Abstract
Background:
Deep brain stimulation of the sub-thalamic nucleus (DBS-STN) reduces symptoms in Parkinson’s disease (PD) patients with motor fluctuations. However, some patients may not feel ameliorated afterwards, despite an objective motor improvement. It is thus important to find new predictors of patients’ quality of life (QoL) amelioration after DBS-STN. We hypothesized that personality dimensions might affect QoL after DBS-STN.
Objective:
To evaluate associations between personality dimensions and QoL improvement one year after DBS-STN.
Methods:
DBS-STN-PD patients (n = 303) having answered the “Temperament and Character Inventory” (TCI) before surgery and the PDQ-39 before and one year after surgery were included, from the cohort study PREDI-STIM. Linear regression models were used to evaluate associations between TCI dimensions and change in PDQ-39 scores after DBS-STN.
Results:
Novelty Seeking and Cooperativeness scores before surgery were positively associated with PDQ-39 scores improvement after DBS-STN (FDR-adjusted p < 0.01). Moreover, paradoxically unimproved patients with deterioration of their PDQ-39 scores after DBS-STN despite improvement of their MDS-UPDRS-IV scores had lower Cooperativeness scores, while paradoxically improved patients with amelioration of their PDQ-39 scores despite deterioration of their MDS-UPDRS-IV scores had higher Reward Dependence scores.
Conclusion:
Some presurgical personality dimensions were significantly associated with QoL amelioration and discrepancy between motor state and QoL changes after DBS-STN in PD. Educational programs before DBS-STN should take in account patient personality dimensions to better deal with their expectations.
INTRODUCTION
Deep brain stimulation of the sub-thalamic nucleus (DBS-STN) is proposed to Parkinson’s disease (PD) patients with motor fluctuations and generally leads to a great improvement of their quality of life (QoL) [1–4]. Nonetheless, some patients do not feel DBS benefits despite objective motor improvement [5, 6]. That is why it seems crucial to find some individual predictive factors of QoL improvement after DBS-STN in order to assure at best patients’ amelioration after this invasive therapy.
QoL improvement is an important outcome when evaluating treatment efficiency. It depends on both objective and subjective factors such as physical and psychological health, as well as personal feeling of well-being and happiness experienced [7, 8].
Because personality is a very individual characteristic and describes how people think, behave and manage their life [9], it seems a factor of interest to predict QoL. To assess personality, several questionnaires have been developed, based on different concepts. From them, the Temperament and Character Inventory (TCI) evaluates personality through seven dimensions [10]. It is derived from the neurobiological and psychological concept of personality made by Cloninger [9]. The TCI was already used in several studies assessing PD personality [11] and was validated by its stability [9, 12–15]. Moreover, our preceding study on PD patients with motor fluctuations has shown the adequacy of the TCI personality questionnaire on predicting QoL in this population before DBS-STN [16].
Our first objective was thus to identify associations between presurgical personality dimensions and QoL improvement one year after DBS-STN. Our second objective was to seek for characteristics of PD patients experiencing discrepancy between motor complications and QoL changes after DBS.
MATERIALS AND METHODS
This study is an ancillary analysis using data collected from the PREDISTIM cohort which principal objective is to define the predictive factors of the therapeutic response to DBS-STN on the long-term QoL in PD. PREDISTIM is an ongoing prospective multicentric cohort (Protocol 2013-A00193-42) sponsored by the University Hospital of Lille, conducted in 17 PD expert centers from the clinical research network in France (NS-Park/F-Crin), approved from the CPP Nord Ouest-IV Ethical Committee and registered in the ClinicalTrials.gov website (NCT02360683).
Patients
Briefly, patients undergoing DBS-STN in one of the participating centers were consecutive included into the study between 11/2013 and 09/2019. Inclu-sion criteria are a diagnosis of PD according to the UKPDSBB, disease duration ≥5 years, age between 18 and 75 and indication of DBS-STN. Exclusion criteria are atypical parkinsonism, severe cognitive impairment (Montreal Cognitive Assessment (MoCA) score < 23), severe psychiatric disorders (following psychiatric evaluation by a psychiatrist), levodopa motor response < 30% and contra-indica-tions to surgery.
Patients gave written informed consent, the study was conducted according to the good clinical practice, local regulations and data collection was compliant with GDPR rules.
Study design and clinical measures
For PREDISTIM, clinical data was collected at baseline, and then at 1-, 3-, and 5-years post-surgery. Clinical assessments at each visit included collection of demographic data, medical and treatment history, MDS-UPDRS part I-IV [17], part-III being performed in OFF and ON state during a standardized acute levodopa challenge, neuropsychological testing, patient and caregivers’ questionnaires. Along our ancillary study, only one personality questionnaire was added to evaluation at baseline: the TCI. Here, only patients evaluated before (V0) and after one year of DBS-STN (V1) were used in this study.
The MDS-UPDRS-III was evaluated for two conditions at V0: OFF and ON medication; and for four conditions at V1: ON stimulation ON medica-tion (stimON-medON), ON stimulation OFF medication (stimON-medOFF), OFF stimulation ON medication (stimOFF-medON) and OFF stimulation OFF medication (stimOFF-medOFF).
The total Levodopa Equivalent Dose (LED) was calculated [18].
