Abstract
Background:
Parkinson’s disease (PD) has been associated with a tendency towards more risky decisions. However, the commonly used paradigms typically neglect the social context.
Objective:
Here, we investigated social decision-making and self-estimation in a competitive experimental task.
Methods:
A computerized experimental setting was used in which 86 PD patients (age = 66.5 [50–79], 62.8% male, H&Y = 2 [1.5–3]) and 44 healthy controls (HC; age = 67 [54–79], 54.4% male) in groups of four performed mathematical addition tasks in which they were asked to calculate as many sums as possible in five minutes. Participants had to choose their preferred compensation scheme (“piece rate” versus “tournament”) and retrospectively rank their performance in comparison to the suspected performance of the others. A comprehensive neuropsychological test battery was also conducted.
Results:
No significant difference was found in overall social decision-making and self-estimation between PD patients and HC. However, for those individuals who made inadequate decisions, PD patients engaged in significantly more risk-averse and HC in more risky decisions. Concerning those inadequate decisions, the PD patients made more extreme decisions (severity of social decision-making) in both directions (risk-averse, risk-seeking).
Conclusion:
Our data indicate that social decision-making behavior and self-estimation are largely intact in PD patients with mild to moderate disease stages and intact global cognition, executive functions, and social cognition. Future studies with more heterogeneous PD samples regarding their neuropsychological profile will have to examine at which state social decision-making may be affected and by which factors this behavior might be influenced.
INTRODUCTION
Beyond the core motor symptoms, Parkinson’s disease (PD) is characterized by various non-motor symptoms [1], among them cognitive and affective dysfunctions, which severely impact quality of life [2]. One frequently affected domain is decision-making [3]. Adequate decisions require considering their future consequences and, especially in uncertain settings, gauging the response options about the true probability of occurrence using reward- and punishment-related learning. In PD, evidence points towards riskier decisions under ambiguity, i.e., in tasks with unclear winning and losing probabilities and rules for reward and punishment such as the Iowa Gambling Task (IGT) [4]. Further, PD patients are impaired in decision-making tasks under risk in which those rules are transparent (Game of Dice Task, GDT) [5]. Altered reward-learning in PD has been related to discrepancies between nigro-striatal and mesolimbic dopaminergic degeneration and administration of dopaminergic drugs which may induce abnormal ‘hyperstimulation’ of the mesolimbic system in PD [6]. Dysfunctional reward-related decision-making has also been associated with impulse control disorders in PD [7].
Notably, research on decision-making in PD has so far almost exclusively used paradigms in which decisions are made “in isolation”, thereby neglecting the social context. However, many decisions in everyday life need consideration of social aspects. Social decision-making may be affected in PD, because dysfunction in several subdomains of social cognition including cognitive and affective theory of mind (ToM) have been reported [8–10]. These deficits may significantly contribute to the impairment in PD, affecting both patients’ and relatives’ quality of life [9, 11]. Additionally, PD symptoms such as communication deficits interfere with social functioning and social roles per se endorsing the risk of inappropriate social behavior [12].
To our knowledge, only one study has analyzed social decision-making and self-estimation in PD. Florin et al. [13] compared patients with and without deep brain stimulation in the subthalamic nucleus (STN-DBS) postoperatively and healthy controls (HC) with an experimental task in which patients had to perform calculations competitively in a group setting of four individuals (without knowing the performance of the others). They could decide upon their compensation by choosing either a “piece rate” or a “tournament” (competitive) compensation and, after performing this task, were asked to make this decision again based on their self-estimation of their performance. The authors found that PD patients without STN-DBS did not show altered social decision-making compared to HC suggesting that this function is not generally affected, while patients with STN-DBS demonstrated increased risk-taking behavior and overestimation of their own performance. Notably, the sample was rather small (29 PD, 30 with PD and STN-DBS, 19 HC), only included male patients, and participants did not receive an elaborate neuropsychological test battery, so that a characterization of their cognitive and affective state, which might be related to decision-making and self-estimation, was not possible.
Hence, our interest was to investigate social decision-making and self-estimation in PD patients without STN-DBS more comprehensively. The aims of this paper were to analyze 1) social decision-making behavior and self-estimation, and 2) potentially relevant factors associated with this behavior in terms of sociodemographic, clinical and neuropsychological parameters in a larger sample of PD patients of both sexes without STN-DBS and without global cognitive impairment. For this purpose, we examined male and female PD patients and age-matched HC with an elaborate neuropsychological test battery as well as with the social decision-making paradigm used by Florin et al. [13]. As outlined above, dysfunctional decision-making, social cognition, and social behavior suggest deficits in social decision-making. However, Florin et al. [13] did not find deficits of PD patients compared to HC. Thus, our study was exploratory, and we did not formulate a directional hypothesis. Also, our analysis regarding potential sociodemographic, clinical, and neuropsychological factors associated with this behavior was exploratory due to a lack of evidence on this topic.
