Abstract
Background:
Parkinson’s disease (PD) and subthalamic nucleus deep brain stimulation (STN-DBS) are both known to induce cognitive changes.
Objective:
The aim of our study was to investigate the impact of STN-DBS on two forms of conditional associative learning (CAL), trial and error or corrective feedback learning, which differed in difficulty to test the load-dependency hypothesis of the cognitive effects of STN-DBS in PD.
Methods:
We recruited two groups of PD patients, those who had STN-DBS surgery bilaterally (n = 24) and a second unoperated group (n = 9) who were assessed on two versions of a task of visual CAL involving either a more difficult trial and error learning or a relatively easier corrective feedback learning. Each task was completed twice by both groups, On and Off STN-DBS for the operated group and a first and second time by the unoperated group.
Results:
With STN-DBS Off, corrective feedback learning was superior to trial and error CAL, but not with STN-DBS On. The unoperated PD group had improved performance during the second assessment. To control for the improvement observed with repeated assessment in the PD control group, we split the STN-DBS group into two subgroups based on the condition of the first assessment (Off first vs. On first). While we found no STN-DBS effects for the Off first subgroup (N = 14), we observed improved performance during the second STN-DBS Off session for the On first subgroup (N = 10).
Conclusion:
The findings suggest that in PD, STN-DBS interferes with use of corrective feedback and its integration in the conditional associative learning process. Also STN stimulation affected the ability of operated patients to resolve proactive interference during learning of the arbitrary visual associations by trial and error or with corrective feedback.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a neurodegenerative movement disorder primarily with motor symptoms. However, attention is increasingly paid to the non-motor signs, and especially to cognitive impairment which is already present in about 30% of the patients during early disease stages, usually in the form of executive dysfunction [1–3]. Among the cognitive deficits in PD, dysfunction in conditional associative learning has also been documented [4]. Treatment of PD targets the motor symptoms and initially involves dopamine-replacing substances, including levodopa and dopamine agonists [5]. When patients develop long-term motor complications and side effects of medication, they are, if eligible, treated surgically with deep brain stimulation (DBS), especially of the subthalamic nucleus (STN) [6, 7]. While STN-DBS has been shown not to result in global cognitive impairment [8, 9], deficits in specific cognitive domains have been documented. These include STN-DBS related deficits in verbal fluency which could be due to a surgical effect [10] even if the exact mechanism is not fully understood, deficits in inhibitory control on tests such as the Stroop [11, 12], load-dependent deficits in action restraint on go no go RT tasks [13, 14] and similar load-dependent deficits in switching from automatic to controlled processing on a random number generation task [15]. Furthermore, in PD, STN stimulation enhances responsiveness of movement initiation speed to high reward value [16] and could induce impulsivity on decision-making tasks which involve reward or response selection conflict [17, 18] or when acting under time pressure of speed instructions [19], whereas no deficits are found when decision-making does not involve conflict, time pressure or reward [20]. The results regarding the effects of STN-DBS on motor inhibition in stop signal tasks are inconsistent; with some reporting delayed inhibition on a conditional stop signal RT task [21], whereas others have found no change [22] or improvement of stop signal reaction time [23, 24] with stimulation.
Among the cognitive functions impacted by both PD and STN-DBS, research on conditional associative learning (CAL) has reported inconsistent results. In PD, no difference between early stage patients and healthy controls on a standard trial and error CAL has been reported [25] whereas others found impaired performance from de novo to advanced disease stages [26, 27]. Nevertheless, the study which found no difference [25] also reported a minority of older patients who did have impaired task performance. One study compared CAL with two different learning instructions in PD [28]. One version of the CAL task involved a typical trial and error learning instruction; whereas the second had a feedback learning instruction such that participants were initially shown the association pairs. Their results highlighted that PD patients were only impaired on the trial and error learning version of the task, attributed to the integrity of the fronto-striatal network.
There is also some research on the effects of STN-DBS surgery on CAL performance. Compared to pre-operative performance, after STN-DBS surgery, increased errors and trials to criterion, were observed, indicating worse learning on the trial and error CAL [29, 30]. Regarding the effects of acute STN-DBS, the findings are inconsistent. On the one hand, it has been reported that on a trial and error CAL task requiring learning of six arbitrary associations PD patients with STN-DBS On produced more errors and required more trials to reach criterion in comparison to the DBS Off state [11]. On the other hand, other studies suggested that CAL with arbitrary associations was improved On compared to Off STN-DBS [31, 32]. However, the latter studies used a CAL paradigm that involved a low cognitive load, which suggests that the effect of STN-DBS on CAL may be load-dependent. Such load-dependency of cognitive deficits with STN stimulation is suggested by other studies using other tasks [13–15].
Previous studies highlighted the importance to differentiate CAL from standard paired-associate paradigms and classical conditioning or discrimination learning paradigms [33]. During paired associate learning (PAL) the correct stimulus pairings are initially presented together thus enhancing the strength of the associations. In addition, the difficulty could be modulated by using non-arbitrary and easy (e.g., baby-cries) or arbitrary and hence difficult (e.g., cabbage-pen) words pairs. By contrast, on standard CAL tasks all arbitrary stimulus-response associations are learned by trial and error, and participants must learn a conditional rule, where they must select a different response for each stimulus. The ‘corrective feedback’ version of the CAL, as used here, is similar to PAL since it also involves initial presentation of the correct but arbitrary stimulus pairings and participants are required to only make one response on each trial and are provided with corrective feedback in case of an error.
