troublesome orthostatic hypotension [19].
We caution against dogmatism since there is also favorable evidence for device therapies in some of these reported contraindications. For example, there are cases where LCIG and CSAI improve ICD, and where LCIG decreases dopamine dysregulation syndrome [20–22]
A relevant factor in decision-making for device-aided therapies is the long-term outcome expectancy. Chronic STN-DBS can cause dysarthric speech, problems in verbal fluency, worsening freezing of gait, and axial symptoms. These are important determinants of quality of life [23–25]. Severe pre-operative gait difficulties might predict limited long-term DBS benefits [23, 24]. Moreover, motor outcomes one year after bilateral STN-DBS are inversely correlated with the rate of progression of motor symptoms [26].
The leading causes of discontinuation of LCIG therapy in long-term follow-up include worsening cognition, dyskinesias, chronic polyneuropathy, weight loss, and hallucinations. Eventually, LCIG may become ineffective [27, 28].
Like LCIG, long-term CSAI may worsen cognition, dyskinesias, postural instability, and hallucinations. CSAI also causes sedation and orthostatic hypotension. These factors may obscure the long-term benefits in some patients [29–32]. Furthermore, a decrease in therapeutic effect may become an important reason for discontinuation within the first four years [33].
In conclusion, it is essential to recognize patient heterogeneity and, if possible, identify biomarkers of short and long-term outcomes. Understanding phenotype-genotype relationships and how variants predict the risk of significant disease milestones [34] may affect the timing, appropriateness, expected outcome, and expectations for device-aided therapies [10].
CLINICAL FEATURES OF CAUSAL MUTATIONS AND GENETIC RISK FACTORS FOR PARKINSONISM
Most PD cases are sporadic, associated with genetic, epigenetic, and environmental risk factors [35]. The most frequent genetic risk factors for sporadic PD are
The severity and risk associated with
Autosomal dominant (AD) PD includes
Autosomal recessive (AR) PD, such as
On the other hand,
Because of scarce evidence, how these causal or risk-modifying variants affect outcomes is debatable. Currently, decision-making for advanced therapies is based on clinical features, which are unreliable for inferring the underlying genetics. Incomplete penetrance (i.e.,
CURRENT EVIDENCE FOR DEEP BRAIN STIMULATION IN MONOGENIC PARKINSONISM AND GBA VARIANTS CARRIERS
Pal et al. analyzed the Consortium On Risk for Early-onset PD (CORE-PD) cohort, emphasizing
Available evidence of outcomes for different device-aided therapies in GBA variants carriers
LEDD, levodopa equivalent daily dose.
Available evidence of outcomes for different device-aided therapies in autosomal dominant monogenic parkinsonism
NR, not reported; LEDD, levodopa equivalent daily dose.
Available evidence of outcomes for different device-aided therapies in monogenic autosomal recessive parkinsonism
NR, not reported; LEDD, levodopa equivalent daily dose.
Available evidence of outcomes for different device-aided therapies in monogenic autosomal recessive parkinsonism presenting with atypical feature
NR, not reported; LEDD, levodopa equivalent daily dose.
Various authors have proposed different categories for motor outcomes. In their systematic review, de Oliveira et al. defined a mean UPDRS-III change of 50% or more as a marked response, a mean change of 30% to 50% as a satisfactory response, and less than 30% change as an unsatisfactory response [79]; on the other hand, Kuusimäki et al., defined an improvement of 30% or more in the UPDRS-III score as a favorable outcome; 20–30% a moderate outcome; and < 20% a poor/mild result [80].