The Parkinson’s Disease Questionnaire 39-items (PDQ-39) is a self-questionnaire evaluating specifically QoL of PD patients [19] and was assessed at V0 and V1. Its eight sub-scales can be divided into a Physical Component Score (“Mobility”, “Activities of Daily Living” and “Bodily Discomfort” sub-scores) and a Mental Component Score (“Emotional Well-Being”, “Stigma”, “Social Support”, “Cognition” and “Communication” sub-scores) of QoL.
The TCI was measured only at V0 in order to serve as predictive factor of QoL improvement after DBS-STN. It is a self-questionnaire of 226 binary questions used to assess patients’ personality through seven dimensions [10]. It includes four temperaments (which are supposed to be genetically determined personality traits [9]): Novelty Seeking (NS), Harm Avoidance (HA), Reward Dependence (RD), and Persistence (P); and three characters (which are developmental personality traits evolving through time and life history [9]): Self-Directedness (SD), Cooperativeness (C), and Self-Transcendence (ST). Each of these seven personality dimensions are measured by an independent score: higher is the score, higher the individual will present the personality characteristic associated. NS corresponds to impulsive and curious temperament; HA to anxious and pessimist traits; RD to socially attached and sensitive individuals; P to hardworking and perseverant personality; and the three characters are associated to levels of maturity: SD to individual maturity, C to social maturity, and ST to spiritual maturity. Except the P, each of these seven dimensions is subdivided into sub-dimensions: NS and HA are formed by 4 sub-dimensions, RD and ST by 3, and SD and C by 5. Each of these twenty-four sub-dimensions represents a specific aspect of the global dimension, also ranging in a spectrum of scores.
Statistical analyses
Descriptive analyses were performed. Moreover, Kruskal-Wallis tests or ANOVA (according to normality of our data) followed by post-hoc analysis were done to control for homogeneity across centers for several variables (age, disease duration, LED at V0, percentage of PDQ-39 total score and sub-scores changes at V1, and TCI dimensions).
Impact of DBS-STN at V1
After DBS-STN, changes between V0 and V1 in LED, MDS-UPDRS, PDQ-39, Hamilton Depression Rating Scale (HAMD), Hamilton Anxiety Rating Scale (HAMA), Lille Apathy Rating Scale (LARS), and MoCA were checked (Wilcoxon Signed Rank Test for Paired Samples).
Associations between TCI dimensions and PDQ-39 scores at V0
Linear regression models were performed to test the association between the different PDQ-39 sub-scores and total score at V0 and the seven TCI dimensions. Models were univariate with only one explanatory variable (each TCI dimensions) by models, resulting in seven models per response variable (each PDQ-39 scores) which were adjusted by age, sex, disease duration and LED at V0. A False Discovery Rate (FDR) correction was applied for 31 comparisons with calculation of q-values (corresponding to FDR adjusted p-values). We have chosen univariate models to study if some TCI personality dimensions were separately associated with QoL. Moreover, because studying TCI sub-dimensions as well as main dimensions seemed of interest, multivariate analysis seemed inappropriate since too many explanatory variables would be used (that is 31 variables –7 dimensions plus 24 sub-dimensions) which can be too much in view of the parsimony rule.
Associations between MDS-UPDRS and PDQ-39 scores at V1
These associations were tested using linear regression models adjusted by age, sex, disease duration, and LED at V1. The PDQ-39 scores at V1 served as response variables (the total and eight PDQ-39 sub-scores) and the MDS-UPDRS (part-I, part-II stimON-medON, part-III stimON-medON and part-IV) scores at V1 as explanatory variables. Models were univariate with only one explanatory variable by models, resulting in four models per response variable. An FDR correction was applied for 4 comparisons.
Associations between TCI dimensions at V0 and PDQ-39 changes at V1
Percentages of changes from V0 to V1 were calculated from the PDQ-39 total and sub-scores. Linear regression models were performed. The response variables corresponded to each calculated percentages of PDQ-39 change. The explanatory variables corresponded to each TCI dimensions and sub-dimensions. As explained above, models were univariate with only one explanatory variable by models, resulting in thirty-one models per response variable, adjusted by age, sex, disease duration and LED at V0, to account for any potential center effect. The same FDR correction was applied with q-values calculation for 31 comparisons.
PD population was also divided into two groups according to the Minimal Clinically Important Difference (MCID) of PDQ-39 total score change [20]: an “improvement” (difference of PDQ-39 total score between V1 and V0 < –4.72) and a “no improvement” group (difference of PDQ-39 total score between V1 and V0≥–4.72). TCI dimensions and sub-dimensions scores were compared in these groups (two-sample Mann-Whitney tests with FDR correction for 31 comparisons).
Differences between groups of responding patients
As supplementary exploratory analysis, we have chosen to study discrepancy between motor complications (MDS-UPDRS-IV) and QoL (PDQ-39 total) evolution. From the two PDQ-39 groups above and two similar MDS-UPDRS-IV groups (an “improvement” group based on MCID [21] (difference of MDS-UPDRS-IV score between V1 and V0 < –0.9) and a “no improvement” group (difference of MDS-UPDRS-IV score between V1 and V0≥–0.9)), PD patients were divided into three final groups of responding patients: a “paradoxically unimproved” group (MDS-UPDRS “improvement” group and PDQ-39 “no improvement” group), a “realistic” group (MDS-UPDRS and PDQ-39 “improvement” groups or MDS-UPDRS and PDQ-39 “no improvement” groups) and a “paradoxically improved” group (MDS-UPDRS “no improvement” group and PDQ-39 “improvement” group). Each group were compared two by two in terms of TCI dimensions and cognitive and behavioral (HAMD, HAMA, LARS, and MoCA) changes after DBS-STN (two-sample Mann-Whitney tests). FDR adjusted p-values (q-values) were calculated for 7 comparisons for the TCI dimensions analysis and for 4 comparisons for the cognitive and behavioral changes analysis.