To date, there has been no study investigating the ‘load-dependency’ hypothesis of the effects of STN-DBS on CAL by comparing how acute STN-DBS influences trial and error and feedback learning versions of this task, with the former making greater demands on cognitive resources during learning and hence involving greater cognitive load. The aim of our study was to investigate the effect of STN-DBS On vs. Off on the trial and error CAL and corrective feedback CAL both using arbitrary stimulus associations and on a verbal PAL with easy and hard associations. Our prediction was that relative to STN-DBS Off, STN stimulation would impair learning on the trial and error CAL and perhaps verbal PAL with difficult associations but not on the feedback CAL or verbal PAL with easy associations; thus, revealing load-dependent effects of stimulation on associative learning.
MATERIALS AND METHODS
Participants
All participants were recruited between 2000 and 2005 and had a clinical diagnosis of Parkinson’s disease according to the UK Brain Bank criteria [34]. As part of the pre-operative screening, the Unified Parkinson’s disease Rating Scale (UPDRS) [35] was used to rate the disease severity and to evaluate the pre-operative responsiveness of all patients to levodopa. UPDRS ratings were also obtained before and after surgery to assess the clinical effects of STN-DBS on the motor symptoms (see Table 1 for the latter). All candidates for surgery, including the unoperated patients on the waiting list, completed a full neuropsychological assessment as part of the screening for DBS-STN surgery. This neuropsychological assessment included the exclusion of dementia with the Dementia Rating Scale-2 and clinical depression with the Beck Depression Inventory. In addition, as part of this study, the Mini-Mental State Examination was used to ensure that the group of patients who had had STN-DBS had not developed any cognitive impairment post-surgery. All operated patients had quadripolar stimulating electrodes (Medtronic, MN, USA) chronically implanted into the STN bilaterally. Twenty-four patients who had bilateral STN-DBS for at least six months (when the acute effects of surgery on the brain have dissipated) and nine unoperated control patients were recruited. Patients were assessed On medication. The group of unoperated patients was recruited from the surgery waiting list with the objective to be as comparable as possible with the operated group regarding their age, disease duration and disease severity. Table 1 shows the demographics and clinical features of both groups. Table 2 shows the stimulation settings for the operated patients. This study was approved by the joint Ethics committee of the UCL Queen Square Institute of Neurology and the National Hospital for Neurology & Neurosurgery and all patients gave informed consent.
Demographic and clinical features of all participants
DBS, deep brain stimulation; F, female; L, left; M, male; MMSE, Mini-Mental State Examination; NA, not applicable; R, right; UPDRS, Unified Parkinson’s Disease Rating Scale.
Stimulation settings for patients with STN-DBS
Hz, frequency; L, left; R, right; V, amplitude; μs, pulse width.
Design and procedure
A 2 (DBS vs. unoperated group)×2 (assessment: DBS On vs. Off or first vs. second) mixed between groups-within subject design was used. All patients were assessed on all three associative learning tasks twice. The operated group was assessed once with their stimulation switched On and a second time with the stimulation switched Off. The order of the stimulation condition was counterbalanced across patients. The Off and On assessments were performed at least half an hour after switching the stimulators Off or On. This time interval is long enough to allow effects of stimulation to be established (On) or dissipate (Off; [36]). The control group was also tested twice to control for the effects of repeated administration and fatigue of the learning tasks.
Tasks
Visual conditional associative learning tasks
The Visual CAL task (VCLT) [4, 37] was administered to assess conditional associative learning of arbitrary non-verbal information. During the task participants were required to learn arbitrary associations between 6 abstract geometric designs and 6 colors (Fig. 1A) within a maximum of 12 blocks. The test material consisted of six cards each showing one color and six cards each showing all six designs presented in random order on each card. Four different sets of abstract designs were used (previously shown to be parallel and equivalent in difficulty in healthy controls); with the order of the two versions of the CAL test and the order of the design sets being counterbalanced across participants.