MATERIALS AND METHODS
Study design
Data analyzed here stem from a longitudinal study on social decision-making behavior after STN-DBS in PD patients in contrast to PD patients without surgery and HC which was registered in the German Clinical Trials Register (DRKS; ID: 00010876) and was approved by the Ethics Committee of the Faculty of Medicine of the University of Cologne, Germany (16–357). The study was conducted in accordance with the Declaration of Helsinki. The reporting of this trial follows the STROBE statement (see Supplementary Material 1) [14]. Data were collected in two German University Hospitals (Cologne, Duesseldorf) between January 2017 and August 2020. Here, we only report cross-sectional data (baseline testing for PD patients with and without planned STN-DBS and HC). Longitudinal data will be reported elsewhere. After written informed consent, participants were screened for eligibility. Clinical evaluation and neuropsychological testing including a computer paradigm in a small group setting was conducted. Participants were financially reimbursed for their travel expenses and were informed about the possibility to win a small amount of money in a randomly chosen condition of the computerized paradigm described below.
Sample size
In total, 86 patients with idiopathic PD and 44 HC participated in the study. Inclusion criteria for PD patients were 1) a clinical diagnosis of idiopathic PD according to the UK Brain Bank criteria [15] and a Hoehn and Yahr (H&Y) stage 1–4 [16]—and for both groups, 2) no dementia indicated by a minimum score of 25 in the Mini-Mental Status Examination (MMSE) [17], 3) age between 50 and 80 years, and 4) all participants had to be eligible to give written informed consent. Exclusion criteria encompassed 1) any other neurological or psychiatric diseases (including impulsive control disorder) as well as any signs of structural cerebral brain damage (according to physician’s letter and/or clinical history), 2) current or former drug or alcohol abuse, and 3) prior implantation of DBS electrodes.
Outcomes and further assessments
Clinical evaluation (only for the PD group)
All assessments and intervention sessions were conducted with patients in their individual medication ON state; short breaks were offered to avoid excessive strain. In order to assess PD disease severity, the Unified Parkinson’s Disease Rating Scale (UPDRS) III [18] as well as the H&Y scale [16] were used. Levodopa equivalent daily dose (LEDD) and percentage of dopamine agonists was calculated according to Tomlinson et al. [19]).
Neuropsychological evaluation
A neuropsychological test battery was performed by experienced psychologists. In both groups, the MMSE [17], the Parkinson Neuropsychometric Dementia Assessment (PANDA) [20], and the Clinical Dementia Rating scale by Mattis [21] were used to assess global cognition. The Beck Depression Inventory-II (BDI-II) [22], the Apathy Evaluation Scale (AES) [23], and the Mania Evaluation Scale (MSS) [24], as well as the Reading the Mind in the Eyes Test (RMET; ToM) [26] were employed to assess psychiatric symptoms and social abilities. For PD patients, the Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease-Rating Scale (QUIP-RS) [27] and the Parkinson’s Disease Questionnaire (PDQ-39) [28] were used to measure symptoms of impulse control disorders and quality of life.
Experimental paradigm: social decision-making and self-estimation
A computerized experimental paradigm developed by Niederle and Vesterlund [29] was used to assess social decision-making and self-estimation (Fig. 1). HC and PD patients were examined in separate groups of four (physically present) participants. Each participant sat in front of a separate computer screen (screen size 15 inch; usage of an optical mouse as a pointing device). All participants were instructed not to exchange any information during or after the experiment. All instructions were displayed on a computer screen and were the same for all participants.
