Abstract
Background:
Dopa-resistant freezing of gait (FOG) and falls represent the dominant motor disabilities in advanced Parkinson’s disease (PD).
Objective:
We investigate the effects of deep brain stimulation (DBS) of the mesencephalic locomotor region (MLR), comprised of the pedunculopontine (PPN) and cuneiform (CuN) nuclei, for treating gait and balance disorders, in a randomized double-blind cross-over trial.
Methods:
Six PD patients with dopa-resistant FOG and/or falls were operated for MLR-DBS. Patients received three DBS conditions, PPN, CuN, or Sham, in a randomized order for 2-months each, followed by an open-label phase. The primary outcome was the change in anteroposterior anticipatory-postural-adjustments (APAs) during gait initiation on a force platform
Results:
The anteroposterior APAs were not significantly different between the DBS conditions (median displacement [1st–3rd quartile] of 3.07 [3.12–4.62] cm with sham-DBS, 1.95 [2.29–3.85] cm with PPN-DBS and 2.78 [1.66–4.04] cm with CuN-DBS; p = 0.25). Step length and velocity were significantly higher with CuN-DBS vs. both sham-DBS and PPN-DBS. Conversely, step length and velocity were lower with PPN-DBS vs. sham-DBS, with greater double stance and gait initiation durations. One year after surgery, step length was significantly lower with PPN-DBS vs. inclusion. We did not find any significant change in clinical scales between DBS conditions or one year after surgery.
Conclusion:
Two months of PPN-DBS or CuN-DBS does not effectively improve clinically dopa-resistant gait and balance disorders in PD patients.
Keywords
INTRODUCTION
Freezing of gait (FOG) and falls represent the dominant motor disabilities in advanced Parkinson’s disease (PD). These signs worsen over time, are associated with increased morbidity, mortality, poor quality of life and high public health cost [1–4], and become unresponsive to dopaminergic treatment or deep brain stimulation (DBS) of the subthalamic nucleus[4–6] or internal part of the globus pallidus [7, 8]. Their pathophysiology is poorly understood, but imaging and post-mortem studies suggest a causal role of cholinergic dysfunction within the pedunculopontine nucleus (PPN) [9–12], located in the mesencephalic locomotor region (MLR), also including the cuneiform nucleus (CuN) dorsally [13–15]. In mice, optogenetic stimulation of cholinergic and glutamatergic PPN neurons modulates and produces slow exploratory locomotion, respectively, whereas stimulation of glutamatergic CuN neurons produces high-speed running [16, 17]. In cats, PPN or CuN stimulation modifies axial tone or elicits locomotion, respectively [15]; and in monkeys, cholinergic PPN lesions provoke increased hindlimb and tail tone with impaired locomotion, whereas CuN lesions increased forelimb and neck tone and gait speed [15]. In humans, neuronal recordings and imaging studies suggest that imagined and simulated gait evoke MLR activation [11, 18–24], with the CuN more involved in initiated high speed gait and the PPN in the automaticity for paced walking [25].
Based on this and on the correlation between PPN cholinergic cell death and falls in PD [10], MLR-DBS has been attempted to alleviate FOG and falls for PD. About half of patients reported a subjective reduction of FOG and falls after MLR-DBS [26–29], but randomized controlled studies including a small number of PD patients generally failed to demonstrate significant objective clinical effects [28–30]. Using gait recordings in a controlled study, we previously observed a small improvement of objective gait initiation parameters in 4 patients with DBS applied in the dorsal MLR, i.e., just above the pontomesencephalic junction (PMJ) [29], and in a postmortem series of 2 patients with progressive supranuclear palsy, a possible better result was also obtained with DBS applied in the dorsal MLR [31]. However, a reduction of FOG during turning was reported in 7 patients with DBS applied in the more ventral MLR, below the PMJ [32]. Finally, the heterogeneous clinical outcomes obtained in previous studies could be due to 1) the unknown optimal target within the MLR, namely the CuN or the PPN that may control different walking parameters; 2) the difficulty in assessing episodic phenomenon such as FOG and falls under experimental conditions, and 3) the unknown PD population that would be the best candidates [33].
Here, we aimed to evaluate the effects of DBS of both PPN and CuN in PD patients with dopa-resistant FOG and/or falls, in a crossover, double-blind randomized trial.
METHODS
Study design and patients
In this exploratory randomized, double-blind, crossover trial with a 6-month blinded phase, we recruited patients from the Pitié-Salpêtrière Hospital. All patients were assessed at the Clinical Investigation Center at the Paris Brain Institute and operated at the Neurosurgery department of the Pitié-Salpêtrière Hospital. Patients were eligible for inclusion if they fulfilled the criteria for idiopathic PD diagnosis based on the United Kingdom Parkinson’s Disease Society Brain Bank, were aged between 18 and 70 years old, had gait and/or balance disorders not improved by dopamine treatment (Movement Disorder Society-Unified Parkinson’s Disease Rating Scale [MDS-UPDRS] [34]: activities of daily living-ADL: part 2: item 2.12-walking / balance > 1 and / or item 2.13-freezing of gait > 1 and / or clinical evaluation item 3.11-freezing of gait > 1 and / or item 3.12-postural instability > 1, On-dopa), had a dopa sensitivity for other motor signs > 40%, had no contraindication to the examination necessary for inclusion, magnetic resonance imaging or for DBS surgical procedure, had no dementia (Mattis Dementia Rating Scale more than 129), were stable on antiparkinsonian drug treatment for at least 3 months at the time of recruitment, agreed to participate and provide written informed consent and affiliation to a social security scheme. For each patient, these evaluations were reviewed by an independent selection committee of 2 expert neurologist and neurosurgeon, who made the final decisions with respect to eligibility.
We performed the study in accordance with the declaration of Helsinki and Good clinical practice guidelines and approval by the ethics committee (CPPRB Ile-de-France Paris 6, Clinical trials.gov registration NCT02931097).
Randomization and blinding
Patients received three DBS conditions, PPN, CuN, or sham, in a randomized order for a 2-month period each (Fig. 1). The randomization sequence was allocated using a block scheme including the 6 order options per block. The randomization sequence was only revealed to the unblinded clinician responsible for the stimulation programming. Patients were blinded to the randomization sequence. To avoid the possibility of a patient being aware of their randomization group, the unblinded neurologist set stimulation parameters below the side-effect threshold and spent the same time adjusting each patient’s stimulator at the start of both active and sham stimulation periods. The DBS parameters were not modified during each double-blind period. The primary and secondary outcomes were assessed by the same blinded investigators throughout the study.
