Abstract
Background:
The satisfaction with life and, in particular, with treatment in Parkinson’s disease (PD) is understudied.
Objective:
To explore a new 7-item rating tool assessing satisfaction with life and treatment (SLTS-7) in PD.
Methods:
In this cross-sectional, multi-center study, including patients screened for advanced therapies, psychometric characteristics of the SLTS-7 were analyzed. An exploratory factor analysis identified the underlying factorial structure of the SLTS-7.
Results:
117 patients were included, and the data quality of the SLTS-7 was excellent (computable data 100%), and acceptability measures satisfied standard criteria. Besides the global assessment (item 1), the exploratory factor analysis produced item 2 (physical satisfaction) as an independent item and two factors among the remaining items: items 3–5 (psycho-social satisfaction), and items 6 and 7 (treatment satisfaction). Cronbach’s alpha was 0.89, indicative of high internal consistency. The SLTS-7 total score correlated moderately with motor symptoms and weakly with non-motor symptoms total scores. SLTS-7 showed the highest correlations with the European Quality of Life with 5 items (EQ-5D) visual analog scale (0.43–0.58, p < 0.01), indicating a moderate convergent validity. The SLTS-7 significantly increased with higher non-motor symptoms burden levels (p = 0.002).
Conclusion:
Life satisfaction in PD covers three specific aspects, namely physical, psycho-social, and treatment satisfaction. The new SLTS-7 is a valid, reliable, and easy-to-use tool to assess satisfaction with life and treatment in patients with PD screened for advanced therapies. Longitudinal studies analyzing the effect of advanced PD treatment on life and treatment satisfaction are warranted.
Keywords
INTRODUCTION
Quality of life (QoL) is one of the most critical patient-reported outcome measures to evaluate treatment efficacy in patients with advanced Parkinson’s disease (PD) [1–3]. Clinical studies mainly assess QoL using the disease-specific Parkinson’s disease questionnaires (PDQ) or the more generic European Quality of Life Questionnaire with 5 dimensions (EQ-5D) [4–7]. However, these tools do not consider the concept of “life satisfaction” which has been described as a relevant component to assess QoL holistically [8]. “Life satisfaction” is regarded as “the degree to which a person positively evaluates the overall quality of his/her life as a whole; in other words, how much the person likes the life he/she leads” [9]. Therefore, life satisfaction covers the cognitive aspects of the well-being of a person triggered by subjective factors rather than external circumstances, as it has been reported for QoL [5, 11]. To date, the literature on life satisfaction in PD remains sparse [12–14].
Ambrosio and colleagues developed and validated the 6-item Satisfaction with Life Scale (SLS-6) in a PD population [12]. This scale surveys life satisfaction overall and in five specific areas: physical health, psychological well-being, social relations, leisure, and financial situation. However, the SLS-6 does not consider aspects of satisfaction with treatment, an essential component of managing patients with PD which is likely to impact life satisfaction. Therefore, understanding the factors that contribute to both satisfaction with life and treatment is critical for improving care and treatment of patients with PD, particularly in advanced stages of PD [13].
Consequently, the main objective of our study was to evaluate the psychometric characteristics of a new tool modified from the SLS-6 that includes aspects on satisfaction with treatment, the so-called Satisfaction with Life and Treatment Scale-7 (SLTS-7). We hypothesized that the SLTS-7 is a valid and reliable tool to assess satisfaction with life and treatment in patients with PD.
MATERIALS AND METHODS
Study design and ethical approval
The analysis was performed cross-sectionally as part of a prospective, observational, multi-center study including patients with PD who were screened for the eligibility of advanced therapies, including deep brain stimulation, apomorphine, and intrajejunal levodopa infusion [15]. Consecutive patients were included between 2013 and 2020 in Cologne and Marburg. All patients gave written informed consent before study inclusion. The study was carried out following the Declaration of Helsinki and protocols approved by local ethics committees (Study numbers Cologne 12/145 and Marburg 155/17) [16].