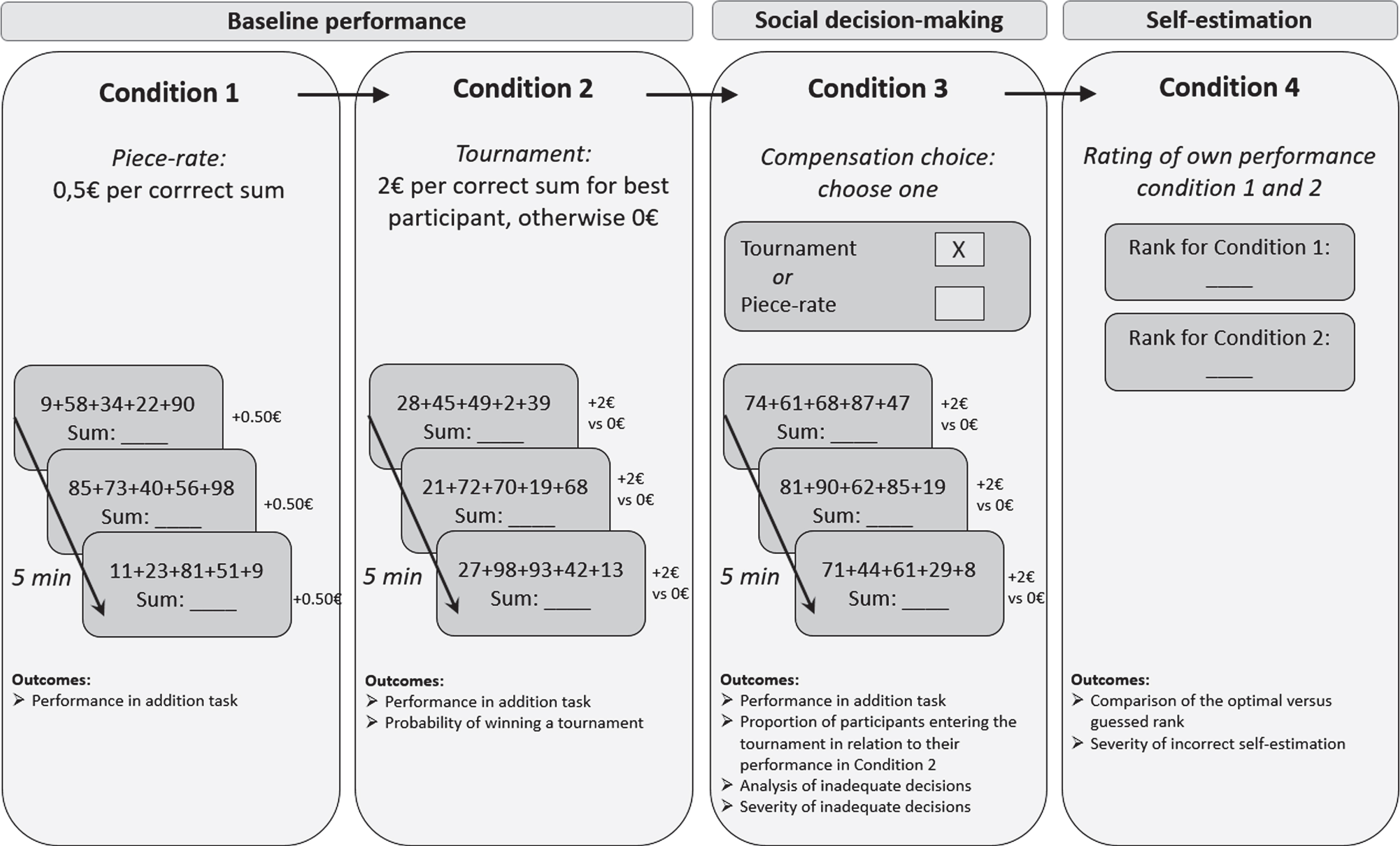
Experimental paradigm.
The paradigm consisted of four conditions (Fig. 1). In condition 1, 2, and 3, participants were asked to perform mathematical addition tasks that entailed adding up as many sets as possible of five random numbers between 1 to 100 within five minutes. In conditions 1, 2, and 3, participants received immediate feedback after each calculation task, indicated by the number of correctly solved addition tasks so far. However, they were not informed about the performance of the other participants. No feedback was given upon how they performed compared to the other participants. They received monetary compensation depending on their performance and the randomly chosen condition. Condition 1 (piece-rate compensation): Participants were asked to calculate as many sums as possible in five minutes. They received 50 cents per correct sum. Condition 2 (tournament compensation): Participants performed the same addition task. However, only the best out of the four participants received 2 € per correct sum. The rest of the group received 0 €. Condition 3 (choice piece-rate vs. tournament compensation): The five minutes addition task was performed again. Before starting the task, participants had to decide whether they wanted to be paid by the piece-rate or tournament compensation scheme. Condition 4 (self-estimation): Participants were asked to rate their own performance for condition 1 and 2 by assigning a rank to themselves from 1 to 4 (1 = if they believed to have had the best performance, 4 = worst performance in the group).
Outcomes derived from the experimental paradigm are also outlined in Fig. 1 and further explained in Table 1.
Outcomes of the experimental social decision-making and self-estimation task
Statistical analysis
Statistical analyses were performed with IBM SPSS Statistics (26.0) and Matlab R2019b. The Kolmogorov-Smirnov test was used to check all variables regarding normal distribution. The original power analysis was based on three groups for a longitudinal design. The a priori power calculation with G*Power (ver. 3.1.9.4) indicated 54 participants necessary for medium effect sizes. However, for the current cross-sectional analysis of two groups (86 PD patients, 44 HC), a post-hoc calculation was carried out with G*power to detect moderate effects sizes (w = 0.30) and achieved power of 87.5%.
For the comparison of the sociodemographic, clinical, and neuropsychological characteristics and the number of correctly solved addition tasks between the two groups, t-tests or Mann-Whitney-U tests were conducted, as appropriate. χ 2 tests were performed to assess group differences in social decision-making and self-estimation.
Spearman’s rank correlation coefficient (rho) was computed between the number of correctly solved addition tasks for all three conditions and education for both groups, as well as the relationship between severity of inadequate social decision-making and self-estimation and relevant demographic, clinical, and neuropsychological variables. Point-biseral correlations (r pb ) were used for correlation analyses between the nominal scaled variable social decision-making and relevant demographic, clinical, and neuropsychological variables. For correlation analyses with the dichotomous variable sex and the nominal scaled variable social decision-making Cramer’s V was used; correlation analyses between the dichotomous variable sex and the metric scaled variable severity of inadequate social-decision making, a point-biseral correlation r pb was conducted; for self-estimation as an ordinal scaled variable, a χ 2 test was used.
RESULTS
Demographic, clinical, and neuropsychological characteristics
The HC (n = 44) group had significantly more years of education compared to the PD group (n = 86), whereas the PD group displayed significantly higher symptoms of depression, apathy, and mania. On all other variables including neuropsychological tests, the two groups did not differ (Table 2).
