Abstract
Background:
Maintaining and manipulating sequences online is essential for language and memory. In Parkinson’s disease (PD), poor performance in sequencing tasks has been associated with basal ganglia dysfunction, especially subthalamic hyperactivity.
Objective:
This study is aimed to investigate the impact of high-frequency subthalamic nucleus (STN) deep brain stimulation (DBS) on sequence processing in PD.
Methods:
Twenty-nine patients with PD (17 women) completed a ‘before/after’ sentence task and a digit ordering task with STN DBS ON and OFF. In the sentence task, patients read a sequence of events expressed in the actual order of occurrence (‘after’ sentences) or reversed order (‘before’ sentences) for comprehension. In the digit task, patients recalled a sequence of ordered digits (ordered trials) or reordered and recalled random digits in ascending order (random trials). Volumes of tissue activated (VTAs) were estimated for the motor and associative STN.
Results:
Patients were slower with STN DBS ON versus OFF in both tasks, although their motor symptoms were significantly improved under DBS. In the sentence task, patients showed higher ordering-related reaction time costs (‘before’ > ‘after’) with DBS ON versus OFF. Moreover, patients with larger left associative VTAs, smaller total motor VTAs, and more daily exposure to dopaminergic drugs tended to show larger reaction time cost increases under DBS. In the digit ordering task, patients with too large or too small right associative VTAs tended to show larger reaction time cost increases under DBS.
Conclusion:
Stimulating the STN, especially its associative part, might impair sequence processing in language and memory.
Keywords
INTRODUCTION
The ability to maintain and manipulate sequences online is essential for language and memory. Temporal connectives such as ‘before’ and ‘after’ give us the freedom to describe a sequence of events in different linguistic orders. In sentences beginning with ‘after’, events are mentioned in their actual order of occurrence (e.g., After the scientist submitted the paper, the journal changed its policy). By contrast, in sentences beginning with ‘before’, events are expressed out of chronological order (e.g., Before the journal changed its policy, the scientist submitted the paper). ‘Before’ sentences are cognitively more challenging than ‘after’ sentences [1, 2], particularly for patients with Parkinson’s disease (PD) [3–5]. Such difficulty may arise from the additional sequence manipulation: the language system has to reverse the linguistic order of clauses to match the serial order of events in reality [3, 6].
PD patients’ poor performance has been observed in diverse sequencing tasks beyond language comprehension. PD patients have difficulties in sorting letters and numbers in a specific order [7–9], arranging scrambled pictures logically to tell a sensible story [10, 11], and organizing sequential steps to achieve goals [12–14]. PD patients’ deficits in sequencing skills have been associated with altered regional activation and functional connectivity of the subthalamic nucleus (STN) [15, 16]. This study aims to provide causal evidence for STN’s role in sequence processing.
Sequence processing in language and memory might be realized by the basal ganglia gating mechanism and prefrontal competitive queuing mechanism [16, 17]. In healthy adults, the prefrontal cortex and STN are more activated for reordering items, accompanied by a lower accuracy in recalling the item’s new position (ordering-related cost) [18, 19]. In PD, the STN is over-activated, associated with a higher ordering-related cost, regardless of the medication state [15, 16].
The STN has been associated with working memory. However, previous STN deep brain stimulation (DBS) studies showed contradictory results in PD and dystonia. Some studies found faster and more accurate responses in maintaining and updating words and locations with DBS ON versus OFF [20–22]. Some studies observed more errors and slower responses in maintaining and updating letters, voices, and locations with DBS ON versus OFF [23–25]. Other studies with similar designs reported no DBS effect on working memory performance [26–29]. The mixed findings may be caused by heterogeneity in patient population, electrode trajectory, and stimulation locus in different studies [30, 31].
Recent advances in biophysical modeling allow us to identify the stimulation locus and estimate the volume of tissue activated (VTA) relative to STN’s motor, associative, and limbic parts [32–34], potentially providing insight into cognitive and affective changes after DBS. For example, Eisenstein et al. showed that stimulating the right posterior STN reduced spatial working memory accuracy in PD [35]. Irmen et al. showed that stimulating the motor STN restored the risk-reward trade-off in PD [36].