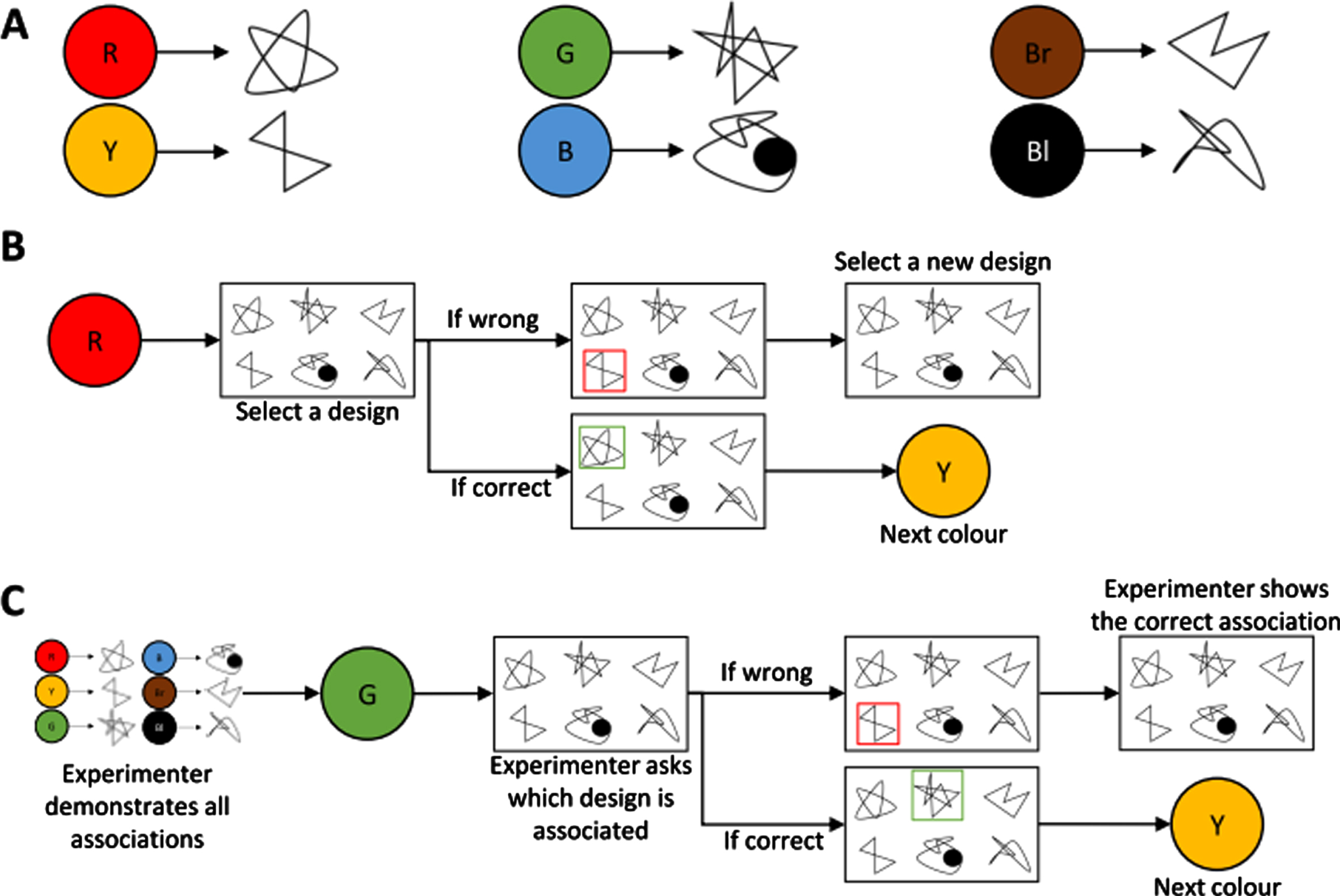
The trial and error and feedback learning versions of the visual conditional associative learning task. Part A represents example associations for one set of abstract designs. Part B represents an example sequence for learning by trial and error. Part C represents an example sequence for learning by corrective feedback. For illustration purposes the same designs are used for both the trial and error and corrective feedback versions of the task. However, in the actual study, different abstract designs were used for the two versions. For black-and-white version: R = Red; Y = Yellow; G = Green; B = Blue; Br = Brown; Bl = Black.
Trial and error learning version (Fig. 1B). During every block of trials, the participants were presented with each color in a predetermined order and asked to indicate which design they thought was associated with that color. For each selection, participants were told if they were correct or not. If the selection was wrong the participants continued to choose other designs until they found the correct association. The task was discontinued when the criterion of two consecutive blocks of trials correct was achieved or 12 blocks were completed.
Corrective feedback learning version (Fig. 1C). The participants were initially shown each color and the design that was associated with it. Following this familiarization period, participants were presented on each trial with one of the six colors in a predetermined order and asked to indicate which design they remembered as being associated with that color. If the selection was correct the next trial was initiated and if it was incorrect the participant was told the correct selection and one error was scored. This was continued until the criterion of two consecutive blocks of trials correct was reached or alternatively until 12 blocks of trials were completed.
The number of errors, the number of trials that were correct first time, the total number of trials and the number of blocks to criterion were recorded. To make the two versions of the task comparable, the scoring scheme previously proposed was used [28]. Therefore, for the total number of trials and errors the first error was counted only.
Paired verbal associative learning task
The Verbal Paired Associates subtest from the Wechsler Memory scale-III [38] was used to assess associative learning of verbal information. The task consisted of six trials. For each trial participants had to listen to eight pairs of associated words, four pairs consisted of related words (easy items: e.g., baby-cries or rose-flower) and four pairs consisted of unrelated words (hard items: e.g., school-grocery or cabbage-pen). After listening to the list of word pairs being presented by the experimenter, participants were presented with the first word of each pair and asked to provide the word that was associated with the given word. This was repeated until they either learned all associations or after six trials had been reached. The number of correct answers and the number of trials until all items were learned were scored for easy and hard items separately.