Participants and clinical assessments
PD was diagnosed according to UK Brain Bank criteria and patients were screened for advanced therapies according to International PD and Movement Disorders Society and national neurological society guidelines [17, 18]. Indication evaluations for advanced treatments were conducted by multi-disciplinary teams comprised of movement disorder neurologists, stereotactic neurosurgeons, neuropsychologists, neuropsychiatrists, and speech therapists [19, 20]. All assessments were conducted in the clinical MedON state.
The following scale was assessed as the primary outcome parameter:
Satisfaction with life and treatment Scale-7
The SLTS-7 is a modified version of the patient-completed SLS-6. Importantly, two questions on satisfaction with treatment were added, and item 6 of the original SLS-6 addressing the financial situation was excluded as previous research in PD has shown that the financial situation has a weak association with life satisfaction [12]. Therefore, the SLTS-7 is a seven-item scale for self-evaluation of the satisfaction with life as a whole (item 1) that assesses five specific areas: physical health (item 2), psychological well-being (item 3), social relations (item 4), leisure (item 5), and additionally, PD treatment (item 6) and expectations met about treatment (item 7). In items 1–6 the stem question is: “All things considered: How satisfied are you with . . . ?” In more detail, in item 6 the question is: “All things considered: How satisfied are you with your Parkinson treatment so far?” Item 7 reads: “All things considered: Does the treatment so far meet your expectations?” [12] Patients were asked to mark on the scales (1–10) their most appropriate current response to the question. Items 1–6 score from 1 (not at all satisfied) to 10 (very satisfied), item 7 scores from 1 (not at all) to 10 (absolutely). Therefore, SLTS-7 total scores range from 7 (minimum) to 70 (maximum). See the Supplementary Material.
Secondary outcomes included
Quality of life
The 8-item Parkinson’s Disease Questionnaire (PDQ-8) is a shortened version of the PDQ-39 for self-evaluation of QoL recommended by the International Parkinson and Movement Disorder Society [6]. It is commonly used in advanced PD cohorts [19, 21]. The scale surveys mobility, activities of daily living, emotional well-being, social support, cognition, communication, bodily discomfort, and stigma. Every question can be answered with a five-level Likert scale. The results are presented as a summary index (SI), which ranges between 0 (no impairment) and 100 (maximum impairment). High correlations have been shown between then PDQ-8 and PDQ-39 when using a Summary Index (PDQ-SI) [4].
The EuroQol Group developed the EQ-5D-3L with 5 dimensions and three levels per item. It is a generic measure that evaluates 5 aspects of QoL: mobility, self-care, daily activities, pain and discomfort, and anxiety and depression. Each item is assessed using a three-level Likert scale resulting in a five digit Health State from “11111” (completely healthy) to “33333” (seriously ill), which can be converted into a SI from 0 (death) to 1 (best health state), although negative values are possible for states valued worse than death. A country-specific conversion was performed for Germany (time trade-off method, TTO) and is here referred to as EQ-5D-3L TTO [22].
The EQ-VAS is a visual analog scale for self-rating current health-related QoL and ranges from 0 (worst imaginable health state) to 100 (best imaginable health state) [22].
Non-motor symptoms
The clinician-rated Non-Motor Symptom Scale (NMSS) contains 30 items divided into nine domains: 1) cardiovascular, 2) sleep/fatigue, 3) mood/apathy, 4) perceptual problems/hallucinations, 5) attention/memory, 6) gastrointestinal tract, 7) urinary, 8) sexual function, and 9) miscellaneous (including pain, inability to smell/taste, weight changes, and sweating). Non-motor symptoms over the last four weeks are surveyed. The NMSS total score ranges from 0 (no non-motor symptoms) to 360 (maximum impairment of non-motor symptoms). The NMSS total score can be applied to grade the severity of the burden of NMS (0 = none, 1–20 = mild, 21–40 = moderate, 41–70 = severe, ≥70 = very severe) [23].
Motor aspects
The Hoehn and Yahr (H&Y) scale classifies the severity of motor symptoms into 5 stages, reflecting disease progression and deterioration, and ranges from 0 (no signs of disease) to 5 (needing a wheelchair or bedridden unless assisted) [24].