DBS in carriers of genetic variants that modify the risk for developing PD or influence PD-related outcomes (GBA)
A recent study screening for
Modeling different datasets, Pal et al. examined global cognition using the Mattis Dementia Rating Scale to compare the rate of change between
Finally, the GPi target may be preferable for
Both GPi-DBS and STN-DBS have similar outcomes on motor function measured by the UPDRS-III in the “on” and “off” medication state [84–86], and both targets have a beneficial effect on levodopa-induced dyskinesias [87]. STN-DBS achieves this goal mainly by a greater reduction in medication dosages [87, 88]; but also, stimulation of the area above the STN can directly suppress levodopa-induced “on”-dyskinesia [86]. In contrast, GPi-DBS may provide greater anti-dyskinetic effects possibly by a direct mechanism [84, 87]. Hence, clinical guidelines recommend GPi as the target, especially when reduction of medication is not anticipated, and there is a goal to reduce the severity of “on” medication dyskinesias [84, 89].
On the other hand, although it seems relatively safe concerning cognitive function, chronic stimulation of STN has been associated with a subtle decline in cognitive domains, exceptionally verbal fluency, and executive function [90, 91]. Despite little data is supporting that STN-DBS has a worse cognitive outcome than GPi-DBS [92], more published information is required for validation [93]; if there is significant concern about cognitive decline, particularly regarding verbal fluency, processing speed, and working memory in a patient undergoing DBS, GPi has been recommended [84, 89].
DBS in autosomal dominant PD (SNCA, LRRK2, VPS35)
A systematic review showed that
Thus far in the literature, five individuals carrying a
In a meta-analysis of
In summary, outcomes appear favorable for the most common
DBS in autosomal recessive PD (PRKN, PINK-1, DJ1)
DBS in autosomal recessive parkinsonism with atypical features
Bilateral GPi-DBS and ventralis intermediate nucleus (Vim)-DBS has been successfully utilized for dystonic storm treatment in a 15-year-old girl with atypical neuroaxonal dystrophy (NAD) phenotype, a subgroup of
A female carrier of a
In a cohort of early-onset sporadic or familial PD, a 46-year-old homozygous
A Caucasian woman with parkinsonism since age 39 had severe dyskinesias under dopaminergic treatment, dysarthria, tremor, mild dementia, hallucinations, dystonia, gait, and gastrointestinal tract problems. She had compound heterozygous canonical splice-site variants in
A Persian male bearing a p.R449Q heterozygous mutation in
EVIDENCE ON THE USE OF LEVODOPA CARBIDOPA INTESTINAL GEL
Autosomal dominant PD and GBA mutations
In a cohort of 12 PD patients on LCIG in the UK, the authors reported one patient with
Autosomal recessive PD with homogeneous presentations
A juvenile PD patient carrying a
A woman with homozygous
EVIDENCE FOR CONTINUOUS APOMORPHINE SUBCUTANEOUS INFUSION
Autosomal dominant PD
In a case series of British
Autosomal recessive PD with homogeneous presentations
Two
Autosomal recessive parkinsonism presenting with atypical features
A Turkish woman with homozygous
As mentioned above, a female
DISCUSSION
The cumulative evidence for device-aided therapies in monogenic-PD and
Along with a regional difference in the prevalence of specific variants, the availability of advanced therapies is critical. Device-aided therapies offered in different countries may vary through healthcare systems, local experience, and center preferences. For instance, we have observed that publications on infusion therapies (i.e., LCIG and CSAI) in monogenic parkinsonism come predominantly from the UK and Middle Eastern countries (Israel, Saudi Arabia, and Turkey), and LCIG in GBA-PD from Israel. On the other hand, DBS-related publications are more widely distributed (i.e., North America, Europe, Middle East, Asia, South America, Australia), possibly because of increasing access to this therapy. Unfortunately, decision-making on device selection is not explicit in most reports from countries where more than one device-aided therapy is available (e.g., Italy, UK, Israel, Turkey, USA). Future reports should explain the selection of a specific device-aided therapy, especially when other alternatives are available.
Systematic reviews and a meta-analysis constitute the best evidence for DBS in monogenic PD. However, these are limited by small sample size, short follow-up, and incomplete data.