Statistical analyses
Statistical analyses consisted of Wilcoxon signed rank tests and series of Mann-Whitney U tests because the measures were not normally distributed. We considered (i) effects of acute STN stimulation in the operated group and the practice effects in the PD control group, (ii) differences between the STN-DBS and PD control group, and (iii) differences between the trial and error learning and the feedback learning versions of the VCLT. For changes that reached statistical significance Cohen’s d was calculated to evaluate the robustness of the change. An effect size of 0.2 is considered a small effect, of 0.5 a moderate effect and of 0.8 a large effect [39]. Regarding demographic and clinical data analyses, we computed paired and unpaired t-tests.
RESULTS
Patients in the operated and unoperated groups were matched in terms of severity of motor symptoms as measured by UPDRS-III, age, disease duration and years of education (p > 0.05).
A paired t-test showed that operated patients had significantly lower UPDRS-III when STN stimulation was On compared to when stimulation was Off (t (23) = –5.7; p < 0.001) confirming the efficacy of STN-DBS for improving their motor symptoms.
Trial and error versus corrective feedback learning of the VCLT
To test the ‘load-dependency’ hypothesis, the effects of STN-DBS On vs. Off on the trial and error and corrective feedback versions of the VCLT were compared. A similar analysis was completed for the two assessments of the PD control group.
When patients with STN-DBS were assessed On stimulation a series of Wilcoxon signed rank tests revealed a significant difference in the total number of trials between the two versions of the task (Z = –2.18; p = 0.029; d = 0.49). The patients required more trials to learn associations on the trial and error learning compared to the feedback learning version of the task (see Fig. 2). The other measures did not differ between the two tasks (all p > 0.05). When the STN-DBS patients were assessed Off stimulation, a series of Wilcoxon signed rank tests revealed significant differences in all measures between the trial and error and feedback learning versions of the task, showing that the patients required more trials (Z = –3.22; p = 0.001; d = 0.82), produced more errors (Z = –2.38; p = 0.017; d = 0.29), had fewer trials correct first time (Z = –2.07; p = 0.039; d = –0.22) and required more blocks to criterion (Z = –4.29; p < 0.001; d = 0.62) on the trial and error learning compared to the feedback learning version of the task. (see Fig. 2).
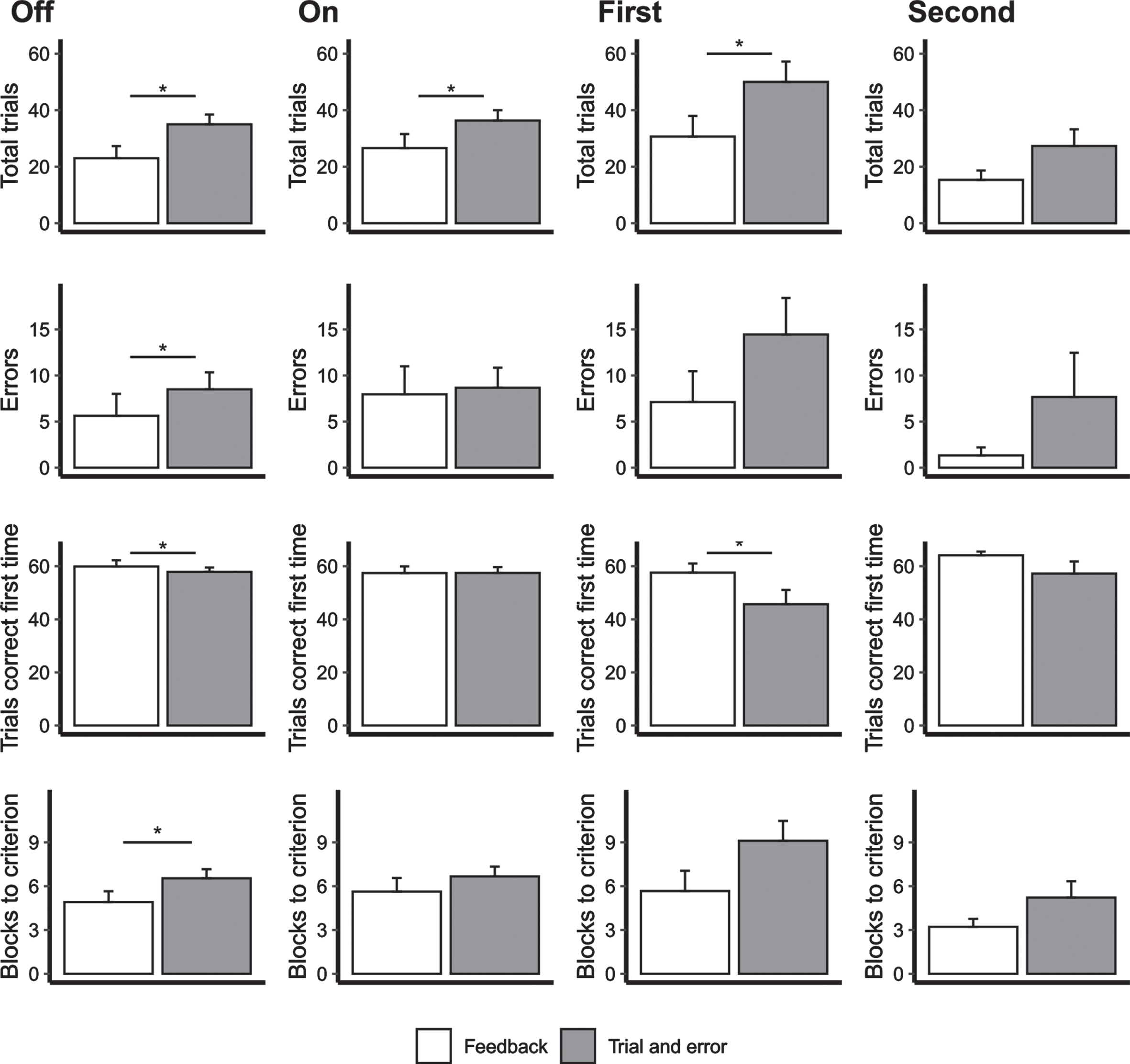
The mean number of total trials, errors, trials correct first time and blocks to criterion on the two versions of the visual conditional associative learning task for the deep brain stimulation group both Off (first column) and On DBS (second column) and for the PD controls for the first (third column) and second assessments (fourth column). Error bars represent standard errors. *p < 0.05.