Motor symptoms were assessed with the SCales for Outcomes in PD-motor (SCOPA-motor), including motor evaluation, activities of daily living, and motor complications. The SCOPA-motor is a modified and abbreviated version of the Unified Parkinson’s Disease Rating Scale (UPDRS), and corresponding domains highly correlate. The SCOPA-motor was used because its assessment time is approximately four times shorter than for the MDS-UPDRS [25–27]. The SCOPA is commonly used in advanced PD cohorts [28, 29]. The SCOPA-motor ranges from 0 (no impairment) to 75 points (maximum impairment) [27].
Statistical analysis
The Shapiro-Wilk test and frequency distribution histograms were applied to test for normal distribution. Descriptive statistics were calculated to show baseline characteristics of the sample. For the validation process of the SLTS-7, firstly, data quality was analyzed by percentage of computable scores and percentage of missing data (criterion: < 5%) [30]. Secondly, acceptability of the scale was analyzed (observed versus possible score range, mean scores closeness to the mid-point (median), floor and ceiling effects as percentage of possible extreme values (criterion:<15%) [11], skewness statistic, and confidence interval of the mean. After that, an exploratory factor analysis with orthogonal rotation (varimax) was conducted to examine whether a division into categories would be appropriate. An initial analysis informed by eigenvalues and scree plots was run to obtain the number of factors. The Kaiser-Meyer-Olkin test (with values≥0.6 indicating adequate sampling) and Bartlet’s sphericity test (with p < 0.05 indicating the existence of an underlying structure to the data) were used to analyze how suited the data are for factor analysis [31]. Following the exploratory factor analysis, we explored internal consistency by analyzing Cronbach’s alpha (α value of≥0.7 acceptable, ≥0.8 good, and≥0.9 excellent), inter-item correlation (criterion value > 0.20 and < 0.75), and corrected item-total correlation (criterion: ≥0.30) [32–34]. The convergent construct validity was expressed as Spearman correlations of the scale components (domains and total score) with other scales measuring similar or related constructs. A distinction was made between rater-based assessments such as H&Y, SCOPA-motor, and NMSS and patient-reported outcomes such as PDQ-8 and EQ-5D-3L. The strength of the correlations were defined as following: ‘weak’ rs = 0.20–0.39, ‘moderate’ rs = 0.40–0.59, ‘strong’ rs = 0.60–0.79, and ‘very strong’ rs = 0.80–1.00 [35, 36]. The internal validity was measured as intercorrelations between domains (item 1, item 2, composite 3 to 5, composite 6 and 7) (criterion values: 0.30–0.70) [34, 37]. Finally, the known-groups validity was explored as the ability of the scale to detect differences between NMSS burden levels. Differences between two groups were tested using the Mann-Whitney U test; differences between several groups were determined using the Kruskal-Wallis rank test. All analyses were conducted using the Statistical Package for Social Science (SPSS version 26.0 for Mac), and p values < 0.05 were considered statistically significant.
RESULTS
Baseline characteristics
In total, 117 patients (62.4%men) were included in this study with a mean age of 62.4±8.36 (range: 38–78) years and a mean disease duration of 9.87±4.57 (range: 2–22). Table 1 shows additional baseline characteristics.
Baseline characteristics
xAdapted to Germany according to TTO conversion published by the EuroQol Group. CISI-PD, Clinical Impression of Severity Index-Parkinson’s Disease; EQ-5D-3L, European Quality of Life Questionnaire with 5 Dimensions and 3 Levels; H&Y, Hoehn and Yahr; IQR, InterQuartile Range; LEDD, Levodopa Equivalent Daily Dose; PDQ-8, Parkinson’s Disease Questionnaire-8; SCOPA-motor, SCales for Outcomes in PD-motor scale; SD, standard deviation; SLTS-7, Satisfaction of Life and Treatment Scale-7; TTO, Time-Trade-Off; VAS, Visual Analogue Scale.
Data quality and acceptability
Missing data was 0%, computable data 100%. Table 2 shows results regarding acceptability. Overall, floor and ceiling effect, median, skewness of items, and confidence interval for the mean met the standard criteria, except for the item social relations with a ceiling effect of 17.1%.