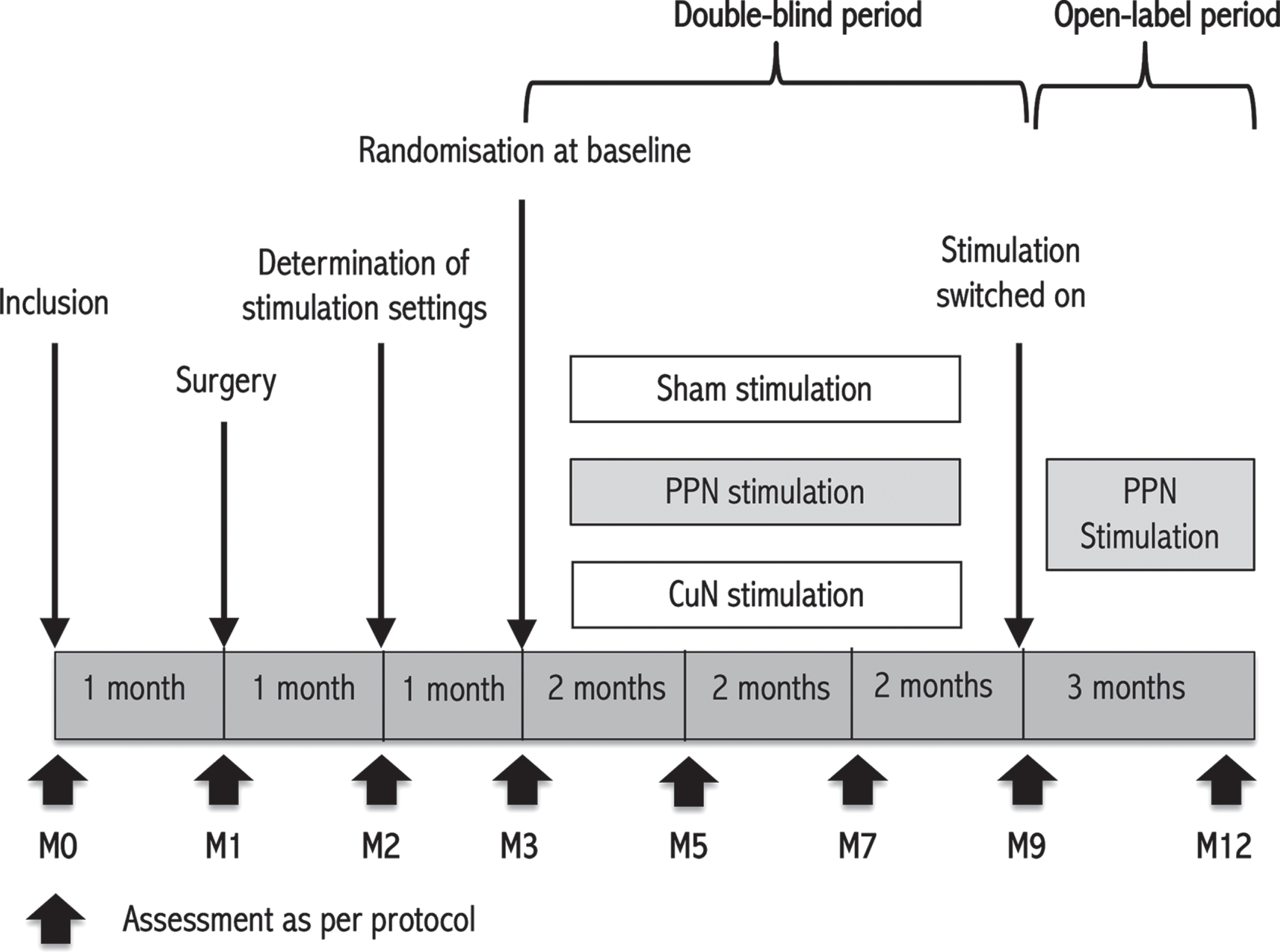
Study design. The study included three 2-month treatment phases (month 3 to month 9). Patients were evaluated at inclusion, less that 2 months before surgery; 2 months after surgery, before sham or active stimulation; 4, 6 and 8 months after surgery, after 2 months with active PPN, active CuN or Sham stimulation; and 11 months after surgery after 3 months with open-label active PPN stimulation. The optimal stimulation settings for the randomization period were determined 1 month after surgery.
Procedures
Patients were assessed at inclusion (baseline), followed 1 month later by bilateral implantation of DBS electrodes into the MLR (Fig. 2). For each patient, we determined the targets after superimposing our 3D histological atlas providing a map of PPN cholinergic neurons and GABAergic neurons of the PPN and CuN [35], adjusted to fit the specific geometry of each patient’s brain MRI (Fig. 2). The trajectory was chosen in order to have an electrode which passes through these 2 MLR areas, with at least one contact located within the PPN and another more dorsally within the CuN. Implantation of the two electrodes with 8 in-line contacts (model DB-2201, contact spacing: 2 mm, diameter: 1.3 mm, Boston Scientific) was performed under general anesthesia in 3 patients and under sedation in 3 patients, with two microelectrodes recordings on each side, with one along the central trajectory and one along the anterior, posterior, or medial trajectories depending on the individual patient’s anatomy. According to per-operative physiological and anatomical checks using X-ray, the central trajectory was chosen for implanting the definitive DBS electrode in 9 cases, the anterior trajectory in 2 cases and the posterior trajectory in one case. A post-operative CT-scan was performed to check the absence of complications and determine electrode locations (Fig. 2). The pulse generator was implanted 1–4 days after (Vercise; Boston Scientific).
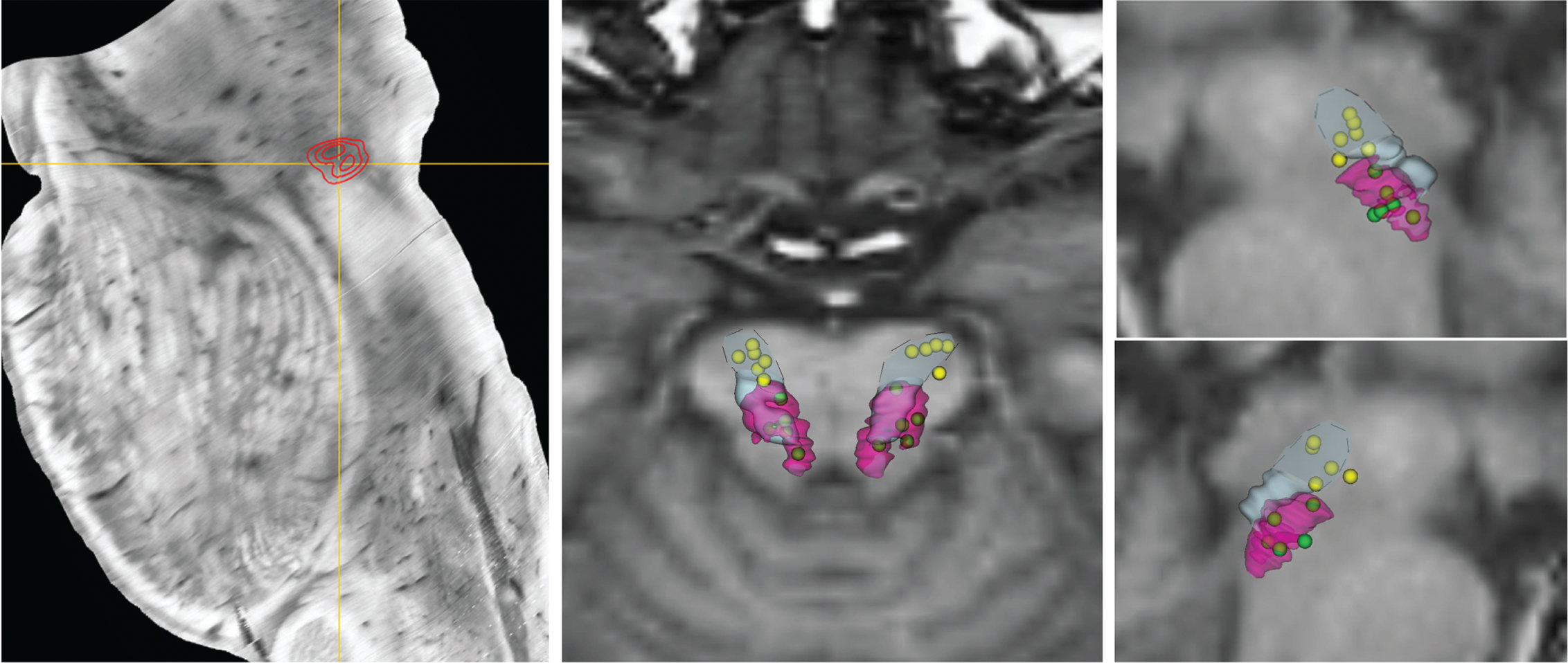
Reconstruction of the PPN and CuN nuclei DBS electrodes location. Left panel: Location of the highest density of cholinergic neurons within the mesencephalic locomotor region corresponding to the pedunculopontine nucleus (PPN, red area). Middle and right panels: Location of contacts used for DBS and reported on 3-D views after fusion with the three-dimensional MRI image (middle panel: posterior view; right panel: right and left sagittal views). The pink and light-blue outline the cholinergic PPN and GABAergic CuN neurons, respectively, with the dashed line delimiting the dorsal part of the CuN. The active contacts used for PPN-DBS during the double-blind period are represented in green and those for CuN DBS in yellow, showing that the electrodes all penetrate the PPN and CuN areas.