For the PD control patients, during the first assessment, the total number of trials (Z = –2.410; p = 0.016; d = 0.922) were significantly higher and the number of trials that were correct first time (Z = –1.956; p = 0.050; d = –1.027) were lower for the trial and error learning version than the feedback learning VCLT. A higher number of blocks to criterion (Z = –1.85; p = 0.064; d = 0.72) at the first assessment and a higher total number of trials (Z = –1.87; p = 0.062; d = –0.54) at the second assessment for the trial and error learning than the feedback version approached significance. The remaining comparisons were not significant (all p > 0.05; see Fig. 2B).
Effect of STN-DBS on the VCLT compared to PD controls
For the unoperated PD control patients, we found differences between the second assessment compared to the first assessment. For the trial and error learning version of the task (Fig. 3A), the second assessment was associated with a lower number of trials (Z = –1.99; p = 0.046; d = 0.89) and a lower number of blocks to reach criterion (Z = –1.99; p = 0.046; d = 1.28). For the feedback learning version (Fig. 3B), the second assessment was associated with fewer errors (Z = –2.02; p = 0.043; d = 0.67) and a decrease in the number of blocks to criterion that approached significance (Z = –1.89; p = 0.06; d = 0.65). By contrast, the effects of acute STN stimulation on either of the two versions of the VCLT were not significant for the group as a whole (p > 0.05; Fig. 3).
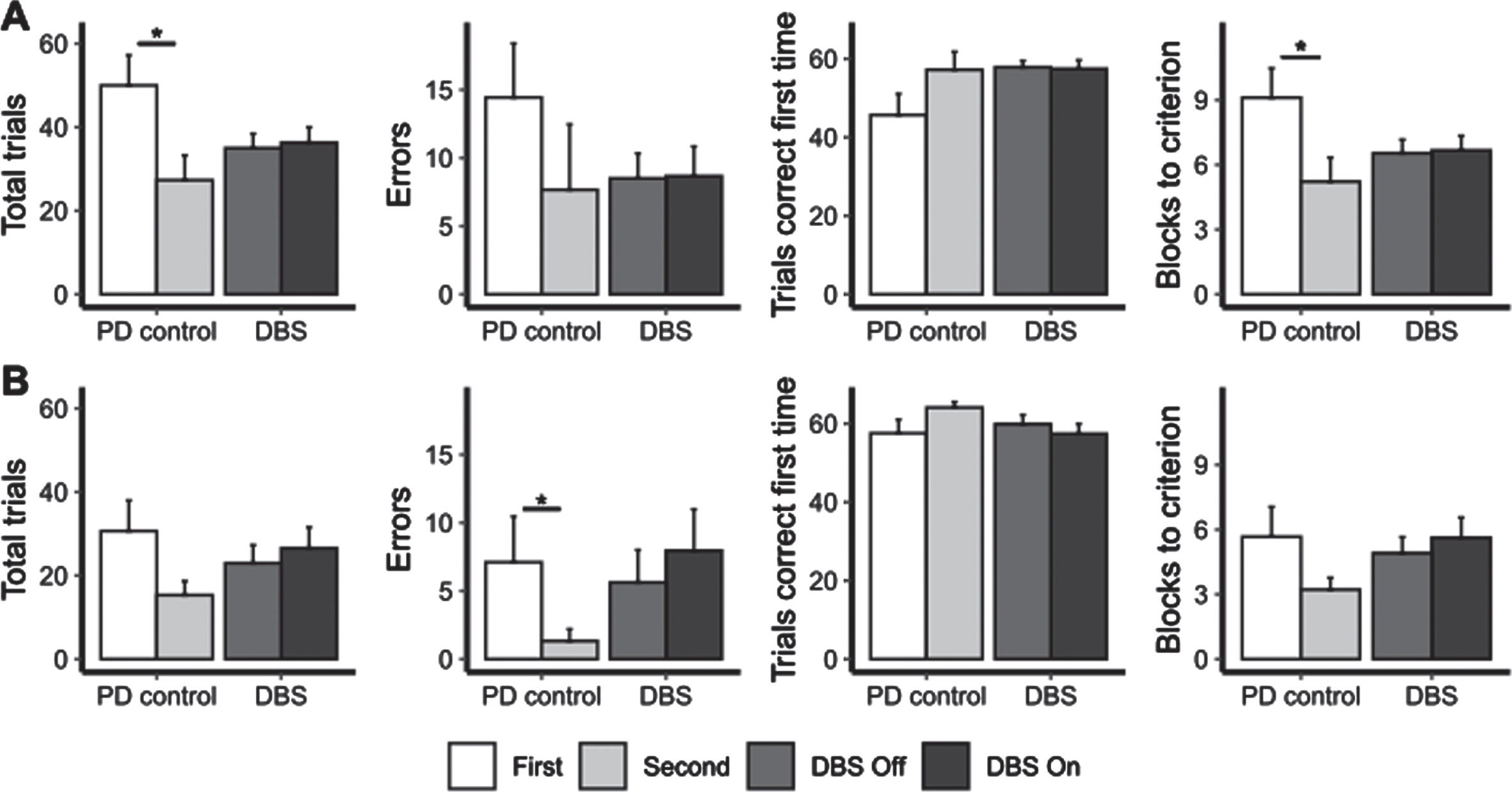
The mean number of total trials, errors, trials correct first time and blocks until criterion was reached on the trial-and-error (part A) and feedback (part B) versions of the visual conditional associative learning task for the first and second assessments of the unoperated patients and with STN-DBS Off and On for operated patients. Error bars represent standard error. * represents significant comparisons.
