Abstract
Monoamine oxidase-B (MAO-B) inhibitors are commonly used for the symptomatic treatment of Parkinson’s disease (PD). MAO-B inhibitor monotherapy has been shown to be effective and safe for the treatment of early-stage PD, while MAO-B inhibitors as adjuvant drugs have been widely applied for the treatment of the advanced stages of the illness. MAO-B inhibitors can effectively improve patients’ motor and non-motor symptoms, reduce “OFF” time, and may potentially prevent/delay disease progression. In this review, we discuss the effects of MAO-B inhibitors on motor and non-motor symptoms in PD patients, their mechanism of action, and the future development of MAO-B inhibitor therapy.
INTRODUCTION
Parkinson’s disease (PD) is a common chronic neurodegenerative disease, characterized by abnormal formation of Lewy neurites and (or) Lewy bodies with intraneuronal aggregates of proteins (mainly α-synuclein) spreading from medulla oblongata to pontine tegmentum, midbrain, mesocortex, and neocortex in an ascending pattern, and a decreased number of dopaminergic neurons in substantia nigra (SN) leading to deficiency of dopamine [1–3]. There are approximately 6.1 million PD patients worldwide, and the prevalence increases with age, reaching 1% to 3% in the population over 65 years of age [4, 5]. Common motor symptoms of PD are bradykinesia, resting tremor, rigidity, postural instability, and gait disorder, which are often accompanied by various non-motor symptoms such as sleep disturbance, autonomic dysfunction, depression, and fatigue that can severely affect daily life [6, 7]. Although the disease cannot be cured, therapeutic strategies have been developed, including drug treatment, surgical techniques, and rehabilitation, which can alleviate symptoms and might have the potential to slow disease progression [8].
Levodopa is considered the most effective symptomatic treatment for PD and is required at some point in the illness by almost all patients [8, 9]. Nevertheless, the long-term use of levodopa can induce motor complications such as motor fluctuations (‘wearing off’; ‘on-off’) and dyskinesia [10]. Dopamine agonists (DAs) or enzyme inhibitors, including catechol-O-methyl-transferase (COMT) inhibitors and monoamine oxidase B (MAO-B) inhibitors, are commonly applied as adjuvants to levodopa to alleviate motor complications. Commonly utilized COMT inhibitors, such as entacapone and opicapone, inhibit levodopa degradation in the periphery while MAO-B inhibitors penetrate the blood-brain barrier, inhibit central MAO activity in the CNS and so reduce dopamine degradation [10]. MAO-B inhibitors have been shown to have excellent efficacy and safety during the early stages of PD as well as when used as adjunctive therapy in advanced illness [11–13]. Clinical studies have shown that a longer duration of MAO-B inhibitor exposure was associated with a reduction in levodopa consumption and a slower rate of clinical decline [14, 15]. Currently approved MAO-B inhibitors include the irreversible inhibitors, such as selegiline and rasagiline, and the reversible inhibitor, safinamide [10–16]. In this review, we will discuss the effect of MAO-B inhibitors on motor and non-motor symptoms of PD, their mechanism of action and the future development of MAO-B inhibitors.