We performed an assessment 1 month after surgery (month 2, Fig. 1). The stimulators were activated, and parameter settings determined for the double-blind period. We randomly assigned patients 1 month later (month 3) to the 3 DBS conditions. At the end of the 6 month double-blind period, as previous studies have demonstrated a link between cholinergic cell death and gait and balance disorders of PD patients [9–12], all patients received PPN-DBS for the subsequent 3 months in an open-label fashion in order to assess more specifically the effects of PPN-DBS over a longer period and with the possibility to adapt parameters settings (month 9 to month 12).
We recommended that patients’ medical treatments for PD remain stable over the study period. We carefully recorded treatment adjustments necessitated by patients’ neurological conditions.
Outcomes
The primary outcome was the difference in the posterior center of foot pressure (CoP) displacement during the anticipatory postural adjustments (APAs) phase Off-dopa between the end of each 2-month period of the randomized double-blind period, i.e., with Sham- vs. PPN- vs. CuN-DBS. We chose this parameter as 1) it has been widely reported to be decreased in PD patients, and more specifically in PD patients with FOG [36], 2) was recorded using validated procedures and calculated independently, and 3) we have already shown it to be modified with MLR-DBS [29].
For this, gait parameters were recorded at each visit using the VICON system with Plug-In-Gait model with markers positioned on the arm and leg joints. The patients, barefoot, initiated gait on a force platform (0.9×1.8 m, Advanced Mechanical Technology Inc. LG6-4-1) and walked for 5-m at a self-paced speed, making a half-turn and returning to the initial position (n = 15–20 trials, Fig. 3). The gait initiation, straight forward walking and turn events were manually identified and the following parameters calculated: 1) APAs phase duration, 2) maximum posterior and 3) lateral CoP displacements during the APAs, 4) length and velocity of the first step, 5) step width, 6) mean step length during a gait cycle, 7) cadence, 8) step length asymmetry, 9) stride time variability, 10) gait cycle, double-stance and half-turn durations, 11) turn amplitude and 11) the number of FOG episodes during the forward path (Fig. 3). These parameters were recorded Off-dopa (after an interruption of 12 hours of the antiparkinsonian treatment) and On-dopa (after receiving a suprathreshold dose of levodopa). For gait parameters, an improvement is reflected by a decrease in temporal parameters (duration of APAs, double-stance and turn) and an increase in spatial parameters (posterior and lateral CoP displacement, step length, velocity, turn amplitude).
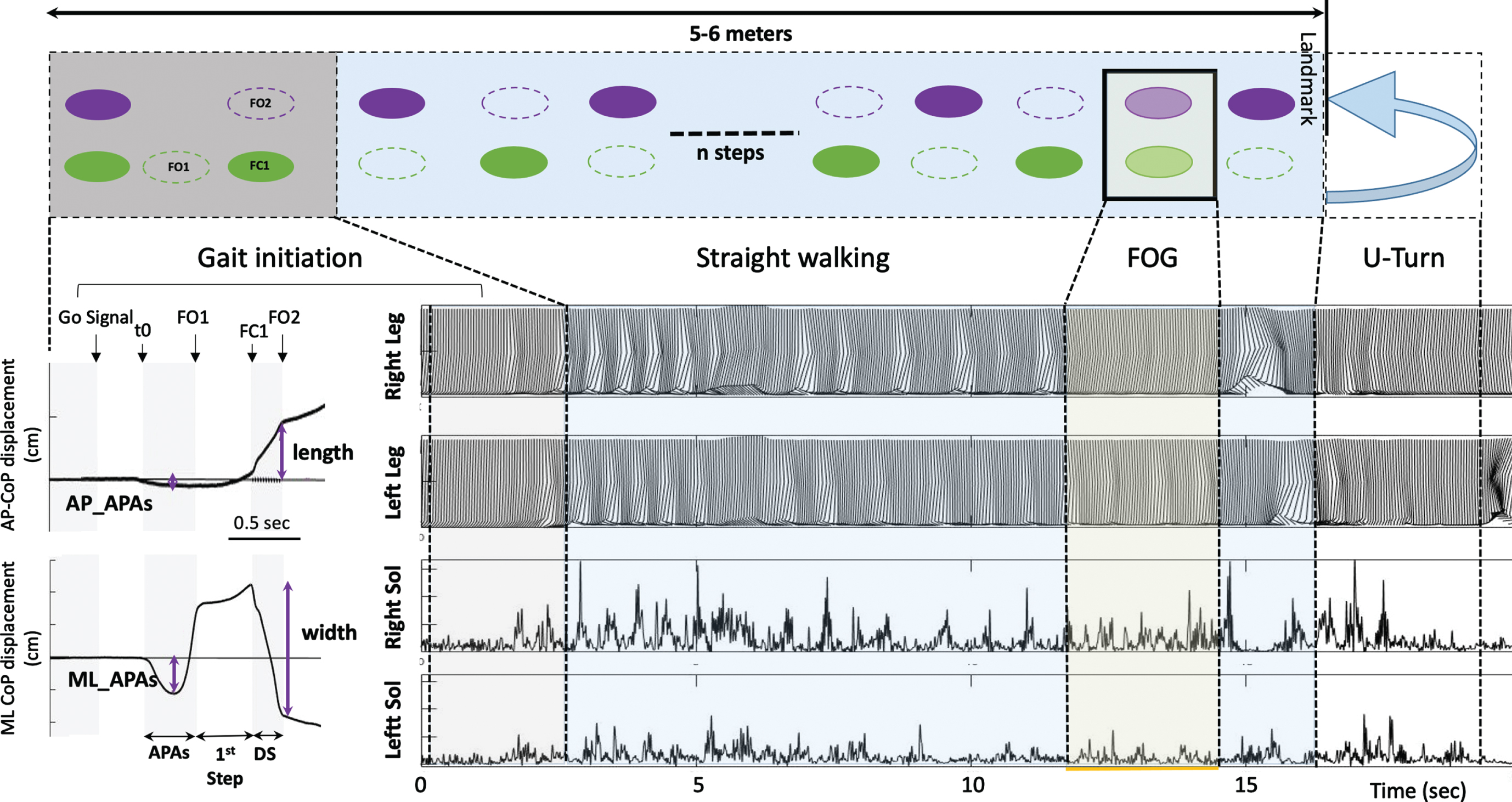
Recordings of gait initiation, straight-line walking and turn using the force platform and VICON system. Top row: The subject stands motionless on the force plate (Grey square) and initiates gait with his right leg (Gait initiation), and then walks straight forward self-paced, makes a half-turn to a landmark taped to the ground and comes back to the starting position. The standardized gait alternation is represented with green and purple filled ellipses when the foot is in floor contact and a dotted outline ellipse during the swing phase of the foot. A freezing of gait episode is represented by the two feet on the ground. Bottom row: Illustration of the AP and ML CoP displacements during the gait initiation phase (left). Events of the gait initiation task are represented with black arrows and grey and white columns. At bottom right, illustration of both pelvis, knee, and ankle kinematics (upper rows) and soleus EMG signal (lower rows) for right and left legs, during gait initiation, straight-line forward walking, and U-turn. A FOG episode is characterized by an absence of forward movement, with attempts to lift the left foot from the ground, and bilateral simultaneous soleus muscle activity. AP, Anteroposterior; APAs, Anticipatory Postural Adjustments; CoP, Centre of foot Pressure; DS, double-stance phase; FC1, Foot Contact of the starting leg; FO1, Foot Off of the starting leg; FOG, Freezing of Gait; ML, Mediolateral; Sol, Soleus; t0, time of the first biomechanical event corresponding to the beginning of the APAs.