All analyses were conducted on R Studio Software Version 1.1.456 and a threshold of bilateral statistical significance of 0.05 was used. As explained above, FDR corrections were applied and interpretations were made carefully according to strength of the results.
RESULTS
Three hundred and three PD patients were enrolled in this study (204 men (67.3%) and 99 women (32.7%)) (Table 1). Statistical testing did not show relevant significant differences across centers: only age and disease duration shown significant differences between centers in the Kruskal-Wallis testing (p = 0.003 and p = 0.03, respectively) which no more reached significance during the post-hoc analysis; LED at V0, percentage of PDQ-39 changes at V1 and TCI dimensions were not different between centers (p > 0.05). In any case, age, disease duration, sex and LED were used as adjustment variables in the linear regression models to control for any potential center effect.
Description of TCI dimensions and clinical characteristics at V0 and V1
Description of TCI dimensions and clinical characteristics at V0 and V1
Mann-Whitney tests: *p < 0.05; **p≤0.01; CI, confidence interval of the difference in means at 95% confidence level; V0, baseline; V1, one year after DBS-STN; †MDS-UPDRS medOFF (V0) vs. stimON-medOFF (V1); ‡MDS-UPDRS medON (V0) vs. stimON-medON (V1); ADL, Activities of Daily Living.
One year after DBS-STN, PD patients had a significant decrease in their dopaminergic treatments (LED). HAMD, HAMA, and MoCA scores significantly decreased after DBS, while LARS scores slightly increased.
All parts of the MDS-UPDRS scores improved significantly after DBS-STN except the part-II-ON which worsened.
Total PDQ-39 scores significantly improved after one year of DBS-STN as well as the “Mobility”, “Activities of Daily Living”, “Stigma”, “Cognition”, and “Bodily Discomfort” sub-scores, whereas the “Emotional Well-Being” sub-score did not sig-nificantly change. The “Social Support” and “Communication” sub-scores significantly worsened after surgery.
Associations between TCI dimensions and PDQ-39 scores at V0
TCI dimensions were mainly associated with the Mental Component Score of the PDQ-39 at V0. Indeed, the linear regression models between TCI dimensions and PDQ-39 sub-scores at V0 showed that HA and ST scores were significantly and positively associated with PDQ-39 total score (q = 1×10–8, R2= 0.16; and q = 9×10–3, R2= 0.07, respectively) whereas SD scores were significantly and negatively associated with PDQ-39 total score (q = 2×10–7, R2= 0.14)).
Associations between MDS-UPDRS and PDQ-39 scores at V1
At V1, the MDS-UPDRS part-I and part-II stimON-medON were positively associated with PDQ-39 total and 8 sub-scores (q < 0.01). The MDS-UPDRS part-III stimON-medON and part-IV were positively associated with PDQ-39 total score (q < 10–4), most of the Physical Component Score (“Mobility” and “Activities of Daily Living”) (p < 10–3) and part of the Mental Component Score (“Emotional Well-Being”, “Stigma”, and “Communication”) (q < 0.05). The “Bodily Discomfort” was associated only with the MDS-UPDRS part-IV (q < 10–3).
Associations between TCI dimensions at V0 and PDQ-39 changes at V1 (n = 303)
There were significant associations between TCI dimensions and PDQ-39 sub-scores change at V1 (Table 2). We mainly considered TCI dimensions having the strongest association with PDQ-39 sub-scores (presenting only significant FDR adjusted p-values that is q≤0.05) to avoid making assumption about lower associations.
Significant associations between TCI dimensions at V0 and percentages of PDQ-39 change at V1 (n = 303)
Significant associations between TCI dimensions at V0 and percentages of PDQ-39 change at V1 (n = 303)
Adjusted linear regression models: *p < 0.05; **p≤0.01; q-values, FDR-adjusted p-values; significant q-values in bold; CI, confidence interval at 95% confidence level; NS, Novelty Seeking; HA4, Fatigability; C3, Helpfulness; NS1, Exploratory excitability; NS4, Disorderliness; C, Cooperativeness; C1, Social acceptance; C4, Compassion; HA, Harm Avoidance; P, Persistence; RD, Reward Dependence; RD3, Dependence; ST3, Spiritual acceptance; ST, Self-Transcendence; SD, Self-Directedness; SD1, Responsibility; C2, Empathy.
The NS1 sub-dimension and some C sub-dimen-sions (C1 and C4) were significantly and negatively associated with the PDQ-39 “Mobility” sub-score change.
PD patients were separated into the PDQ-39 total “improvement” (n = 150) and the “no improvement” (n = 153) groups according to the MCID [20]. Among the “no improvement” group, 63% of patients had a no clinically relevant amelioration (that is a delta PDQ-39 total score between the MCID of –4.72 and 0) and 37% were not ameliorated (that is a delta PDQ-39 total score superior to 0). Uncorrected significant differences of HA4, C and C3 scores were found between the “improvement” group and the “no improvement” group which did not maintain significance with FDR correction (Table 3).
Differences of TCI dimensions scores between PDQ-39 total improvement groups at V1 (n = 303)
Mann-Whitney tests: *p < 0.05; **p≤0.01; q-values, FDR-adjusted p-values; CI, confidence interval of the difference at 95% confidence level; HA4, Fatigability; C, Cooperativeness; C3, Helpfulness.