MECHANISM OF ACTION OF MAO-B INHIBITORS
MAO-B inhibitors regulate the MAO-B activity
Monoamine oxidase (MAO) is a riboflavin protein distributed on the outer membrane of mitochondria. It catalyzes the oxidative deamination of tyramine and monoamine neurotransmitters such as dopamine, phenethylamine, 5-hydroxytryptamine, and norepinephrine [17, 18]. There are two MAO isoenzymes: MAO-A and MAO-B. MAO-A is mainly distributed in the gastrointestinal tract, platelets, and heart, and can promote the metabolism of tyramine-containing substances in food so avoiding hypertensive crises caused by the accumulation of tyramine (“cheese reaction”). MAO-A also exists in catecholaminergic neurons, such as dopaminergic neurons in SN, norepinephrine neurons in locus coeruleus, etc. [18]. MAO-B is mainly distributed in platelets and glial cells, and total MAO activity within the brain is composed of approximately 20% MAO-A and 80% MAO-B [19–22]. Both MAO-A and MAO-B regulate the amine neurotransmitters, including dopamine. MAO-A metabolizes dopamine in presynaptic neurons, while MAO-B metabolizes dopamine released to synaptic cleft and taken up by glial cells. The number of glial cells was shown to increase with age, and in neurodegenerative diseases, as expected, the activity of MAO-B also increased [23–25]. MAO-B inhibitors inhibit MAO-B activity in the brain, block dopamine catabolism, enhance dopamine signaling, and selectively enhance dopamine levels at synaptic cleft [21]. Preclinical studies have shown that rasagiline, a second-generation MAO-B inhibitor, is 3–15 times more potent than selegiline in
MAO-B inhibitors may have disease modifying effects
For PD patients, disease modification is defined as the ability of a given agent to slow, arrest, or rescue the process of nigral dopaminergic cell death, and thus slow clinical progression and deterioration [22, 34]. MAO-B metabolism of dopamine may lead to the formation of toxic metabolites, such as hydrogen peroxide and dihydroxyacetaldehyde, thus MAO-B inhibitors may protect neurons by inhibiting these toxic processes [22]. Both selegiline and rasagiline have neuroprotective effects related to their propargylamine structure which is independent of their inhibition on MAO-B [35–37]. Selegiline and rasagiline can increase the expression level of nerve growth factor, glial cell-derived neurotrophic factor, brain-derived neurotrophic factor, and other nerve growth factors, therefore exerting a neuroprotective role [16]. Rasagiline can inhibit apoptosis by directly or indirectly activating anti-apoptotic factors such as Bcl-2 and Bcl-xL and inhibiting the expression of proapoptotic factors such as Bax [38, 39]. Rasagiline may have a neuroprotective advantage over selegiline due to a different pathway of metabolism. For example, both selegiline and rasagiline can reduce oxygen-glucose deprivation-induced cell death in PC12 cell lines. However, L-methamphetamine, a metabolite of selegiline, not aminoindan, the major metabolite of rasagiline, enhanced oxygen-glucose deprivation-induced cell death by 70% [37]. In addition, brain SPECT scans showed that rasagiline had a potential neuroprotective effect by stabilizing regional cerebral blood flow perfusion in patients with PD [40]. Unlike selegiline and rasagiline, safinamide blocks sodium and calcium channels, regulates glutamate release, and reduces excitotoxicity to dopaminergic neurons [30]. Animal studies have shown that safinamide inhibits microglial cell activation and protects dopaminergic neurons from degeneration [41].