To control for the improvement with the repeated administration of the VCLT that was observed in the PD control group, the above analysis was repeated separately for those patients who completed the assessment On stimulation first (N = 10) and Off stimulation first (N = 14). We observed differences in the measures of the trial and error and feedback learning versions of the VCLT only for patients who was assessed On stimulation first.
For the trial and error learning version (Fig. 4A), the patients who were assessed On stimulation first had fewer trials (Z = –1.97; p = 0.05; d = 0.82), fewer errors (Z = –2.31; p = 0.02; d = 0.82), more trials correct first time’ (Z = –2.32; p = 0.02; d = –0.82) and a tendency for lower number of blocks to criterion (Z = –1.79; p = 0.07; d = 0.71) when stimulation was Off (i.e., for the second assessment) compared to when DBS was On.
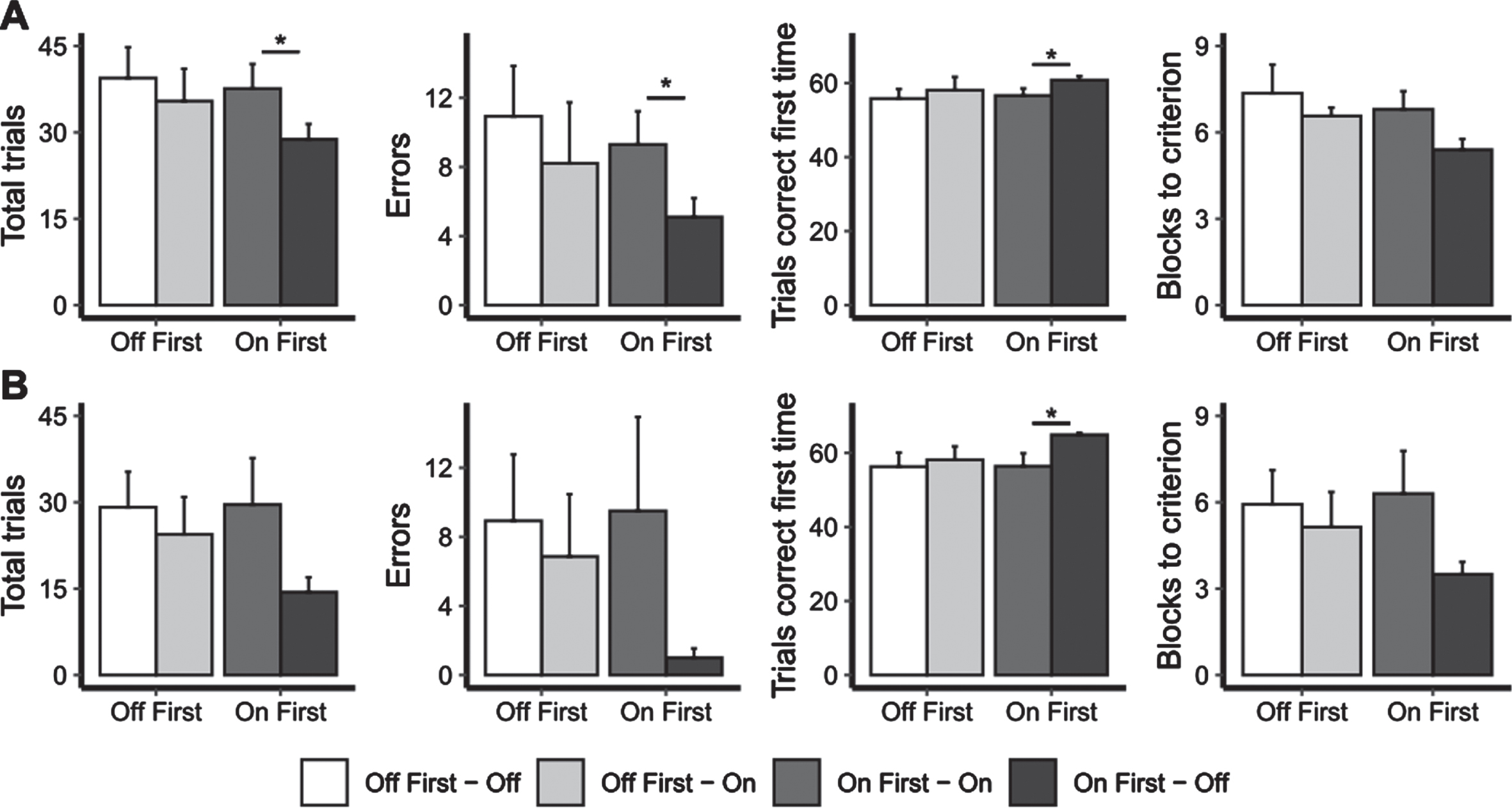
The mean number of total trials, errors, trials correct first time and blocks until criterion was reached on the trial-and-error (part A) and feedback (part B) versions of the visual conditional associative learning task for the DBS First Off and DBS First On groups, both with STN-DBS Off and On. Error bars represent standard error. * represents significant comparisons.
For the feedback learning version (Fig. 4B), the patients who were assessed On stimulation first had a lower number of trials correct first time when stimulation was Off compared to when it was On (Z = –1.96; p = 0.05; d = –0.93). The other measures also showed change in the same direction, better performance with stimulation Off compared to On but the differences were not significant because of individual variability.
The analyses did not reveal any differences in any of the measures of the VCLT between the conditions DBS On first, DBS Off first and the PD control group either at the first or second assessments (p > 0.05).
Paired verbal associative learning task