For this second exploratory objective, because of missing data in the MDS-UPDRS-IV, only 264/303 PD patients were used to form the three groups of responding patients according to their PDQ-39 and MDS-UPDRS-IV changes: 97 patients were in the “paradoxically unimproved” group, 149 in the “realistic” group, and 18 in the “paradoxically improved” group.
Reward Dependence scores were significantly higher in the “paradoxically improved” group compared to the “paradoxically unimproved” group (q = 0.04 and CI = [1.0; 4.0]) (Fig. 1). Cooperativeness scores were significantly lower in the “paradoxi-cally unimproved” group compared to the “realistic” and “paradoxically improved” groups (q = 0.03 and CI = [1.0; 3.0]; q = 0.04 and CI = [1.0;5.0], respectively) (Fig. 2).
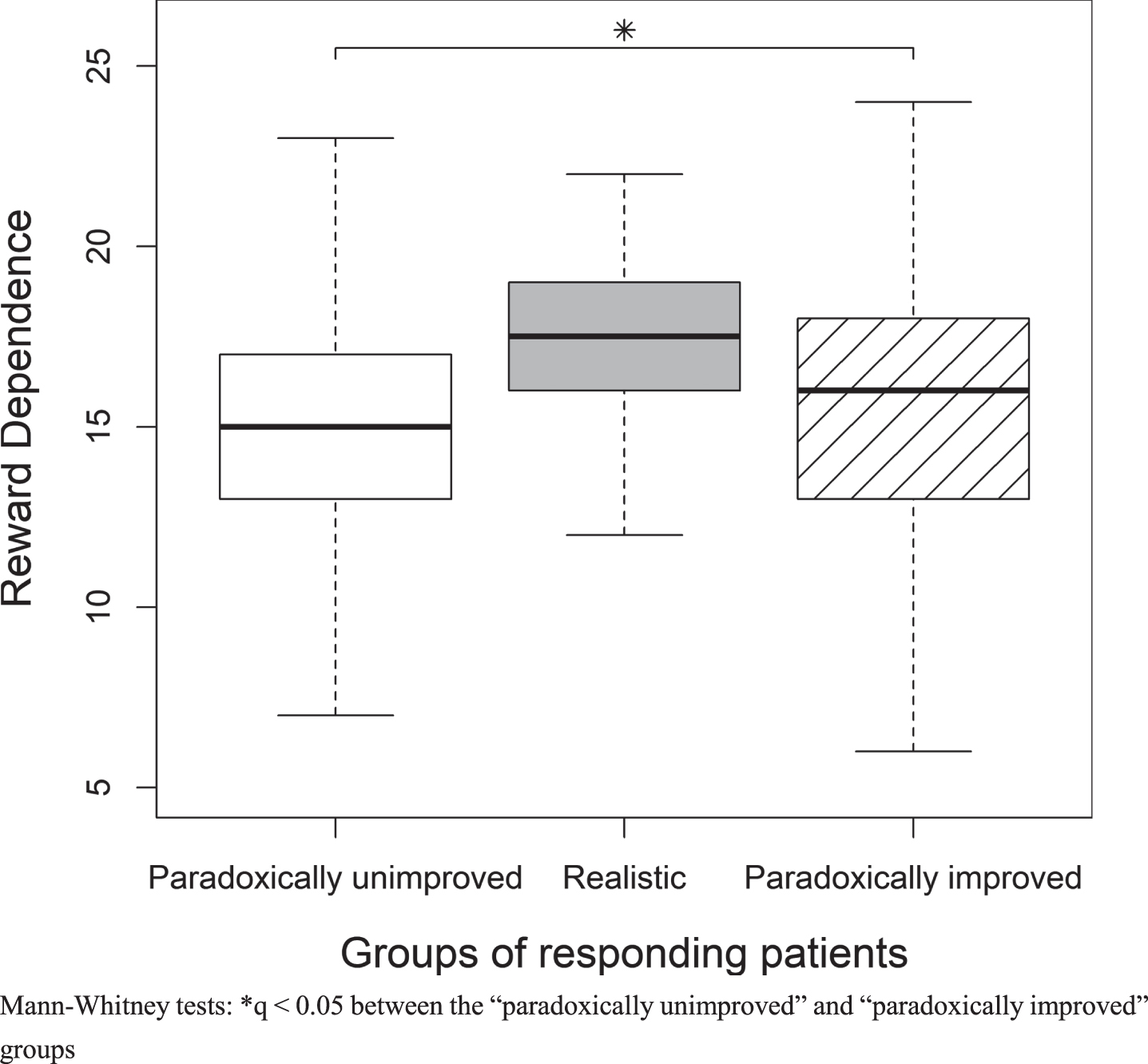
Differences in Reward Dependence scores between groups of responding patients.