Prespecified secondary outcomes were differences in the other gait initiation and spatiotemporal parameters, and clinical scales between the end of each 2-month period of the randomized double-blind period (with Sham vs. CuN vs. PPN DBS). Clinical assessments included the following scales: the MDS-UPDRS (part 1: mental status, part 2: ADL, part 4: levodopa-related motor complications, and part 3: motor disability score assessed both Off- and On-dopa, comprising the “axial” subscore) [34]; the gait and balance scale (GABS) [37], also assessed both Off and On-dopa; the Rating Scale for Gait Evaluation (RGSE) [38]; the Tinetti scale [39]; the freezing of gait questionnaire (FOG-Q) [40]; the Activities-Balance Confidence (ABC) scale [41]; the Parkinson’s Disease Questionnaire (PDQ-39); the Mattis Dementia Rating Scale (MDRS) [42]; the Comprehensive Psychopathological Rating Scale [43], which includes the Montgomery and Asberg Depression rating Scale (MADRS) [44] and Brief Anxiety Scale (BAS) [45].
We also assessed the differences in gait initiation and clinical parameters between inclusion and the end of each 2-month double-blind period (inclusion vs. Sham-, CuN- or PPN-DBS); between inclusion and the end of the open-label period (inclusion vs. month 12); and between inclusion and post-surgery (inclusion vs. month 3) to assess the effects of the surgery itself.
We assessed safety in all patients who were enrolled and received MLR-DBS surgery. We classified any new symptoms as adverse events. We classified an adverse event as serious if the patient required hospital admission, if sequelae were present, or if the clinician considered the event to be serious.
Statistical analysis
This is a phase II study, and all outcomes are exploratory. We performed statistical analyses including all randomly allocated patients who received sham or active DBS during the double-blind period. All quantitative data are described with mean and SDs.
To assess a difference in the primary outcome, we used the non-parametric Friedman rank sum test with stimulation target (Condition, i.e., Sham-, PPN- or CuN-DBS) as the treatment or grouping variable and patient as the blocking factor. We examined the differences in other gait parameters and clinical scores by modelling each outcome using a linear mixed-model. We included treatment condition (baseline, month 3, Sham-, CuN-, or PPN-DBS, and month 12) and a period effect as fixed effects, and patient intercepts as random effects. We used the R package (version 3.3.1, R Core Development Team) for the statistical analysis, with the LmerTest package for the linear mixed-effect model testing. We used a significance threshold of 0.05. Post-hoc comparisons were FDR-corrected.
RESULTS
Cohort analysis
Between October 2016 and June 2018, we assessed 9 patients for eligibility, enrolling 8 in the study (Fig. 4). Two patients were withdrawn before surgery because of non-inclusion criteria: one with dementia (MMSE = 18) and one showed dopaminergic responsive axial motor signs (improvement of 72%). Thus, 6 patients were operated, randomly allocated and completed the study (Table 1). Medication was held constant during the double-blind period except in one patient (P03). Patients inclusion was stopped after completion of the randomized double-blind period by the sixth patient following a decision by the independent supervisory committee. This decision was based on the absence of an objective or subjective individual improvement in clinical scores in the 6 operated patients.