Baseline demographic, clinical, and neuropsychological characteristics of the total sample, separated by group
Values are presented as the mean±standard deviation or median and range or frequency with percentages. For baseline comparison between groups, p-values of Mann-Whitney-U tests or independent sample t-tests are reported as appropriate. HC, healthy control; PD, Parkinson’s disease; UPDRS, Unified Parkinson’s disease rating scale; LEDD, Levodopa equivalent daily dose; MMSE, Mini-Mental State Examination; PANDA, Parkinson Neuropsychometric Dementia Assessment; CDR, Clinical Dementia Rating; TMT, Trail Making Test; MCST, Modified Wisconsin Card Sorting Test; RMET, Reading the Mind in the Eyes Test; BDI II, Beck Depression Inventory-II; AES, Apathy Evaluation Scale; MSS, Mania Self-evaluation Scale; QUIP-RS, Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease-Rating Scale; PDQ-39, Parkinson’s Disease Questionnaire; level of significance: *p < 0.05; **p < 0.01; ***p < 0.005.
Experimental paradigm
Performance in addition tasks
In all three conditions, HC solved significantly more addition tasks correctly than PD patients (Table 3). As HC had significantly more years of education, we correlated the number of solved addition tasks in each condition with years of education. There were no significant correlations between any of the conditions and the years of education (p = 0.339–0.976 for HC; p = 0.336–0.777 for PD).
Correctly solved addition tasks for both groups and all three conditions
Values are presented as the mean±standard deviation (normal distribution) or median and range or (non-normal distribution). For baseline comparison between groups, p-values of Mann-Whitney-U tests or independent sample t-tests are reported as appropriate. HC, healthy controls; PD, Parkinson’s disease; level of significance: ***p < 0.005.
Social decision-making
Proportion of participants entering the tournament in relation to their performance
Given the same amount of correctly solved addition tasks, the probability to win in condition 2 is different between PD patients and HC, because the results are based on the number of correctly solved addition tasks in each group (which is lower in PD; Fig. 2A, see Supplementary Material 2 for details).
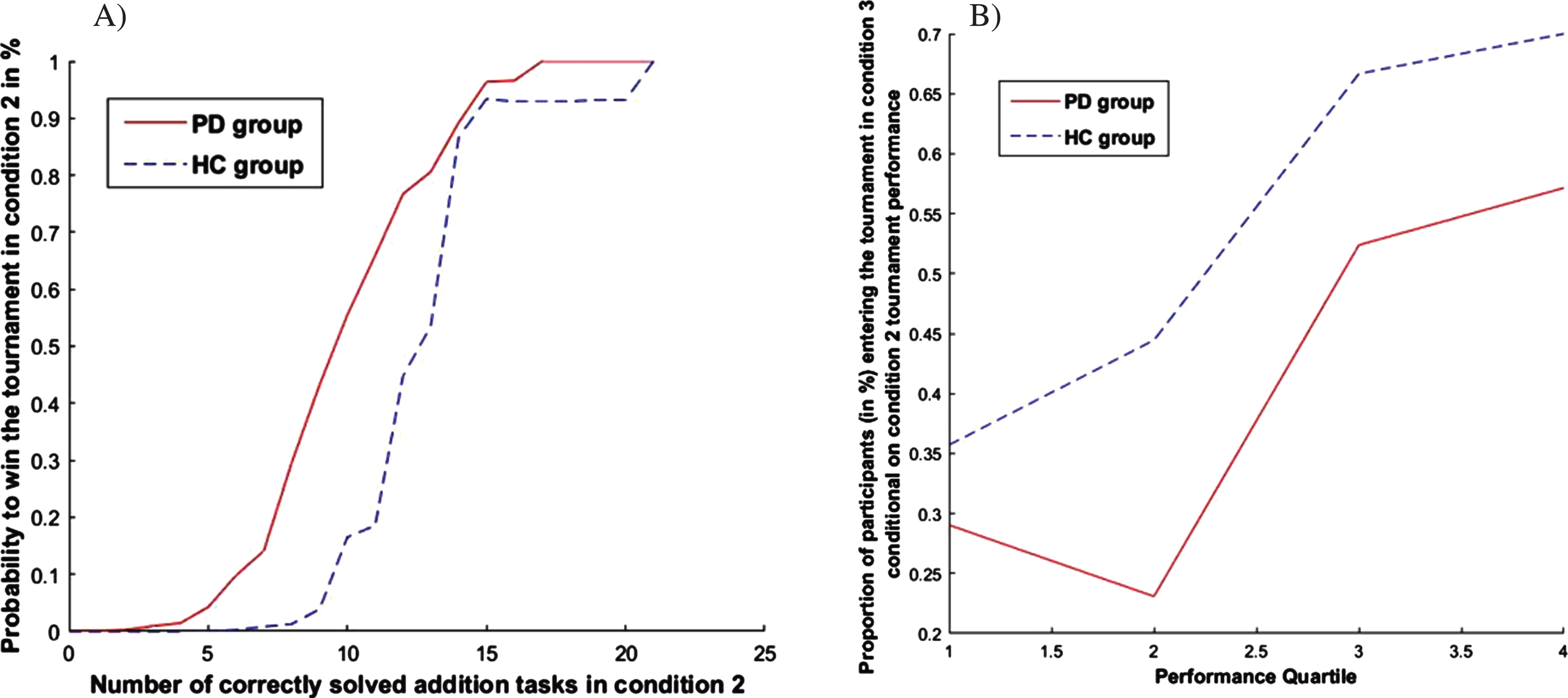
A) Probability of winning a tournament based on the number of correctly solved addition tasks in condition 2. B) Proportion of participants entering the tournament in condition 3 conditional on the tournament performance in condition 2 (1 = worst quartile, 4 = best quartile).