This study investigates the impact of STN DBS on sequence processing in PD. Patients completed a ‘before/after’ sentence task and a digit ordering task with DBS ON and OFF (Fig. 1). In the sentence task, patients read two events and judged which event happened first/last. The events were expressed in the order of occurrence (‘after’ sentences) or reverse chronological order (‘before’ sentences). In the digit task, patients memorized four digits in ascending order over a short delay. The digits were presented in ascending order (ordered trials) or fully randomized (random trials). Both tasks emphasized the processing of serial order in working memory. VTA was estimated for the motor and associative STN. First, we aimed to examine whether STN DBS (ON versus OFF) improves or impairs task performance (accuracy or reaction times). Second, we sought to explore whether the DBS effect on sequence processing correlates with the associative or motor STN. This study would provide information at the subregional level beyond the capacity of regular human functional neuroimaging.
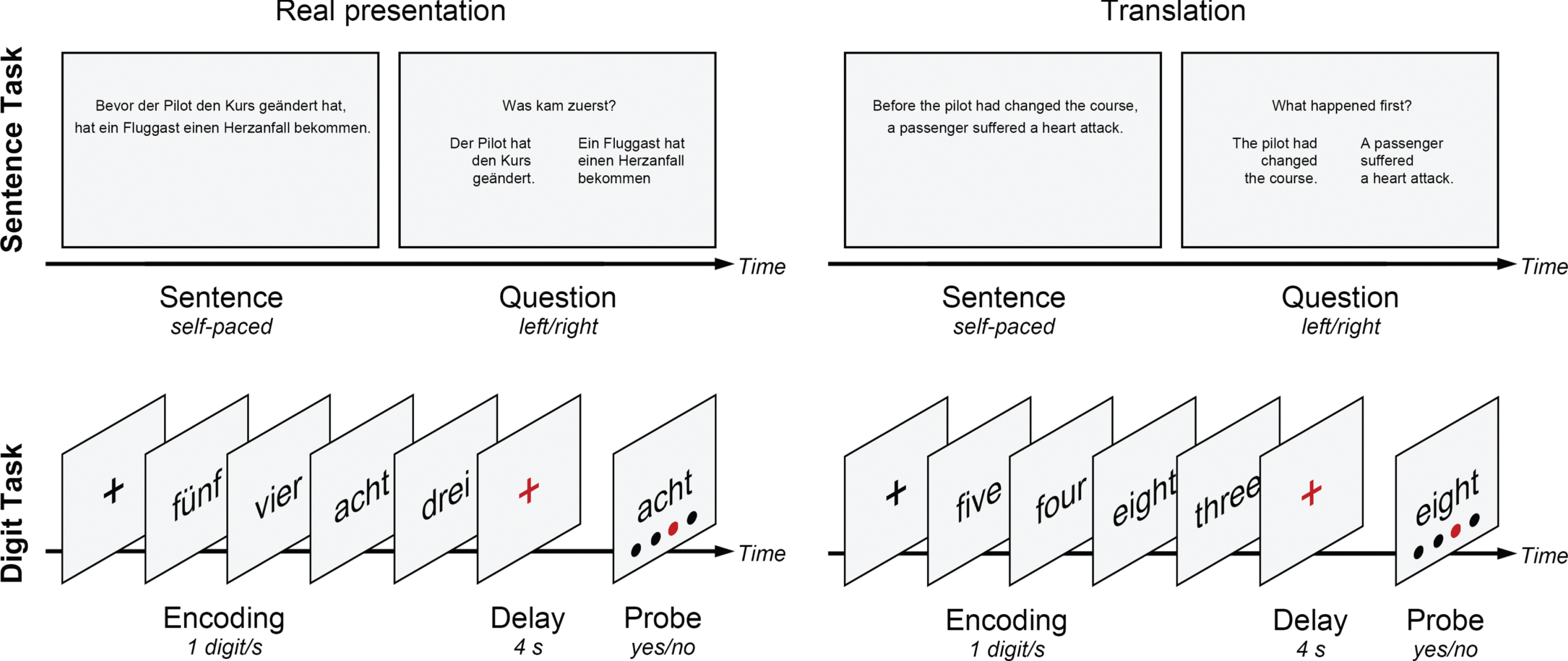
Real presentation and English translation of the ‘before/after’ sentence task and digit ordering task. In the sentence task, patients read two events expressed in the order of occurrence (‘after’ sentences) or in reverse chronological order (‘before’ sentences). They judged which event happened first or last by pressing the left/right buttons. In the digit task, patients memorized four digits in ascending order over a delay. The digits were presented in ascending order (ordered trials) or fully randomized (random trials). They judged whether a pair of digit and position matched by pressing the yes/no buttons.