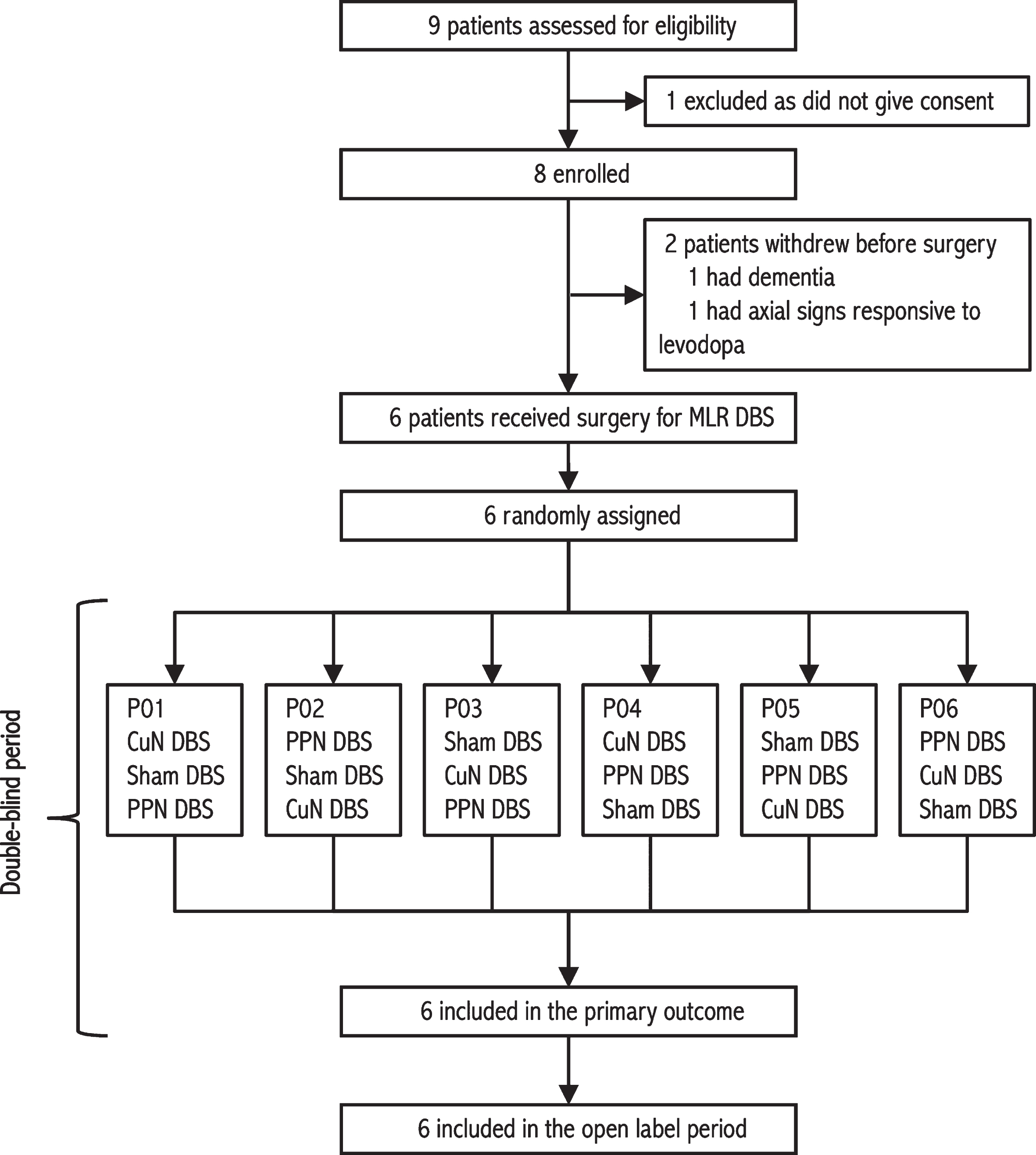
Trial profile. DBS, deep brain stimulation; CuN, cuneiform nucleus; MLR DBS, mesencephalic locomotor region deep brain stimulation; PPN, pedunculopontine nucleus.
Baseline demographic and clinical characteristics of 6 PD patients and stimulation parameters setting used during the blinded period
*The axial score is the sum of the following items: “arising from chair”, “gait”, “freezing of gait”, “postural stability”, and “posture”. DBS, deep brain stimulation; FOG-Q, freezing of gait questionnaire; GABS, gait and balance scale; LED, levodopa-equivalent daily dosage; MDRS, Mattis Dementia Rating Scale; MDS-UPDRS, Movement Disorders Society-Unified Parkinson’s Disease Rating. Electrodes coordinates are expressed in the lateral (X, relative to the midline, absolute values), anteroposterior (Y, relative to the posterior commissure) and depth (Z, relative to the ponto-mesencephalic junction with negative values indicating electrodes placed below the PMJ) axes, in mm, relative to the fourth ventricle floor and the PMJ line.
Electrode locations and stimulation parameters
Electrodes were accurately implanted in the PPN and the CuN on both sides except for one side in P04 (Fig. 2, Table 1). We used bipolar DBS using adjacent contacts as anode and cathode, for both CuN and PPN targets with 30 Hz stimulation frequency, 60μs pulse width and 1.7 to 4.2mA amplitude.
Changes in gait parameters and clinical scales during the double-blind randomized period
Gait parameters
Changes in the anteroposterior CoP displacement during the APAs were not significantly different between the end of each 2-month DBS condition during the double-blind period Off- and On-dopa (median displacement [1st–3rd quartile] of 3.07 [3.12–4.62] cm with Sham-DBS, 1.95 [2.29–3.85] cm with PPN-DBS and 2.78 [1.66–4.04] cm with CuN-DBS; Friedman test, p = 0.25, Fig. 5). In secondary analyses, using linear mixed models including the dopa treatment conditions, we found that the APAs anteroposterior CoP displacement was lower with PPN- relative to both Sham- and CuN-DBS in the On-dopa condition, and lower with Sham-, CuN-, and PPN-DBS relative to before surgery (On-dopa, Fig. 5).
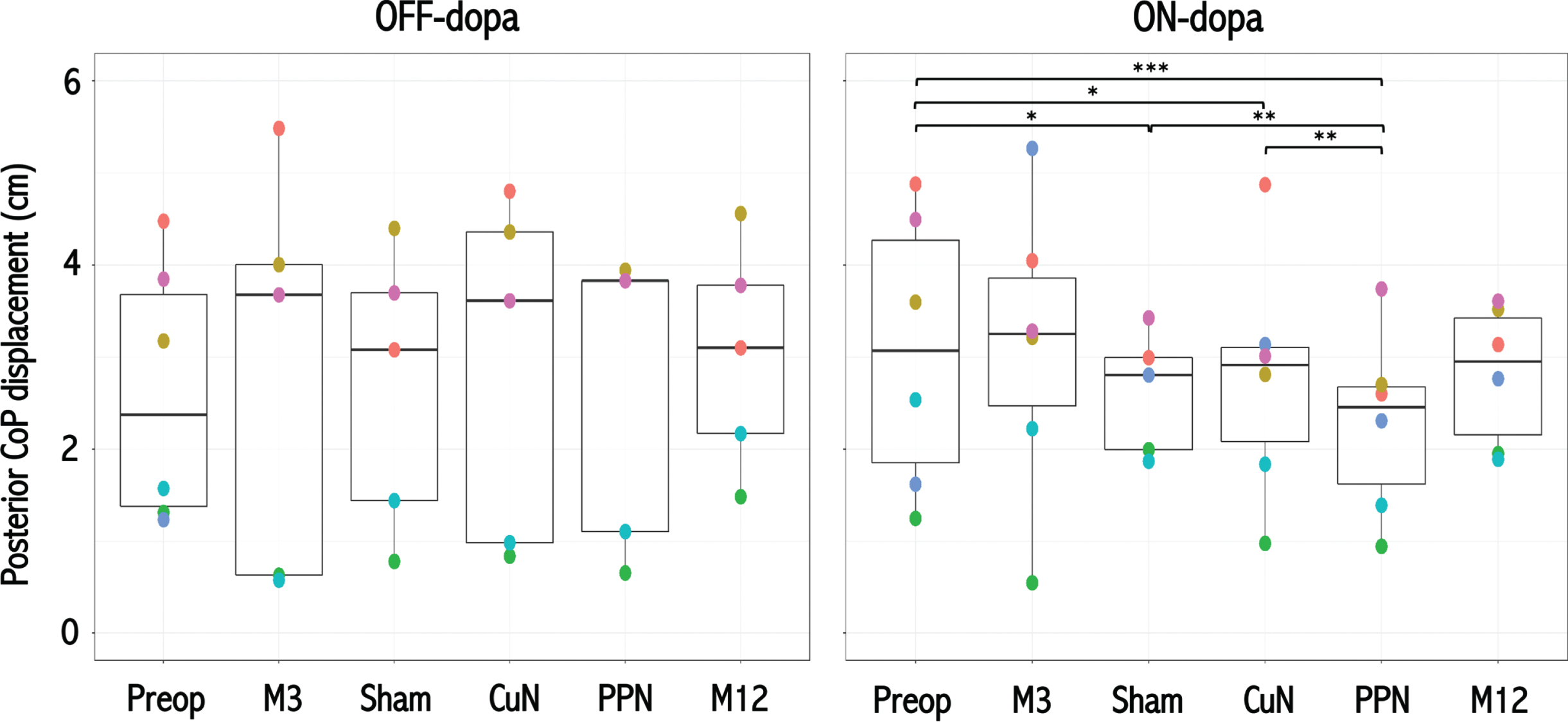
Effects of MLR DBS on posterior APAs displacement of the center of foot pressure. Posterior displacement of the CoP during the APAs of the gait initiation in the 6 PD patients before surgery (Preop), 3 months after surgery without active DBS (M3); and during the randomized double-blind period with Sham, CuN-DBS, or PPN-DBS, and at the end of the open label period with PPN-DBS (M12); OFF (left graph) and ON-dopa (right graph). Each color point represents an individual patient. One patient (P05, blue circle) could not perform gait initiation Off-dopa during the randomized and open-label periods. Horizontal black lines represent the estimated marginal mean for each condition, and the upper and lower bounds of the rectangles represent the corresponding 95%confidence intervals. APAs, anticipatory postural adjustments; CoP, centre of foot pressure; M, month; PPN, pedunculopontine nucleus; CuN, cuneiform nucleus. *p < 0.05; **p < 0.01; ***p < 10–4
During the randomized period, Off-dopa, we found that the first step length was higher with PPN-DBS relative to Sham-DBS (Fig. 6); and that the number of FOG episodes was lower with CuN- relative to PPN-DBS (Supplementary Figure 2). In the On-dopa condition, we found that the APAs, double-stance and turn durations were lower with Sham- relative to both PPN- and CuN-DBS (Fig. 6, Supplementary Table 1, Supplementary Figure 2), and the mediolateral CoP displacement during the APAs, step width, cadence, and turn amplitude were higher (Fig. 6, Supplementary Table 1, Supplementary Figure 2). On-dopa, the first step length, velocity, and mediolateral CoP displacements were also significantly higher (Fig. 6, Supplementary Table 1) and the gait cycle duration lower (Supplementary Figure 2) with CuN- relative to PPN-DBS. The first step length (On-dopa) was also significantly higher with CuN- relative to Sham-DBS (Fig. 6) and the number of FOG lower with Sham- relative to PPN-DBS (Supplementary Figure 2). Lastly, we also found a significant interaction between DBS conditions and the effects of the dopaminergic treatment on gait parameters during the randomized period (p < 0.001, Supplementary Material, Results).
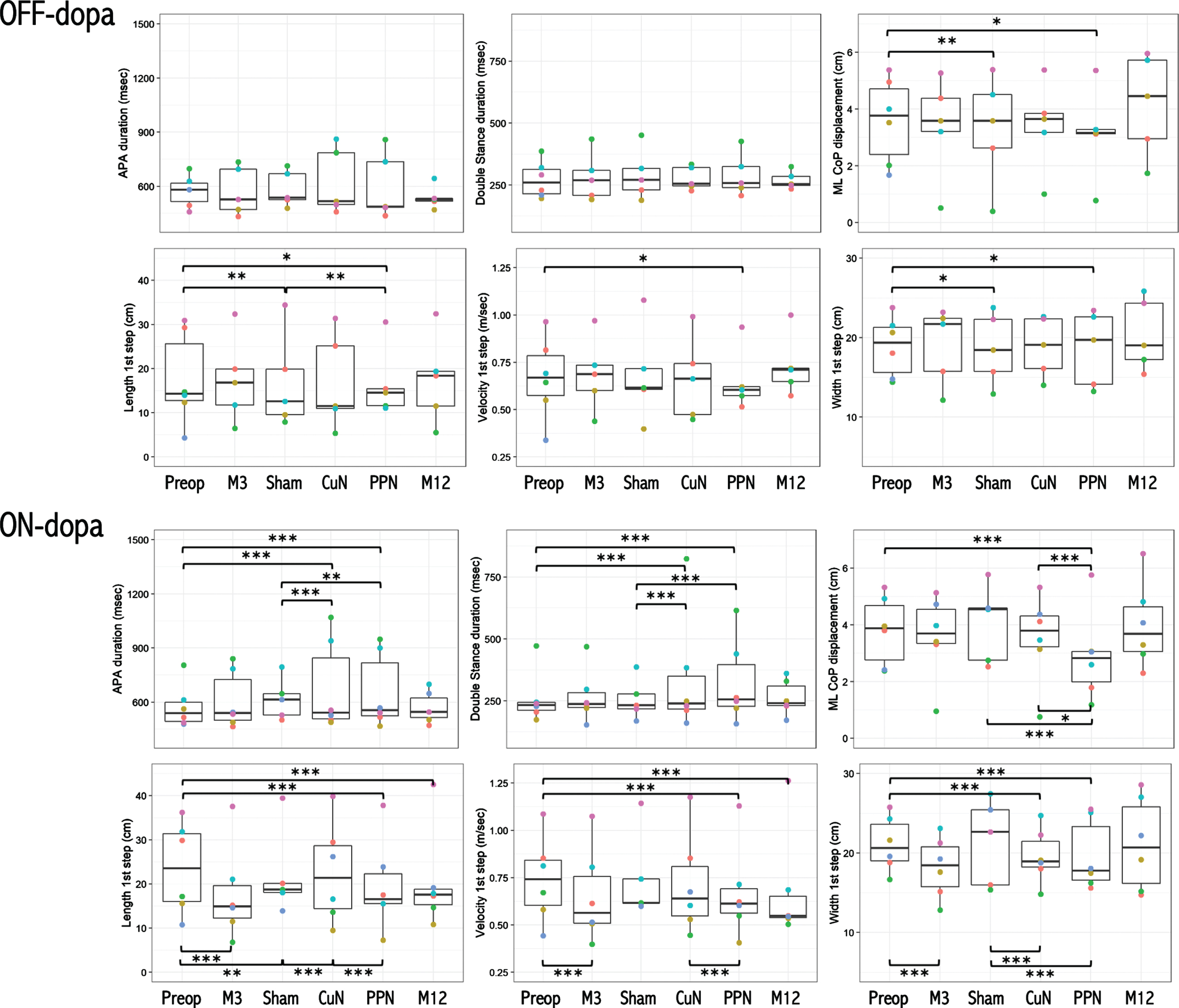
Effects of CuN, PPN, and Sham DBS on initiation gait parameters. Anticipatory postural adjustments (APA) and double-stance durations, APAs mediolateral CoP displacement, first step length, velocity, and width during gait initiation in the 6 PD patients before surgery (Preop), 3 months after surgery without active DBS (M3); and during the randomized double-blind period with Sham, CuN-DBS, or PPN-DBS, and at the end of the open label period with PPN-DBS (M12). Each color point represents an individual patient, obtained in the Off-dopa (upper panel) and On-dopa (bottom panel) conditions. One patient (P05, blue circle) could not perform gait initiation Off-dopa during the randomized and open-label periods. Horizontal black lines represent the estimated marginal mean for each condition, and the upper and lower bounds of the grey rectangles represent the corresponding 95%confidence intervals. CoP, center of foot pressure; ML, mediolateral. *p < 0.05; **p < 0.01; ***p < 0.001 between treatment conditions.