Moreover, several investigators have used different categories and cut-off values when defining DBS responses in gene-related PD populations; because of this heterogeneity, the same percentage of change in UPDRS-III would be qualified differently by distinct authors. An explicit limitation of this approach is the lack of consensus, adding difficulties when interpreting the literature. In addition to arbitrariness in establishing cut-off values, the effectiveness of these therapies has been firmly focused on the change in UPDRS-III scores, in our opinion lacking adequate emphasis on non-motor symptoms or changes in quality of life, which can be decisive in decision-making and in establishing the benefits of these therapies. Further, with some exceptions, reports on LCIG or CSAI lack objective and detailed results making a similar analysis difficult.
Mutation carriers seem to be overrepresented in DBS-cohorts compared to non-carrier PD populations.
This should be kept in mind when discussing prognosis, timing, and expectations for DBS.
Publications on DBS in autosomal recessive variants with atypical features are mainly limited to individual cases. Some patients have reported benefits, but outcomes are incompletely reported, and long-term data is scarce. Dysarthric speech, swallowing disturbances, freezing of gait, and balance problems are frequent features of atypical autosomal recessive parkinsonism (e.g.,
Small genetic screening studies are the primary source of evidence for LCIG. There is no significant difference in motor outcomes between
CSAI has the most limited evidence of the three therapies in monogenic PD and
The available information regarding individual monogenic variants and device-aided therapies is far from comprehensive. The data are limited to small numbers of patients, short follow-ups, and observational reports. Multicenter prospective cohort studies are needed to guide our knowledge and improve decision-making for device-aided therapies and PD-related variants.
In addition, we recommend that several key elements be included when reporting outcomes from device-aided therapy amongst genetic PD populations (Fig. 1).
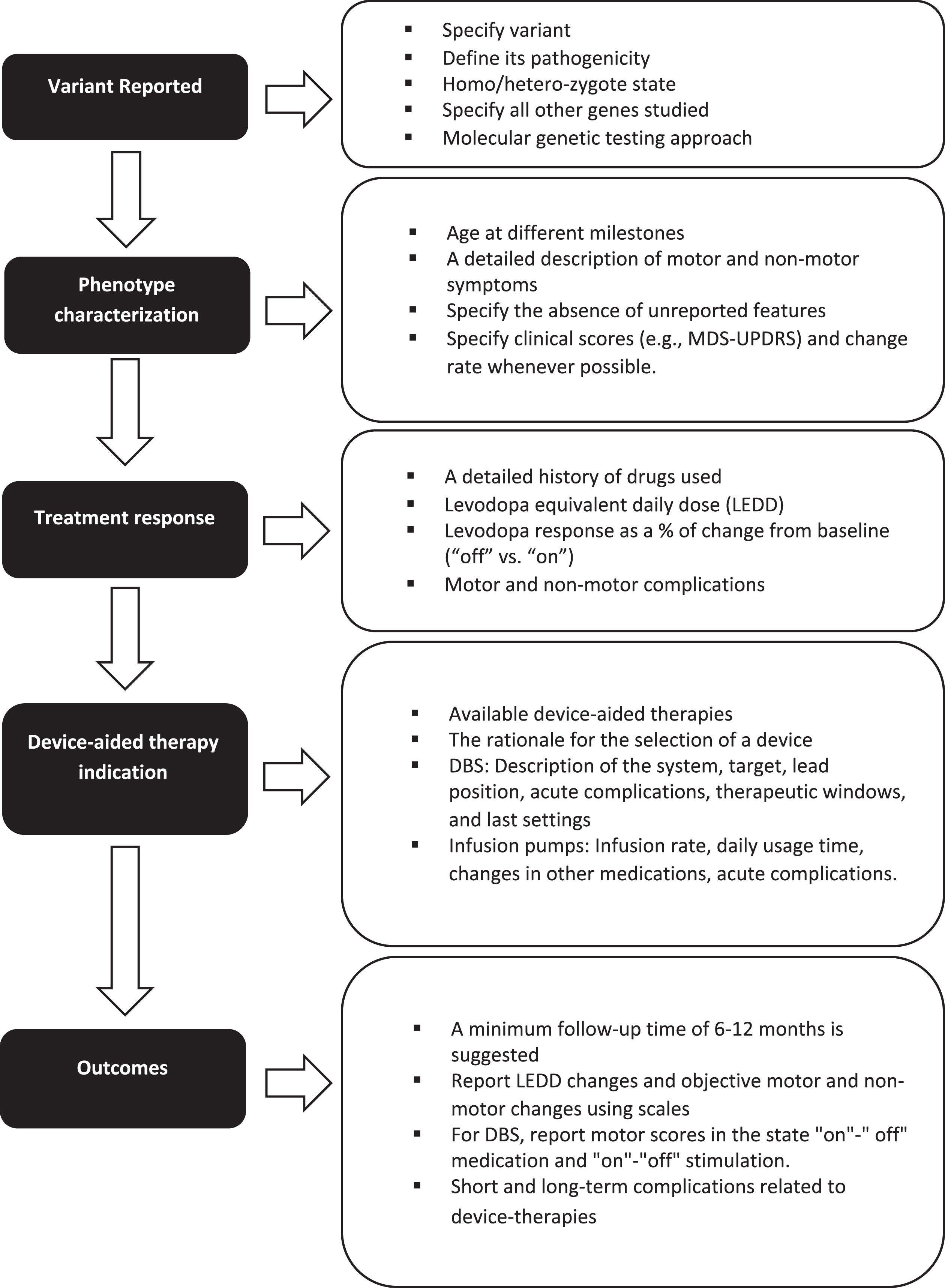
Key elements to consider when reporting the response to device-aided therapies in patients with monogenic parkinsonism or GBA variants carriers.
First, when discussing the genetic variant, the type of variant, its pathogenicity, and its homozygote or heterozygote state should be included. When using panels, all the genes studied should be mentioned, especially in patients belonging to ethnicities at risk for more than one type of variant (for example,