Acceptability of the Satisfaction with Life and Treatment Scale-7 (SLTS-7)
Exploratory factor analysis
We conducted an exploratory factor analysis with the specific items 2–7 to identify domains within the items. The Kaiser-Meyer-Olkin test with a value of 0.77 and the Bartlett’s sphericity test (p < 0.001) were adequate for factor analysis. The exploratory factor analyses revealed a two-factor structure explaining variance of 88.66%, which grouped items 3,4, and 5 into one domain (psycho-social satisfaction) and items 6 and 7 into another domain (treatment satisfaction). Item 2 remained independent, i.e., ungrouped with any other item (Table 3).
Factor loadings after rotation in exploratory factor analysis
Items assigned to specific factor are highlighted in bold font.
Internal consistency
For all seven single items of the SLTS-7, Cronbach’s alpha index was 0.89. Inter-item correlations were between 0.35 and 0.84, whereby items 6 and 7 showed the highest correlation (0.84). The corrected item-total correlations were between 0.62 and 0.79.
Convergent validity
Figure 1 illustrates Spearman correlations between the main outcomes. The SLTS-7 total score was moderately correlated with the SCOPA-motor and weakly with the NMSS total scores (both p < 0.01). In contrast, the PDQ-8 SI was weakly with the SCOPA-motor and moderately correlated with the NMSS total scores (both p < 0.01). The EQ-5D-3L TTO was weakly correlated with the H&Y as well as the NMSS and SCOPA-motor total scores (all p < 0.01).
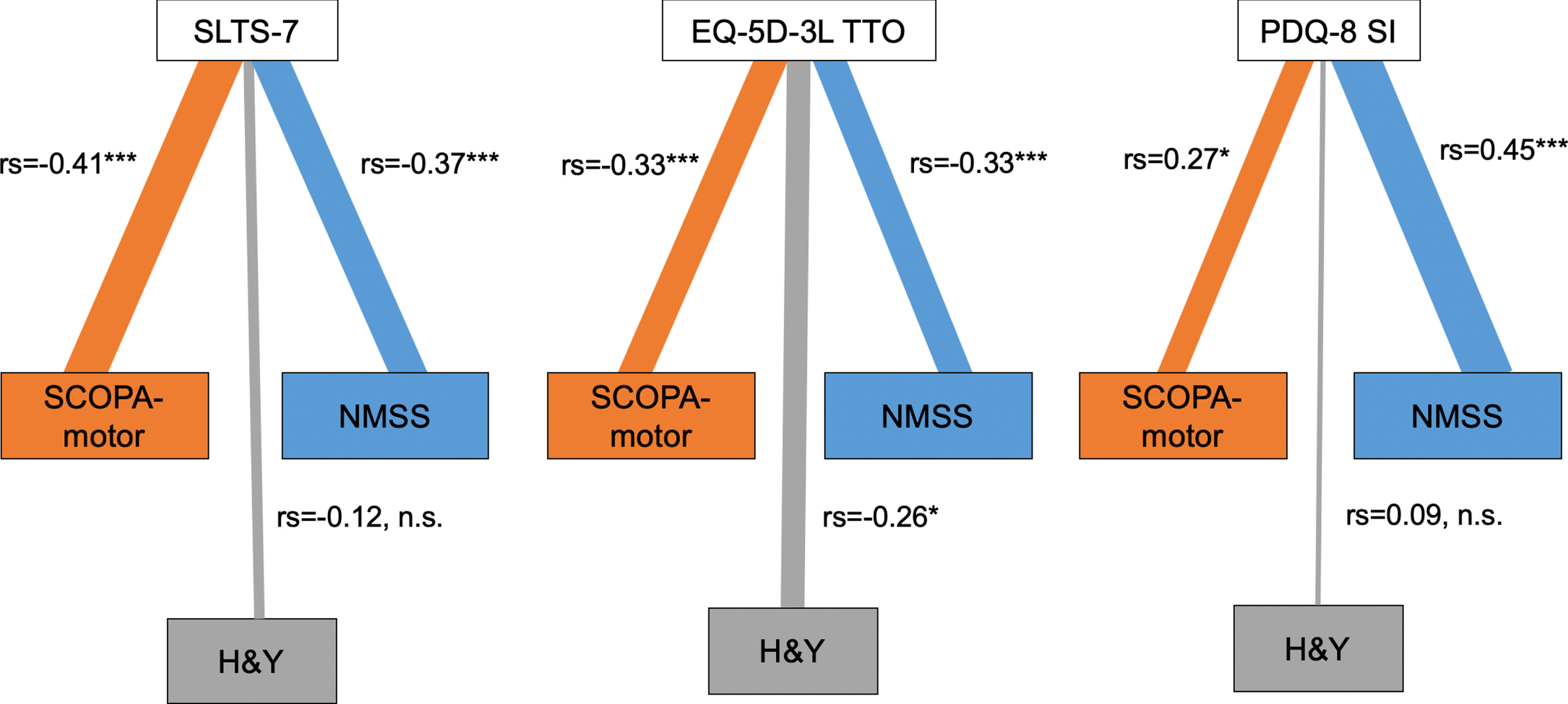
Distinct non-motor and motor correlation profiles for SLTS-7, EQ-5D-3L TTO, and PDQ-8 SI. Figure 1 shows the correlations between SLTS-7, EQ-5D-3L TTO, and PDQ-8 SI and SCOPA-motor (orange), H&Y (grey), and NMSS (blue). The line thickness corresponds to the strength of correlations, i.e., Spearman rank correlation coefficients (‘very weak’ rs = 0.00–0.19, ‘weak’ rs = 0.20–0.39, ‘moderate’ rs = 0.40–0.59, ‘strong’ rs = 0.60–0.79, and ‘very strong’ rs = 0.80–1.00); *p < 0.05; **p < 0.01; ***p < 0.001; n.s. = not significant. The