Comparing DBS conditions to before surgery, Off-dopa, we found that the APAs mediolateral CoP displacement, first step length and width were decreased with both Sham and PPN-DBS, and the velocity also decreased with PPN-DBS relative to before surgery (Fig. 6). On-dopa, we found that the APAs and double-stance durations were significantly higher and the APAs mediolateral CoP displacement and first step width lower with both CuN- and PPN-DBS relative to before surgery (Fig. 6). On-dopa, the APAs mediolateral CoP displacement, first step length and velocity were also significantly lower with PPN-DBS relative to before surgery; and the first step length significantly lower with Sham-DBS relative to before surgery (Fig. 6). Lastly, the first step length and velocity (On-dopa) were also significantly decreased one year after surgery with open-label PPN-DBS relative to before (Fig. 6).
Lastly, looking for differences in the clinical scores and gait parameters with exclusion of the patient with unilateral MLR-DBS (P05), we found no differences in the results obtained, except a lower step length with Sham- relative to CuN-DBS (On dopa, not shown).
Clinical scales
We found no significant differences in the MDS-UPDRS, “axial” subscore, GABS, RSGE, ABC, FOG-Q, PDQ-39, MDRS, MADRS, and BAS scores at the end of each DBS condition (Off and On-dopa conditions, Table 2), and relative to pre-surgery scores (Table 2). During the randomized period, 3/6 patients had lower scores for FOG-Q with CuN-DBS and 3/6 with sham-DBS, 3/6 patients had higher ABC-scale scores with PPN-DBS, 2/6 patients with CuN-DBS and one with sham-DBS (Fig. 7).
Changes in gait and balance disorders, quality of life, cognition and psychiatric signs in 6 PD patients with PPN, CuN, or sham deep brain stimulation
Values are mean±standard deviation. ABC, Activities Balance Confidence; BAS, Brief Anxiety Scale; FOG-Q, Freezing of Gait Questionnaire; GABS, Gait And Balance Scale; MADRS, Montgomery and Asberg Depression rating Scale; MDRS, Mattis Dementia rating; MDS-UPDRS, Movement Disorders Society-Unified Parkinson’s Disease Rating Scale; PDQ-39 SI, Parkinson’s Disease Questionnaire Summary Index; RSGE, Rating Scale for Gait and Equilibrium. *p values for the comparison between values with Sham vs. CuN-DBS vs. PPN-DBS, $p values for the comparison between values at Month 12 with PPN-DBS relative to baseline. £p < 0.05 On relative to Off-dopa condition.
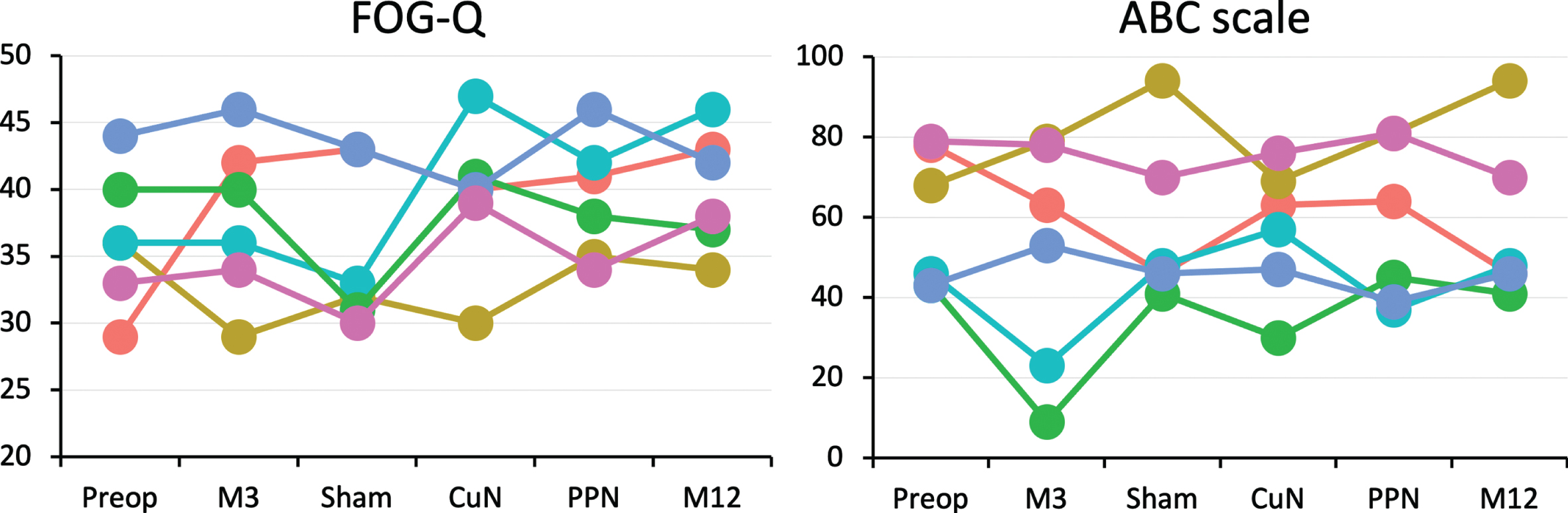
Effects of CuN, PPN and Sham DBS on freezing of gait and feelings of imbalance scores. Effects of surgery (M3), Sham, CuN and PPN-DBS during the randomized controlled period and after 3 months with PPN-DBS during the open-label period (M12) on the FOG severity and imbalance feelings. FOG-Q, freezing of gait questionnaire; ABC, Activities Balance Confidence.
Comparing clinical scales obtained Off vs. On-dopa during the randomized period only revealed a significant decrease in the motor disability score (MDS-UPDRS part 3) at baseline, after surgery without DBS (month 3), with no other significant change On vs. Off-dopa for the “axial”, GABS and Tinetti scales (Table 2).
Changes in gait parameters and clinical scales with PPN-DBS during the open-label period
At the end of the open-label follow up period (month 12) with PPN-DBS vs. before surgery, On-dopa, the first step length and velocity were significantly lower (Fig. 5), with no other significant change in gait parameters or clinical scales (Table 2). At the individual level, 3 patients (P01, P03, and P06) showed higher FOG-Q and lower ABC scale scores at month 12, relative to inclusion; with small or no change in the three remaining patients (Fig. 7). Lastly, comparing Off vs On-dopa at month 12, we found that the motor disability score was lower On vs Off-dopa with no other significant difference (Table 2).