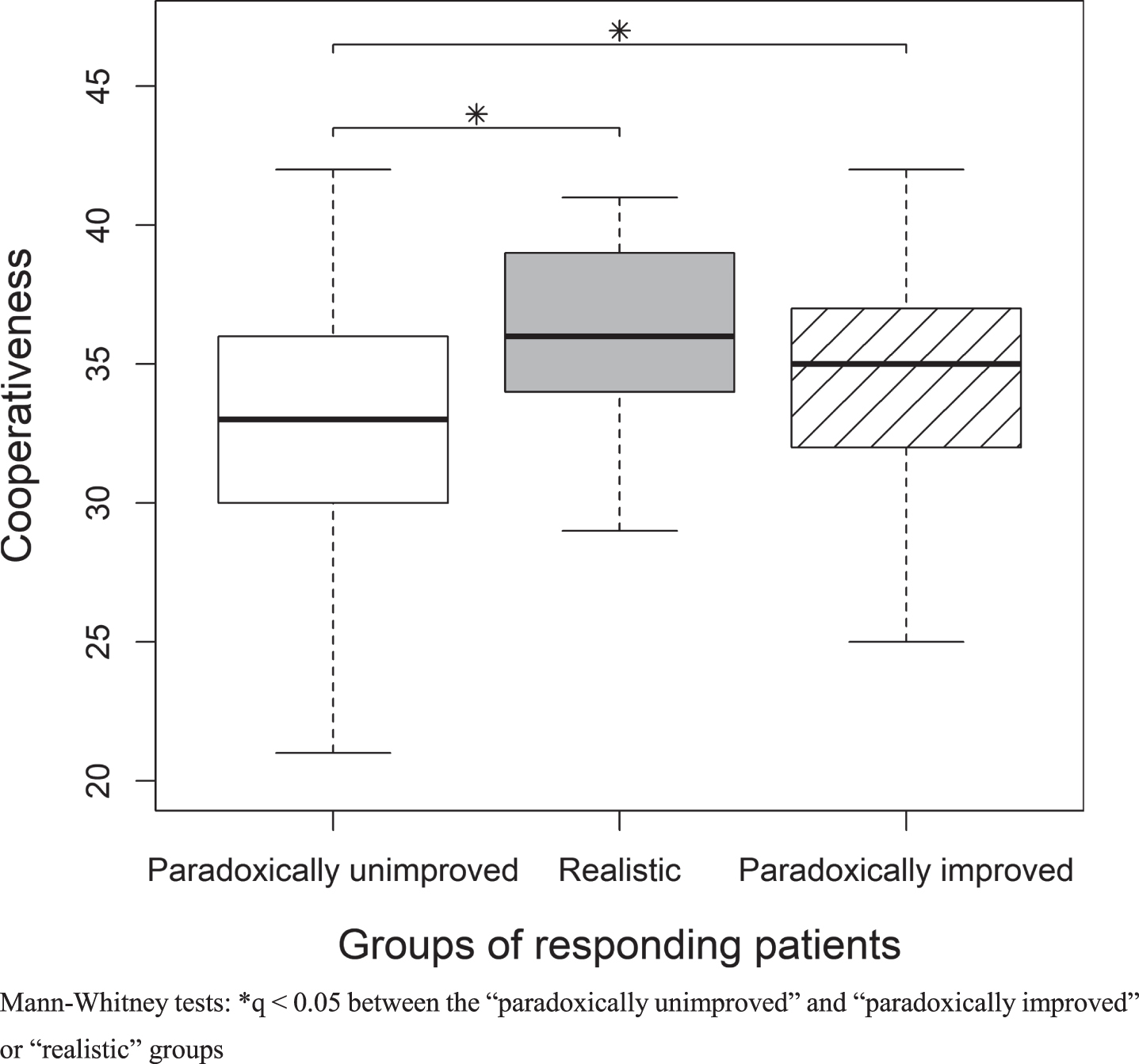
Differences in Cooperativeness scores between groups of responding patients.
Concerning the HAMA, HAMD, LARS, and MoCA changes, there were no significant differences between groups.
In this study, presurgical TCI personality dimensions such as higher Cooperativeness and Novelty Seeking sub-dimensions were associated with QoL improvement after one year of DBS-STN in PD population. This favorable outcome affected mainly the physical component of QoL (“Mobility” evolution). Secondly, patients with lower Cooperativeness scores were more paradoxically unimproved after DBS-STN: their objective motor complications improvement was not translated into QoL improvement; while patients with higher Reward Dependence scores were more paradoxically improved after DBS-STN: they had QoL improvement despite the absence of motor complications reduction.
According to the literature [1–3, 23], this study reported an improvement in MDS-UPDRS and PDQ-39 scores after one year of DBS-STN and a significant reduction of dopaminergic treatments. A slight amelioration of depression and anxiety was also shown after DBS, whereas apathy and global cognition slightly deteriorated while remaining below the threshold of apathy (LARS scale) and cognitive deficit (MoCA). Thus, DBS did not affect much behavior of our PD patients which globally did not develop either anxio-depressive state or apathy or cognitive decline.
First, we confirm the results of our previous study showing that personality dimensions are associated with different QoL sub-scores before DBS-STN in PD [16].
Second, we found associations between personality dimensions and QoL improvement after one year of DBS-STN. Some Cooperativeness and Novelty Seeking sub-dimensions (C1 (Social Acceptance), C4 (Compassion), and NS1 (Exploratory Excitability)) were strongly related to the physical component of QoL (“Mobility”): PD patients with high Cooperativeness and Novelty Seeking before DBS-STN had the best QoL improvement (physical component) after DBS-STN.