Figure 2B then indicates the proportion of participants entering the tournament in condition 3 dependent on the condition 2 performance quartile (quartile 1 = worst performance, 4 = best performance). An adequate social decision-making behavior would lead to a low number of participants with poor performance and a high number of participants with good performance entering the tournament condition. The largely steadily increasing pattern indicates that both groups mostly follow the aforementioned logic by basing their tournament entry decision on their past performance (Fig. 2B). According to their lower performance level, the proportion of PD patients entering the tournament is lower.
Analysis of inadequate decisions
About 60% of the participants in both groups (PD n = 54, HC n = 25) showed adequate social decision-making in condition 3, whereas the rest showed inadequate social decision-making (risk-averse: PD n = 20, HC n = 5; risk-seeking: PD n = 12, HC n = 12; Fig. 3A). Overall, there was no significant difference between the two groups in social decision-making (χ 2 (2) = 5.126, p = 0.077, φ= 0.200). Yet, for inadequate decisions (risk-averse, risk-seeking), the two groups differed significantly (χ 2 (1) = 4.864, p = 0.027, φ= 0.315). PD patients made more risk-averse decisions, whereas HC made more risk-seeking decisions.
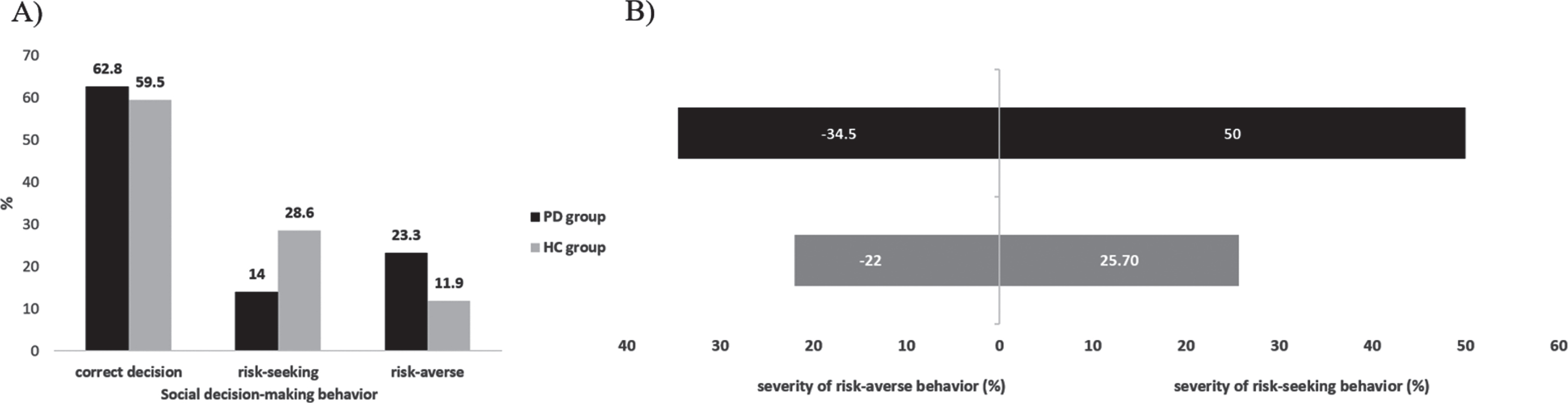
A) Distribution of social decision-making behavior in both groups. B) Severity of maladaptive social decision making in a subsample for both groups. Note that this analysis only refers to the subgroup of individuals who showed inadequate social decision-making.
In terms of the severity of maladaptive decision (the deviation of the individual performance from a cut-off score for rightly entering the tournament based on the group mean of correctly solved addition tasks), the groups differed significantly (Fig. 3B). Wrong decisions in terms of deviation from the optimal decisions (choosing tournament compensation scheme when supposed to choose piece-rate compensation scheme and vice versa) was significantly higher for the PD group for both, risk-averse and risk-seeking decisions (U = –2.172, p = 0.030).
Self-estimation
About three-fourths of the participants correctly estimated their own rank (Fig. 4). For both groups, more participants overestimated their rank than underestimated it. However, self-estimation was not significantly different between the two groups (p = 0.245, φ= 0.215).