Adverse events
All patients were ambulatory the day after surgery. Three serious adverse events were reported in 3 patients (Table 3). One was related to surgery with one electrode displacement of one hemisphere (patient P04). One patient had recurrent falls two months after surgery with the occurrence of a subdural hematoma revealed by a mental confusion that necessitated hospitalization and reduction of the apormorphine daily dosage and resolved spontaneously (P03). One patient had an abdominal wall abscess necessitating surgery 7 months after DBS surgery (P01). Twenty-seven non-serious adverse events were reported (Table 3). Falling was the most common adverse event (24/27) and reported in all patients, leading to shoulder injury in one patient (P02), and scalp or face wound in 3 patients (P02, P05, and P07). Lastly, comparing gait parameters before (inclusion) vs. post-surgery (month 3), we observed significant decreases in the step length (On-dopa Fig. 4), width and velocity (On-dopa, not shown).
Adverse events
Data are the number of adverse events, with the patients who had the adverse events in parentheses. *3 serious adverse events occurred in these 6 patients. ‡The safety committee advocated no reimplantation. $29 non-serious adverse events occurred in these 6 patients.
DISCUSSION
In this randomized, double-blind, controlled trial of 6 PD patients with severe and dopa-resistant gait and balance disorders, we assessed for the first time the effects of a 2-month period of PPN- vs. CuN- vs. sham-DBS, using a new individually-adjustable digital histological atlas [35]. We found better anteroposterior APAs, velocity and step length with CuN- vs. sham-DBS, but no significant improvement in any clinical outcome or quality of life was observed with either CuN- or PPN-DBS. We even quantified a better effect of sham-DBS on step cadence, double stance duration, FOG episodes and turn task during gait recordings. One year after surgery, we did not observe any significant improvement with PPN-DBS (open label) relative to pre-surgery levels.
These results partly reproduce those obtained in our previous controlled study of 4 patients with higher APAs and step length with MLR-DBS applied above the PMJ [29]. However, these changes did not translate into a significant clinical benefit, consistent with two controlled studies with MLR-DBS applied just above or at the level of the PMJ [28–30]. In these previous studies and other open-label studies, about half of patients described however a subjective improvement in FOG or falls that could last more than 2 years after surgery [46, 47]. In the present study, 3/6 patients also had subjective lower scores for FOG-Q with CuN-DBS suggesting that stimulation applied in the dorsal MLR may be beneficial. This is in line with the demonstrated role of the CuN in the triggering of gait initiation and high speed locomotion in animals [16, 17]. Using fMRI, the CuN-cortical-cerebellar functional network has been also shown to be more involved in dual-task walking [25], a locomotor activity altered in freezing PD patients [48]. However, 3/6 of our patients had better FOG-Q scores with sham-DBS, with fewer number of FOG episodes during forward gait. Finally, these results suggest that continuous 2-month CuN-DBS is not effective to promote clinical and daily-life benefit on FOG and/or falls for advanced PD patients.
With PPN-DBS, we found no objective improvement in any gait parameters during the randomized period, with conversely higher FOG episodes during forward gait, lower step length and turn amplitude; and significant lower step length after open label PPN-DBS. This result is not in accordance with decreased time and increased cadence during the turn task found in 7 PD patients with MLR-DBS applied below the PMJ, with however, no significant change in spatiotemporal parameters of straight-line gait [32]. The DBS electrodes in that study were also located more deeply within the pons, thus possibly modulating the activity of the descending MLR-spinal pathways, also involved in locomotion and postural controls in mammals [13]. The absence of improvement with PPN-DBS found in our patients could also be related to the differences in the patient selection, with On-dopa FOG and falls suggesting a high proportion of cholinergic cell death preventing to obtain benefit using DBS [10, 33]. Finally, one should also take into consideration that even though the surgery was well tolerated, MLR surgery has potential risks. The fact that these advanced PD patients had recurrent falls represent an additional risk factor with implanted electrodes and one of our patients indeed presented a non-surgical subdural hematoma following falls.
Differences in DBS programming may also explain the variability of MLR-DBS clinical effects, and the parameter settings used during the double-blind period could be not completely optimal. The duration of DBS with a 2-month period may not be long enough, a sustained improvement or no aggravation of FOG/falls being reported two years after surgery suggesting a possible gradual alleviation [26, 49]. In our study, 4/6 patients reported a subjective improvement in the hours following DBS parameters changes, with two patients after PPN-DBS and two after CuN-DBS (not shown). A short, 30-minute duration of PPN-DBS has been reported to induce positive effects on gait [32] and such subjective short duration effects were also previously reported [28], thus leading the proposal of intermittent or diurnal cycling DBS, [50] but without significant objective improvement. Finally, no evidence of the “good” parameters have been determined up to now [33], and we used low frequency stimulation as also proposed by others teams, with similar amplitude [51].
Limitations
Several limitations of this study need to be pointed out. Firstly, these results were obtained in a small group of patients, thus limiting the generalizability of our data. Indeed, at the request of the safety board the study was prematurely halted and only half of the patients previously plan to be included in this study were finally included, randomized, and assessed. This under-powered the study and prevented us to fully test the efficacy of MLR-DBS for these severe PD patients. However, the fact that all the patients presented a similar form of the disease, were precisely assessed using specific tools to measure gait and balance controls, and that all the electrodes were precisely positioned within both the CuN and PPN using a histological individualized atlas of the MLR, with no dramatic clinical change in gait and balance disorders at an individual level favored the robustness of these results. Secondly, the occurrence of side effects also rendered the stimulation parameter selection difficult, and the double-blind design of our study may also have prevented us from optimally setting the DBS parameters, with stimulation amplitude being potentially too low to produce significant clinical effect.
CONCLUSION
This study suggests that both PPN- and CuN-DBS are not clinically efficient to improve severe and dopa-resistant gait and balance disorders in PD patients. Further research is needed to test other parameter settings to modulate this particular anatomical area, such as intermittent stimulation and longer double-blind period of stimulation, and using different protocol designs allowing setting optimal parameters before blinded assessments or parallel group study to compare the changes in gait and balance disorders over time [52]. Finally, we need to further understand the role of the MLR in gait and balance control in humans using new imaging or neurophysiological approaches, and also its dysfunction in FOG or falls using animal models research.
Footnotes
ACKNOWLEDGMENTS
We extend our deepest thanks to the patients who participated in this study. We also thank the nurses and administrative staff of the Clinical Investigation Centre for their help in the conduct of the study. We thank Prs J.P. Azulay, S. Chabardes, S. Derrey, D. Guehl, M. Guenot, and C. Moreau for their independent contributions to the study with respect to ethical and safety issues.
This study was supported by the Institut National de la Recherche Médicale (INSERM) and the ‘Investissements d’avenir’ program (ANR-10-IAIHU-06 and ANR-11-INBS-0006) and grants from the Michael J Fox Foundation for Parkinson’s disease (grant number: 10019). JB was supported by a grant from the Fondation pour la Recherche MMédicale. YM was supported by a grant from the European Union’s Horizon 2020 research and innovation programme under the Marie Slodowska-Curie-Horizon 2020 (H2020-MSCA-IF-2019, grant agreement No 898265).
CONFLICT OF INTEREST
The authors declare to have no conflict of interest relative to the research.