SLTS-7 total score was moderately significantly correlated with the SCOPA-motor and weakly with the NMSS. In contrast, the PDQ-8 SI was weakly with the SCOPA-motor and moderately correlated with the NMSS. The EQ-5D-3L TTO was weakly correlated with the H&Y, SCOPA-motor, and NMSS. H&Y, Hoehn und Yahr; EQ-5D-3L TTO, European Quality of Life Questionnaire with 5 Dimensions and 3 Levels Time-Trade-Off; NMSS, Non-Motor Symptom Scale; PDQ-8 SI, Parkinson’s Disease Questionnaire-8 Summary Index; SCOPA-motor, SCales for Outcomes in PD-motor scale; SLTS-7, Satisfaction with Life and Treatment Scale-7.
Furthermore, Table 4 shows the correlations between the identified SLTS-7 domains and other scales. Notably, the SLTS-7 item-domain “Satisfaction with life as a whole” and “psycho-social” domains correlated moderately with PDQ-8 emotional well-being item. Further, the “psycho-social” SLTS-7 domain also correlated moderately with the PDQ-8 SI. Furthermore, correlations between all SLTS-7 domains and the EQ-VAS were moderate, whereas they were weak for EQ-5D-3L TTO.
Convergent construct validity of the Satisfaction with Life and Treatment Scale-7 (SLTS-7)
Spearman rank correlation coefficients. *p < 0.05; **p < 0.01; ***p < 0.001; except (a) not significant. xAdapted to Country according to EuroQol. H&Y, Hoehn und Yahr; EQ-5D-3L, European Quality of Life Questionnaire with 5 Dimensions and 3 Levels; NMSS, Non-Motor Symptom Scale; PDQ-8, Parkinson’s Disease Questionnaire-8; SCOPA-motor, SCales for Outcomes in PD-motor scale; TTO, Time-Trade-Off; VAS, Visual Analogue Scale.
Internal validity
The intercorrelations between the domains were between 0.38 and 0.73 and are shown in Table 5.
Internal validity of the Satisfaction with Life and Treatment Scale-7 (SLTS-7)
Spearman rank correlation coefficients. All p < 0.001.
Known-groups validity
Table 6 summarizes the results of the known-groups validity exploring differences in SLTS-7 total score according to the NMSS burden levels. There were no significant differences in the SLTS-7 total score per sex (p > 0.05).