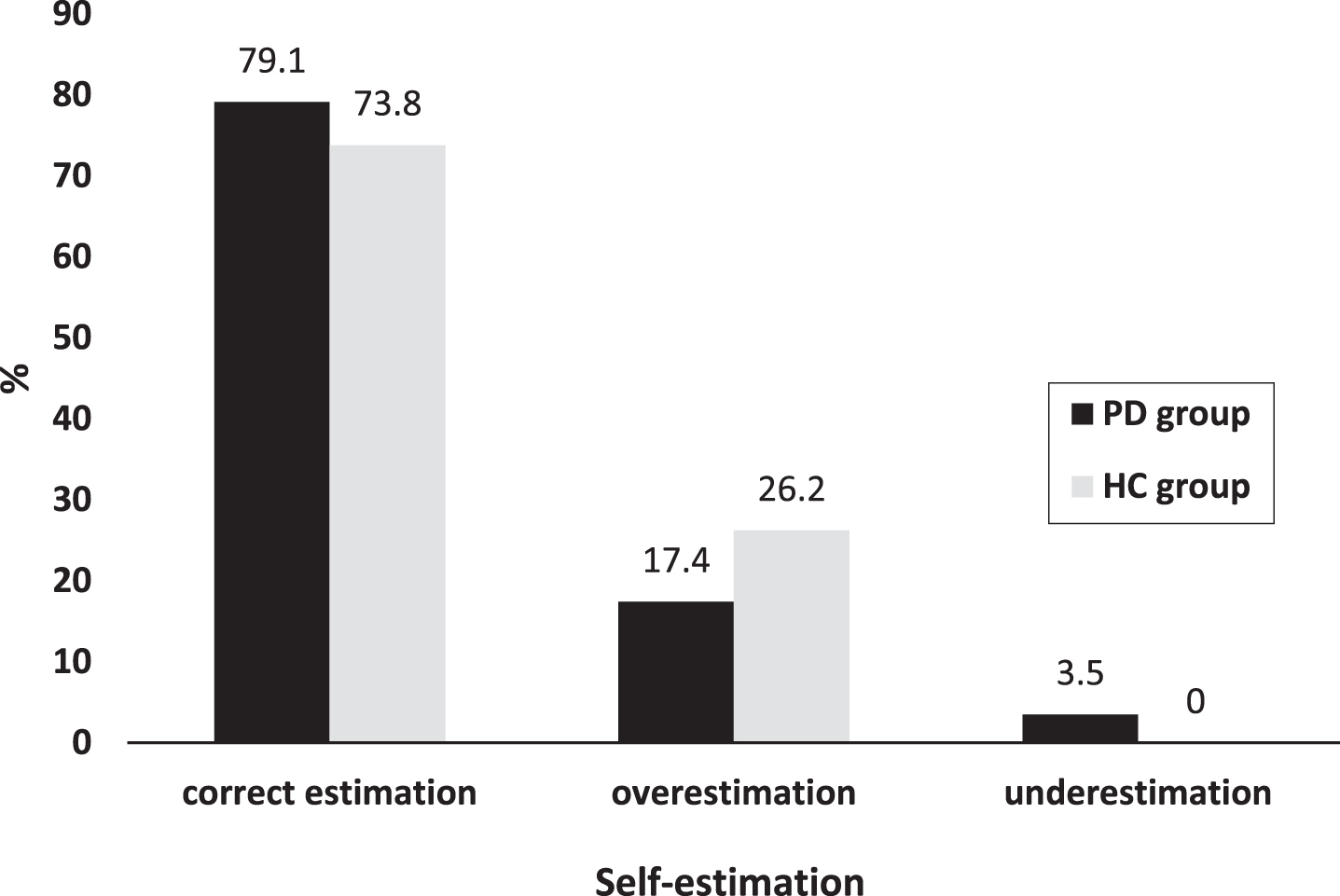
Distribution of self-estimation in both groups for the total sample.
Correlation analyses
Correlation analyses (Table 4) revealed only one significant correlation between self-estimation and education indicating that more years of education are related to incorrect self-estimation in HC (p = 0.016). There were no significant correlations between the total amount of dopaminergic medication and any aspects of performance (Table 4).
Correlation analysis of social decision-making and self-estimation with sociographic, clinical, and neuropsychological variables for the PD and HC group
*p<0.05; **p<0.01; ***p<0.001. aInterval scaled variables indicating severity of inadequate social decision-making; bOrdinal scaled variable for the difference between optimal rank and guessed rank. 1Higher values indicate better/adaptive performance; 2Lower scores indicate better/adaptive performance. AES, Apathy Evaluation Scale; BDI-II, Beck Depression Inventory II; EF, Executive functions; HC, healthy controls; LEDD, Levodopa Equivalent Daily Dose; MCST, Modified Card Sorting Test; MSS, Mania Self-evaluation Scale; PANDA, Parkinson Neuropsychometric Dementia Assessment; PD, Parkinson’s disease; QUIP-RS, Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease–Rating Scale; r pb , point-biseral correlation; rho, Spearman’s rank correlation coefficient; RMET, Reading the Mind in the Eyes Test; ToM, Theory of mind; TMT, Trail-Making-Test; UPDRS, Unified Parkinson Disease Rating Scale; χ2, Chi-Quadrat.