Cooperativeness is a character reflecting social maturity and described as a tendency to consider and accept others [24, 25]. PD patients with higher Cooperativeness probably better listen to their relatives and believe them, which can help them to remember their past difficulties and to realize the strong motor improvement they benefit from DBS-STN. Indeed, it is usual that PD patients forget their past difficulties after DBS and need a reminder from their relatives or caregivers to become aware of their improvements [26]. In literature, low Cooperativeness was associated with treatment-resistance in depressive patients [27], and high Cooperativeness was also linked with good QoL or life satisfaction in different populations [28–30]. Also, this hypothesis makes sense because Cooperativeness mostly impact physical QoL improvement after DBS, only part of QoL that can be assessed by patients’ relatives. Therefore, relatives probably have a more objective feeling on these improvements which may contrast with patients’ subjective one. Moreover, it is interesting to compare personality-QoL associations before and after DBS. Before, Self-Directedness (representing the individual maturity) was associated with better QoL [16]; whereas, after, Cooperativeness (social maturity) becomes associated with better QoL. This may reflect that, before surgery, PD patients were used to their disease and to their relatives’ comments because of adaptive process through chronic disease; whereas, after DBS, PD patients are confronted to abrupt changes [31] and motor improvement are often obvious. Thus, their relatives can appreciate these improvements and inform the patients, helping them to become more aware of these changes. Hence, before surgery, in case of a slowly evolving chronic disease, PD patients may rely more on their personal resources (individual maturity) to adapt and have a better QoL [16]; whereas, after DBS, they focus more on the external comments (social maturity) to adapt to the abrupt changes. It would thus be interesting to see the impact of Cooperativeness on QoL after long-time of DBS, when PD patients would have become used to these changes, with the PREDISTIM 5-year QoL evaluation.
Novelty Seeking is a temperament representing a tendency to respond positively and with excitation to new stimuli [24, 25]. It reflects good adaptability to new situations and motivation facing change. PD patients with higher Novelty Seeking may thus better adapt to the abrupt changes caused by DBS-STN and so really feel the strong motor improvement they benefit from the stimulation. In literature, higher Novelty Seeking was also associated with better global QoL and physical health in patients with schizophrenia [32].
Interestingly, Novelty Seeking and/or Cooperativeness are two personality dimensions potentially implicated in placebo responses [33–35]. Therefore, these two personality dimensions may enhance the part of placebo effect induced by DBS-STN [36]. For example, the sub-dimension C3 (Helpfulness) reflects solidarity and good level of team work which may enhance placebo effect [37].
Moreover, our study has shown good associations between the more objective evaluation of PD symptoms (MDS-UPDRS) and the subjective evaluation of PD QoL (PDQ-39). Mainly, the objective motor examination, MDS-UPDRS-III, as well as the motor complications assessed by the MDS-UPDRS-IV, were mostly associated with physical components of the PDQ-39 evaluation, assuring that both scales are reliable and evaluate PD patients in the same way [38]. Nonetheless, some PD patients (43.6%) had a feeling of evolution after DBS-STN that differs from the objective motor complications examination (MDS-UPDRS-IV). Indeed, eighteen patients (6.8%) felt “paradoxically improved” after DBS: they felt improvement of their QoL, whereas the MDS-UPDRS-IV showed no improvement of their motor complications. Also, ninety-seven patients (36.7%) felt “paradoxically unimproved” after DBS: they did not feel any improvement of their QoL, whereas the MDS-UPDRS-IV showed an improvement of their motor complications. This dissociation between reality of PD symptoms improvement after DBS and PD patients’ feelings has already been shown in several studies [5, 6] and can be explained by differences between patients’ expectations and reality, difficulties of psychosocial adjustment, mood and behavioral changes [26, 40], etc. Moreover, we have decided to focus our analysis on discrepancy between QoL evolution and MDS-UPDRS-IV change since DBS preferentially improves motor complications.
Here, paradoxically unimproved PD patients had lower Cooperativeness scores than realistic and paradoxically improved ones. This result seems to be congruent with our previous observation that lower Cooperativeness scores before surgery lead to less improvement of QoL after DBS-STN. Moreover, paradoxically improved PD patients had higher Reward Dependence scores than the paradoxically unimproved ones. Reward Dependence temperament is associated with sensitive, dependent and social individuals [41]. It is a system facilitating conditioning to reward stimuli [25] and therefore may be associated with placebo effect. Indeed, some studies have found associations between higher Reward Dependence scores and better outcomes after interpersonal therapy [42] or antidepressants [43]. Moreover, treatment adhesion was shown to be related to higher Reward Dependence temperament in a weight-loss therapy, probably thanks to better collaborative relationship with the clinicians [44]. Nonetheless, these differences between realistic, paradoxically improved or unimproved patients could not be explained by differences in cognitive or behavioral changes after DBS-STN.