Known-groups validity
Kruskal-Wallis rank test was used to test differences between the groups, p = 0.002; there were no patients who reported no non-motor symptoms. NMSS, Non-motor Symptom Scale; SD, standard deviation.
DISCUSSION
In the present study, we addressed the psychometric characteristics of the new SLTS-7, assessing satisfaction with life and treatment. Our results provide evidence that the SLTS-7 is a valid and useful tool that can be easily applied in a PD population screened for advanced treatments to assess both satisfaction with life and treatment.
Clinimetric properties
The data quality of the SLTS-7 was excellent with 100%computable data. We observed adequate values for the floor and ceiling effect, median, skewness of items, and confidence interval for the mean, providing evidence that the SLTS-7 is a valid tool to be applied in our advanced PD population. The data distribution is comparable to the findings of the validation study of the SLS-6 [12].
In addition to item 1 representing the overall satisfaction with life, we conducted an exploratory factor analysis among the specific components (items 2–7). We identified that item 2 (physical satisfaction) remained as an independent domain not linked to other factors. In contrast, items 3 to 7 corresponded to two additional domains: items 3, 4, and 5 in a psycho-social satisfaction domain and items 6 and 7 in a treatment satisfaction domain. The distinction between physical (motor) and psycho-social (non-motor) satisfaction is not surprising as QoL in PD is influenced independently by motor and non-motor aspects [16, 38–41].
Regarding the internal consistency, Cronbach’s alpha was 0.89, indicative of high internal consistency. Also, the inter-item correlations and the corrected item-total correlations showed satisfactory values [12]. All intercorrelations between SLTS-7 domains were between 0.38 and 0.73, showing acceptable internal validity and supporting the fact that life as a whole is positively significantly associated with all three identified domains. The highest association was found between life as a whole and the domain psycho-social satisfaction (rs = 0.73).
Concerning known-groups validity, the SLTS-7 scores were significantly worse with a more severe NMS burden. Male and female patients experienced no significant difference in satisfaction with life and treatment. Further studies are needed to explore how sensitive SLST-7 is to change over time or after an intervention.
Satisfaction with life as a whole and all items
Overall, the mean “satisfaction with life as a whole” was 6.8 (with a possible range score between 1–10), and the highest satisfaction scores were found for the item “social relations” (mean 7.2), which is in line with the findings of Ambrosio and colleagues in a general PD population [12]. The lowest satisfaction score was found for the item “physical health” (mean 5.0), which is slightly lower than the findings by Ambrosio and colleagues (mean 5.7) [12]. This discrepancy might be explained by the fact that all patients in our population were referred for advanced treatment and, therefore, more affected by motor symptoms.
Following previous studies in the general population, we observed that satisfaction with life as a whole is closely linked to the self-perceived health status measured with the EQ-VAS [10].
The SLTS-7, PDQ-8, and EQ-5D-3L address different constructs: Satisfaction with life and treatment, specific health-related quality of life in PD, and generic health-related quality of life. Our results underpin these differences in constructs as (1) only the SLTS-7 contains treatment-related items, and (2) we observed distinct non-motor and motor correlation profiles for all three tools (see Fig. 1): The SLTS-7 was moderately correlated with motor and weakly with non-motor symptoms total burden. In contrast, the PDQ-8 SI was weakly correlated with motor and moderately with non-motor symptoms total burden, which is in line with previous studies [42]. The EQ-5D-3L TTO was the only scale significantly correlated with motor disease progression (H&Y) [43].
Physical satisfaction
We found a weaker association between physical satisfaction and PDQ-8 SI, which is in line with previous observations that motor symptoms are associated with QoL rather weakly [44].
Psycho-social satisfaction
The highest correlation was found between the domain “psycho-social satisfaction” and PDQ-8 SI, which confirms previous findings that psychological well-being is closely connected with QoL [42]. To our knowledge, this is the first study to provide evidence that “psycho-social satisfaction” separately contributes to life satisfaction. Furthermore, we found the NMSS total score had the highest correlation with the domain “psycho-social satisfaction” (-0.39) and the domain “psycho-social satisfaction” was moderately correlated withe the PDQ-8 emotional well-being item, underlining that this domain captures non-motor aspects of life satisfaction.