Statistical analyses revealed no changes on the four measures (i.e., number of correct answers and number of trials until all items were learned, for either easy or hard items) between the On and Off stimulation assessments for the operated group or between the first and second assessments for the unoperated control group (p > 0.05). In addition, we found no differences between the STN-DBS group and the PD control group in any of the PAL measures.
DISCUSSION
STN-DBS interferes with use of corrective feedback in the conditional associative learning process
Based on previous studies [11, 40] and on the ‘load-dependency’ hypothesis, we predicted that STN-DBS would have a negative effect on the more cognitively demanding CAL by trial and error, based on the assumption that this form of learning would be sensitive to altered output from the basal ganglia to the frontal cortex [41], whereas feedback learning would not be [28]. For the STN-DBS group the effect of the learning instruction differed between the On and Off stimulation assessments. When stimulation was switched On, patients with STN-DBS required fewer trials for completing the feedback learning compared to the trial and error learning version of the task with the other measures showing no differences. Whereas when stimulation was switched Off, learning and performance was superior on all measures for the feedback relative to the trial and error version of the task. Thus, they required fewer trials and fewer blocks to criterion, produced fewer errors and had more trials correct first time for the feedback than the trial and error CAL when the STN stimulation was Off. These findings suggest that STN-DBS diminishes the previously described effect of learning instruction and the advantage of the corrective feedback over trial and error learning [28]. Furthermore, it suggests that STN stimulation interferes with the patients’ ability to use and integrate corrective feedback to guide learning. Thus, these results support a ‘load-dependency’ of STN-DBS effects in the sense that only with stimulation Off the operated patients showed the superiority of feedback over trial and error CAL and more specifically STN stimulation seems to interfere with the use and integration of corrective feedback during CAL.