DISCUSSION
We aimed at investigating social decision-making behavior and self-estimation in PD patients compared to HC. We found that 1) there was no significant difference between PD and HC in overall social decision-making behavior and self-estimation. In the subgroups of individuals who made inadequate decisions, 2) PD patients made significantly more risk-averse decisions, whereas HC made significantly more risky-seeking decisions. The two groups 3) differed significantly concerning the severity of inadequate decision-making, with the PD group showing more extreme decisions regarding both directions (risk-averse and risk-seeking) compared to HC. Finally, 4) the severity of inadequate decisions was not significantly related to any of the included sociodemographic, clinical, or neuropsychological variables in the PD group.
Functional social decision-making and self-estimation in PD patients
Our finding that social decision-making and self-estimation in PD was comparable to that of HC corroborates the findings by Florin et al. [13] who also did not find differences between PD and HC using the same computerized paradigm. These results are remarkable, as vast evidence exists for decision-making impairments in PD in various paradigms [4, 30]—albeit typically in tasks without a social context. However, this impairment has been shown to be dependent on both the type of task and the severity of PD symptomatology including cognitive symptoms. For example, Euteneuer et al. [5] found that non-demented PD patients showed dysfunctions in a decision-making task under risk with explicit rules for risk and reward/punishment (GDT), while performance in a decision-making task under ambiguity with implicit rules (IGT) was unimpaired. Both tasks have been associated with the limbic-orbitofrontal-striatal loop, involved in emotional processing. Additionally, the GDT relies on the dorsolateral prefrontal loop providing executive function, enabling individuals to act strategically. In fact, performance in the GDT and executive tasks was related, indicating that patients with poorer executive functions made more risky decisions [30]. Our decision-making paradigm resembles both the GDT in that rules for risk and reward/punishment were explicit and the IGT in that ambiguity exists regarding the estimation of one's own performance in comparison to that of the others. Although our PD group performed worse in the addition tasks of the decision-making paradigm compared to the HC, they performed within the normal range in measures for global cognition and for executive function. Furthermore, even in the correlation analysis of inadequate decisions, no relationship was found with any cognitive measure. Thus, future studies will have to investigate whether PD patients with executive dysfunctions do perform below the normal range in a social decision-making task as used in our study, and whether or not these two functions are related. Notably, adequate social decisions in our experiment required social cognition or, more specifically, good self-estimation and cognitive ToM performance as well as adequate social comparisons based on an integration of this information. As deficits in social cognition in PD are well known [8, 10], our findings of functional social decision-making appear surprising. In PD, the alterations of social functioning including inadequate social decision-making in PD have been attributed to frontal-executive dysfunctions due to a subversion of online action-monitoring leading to disinhibition with volition [31, 32]. Morese et al. [31] suggest this to be due to genetic polymorphisms and alterations of the nigrostriatal pathway, the medial prefrontal cortex, and the default mode network. However, our PD group showed neither executive dysfunctions, as outlined above, nor ToM dysfunctions. Furthermore, self-estimation in our PD group did not differ from that of HC, a finding which is in line with those in the study of Coundouris et al. [8] who found that the ability to correctly estimate one’s own performance within a social group might still be intact in PD patients without cognitive impairment. Admittedly, it is difficult to evaluate whether and how our negative (non-significant) results concerning group differences in overall social decision-making and self-estimation relate to the experimental paradigm used. To the best of our knowledge, this is only the second study after Florin et al. [13] investigating social decision-making in those groups with this paradigm. It might be that PD patients act differently and closer towards how healthy controls make decisions because of the direct peer interaction. As stated earlier, the majority of decision-making paradigms have focused solely on the decision-making part, without any direct social interaction. More empirical values using this paradigm (or others taking into consideration the social context of decision-making) in PD are needed to better understand social decision-making in PD. Future studies will have to indicate whether patients with cognitive impairment in executive functioning, social cognition or self-estimation do show inadequate social decision-making.
Inadequate social decision-making and self-estimation
Inadequate social decisions (risk-averse or risk-seeking) and self-estimations (under- vs. overestimation) were made by several participants in both groups (approximately 40% in social decisions and 20% in self-estimation). PD patients made significantly more risk-averse decisions, while HC made significantly more risk-seeking decisions. This result is rather unexpected, as inadequate decisions in reward-related test paradigms have typically demonstrated riskier decisions in PD [33, 34]. Also, PD patients are at risk for impulse control disorders, due to dopaminergic treatment, especially dopamine agonists, but also other demographic, genetic, behavioral and clinical factors, and being related to an increased activity in the ventral striatum and orbitofrontal cortex and decreased activity in the anterior cingulate cortex [35].