This study has some limits. Being the first study on this subject, only analyses of associations between QoL outcomes and personality dimensions were done. Thus, our results are not predictive but rather suggestive. Hence, they should be used in order to prevent disappointment of PD patients after DBS-STN (not to select PD patients for DBS) acting in two ways: 1) by supporting them upstream and explaining them at best what they should expect of DBS, and 2) by preparing and encouraging their relatives to be really present with the patients after the operation to enhance their cooperation (which seems to facilitate their positive outcomes). Additionally, our models explaining less than ten percent of the variance (R2) of QoL improvement after DBS-STN, we are aware that personality dimensions are not enough to guaranty PD patients’ QoL improvement after stimulation and that other variable are needed to explain at best QoL outcome after surgery. However, our focus being concentrated on personality dimensions’ evaluation, we did not look for other potential predictive variables as it will be the aim of the main PREDI-STIM study. Finally, only personality dimensions before DBS were evaluated (there was no re-evaluation of personality after stimulation), and it remains possible that some personality dimensions evolve after DBS. Even if temperaments are not supposed to change with time [9], personality in PD patients seems to evolve with the disease course particularly with the introduction of dopaminergic treatments [45]. DBS could also impact personality as shown by some studies [46–50]. In any case, our aim was to find “predictive” factors of QoL improvement after DBS. Thus, even if personality changes after stimulation, it is not supposed to affect our results. Lastly, our three groups of responding PD patients (“paradoxically unimproved”, “realistic” and “paradoxically improved”) were done according to their motor complications improvement (reflected by MDS-UPDRS part-IV) since it is the main purpose of DBS-STN action. Nonetheless, improvements of non-motor symptoms (pain, fatigue etc.) were not accounted in these analyses, although non-motor symptoms can benefit of DBS-STN [51] and are best predictors of QoL in PD [52], because we were interested in more objective assessments than MDS-UPDRS parts I and II. Moreover, we are aware that using MCIDs to classify into groups of “improvement” or “no improvement” may not precisely reflect the evolution of patients after DBS, mainly since at this stage of motor fluctuations a change of 0.9 points in the MDS-UPDRS part-IV may not be sufficient to be clinically significant. Nonetheless, the choice of this MCID was done accordingly with literature. Ultimately, we are aware that there are other models of personality that could have been explore in this study to depict a more complete exploration of personality associated with QoL. Nonetheless, the TCI being a long questionnaire, we thought that using a multilayer approach (through different personality investigations) would have been too complex in clinical practice and too tiring for the PD patients.
In conclusion, PD patients with higher Cooperativeness and Novelty Seeking scores at baseline seem to have the best QoL outcome after one year of DBS-STN in the physical component of QoL. A tendency of being more trustful towards relatives and more motivated by changes could explained the ability of these PD patients to feel a better QoL improvement following the abrupt changes after surgery. Moreover, PD patients with low Cooperativeness were more paradoxically unimproved after DBS, whereas PD patients with high Reward Dependence were more paradoxically improved after DBS. Therefore, systematic TCI examination could be proposed as standard evaluation before DBS. Subsequently, according to PD patients’ scores at the Novelty Seeking and Cooperativeness dimensions, specific educational programs should be proposed to patients with higher risk of QoL unimprovement after DBS to prepare them at best for the stimulation. For this, we are now looking for cut-off scores for these dimensions.
Footnotes
ACKNOWLEDGMENTS
The study was funded by the France Parkinson charity and French Ministry of Health (PHRC national 2012). This is an ancillary study to Protocol ID: 2013-A00193-42; ClinicalTrials.gov: NCT02360683.
We thank all participants for their cooperation, and Déborah Meligne from ToNIC (Toulouse NeuroImaging Center–INSERM, University of Toulouse III) for her reflection. The authors are also grateful for support from the French clinical research network NS-Park/F-Crin and the Fédération de la Recherche Clinique du CHU de Lille (with Anne-Sophie Rolland, Alain Duhamel, Maeva Kheng, Julien Labreuch, Dominique Deplanque, Edouard Millois, Nolwen Dautrevaux, Victor Laugeais, Maxime Caillier, Aymen Aouni, Pauline Guyon, Francine Niset, Valérie Santraine, Marie Pleuvret, Julie Moutarde and Laetitia Thibault).