Effects of MAO-B inhibitors on mitochondria and cell apoptosis
Mitochondrial dysfunction occurs in PD characterized by decreased activity of mitochondrial complex I and increased reactive oxygen species production [42].
Effects of MAO-B inhibitors on alpha-synuclein aggregation
Alpha-synuclein is a major component of Lewy bodies.
PD SYMPTOMS TREATED BY MAO-B INHIBITORS
PD-related symptoms treated with MAO-B inhibitors are detailed in Table 1.
PD-related symptoms covered by this review and selection of corresponding MAO-B inhibitors
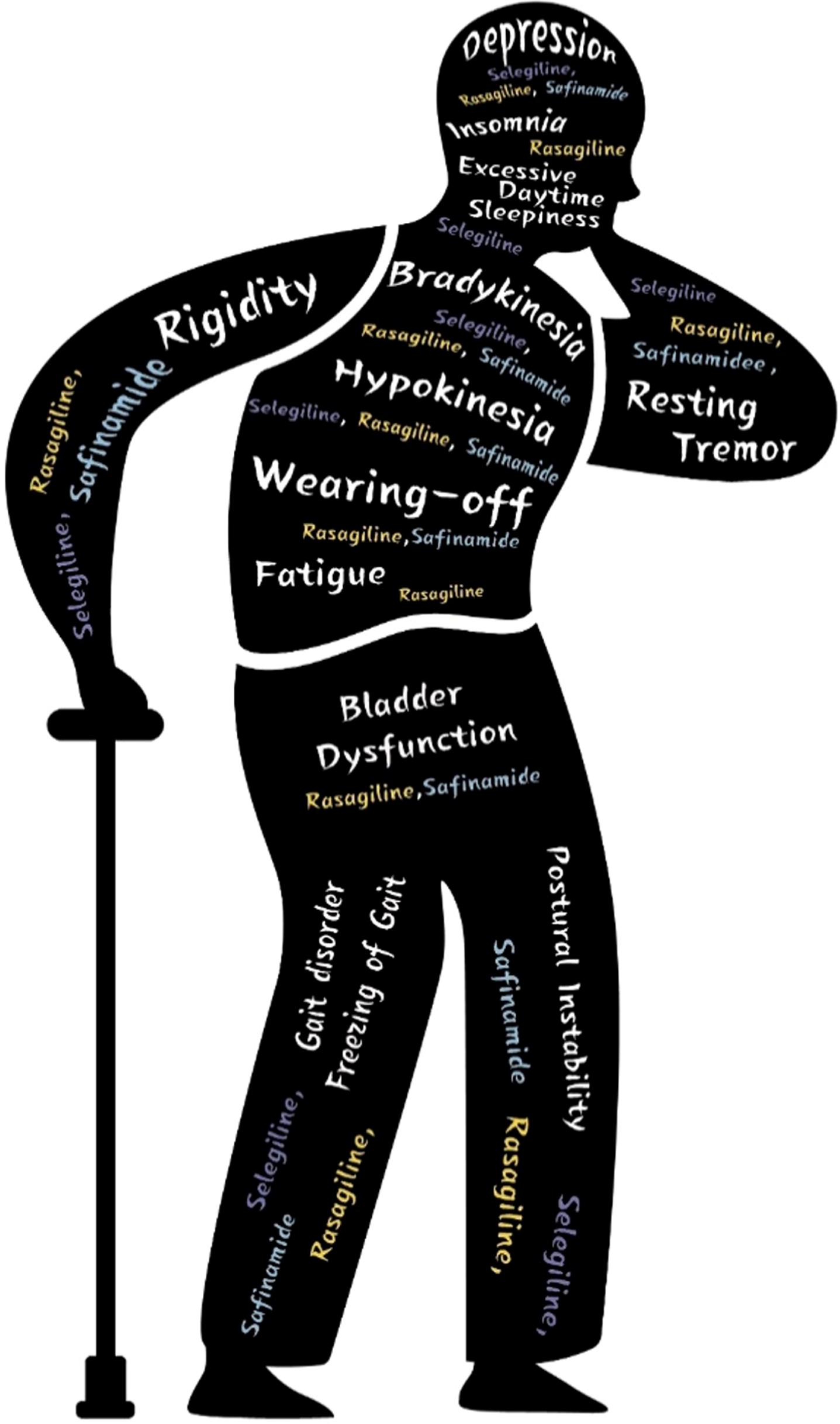
Selection of MAO-B inhibitors according to PD related symptoms.
Improvement of motor symptoms
According to the Guidelines for Adult Parkinson’s Disease published by National Institute for Health and Care Excellence (NICE) in 2017 [9], MAO-B inhibitors are recommended as first-line therapies for patients with early PD whose motor symptoms are not affecting their quality of life. For PD patients with dyskinesia or motor fluctuations, MAO-B inhibitors may be selected as adjuncts to levodopa. Treatments for the Motor Symptoms of PD from the International Parkinson and Movement Disorder Society Evidence-Based Medicine Review in 2018 [49] recommend the use of MAO-B inhibitor selegiline and rasagiline as effective monotherapy for early PD. Yet, it seems that rasagiline is clinically effective in adjunct to DA therapy for early/stable PD, but safinamide is ineffective as an adjunct to DA. Both rasagiline and safinamide are effective in the treatment of motor fluctuations, and there is insufficient evidence for selegiline. 2020 Chinese guidelines for the Treatment of Parkinson’s disease (fourth edition) states that MAO-B inhibitors can be used in early PD patients and add-on therapy for advanced PD patients and that rasagiline is a better choice compared to selegiline in improving motor complications [50].
Monotherapy in early PD (first-line therapy)
The PD-MED study [51] was a long-term, large, pragmatic, randomized controlled study (real-world study) that compared the long-term control of symptoms and improvement in the quality of life of patients with early PD treated with levodopa, DAs, or MAO-B inhibitors. For levodopa-sparing therapy, participants treated with MAO-B inhibitors were significantly more likely to discontinue their allocated drug class than those allocated DAs (72% vs. 50%). Discontinuation was mainly due to side effects, and a small group was due to an absence of efficacy. However, after exposure to levodopa, initial treatment with MAO-B inhibitors showed small but significantly better benefits in PDQ-39 mobility scores (1.4 points better), cognition, PDQ-39 summary index (0.8 points better) but not the quality of life from EuroQol EQ-5D scale compared with initial treatment with DAs. Rates of dyskinesia were similar between the MAO-B inhibitors group and the DAs group, but motor fluctuations were higher in the DAs group. A possible explanation for better efficacy of MAO-B inhibitor with lower levodopa equivalent doses (LED) compared with DAs (695 mg/day vs. 768 mg/day) might be that levodopa could be less effective when added to DAs than MAO-B inhibitors, which needs to be further validated [51]. DAs are generally considered to be more effective than MAO-B inhibitors, but according to the PD MED study, MAO-B inhibitors as initial levodopa-sparing therapy were at least as effective as DAs [51].