However, our PD patients did not show impulsive behavior as assessed with the QUIPS-RS. A possible reason for the more risk-averse behavior of our PD patients compared to HC may be due to the nature of the task used. Although global cognition was not reduced in our patients, the number of correctly solved calculation tasks was significantly lower for the PD group. Possibly, patients might have had a lower self-esteem in this task based on their lower education. However, this interpretation remains speculative (especially, as a paradoxical significant correlation was found between lower self-estimation and higher education in our HC group). Computer literacy might be another reason for the reduced performance of PD patients. In addition, patients with bradyphrenia with yet intact cognition might still work slower during the calculation tasks as compared to HC. A further speculative explanation for the more risk-averse behavior might be related to the social context per se as the setting might have a correctional function. Finally, it cannot be excluded that patients simply solved less addition tasks due to motor slowness as the answer format required using a keyboard and an optical mouse. Taken together, a study including social and non-social decision-making tasks might reveal the influence a social competitive situation on decision-making, and self-estimation.
A further interesting finding was that PD patients’ inadequate social decisions were more extreme as they deviated more significantly from the calculated “correct” decision compared to HC. However, as these decisions were observed for both directions (risk-averse and risk-seeking decisions), this behavior may not follow a logical pattern and be interpreted as “random” behavior rather than being related to the level of social risk-taking. Such “random behavior” could be derived from less strategy use or reasoning as executive functions, but as no correlation was found with executive tests, this aspect remains unclear.
Strengths and limitations
To our knowledge, this is the first study investigating social decision-making and self-estimation in PD using a computerized paradigm, employing a large sample size, a well-characterized PD group and a comprehensive neuropsychological test battery. The use of HC is a further strength to identify potential PD-specific changes in social decision-making and self-estimation. However, the study has some limitations: First, despite the relatively large total sample size, subgroup analyses (severity of inadequate social decision-making) was carried out with a subset of the total sample which was then rather small. For these subgroup analyses, future studies would need an even larger sample to ensure higher statistical power. Second, future studies should include control samples which are matched regarding sociodemographic variables including education which was not achieved in our study. Third, a larger heterogeneity in the clinical sample regarding the clinical and neuropsychological profile (especially executive functions and social cognition) might allow identifying variables influencing social decision-making in PD patients with more advanced symptoms. In this context, it would also be of interest to compare who do or do not take dopamine agonists. Fourth, as we decided for a PD-specific impulsivity scale, no comparisons were possible for PD patients and HC. However, as impulsivity was not present in our patient group, this aspect is irrelevant for our analysis. Yet, future studies should use an impulsivity scale that is suitable for patient as well as non-patient groups, therefore allowing between group comparisons. Fifth, the use of an experimental paradigm in a laboratory setting might be regarded as limited concerning ecological validity and should be taken into consideration by future studies.
Conclusion
Our data do not support general tendencies for risk-seeking behavior in social decision-making in PD and show intact self-estimation. However, as cognitive functioning, including, ToM as an important social subfunction, and impulsivity were within the normal range in our PD sample, the findings from this study might hold true for PD with the aforementioned neuropsychological profile, while it cannot be generalized to PD patients with more advanced symptoms. Thus, future studies with more heterogeneous PD samples are needed to investigate social decision-making and self-estimation to understand at which state PD patients may show impairment in this domain as well as possible influencing factors on social decision-making. Another important endeavor will be to investigate the impact of STN-DBS which is discussed to possibly modulate aspects of social behavior [13, 37].
Footnotes
ACKNOWLEDGMENTS
We thank all participants and their relatives for their participation in our study. Furthermore, we give our thanks to Alice Schamong, Katharina Göke, Maren Herrnberger, Paulina Olgemöller, and Pia Walger for helping with patient recruitment and data collection.
This study was financed by the participating study centers.
CONFLICT OF INTEREST
AZ declares no conflict of interest.
AKF has received grants from the German Parkinson Society and the German Alzheimer’s Society, as well as honoraria from: Springer Medizin Verlag GmbH, Heidelberg, Germany; Springer-Verlag GmbH, Berlin; ProLog Wissen GmbH, Cologne, Germany; pro audito Switzerland, Zürich, Switzerland; Seminar- und Fortbildungszentrum Rheine, Germany; LOGOMANIA, Fendt & Sax GbR, Munich, Germany. AFK is author of the cognitive intervention programs “NEUROvitalis” but receives no corresponding honoraria.
LK declares no conflict of interest.
AS has received grants from the German Research Council; honoraria from Medtronic Inc., Boston Scientific, Abott Medical, UCB, Abbvie.
PR has received a travel grant from AbbVie.
MTB has received speaker’s honoraria from Medtronic, Boston Scientific, Abbott (formerly St. Jude), GE Medical, UCB, Apothekerverband Köln e.V. and Bial as well as research funding from the Felgenhauer-Stiftung, Forschungspool Klinische Studien (University of Cologne), Horizon 2020 (Gondola), Medtronic (ODIS), and Boston Scientific and advisory honoraria for the IQWIG.
EF has received grants from the Volkswagen Foundation and the German Research Association (DFG).
EK has received grants from the German Ministry of Education and Research, ParkinsonFonds Deutschland gGmbH, the German Parkinson Society, the German Alzheimer’s Society; honoraria from: Oticon GmbH, Hamburg, Germany; Lilly Pharma GmbH, Bad Homburg, Germany; Bernafon AG, Bern, Switzerland; Desitin GmbH, Hamburg, Germany. EK is author of the cognitive training program NEUROvitalis but receives no corresponding honoraria.