STN-DBS impairs visual conditional associative learning through proactive interference
The unoperated PD control group improved on several measures for both versions of the VCLT from the first to the second assessment indicating a practice effect as a result of repeated administration. To control for such a potential practice or order of testing effect in the operated group, patients were separated into two subgroups, those who were tested with stimulation ‘On First’ and those who were tested with stimulation ‘Off First’. Interestingly when the effects of STN-DBS were analyzed for the two groups separately, there were several significant differences between the On and Off stimulation assessments for the group that was tested ‘On First’, while no significant effects were found for the ‘Off First’ group. On the trial and error learning version of the VCLT, patients required more trials to criterion, produced more errors and had fewer first correct trials when stimulation was On compared to when stimulation was switched Off. On the feedback learning version of the task, patients had fewer first correct trials when they were tested On stimulation compared to when stimulation was switched Off. It may be suggested that these findings reflect a practice effect only for the group of patients who were tested ‘On First’, whose learning improved on all the measures when assessed on the second occasion Off stimulation. Conversely, they may also be the result of increased proactive interference induced by STN-DBS when patients were assessed with the stimulation On. This would be consistent with evidence of increased proactive interference, earlier learning interfering with subsequent learning, with STN stimulation On compared to Off in PD patients [42] as well as previous evidence of increased proactive interference following STN-DBS operation [29] or pallidotomy [43] in PD patients compared to before surgery. As the STN has been largely described as playing a role in inhibitory processes [44–46], this influence could also be generalized to the interference of a prior learning with the next one, corresponding to a proactive interference, especially for learning involving a high load-dependency. Studies implementing functional imaging to identify brain structures underlying the resolution of proactive interference for such tasks reported frontal lobe involvement [47], and especially an increased activation in the left mid-ventrolateral prefrontal cortex [48–54], but also an association between the right STN and the caudate nucleus, left inferior frontal cortex and bilateral frontopolar cortex [55]. This is consistent with previous evidence of increased proactive interference following STN-DBS surgery [29] or pallidotomy [43] in PD patients. Taking the above findings into account when interpreting the present results, it may be argued that acute STN-DBS interrupted the process of resolving proactive interference or increased the level of proactive interference when learning the VCLT by trial and error or corrective feedback for those assessed with stimulation ‘On First’. By contrast, there was no STN-DBS effect on the VCLT in the subgroup assessed ‘Off First’, possibly because the increased proactive interference induced by the On stimulation condition performed second, cancelled out the advantages of any improvement of performance relating to repetition of task performance during the second assessment.
Our findings contribute to the ongoing debate on the effects of STN-DBS on VCLT since previous studies reported both impaired [11] and improved [31, 32] CAL with STN-DBS On. In light of these and the current results, we hypothesize that STN-DBS impairs the ability to learn new material when this new learning involves a high cognitive ‘load’ and when learning this new material occurs close in time to a previous learning.
These results suggest that STN-DBS interferes with the ability to benefit from corrective feedback to guide learning which may influence the outcome of speech therapy or cognitive behavioral therapy following STN-DBS surgery. Deficits in CAL only occur when there is a high cognitive load and when learning is disrupted through enhanced proactive interference when the stimulators are On. This suggests that during adjunct therapies such as speech therapy involving sequential learning, cognitive load should be minimized and learning episodes sufficiently spaced in time to prevent proactive interference of earlier with subsequent learning.
The effects of acute STN stimulation on verbal paired associative learning
Analysis of the effects of acute STN stimulation on the PAL task did not reveal any significant changes in the number of correctly named associates between the On and Off stimulation assessments for either the hard or the easy items. There was also no change for the PD control group in the number of correctly learned associations between the first and second assessments.
We predicted a negative effect of acute STN-DBS on the number of correctly named hard associates only. This prediction was made based on research on the effects of acute stimulation on several aspects of cognition that found detrimental effects on performance for tasks that require higher levels of cognitive control [11, 56–60], whereas similar tasks requiring less cognitive control remained unchanged or improved with stimulation On relative to when stimulation was Off.
The reasons for the absence of the predicted effect in the present results may relate to the nature of the task. On the PAL, the patients’ learning was externally driven as paired associates were presented several times. Research into the effects of STN-DBS on both paired associative conditional learning and language function separately reported detrimental effects of STN stimulation only if patients had to generate responses internally [11]. The present findings suggest that the PAL was not subject to proactive interference as it might have been expected from the results on the VCLT mentioned above. This may be explained by the fact that the parallel forms of the PAL that were used included associates that were semantically unrelated. Proactive interference usually arises when new information has features similar to previously learned information [49, 51–53]. One last point pertinent to the present results may be the brain activity underlying PAL. Functional imaging studies reported medial temporal lobe activation when participants learned verbal associate pairs [61]. Research implementing single neuron recordings reported activity of neurons in the amygdala, entorhinal cortex and hippocampus during such tasks [62]. Considering that acute STN stimulation mainly relates to activity changes in the frontal lobe [63], it may not lead to changes in word-pair learning.
Limitations
The present study had some limitations. First, despite use of “parallel” forms of the VCLT, it might have been better to perform the two assessments for each group on different days to avoid fatigue and control for eventual learning effects. However, some of the patients came from outside London and it would have been inconvenient for them to have to travel to the hospital twice. Second, the PD control group was less than half the size of the operated group, making comparisons more difficult. However, the two groups were matched in terms of disease duration and other pertinent clinical factors. Third, the Levodopa equivalent dose was not matched in the two groups, but is not likely to have influenced the results since both groups were assessed On medication, with a stable dopamine medication throughout the study. STN-DBS was the only parameter to have been changed.
CONCLUSIONS
In conclusion, this study was the first to investigate the effects of acute STN-DBS on CAL under different learning instructions. With STN-DBS Off, corrective feedback learning was superior to trial and error CAL, but not with STN-DBS On; which suggests that STN stimulation may interfere with the patients’ ability to use corrective feedback during CAL. There also seems to be an effect of STN stimulation on the patients’ ability to resolve proactive interference. There was no effect of STN stimulation on the verbal PAL task, suggesting no effect on externally driven verbal associative learning.
Footnotes
ACKNOWLEDGMENTS
This work was supported by an NIH R01 grant (NS40862-02) to MJ.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