The 2009 ADAGIO study [57] evaluated the disease-modifying effect of rasagiline monotherapy in patients with early treatment-naive PD. Three stratified primary endpoints based on UPDRS were set to determine the disease-modifying effect of 1 mg/2 mg rasagiline by a study design of early-start treatment with rasagiline for 72 weeks and delay-start treatment with placebo for 36 weeks followed by 1 mg/2 mg rasagiline till 72 weeks. The first endpoint was that rate of total UPDRS change in 1 mg/2 mg rasagiline group had superiority to placebo during 12-36 weeks treatment; the second endpoint was a better change of UPDRS score between baseline and 72 weeks in the early-start treatment than delayed treatment groups; the third endpoint was that rate of UPDRS change during 48 to 72 weeks in the early-start treatment was noninferiority to delayed treatment groups. The study results showed that the 1 mg rasagiline group met all 3 stratified endpoints but not the 2 mg rasagiline group. Between 12 and 36 weeks of treatment, rates of UPDRS scores changes per week were significantly smaller in the 1 mg and 2 mg rasagiline groups compared with placebo group (0.09±0.02 vs. 0.14±0.01,
The CHORUS study [58] published in 2018 evaluated the efficacy of rasagiline in Chinese patients with early PD. A total of 127 patients with early treatment-naive PD were included in this randomized controlled study. The results showed that the decrease in total UPDRS scores was significantly greater in the 1 mg/day rasagiline group than the placebo group (–3.18±0.95 vs. –0.18±0.98,
Symptomatic adjunct therapy in early or stable PD
In patients with early or stable PD, MAO-B inhibitors could relieve symptoms and reduce levodopa dosage and might have beneficial effects on neuroprotection which still need investigation because of a lack of sufficient evidence.
Some studies have explored the feasibility of selegiline combined with DAs. In 1991, Nappi et al. studied the effects of selegiline added on lisuride therapy and found that mean lisuride dosage could be reduced without deterioration of the clinical effects [64]. In 1995, Calzetti et al studied that L-deprenyl (10 mg daily) as an adjunct to low-dose bromocriptine (up to 25 mg daily) compared with bromocriptine alone or bromocriptine plus placebo and found that selegiline combined regimens produced a mildly significant improvement in the majority of clinical rating scales, a stabilization of clinical status up to 12 months, and a smaller proportion of patients in need of levodopa at 24 months [65]. However, both lisuride and bromocriptine are ergot-related dopamine agonist, and bromocriptine has the risk of fibrotic cardiac valvulopathies from long-term use. Lyons et al. reported a study of PD patients with levodopa-induced motor fluctuations and pramipexole related adverse events (AEs) of excessive daytime sleepiness, pedal edema, hallucinations and impulse-control disorders, investigating the effect of adding orally disintegrating selegiline on the reduction of DA-related AEs [66]. Results showed that DA-related AEs were reduced or resolved at week 12 in most subjects, while UPDRS activities of daily living scores were improved obviously (
Adjunct therapy in advanced PD
A randomized controlled study from 2013, which investigated the efficacy of rasagiline as adjunctive therapy to levodopa treatment in Chinese patients with PD [75], showed that the mean adjusted total daily “OFF” time shortened more significantly in the rasagiline group than in the placebo group (–1.75±1.69 h vs. –0. 69±1.44 h,
Furthermore, two studies conducted in 2019 in Japanese PD patients who received levodopa and experienced wearing-off phenomena showed that patients treated with rasagiline 1 mg or 0.5 mg for 26 weeks had a 0.84 h and 0.60 h decrease in “OFF” time compared with placebo (
Non-motor symptoms
The non-motor symptoms of PD involve multiple types and may occur at all stages of the disease, particularly sleep disturbance, fatigue symptoms, and neuropsychiatric symptoms.