MATERIALS AND METHODS
This study was approved by the ethics committee of the University of Lübeck following the Declaration of Helsinki. Each participant signed a written informed consent before participating in this study.
Patients and clinical assessment
We included 29 patients with idiopathic PD at the University of Lübeck Department of Neurology between 2018-2019. Inclusion criteria were (a) diagnosed PD according to the UK Parkinson’s Disease Society Brain Bank Clinical Diagnostic Criteria [37], (b) receiving STN DBS, (c) Hoehn and Yahr stage 1–4 in the ON state, (d) age 45–75 years, (e) education ≥11 years, (f) right-handed, (g) native German speakers. Exclusion criteria were (a) a history of other neurological or psychiatric diseases, (b) brain lesions, (c) alcohol or drug abuse, (d) possible dementia (Montreal Cognitive Assessment < 21/30). Three additional patients were excluded because of severe motor symptoms and concentration difficulty with DBS OFF.
All patients received bilateral high-frequency STN DBS (130±19 Hz) except for one patient who received left unilateral STN DBS (Table 1). All patients were assessed on their regular anti-parkinsonian drugs. The severity of motor symptoms was assessed with DBS ON and OFF using the Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) Part III subscale. Other clinical assessments were conducted with DBS ON.
Medication and stimulation parameters of individual patients
LEDD, levodopa equivalent daily dose; L, left; R, right.
Study design and procedure
Twenty-eight patients completed the ‘before/after’ sentence task (15 min), and twenty-eight patients completed the digit ordering task (14 min) in both ON and OFF sessions. The session order was counterbalanced across patients. Each session took 40 min with a wash-out interval of 30 min.
The ‘before/after’ sentence task [4, 6] included 40 German sentences beginning with ‘bevor’ (‘before’) and 40 sentences beginning with ‘nachdem’ (‘after’, Fig. 1). German is similar to English concerning the ‘before/after’ distinction. In ‘after’ sentences, events are mentioned in their actual order of occurrence. In ‘before’ sentences, events are mentioned in reverse chronological order, and a comprehender has to rearrange the events to construct a temporally coherent story. Each sentence had two clauses (seven words in the first clause and six words in the second clause). Each clause described a specific event that was not logically, causally, or referentially related to the other. A full list of ‘before/after’ sentences has been given in Ye et al. [6]. For example:
Bevor/Nachdem der Pilot den Kurs geändert hat, hat ein Fluggast einen Herzanfall bekommen. (Before/After the pilot had changed the course, a passenger suffered a heart attack.)
In each trial, participants read a sentence for comprehension and pressed a button when they finished. They then saw one of two possible questions (“What happened first?” or “What happened last?”) and the two events side by side. They selected the correct event by pressing the left/right button with the right hand. Half of the sentences were followed by “What happened first?” and the other half by “What happened last?” There was no time limit for making a response.
The digit ordering task [18] included interleaved 30 ordered trials and 32 random trials (Fig. 1). In each trial, participants memorized four different digits in ascending order over a short delay. In ordered trials, the digits were presented already in ascending order (e.g., 3-4-5-8). In random trials, the digits were fully randomized, and participants always had to reorder them (e.g., 5-3-8-4). After the delay, participants saw a pair of digit and position and judged whether the digit matched the position in the target order by pressing the Yes/No buttons with the right hand. There was no time limit for making a response.