Treatment satisfaction
Life and treatment satisfaction have been studied in patients treated with deep brain stimulation for movement disorders in general. For instance, Kuehler and colleagues have developed a quality of life questionnaire including life and treatment satisfaction (Questions on Life Satisfaction (QLSM) [45]. However, this questionnaire was examined in mixed cohorts with several movement disorders, not specifically PD. A study by Ferrera and colleagues provided evidence for an improvement of life and health satisfaction, particularly satisfaction with motor function and independence, 17 months after subthalamic stimulation in a PD population [46]. However, the sample size was small (21 patients), and the applied QLSM tool was not explicitly validated for patients with PD [46]. Reddy and colleagues developed a tool (PRO-APD) to measure the perceived problem severity before initiating advanced PD treatment and the associated expectations. However, they did not specifically address and measure satisfaction [47].
To our knowledge, the SLTS-7 is the first scale for combined assessments of life and treatment satisfaction validated specifically in a PD population. The treatment domain covers two aspects, PD treatment (item 6) and expectations met concerning the treatment (item 7). Both items showed a robust significant inter-correlation. Correlation analyses indicate the close relationship between “treatment satisfaction” and self-reported health status (EQ-VAS).
As suggested by Ambrosio and colleagues, our results support the notion that besides a global assessment, life satisfaction should be addressed in different domains to reflect a comprehensive real-life representation of the different life satisfaction components for each patient [12].
Satisfaction with life and quality of life
QoL and life satisfaction are two distinct entities that complement each other.[48] While QoL appears to be a broader concept focusing on external factors, life satisfaction is influenced more by internal and subjective aspects. In this context, it has been termed “subjective QoL” [11, 14]. As outlined previously, patients with PD might report a low QoL, whereas their life satisfaction can be reasonably good if they are satisfied, e.g., with their personal and family life [49]. Our observation that the correlation between satisfaction with life as a whole and specific satisfaction domains was most prominent for “psycho-social satisfaction” highlights the importance of internal factors for this construct.
Limitations
The present study has several limitations. Our sample size was smaller than in the validation study of the SLS-6. However, we conducted the validation in a highly selected patient population undergoing advanced PD therapies, and our sample size was comparable to other validation studies in cohorts undergoing advanced therapies in PD [15, 50–53]. As this analysis was conducted in a highly selected group of patients with advanced PD, our results might not be applicable to a general PD population at an earlier phase of their disease as well as populations with other medical conditions. We did not apply the Montreal Cognitive Assessment (MoCA) as part of this study. However, all patients were assessed by expert neuropsychologists as part of the clinical routine preoperative DBS indication evaluations using specific neuropsychological tests, such as the MoCA, the Parkinson’s Neuropsychometric Dementia Assessment or the Mattis Dementia Rating Scale. Neuropsychological assessments focused on the presence of clinically relevant cognitive impairment including multi-domain mild cognitive impairment or dementia. Cross-validations of the SLTS-7 in independent cohorts are warranted to extend our results. An easy and valid assessment of life satisfaction, including treatment aspects, is relevant not only to PD but also to other chronic diseases. Therefore, cross-validations should be conducted in other conditions. Furthermore, the multi-center design of our study increases external validity and reduces a potential bias that may be introduced by a single center. In this study, we only conducted an exploratory factor analysis in a cross-sectional design without performing a confirmatory factor analysis. Additional studies should confirm our findings in longitudinal designs to further validate the use of the SLTS-7 for measuring PD treatment effects.
CONCLUSIONS
In the present study, we have shown that the SLTS-7 is a valid, reliable, and easy-to-use tool to assess satisfaction with life and treatment in patients with PD screened for advanced therapies. Our study provides the first evidence that life satisfaction in patients with PD includes three aspects, i.e., physical, psycho-social, and treatment satisfaction. To our knowledge, the SLTS-7 is the first scale for a combined assessment of these aspects validated specifically in a PD population. Further longitudinal studies analyzing the effect of advanced PD treatment on satisfaction with life and treatment are warranted.