Sleep disturbance
Approximately half of the newly diagnosed PD patients have subjective sleep discomfort, which is the second most common non-motor symptom in PD [83]. Sleep disturbances of PD patients comprise insomnia, excessive daytime sleepiness, rapid eye movement behavior disorder and sleep-related movement disorders, namely restless legs syndrome, etc. Disease progression, motor complications, dopaminergic medications, severe autonomic dysfunction, presence of hallucination were reported to be associated with sleep disorders for PD patients [84, 85].
Selegiline metabolites amphetamine derivatives have potential sympathomimetic effects and side effects affecting sleep. When combining the AE data from the long-term studies, insomnia was included in the most common AEs [86]. One may assume that amphetamine-like derivatives of selegiline worsen sleep quality supported by the onset of insomnia during chronic selegiline intake in PD patients [87], thus it needs to be taken in the morning and at noon. Selegiline may be tried in patients with excessive daytime sleepiness [50]. Gallazzi et al. reported that 45 PD patients with excessive daytime sleepiness, usually developed after the introduction of DAs, were treated with 10 mg selegiline and the results showed a statistically significant improvement of somnolence but no change in the UPDRS III scores [88].
Rasagiline can improve sleep in PD patients when used as monotherapy or adjunct therapy. A 12-week prospective observational study showed that rasagiline adjunct to levodopa improved sleep outcomes compared with baseline, reduced mean sleep latency compared with levodopa alone (–1.68±1.21 h vs. –0.55±0.69 h,
So far, only a few studies have reported on the effect of safinamide on sleep in PD patients. Some investigators evaluated the effects of safinamide treatment on non-motor symptoms of PD patients [91–94]. Patients treated with safinamide showed a significant reduction of PDSS2 and ESS scores [92]. SAFINONMOTOR study showed 6 months after starting with safinamide, sleep (NMSS-domain 2 and PSQI) was significantly improved in PD patients [93]. The efficacy of safinamide in PD patients with sleep disorders still needs further research and exploration in the future.
Fatigue symptoms
Fatigue is defined as an overwhelming sense of tiredness, lack of energy, and feeling of exhaustion, which is unrelated to physical activity. It is described as one of the most disabling symptoms of PD, occurring in about 50% of patients even at the early stages, negatively impacting the quality of life [95, 96]. A
Depression
Depression is common in PD patients and frequently involves apathy and anhedonia. Disease duration, severity of motor symptoms, occurrence of motor complications, cognitive decline and dementia, anxiety, sleep disturbance, autonomic symptoms, etc., have been linked to increased risk of depression in PD patients [101]. Meta-analysis showed that MAO-B inhibitors could reduce the severity of depression symptoms in PD patients, especially in patients with early PD [102]. A French multicenter, double-blind, randomized, controlled study published in 1991 included a total of 93 PD patients who were randomized to receive either 10 mg/day selegiline or placebo for 3 months [103]. The results showed that the 90-day Hamilton Depression Rating Scale (HDRS) score and mental UPDRS Scale score were more improved in the selegiline group than in the placebo group (
Bladder dysfunction
Bladder dysfunction is one of the most common autonomic dysfunctions in PD patients, with an estimated incidence of 55% –80% [107]. In 2014, a 2-month open-label study evaluated changes in urodynamic parameters in patients with early mild PD before and after treatment with 1 mg/day of rasagiline. The results showed that rasagiline significantly improved bladder function, increased bladder capacity (16%), and first micturition sensation (34%), and reduced residual urine volume (–53%) in PD patients [107]. In 2021, a small retrospective analysis of urological symptoms in PD patients treated with safinamide [108] showed that the total score of Scale for Outcomes in Parkinson’s disease for Autonomic Symptoms-Urinary subscale (SCOPA-AUT-U) and subscale scores for micturition urgency, urinary incontinence, urinary frequency, and nocturia were significantly improved compared to baseline in patients receiving safinamide add-on therapy, suggesting that safinamide may contribute to the improvement of urinary symptoms of PD.