Statistical analysis
First, as a validity check, we examined whether the MDS-UPDRS III score was lower with DBS ON versus OFF using a paired t-test (p < 0.05).
Second, we examined whether the patients performed with lower accuracy or longer reaction times with DBS ON versus OFF using repeated-measures ANOVAs (p < 0.05). The ANOVA had two factors, DBS (ON, OFF) and Trial Type (sentence task: ‘after’, ‘before’; digit task: ordered, random). We controlled the quality of behavioral data by monitoring premature (reaction time shorter than 0.1 s) and inattentive responses (reaction time longer than 2.5 standard deviations above the individual mean). The patients made only a few premature (sentence task: 0.7%, digit task: 0%) and inattentive responses (sentence task: 1.6%, digit task: 2.0%). The premature and inattentive responses were excluded from further analysis.
Third, we examined whether the DBS effect correlated with the STN VTA using the Lead-DBS toolbox following a standard pipeline [32]. The VTA analysis was conducted for 18 patients with pre- and post-operative MRIs (17 patients completed both tasks, and one patient completed the digit ordering task in both sessions). Eleven patients were excluded from the VTA analysis due to the lack of high-quality MRI. Pre- and post-operative MRIs were coregistered, normalized to the Montreal Neurological Institute (MNI) space [38], and corrected for brain shift with a coarse mask [39]. The quality of coregistration and normalization was visually monitored. Locations of ring electrodes (e.g., Medtronic 3389) were reconstructed with PaCER [40] and manually refined. VTA was estimated for the motor, associative, and limbic STN on the DISTAL Atlas in MNI space [41]. For monopolar and double monopolar stimulation (left: 11/18 electrodes, right: 11/17 electrodes), VTA was calculated with the Dembek model [33] using clinically applied stimulation parameters (e.g., current, impendence). However, most VTA models of ring electrodes do not support bipolar stimulation [34, 43]. Therefore, for bipolar estimation, the most upper contact was treated as the negative contact of the monopolar stimulation to generate an approximate measure of VTA.
As a validity check, we compared the motor, associative, and limbic VTAs (repeated-measures ANOVA, p < 0.05) and examined whether the MDS-UPDRS III score change (medication ON, DBS ON versus OFF) correlated with the total motor VTA (sum of two hemispheres) or levodopa equivalent daily dose [44]. For each task, we then examined whether the baseline reaction time change or reaction time cost change between ON and OFF sessions correlated with the associative or motor VTA, levodopa equivalent daily dose, or disease duration (step-wise regression, p < 0.05).
RESULTS
Improved motor functions with DBS ON versus OFF
Table 2 presents demographic and clinical data of the patients. The MDS-UPDRS III score was lower with DBS ON versus OFF (–67.5∼3.8%change relative to the MDS-UPDRS III score in the OFF session, paired t-test: t(28) = –7.61, p < 0.001). It confirmed the DBS’ effectiveness in treating motor symptoms in medicated patients with PD.
Demographic and clinical data of the patients (means and standard deviations)
MDS-UPDRS, Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale.