Footnotes
ACKNOWLEDGMENTS
The authors wish to thank their patients for participating in this study. The London center (KRC, AR, KA) wishes to thank Dr. M. Samuel for attending to patients treated with deep brain stimulation in clinical routine. The Marburg center (LT, PAL, CN) wishes to thank Dr. D. Pedrosa for allocating patients to specific studies and attending to patients treated with deep brain stimulation in clinical routine.
This paper presents independent research funded by the National Institute for Health Research (NIHR) Mental Health Biomedical Research Centre and Dementia Unit at South London and Maudsley NHS Foundation Trust and King’s College London. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
CONFLICT OF INTEREST
Anna Sauerbier is funded by the Gusyk program and the Advanced Cologne Clinician Scientist program of the Medical Faculty of the University of Cologne and has received funding from the Prof. Klaus Thiemann Foundation.
Philipp A. Loehrer was funded by the SUCCESS-Program of the University of Marburg, the Parkinson’s Foundation, and the Stiftung zur Förderung junger Neurowissenschaftler.
Alexandra Rizos was supported by a grant from Medtronic, The International Parkinson and Movement Disorder Society and the National Institute for Health Research Clinical Research Network South London and received honorarium from UCB and Britannia Pharmaceuticals.
Stefanie T. Jost was funded by the Prof. Klaus Thiemann Foundation.
Michael T. Barbe received speaker’s honoraria from Medtronic, Boston Scientific, Abbott (formerly St. Jude), GE Medical, UCB, Apothekerverband Köln e.V. and Bial as well as research funding from the Felgenhauer-Stiftung, Forschungspool Klinische Studien (University of Cologne), Horizon 2020 (Gondola), Medtronic (ODIS), and Boston Scientific and advisory honoraria for the IQWIG.
Gereon R. Fink serves as an editorial board member of Cortex, Neurological Research and Practice, NeuroImage: Clinical, Zeitschrift für Neuropsychologie, and DGNeurologie; receives royalties from the publication of the books Funktionelle MRT in Psychiatrie und Neurologie, Neurologische Differentialdiagnose, and SOP Neurologie; received honoraria for speaking engagements from Bayer, Desitin, Ergo DKV, Forum für medizinische Fortbildung FomF GmbH, GSK, Medica Academy Messe Düsseldorf, Medicbrain Healthcare, Novartis, Pfizer, and Sportärztebund NRW.
Keyoumars Ashkan has received honoraria for educational meetings, travel and consultancy from Medtronic, St. Jude Medical and Boston Scientific.
Christopher Nimsky is consultant for Brainlab and received speaker’s honoraria.
Veerle Visser-Vandewalle is a member of the advisory boards and reports consultancies for Boston Scientific, Medtronic, and LivaNova.
K. Ray Chaudhuri has received funding from Parkinson’s UK, NIHR, UCB, and the European Union; he received honoraria from UCB, Abbott, Britannia, US Worldmeds, and Otsuka Pharmaceuticals; and acted as a consultant for AbbVie, UCB, and Britannia.
Lars Timmermann reports grants, personal fees and non-financial support from SAPIENS Steering Brain Stimulation, Medtronic, Boston Scientific and St. Jude Medical.
Pablo Martinez-Martin has received honoraria from Editorial Viguera and Takeda Pharmaceuticals for lecturing in courses; from Britannia for writing an article in their Parkinson’s Disease Medical Journal-Kinetic; and from the International Parkinson and Movement Disorder Society (MDS) for management of the Program on Rating Scales. Grants from the MDS for development and validation of the MDS-NMS.
Haidar S. Dafsari was funded by the EU Joint Programme –Neurodegenerative Disease Research (JPND), the Prof. Klaus Thiemann Foundation in the German Society of Neurology, the Felgenhauer Foundation, the KoelnFortune program of the Medical Faculty of the University of Cologne, and has received honoraria by Boston Scientific, Medtronic and Stadapharm.
Pia Bachon, Leire Ambrosio, Alexandra Gronostay, and Agni Konitsioti report no financial disclosures.