Gait disorder
With disease progression, severe gait disorders can lead to disability and quality of life decline in PD patients, where freezing of gait (FOG) is a common type of gait disorder in PD patients [109]. The prevalence of FOG is 46.62% in Chinese PD patients [110]. Studies suggested that MAO-B inhibitors selegiline and rasagiline can reduce the risk of developing FOG [111]. Analysis of data from 800 patients with early PD in the DATATOP study [112] showed that the application of selegiline could reduce the risk of developing FOG by 53% (
To sum up, MAO-B inhibitors show good efficacy in treating patients with early and advanced PD. Compared with selegiline monotherapy or adjunct therapy mainly used in patients with early or stable PD and safinamide adjunct therapy mainly used in patients with advanced PD, rasagiline had high evidence-based efficacy in the monotherapy and adjunct therapy for early and advanced PD and significantly improved various motor and non-motor symptoms in PD patients. Binde et al. reported a meta-analysis that included 27 trials assessing the most effective MAO-B inhibitors for early or later PDs by evaluating both UPDRS score and serious AEs [116]. They found that all included MAO-B inhibitors were effective compared to placebo when given as monotherapy, while selegiline became more efficient than other MAO-B inhibitors when combined with levodopa, whether or not the duration of the disease was taken into account. In another subsequent meta-analysis of 79 publications, Binde et al. included DAs (cabergoline, pramipexole, ropinirole, rotigotine) and MAO-B inhibitors in the comparison, and interestingly, they found that all the investigated drugs to be effective compared with placebo when given as monotherapy except safinamide, but selegiline remained the best option in combination with levodopa [117]. Due to the lack of exact data from the head-to-head RCTs which have compared one MAO-B inhibitor to another, the conclusions drawn from meta-analyses are still needed to be validated in further trials.
SAFETY OF MAO-B INHIBITORS
The overall conclusions from evidence-based medicine recommendations are that the number of side-effects and the proportion of patients withdrawing from treatment because of poor tolerability are higher with DAs than with MAO-B inhibitors or levodopa in early PD [118]. MAO-B inhibitors have a low frequency of specific adverse events, and DAs are at a higher risk of excessive somnolence, hallucinations, and impulse-control disorders [119]. Regarding monotherapy, one randomized controlled trial suggested that the incidences of treatment-emergent adverse events (TEAEs) leading to withdrawal were slightly lower in the rasagiline group (4.6%) than in placebo-treated individuals (7.7%) [58]. Regarding combination treatments, moderate-quality evidence suggested that COMT inhibitors and DAs significantly increase the rates of AEs compared with placebo [9]. In addition, COMT inhibitors increased the rates of AEs compared with MAO-B inhibitors and DAs. The PD-MED study showed that in PD patients with DAs or MAO-B inhibitors, the rates of dyskinesia were similar (HR 0.85, 95% CI 0.60–1.22,
Selegiline
It has been reported that PD patients treated with selegiline had a risk of “cheese reaction” and toxic effects on the cardiovascular system [120, 121], which was reported leading to increased mortality when selegiline was combined with levodopa [122]. However, this finding of increased mortality caused by selegiline is controversial. No significant increase in mortality was observed in later meta-analysis and good tolerability of MAO-B inhibitors was confirmed. A meta-analysis did not find that excess deaths occurred with MAO-B inhibitors compared with the control arm (20% vs. 21% deaths, OR 1.13, 95% CI [0.94, 1.34],
A study investigating the effects of selegiline in adjunct with levodopa on the cardiovascular system in PD patients showed a significant increase in plasma norepinephrine levels in patients acutely loaded with selegiline in adjunct with levodopa compared with levodopa monotherapy and healthy controls (
Rasagiline
Rasagiline is well tolerated both in monotherapy and in combination with levodopa, with nasopharyngitis, fall, eczema, headache, and contusion being the most frequently reported adverse effects in monotherapy, and fall, nasopharyngitis, and dyskinesia in add-on therapy [60, 78]. With no risk of tyramine interaction or sympathomimetic effects, the long-term safety and efficacy of rasagiline have been proven either as monotherapy or adjunct to levodopa treatment. Up to 52 weeks, rasagiline was well tolerated with sustained motor symptom improvement [60, 78]. On the association between rasagiline and antidepressants in PD patients, retrospective analysis of data from 1504 PD patients in the STACCATO study [128] showed that the mean duration of antidepressant use was 50.5 to 53.5 weeks in patients treated with rasagiline in adjunct with antidepressants, among which, SSRIs were used in up to 77% of these patients, and patients did not develop serotonin syndrome during adjunct therapy. However, the risk of the combination cannot be completely eliminated.