We also thank all the members of the PREDI-STIM study group: Dr Caroline Moreau, Pr Luc Defebvre, Dr Nicolas Carriere, Dr Guillaume Grolez, Dr Guillaume Baille, Dr Kreisler, Pr Jean-Pierre Pruvo, Pr Leclerc, Dr Renaud Lopes, Dr Romain Viard, Dr Gregory Kuchcinski, Mr Julien Dumont, Pr Kathy Dujardin, Mme M Delliaux, Mrs M Brion, Dr Gustavo Touzet, Pr Nicolas Reyns, Pr Arnaud Delval, Mrs Valerie Santraine, Mrs Marie Pleuvret, Mrs Nolwen Dautrevaux, Mr Victor Laugeais, Thavarak Ouk, Camille Potey, Celine Leclercq and Elise Gers (for Lille University Hospital); Jean-Christophe Corvol, Marie-Vidailhet, Elodie Hainque, Marie-Laure Welter, Lucette Lacomblez, David Grabli, Emmanuel Roze, Yulia Worbe, Cécile Delorme, Hana You, Jonas Ihle, Raquel Guimeraes-Costa, Florence Cormier-Dequaire, Aurélie Méneret, Andréas Hartmann, Louise-Laure Mariani, Stéphane Lehericy, Virginie Czernecki, Fanny Pineau, Frédérique Bozon, Camille Huiban, Eve Benchetrit, Carine Karachi, Soledad Navarro, Philippe Cornu, Arlette Welaratne, Carole Dongmo-Kenfack, Lise Mantisi, Nathalie Jarry, Sophie Aix and Carine Lefort (for the AP-HP, Paris); Dr Tiphaine Rouaud, Pr Philippe Damier, Pr Pascal Derkinderen, Dr Anne-Gaelle Corbille, Dr Elisabeth Calvier-Auffray, Mrs Laetitia Rocher, Mrs Anne-Laure Deruet, Dr Raoul Sylvie, Dr Roualdes Vincent and Mrs Le Dily Séverine (for Nantes University Hospital); Dr Ana Marques, Dr Berangere Debilly, Pr Franck Durif, Dr Philippe Derost, Dr Charlotte Beal, Carine Chassain, Laure Delaby, Tiphaine Vidal, Pr Jean Jacques Lemaire, Isabelle Rieu and Elodie Durand (for Clermont-Ferrand University Hospital); Pr Alexandre Eusebio, Pr Jean-Philippe Azulay, Dr Tatiana Witjas, Dr Frédérique Fluchère, Dr Stephan Grimaldi, Pr Nadine Girard, Eve Benchetrit, Marie Delfini, Dr Romain Carron, Pr Jean Regis, Dr Giorgio Spatola and Camille Magnaudet (for the AP-HM, Marseille); Dr Ansquer Solène, Dr Benatru Isabelle, Dr Colin Olivier, Pr Houeto JL, Pr Guillevin Remy, Mrs Fradet Anne, Mrs Anziza Manssouri, Mrs Blondeau Sophie, Dr Richard Philippe, Dr Cam Philippe, Dr Page Philippe, Pr Bataille Benoit, Mrs Rabois Emilie and Mrs Guillemain Annie (for Poitiers University Hospital); Dr Drapier Sophie, Dr Frédérique Leh, Dr Alexandre Bonnet, Pr Marc Vérin, Dr Jean-Christophe Ferré, Mr Jean François Houvenaghel, Pr Claire Haegelen, Mrs Francoise Kestens and Mrs Solenn Ory (for Rennes University Hospital); Pr Pierre Burbaud, Dr Nathalie Damon-Perriere, Pr Wassilios Meissner, Pr Francois Tison, Dr Stéphanie Bannier, Dr Elsa Krim, Pr Dominique Guehl, Sandrine Molinier-Blossier, Morgan Ollivier, Marion Lacoste, Nicolas Auzou, Marie Bonnet, Pr Emmanuel Cuny, Dr Julien Engelhardt, Olivier Branchard, Clotilde Huet and Julie Blanchard (for Bordeaux University Hospital); Pr Rascol Olivier, Dr Christine Brefel Courbon, Dr Fabienne Ory Magne, Dr Marion Simonetta Moreau, Pr Christophe Arbus, Pr Fabrice Bonneville, Dr Jean Albert Lotterie, Marion Sarrail, Charlotte Scotto d’Apollonia, Pr Patrick Chaynes, Pr François Caire and Estelle Harroch (for Toulouse University Hospital); Pr David Maltete, Dr Romain Lefaucheur, Dr Damien Fetter, Dr Nicolas Magne, Mrs Sandrine Bioux, Mrs Maud Loubeyre, Mrs Evangéline Bliaux, Mrs Dorothée Pouliquen, Pr Stéphane Derrey, Mrs Linda Vernon and Dr Frédéric Ziegler (for Rouen University Hospital); Mathieu Anheim, Ouhaid Lagha-Boukbiza, Christine Tranchant, Odile Gebus, Solveig Montaut, S Kremer, Nadine Longato, Clélie Phillips, Jimmy Voirin, Marie des Neiges Santin, Dominique Chaussemy and Dr Amaury Mengin (for Strasbourg University Hospital); Dr Caroline Giordana, Dr Claire Marsé, Lydiane Mondot, Bruno Giordana, Robin Kardous, Bernadette Bailet, Héloise Joly, Denys Fontaine, Dr Aurélie Leplus, Amélie Faustini and Vanessa Ferrier (for Nice University Hospital); Pr Pierre Krystkowiak, Dr Mélissa Tir, Pr Jean-Marc Constans, Sandrine Wannepain, Audrey Seling, Dr Michel Lefranc, Stéphanie Blin and Béatrice Schuler (for Amiens University Hospital); Pr Stephane Thobois, Dr Teodor Danaila, Dr Chloe Laurencin, Pr Yves Berthezene, Dr Roxana Ameli, Helene Klinger, Dr Gustavo Polo, Patrick Mertens, A Nunes and Elise Metereau (for Lyon University Hospital); Dr Lucie Hopes, Dr Solène Frismand, Dr Emmanuelle Schmitt, Mrs Mylène Meyer, Mrs Céline Dillier, Pr Sophie Colnat and Mrs Anne Chatelain (for Nancy University Hospital); Dr Jean- Philippe Brandel, Dr Cécile Hubsch, Dr Patte Karsenti, Dr Marie Lebouteux, Dr Marc Ziegler, Dr Christine Delmaire, Dr Julien Savatowky, Mrs Juliette Vrillac, Mrs Claire Nakache, Dr Vincent D’Hardemare and Mr Lhaouas Belamri (for the Rotschild fondation of Paris); Dr Philippe Graveleau, Dr Camille Decrocq, Dr Frédéric Bourdain, Dr Vadim Afanassiev, Dr Anne Boulin, Mrs Elodie Dupuy, Dr Bérénice Gardel, Pr Béchir Jarraya, Mrs Delphine Lopez and Mr Christophe Fruit (for the Foch Hospital of Paris-Saclay University); David Gay, Robin Bonicel, Fouzia El Mountassir, Clara Fischer, Jean-François Mangin, Marie Chupin and Yann Cointepas (for CATI (MRI acquisition management, preprocessing and data management)); Bertrand Accart, Patrick Gelé, Florine Fievet, Matthieu Chabel, Virginie Derenaucourt, Loïc Facon, Yanick Tchantchou Njosse and Dominique Deplanque (for CRB of Lille (Center of Biological Resources)); and Alain Duhamel, Lynda Djemmane and Florence Duflot (for Data management of Lille).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