Second, when discussing phenotype, characterization must be rigorous, including the age at symptom onset, age at diagnosis, disease duration, initial clinical manifestation, presence of falls, freezing of gait, cognitive profile, neuropsychiatric manifestations, and other non-motor symptoms. The absence of unreported features should be specified. Individuals may be classified according to the MDS-UPDRS-III score (i.e., tremor dominant, intermediate, or postural instability/gait difficulty). Levodopa response should be described as % of change from baseline. Whenever possible, the rate of progression of motor and non-motor symptoms (i.e., cognitive decline) in the pre- and post-device-aided therapy stage should be included. A detailed history of the drugs used, related side effects and levodopa equivalent daily dose should also be included.
Third, the indication and rationale for each specific advanced device-aided therapy should be documented. In addition, for DBS, it is essential to define whether the surgery is uni- or bilateral, which commercial device was implanted, the target, lead position information, therapeutic window, and final stimulation parameters. The infusion rate, daily usage time, and changes in other medications should be indicated for infusion pumps.
Finally, long-term motor and non-motor outcomes should be measured objectively using, for example, the MDS-UPDRS scale administered at multiple time points. In the case of DBS, it is essential to report motor scores in the state “on"-” off” medication and “on"-“off” stimulation. Follow-up time should be sufficient for the device settings to reach a steady-state and assess disease progression, treatment efficacy, and long-term adverse effects. While there is no specific time, a reasonable minimum follow-up time would be greater than 6–12 months.
CONCLUSION
Based on current studies, it is unfeasible to establish evidence-based decision-making guidelines for device-aided therapies in monogenic parkinsonism. So far, an added prognostic value of genetic testing beyond a careful clinical assessment when patients are evaluated for device-aided therapies is yet to be demonstrated for monogenic parkinsonism. Large prospective cohorts combining genetic profiling with deep phenotyping, and randomized studies, can provide relevant data to address this question.
Although no randomized trials are available, based on accumulated evidence on the natural history and probable deleterious cognitive outcomes after STN-DBS in carriers of pathogenic variants in
We call for the development of guidelines that allow us to improve the quality and number of reports and randomized clinical studies that optimize our decision-making on device-aided therapies in monogenic parkinsonism and
CONFLICT OF INTEREST