Safinamide
Safinamide was well tolerated in different randomized controlled trials and observational studies with similar incidences of TEAEs compared with placebo, most of which were mild to moderate [129]. A long-term safety and efficacy study of safinamide as add-on therapy to levodopa enrolled 203 Japanese PD patients with wearing-off, 142 of whom completed 52 weeks of treatment. The results showed that the incidence of adverse reactions was 78.3%. The most common adverse reactions were nasopharyngitis (20.7%) and dyskinesia (17.7%) [130]. Another randomized, double-blind, placebo-controlled Phase II/III study in Japanese PD patients [82] showed that the incidence of adverse reactions was 58.9%, 60.2%, and 61.4% in the placebo group, 50 mg/day and 100 mg/day safinamide groups, respectively, and the most common adverse reactions were dyskinesia (2.1%, 8.3% vs. 10.6%) and visual hallucinations (1.4%, 3.0% vs. 4.5%). In 2021, a European real-world study to assess the safety of safinamide in PD patients [131] showed that the adverse events in treated patients were predominantly mild (62.0%) or moderate (28.0%), and only 2% were related to safinamide, and in most cases, these adverse events did not need to be treated. As an adjuvant therapy, safinamide is safe and well-tolerated in patients with fluctuating symptoms and in special populations.
FUTURE OF MAO-B INHIBITORS
Despite numerous clinical trials, PD remains an untreatable disease. Neuroprotection in PD has intended to halt or reverse cell death of nigrostriatal dopaminergic neurons and prevent disease progression, but clinical studies have not presented enough supporting results, except for the trial of rasagiline by delayed start design at a low dose of only 1 mg/day. MAO-B inhibitors selegiline and rasagiline can both exhibit potent neuroprotective function by regulating mitochondrial apoptosis cascade, maintaining the mitochondrial function, and increasing gene coding expression of antioxidant enzymes, anti-apoptotic Bcl-2, and pro-survival neurotrophic factors, and suppressing oligomerization and aggregation of α-synuclein and cell toxicity [132]. In addition to the traditional oral dosage forms, other novel galenic forms of MAO-B inhibitors were explored. Pharmacokinetic studies in healthy Chinese populations showed that the transdermal rasagiline patch was able to prolong the duration of administration with good safety and tolerability, providing a continuous treatment modality for patients who were not able to routinely take oral medications [133]. Furthermore, animal studies showed that injectable rasagiline based on an injectable long-acting formulation system significantly increases dopamine levels in PD model rats (
MAO-B inhibitors have now evolved into the third generation with higher selectivity, reversible effects, wider safety margins, and potential for neuroprotection, and their clinical efficacy is worth exploring [136]. Recent data show that monoamine oxidase remains a preferred molecular target in the study of neurodegenerative diseases [137]. Many novel MAO-B inhibitors with high reversibility, selectivity, and fewer adverse effects are still under development [137, 138]. There are also a large number of selective MAO-B inhibitors in natural products waiting to be discovered, including flavonoids, xanthones, proanthocyanidins iridoid, glucosides, curcumin, alkaloids and their derivatives [24]. Danshensu (3-(3,4-dihydroxyphenyl)-(2R)-lactic acid, salvianic acid A), which was isolated from several herbs, such as
In addition to the treatment of PD patients, MAO-B inhibitors have attracted extensive attention in other diseases related to aging and apoptosis. Rasagiline was well tolerated in randomized controlled studies in patients with amyotrophic lateral sclerosis. The results of a
SUMMARY/EXPERT OPINIONS
Many studies have shown that MAO-B inhibitors are effective and safe for patients with early and advanced PD. This medication can effectively improve patients’ motor and non-motor symptoms and reduce the “OFF” time, with neuroprotective potential at the same time. It can also meet the patients’ need for long-term and standardized treatment and provide more options for the whole course management of PD patients [146, 147]. In addition, a variety of new MAO-B inhibitors with higher selectivity, safety, and neuroprotection are under development. It is believed that with further study, MAO-B inhibitors will bring more profound benefits to patients being treated for neurodegenerative diseases such as PD.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the National Natural Science Foundation of China, Grant/Award Number: 81430022, 81771374, 81971187, 81971183; Shanghai Municipal Science and Technology Major Project, Grant/Award Number: 2018SHZDZX05; Innovation Program of Shanghai Municipal Education Commission, Grant/Award Number: 2017-01-07-00-01-E00046; Foundation of Shanghai Municipal Education Commission, Grant/Award Number: 2017NKX001.
CONFLICT OF INTEREST
The authors have no conflict of interests.