Impaired task performance with DBS ON versus OFF
Figure 2 presents behavioral data of the ‘before/after’ sentence task and digit ordering task. We examined the DBS effect on accuracy and reaction time using ANOVAs with two factors, DBS (ON, OFF) and Trial Type (sentence task: ‘after’, ‘before’; digit task: ordered, random). In the sentence task, for accuracy, a main effect of Trial Type was found (F(1, 27) = 4.90, p = 0.035,
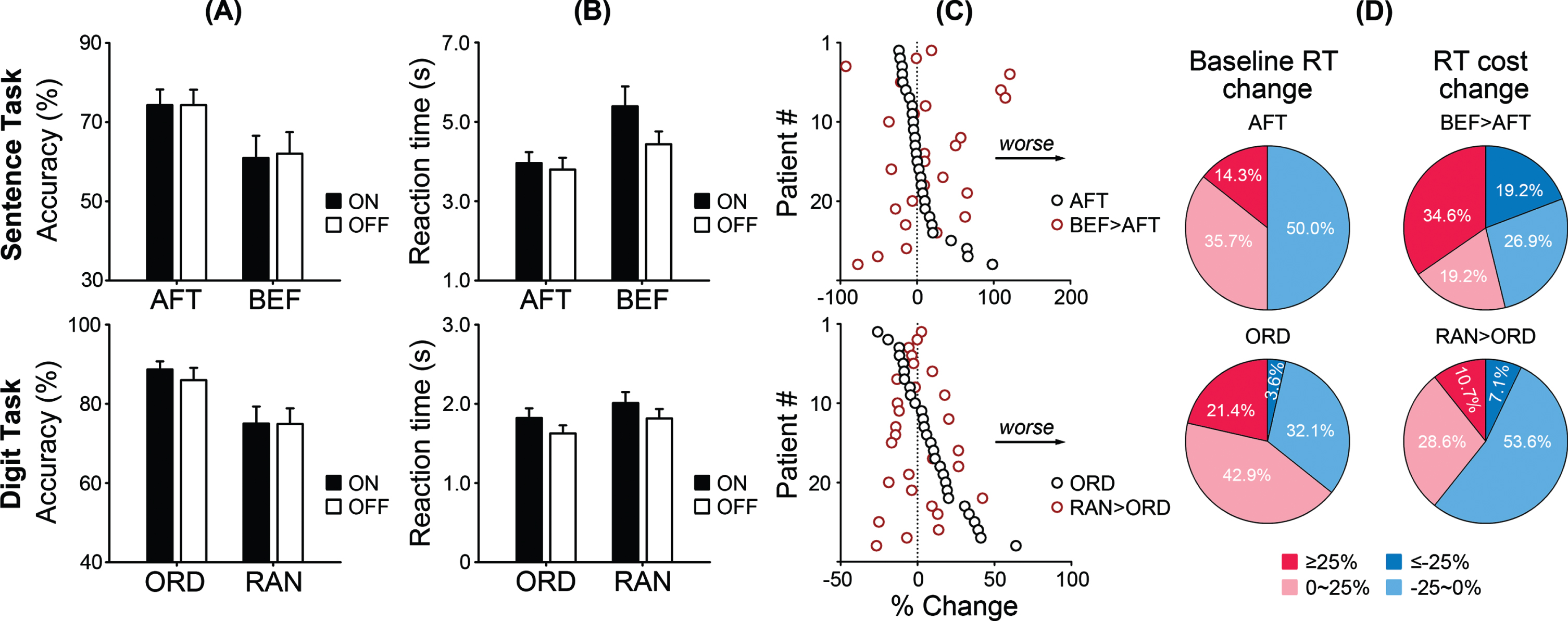
Behavioral data of the ‘before/after’ sentence task and digit ordering task. Means and standard errors of (A) the accuracy and (B) reaction times in ON and OFF sessions. AFT, ‘after’ sentences; BEF, ‘before’ sentences; ORD, ordered trials; RAN, random trials. C) Individual patients’ normalized baseline reaction time changes (AFT/ORD) and reaction time cost changes (BEF > AFT/RAN > ORD) between ON and OFF sessions. D) Distribution of the DBS effect across patients, with a benchmark of 25%.
A similar pattern was observed in the digit ordering task. For accuracy, a main effect of Trial Type was found (F(1, 27) = 11.03, p = 0.003,
Figure 2C presents individual patients’ baseline reaction time changes (‘after’/ordered) and reaction time cost changes (‘before’ > ‘after’/random > order) between ON and OFF sessions, as percentages of the corresponding baseline reaction time in the OFF session. The DBS effect varied significantly across patients. Figure 2D presents the distribution of the DBS effect across patients, using 25%as a benchmark. 10–34%of patients were significantly impaired in performance, while 3–19%were significantly improved.
DBS effects on motor symptoms correlated with the motor VTA
Figure 3A presents the motor, associative, and limbic VTAs of each hemisphere. The ANOVA revealed a main effect of Subregion (F(2, 34) = 7.02, p = 0.006,
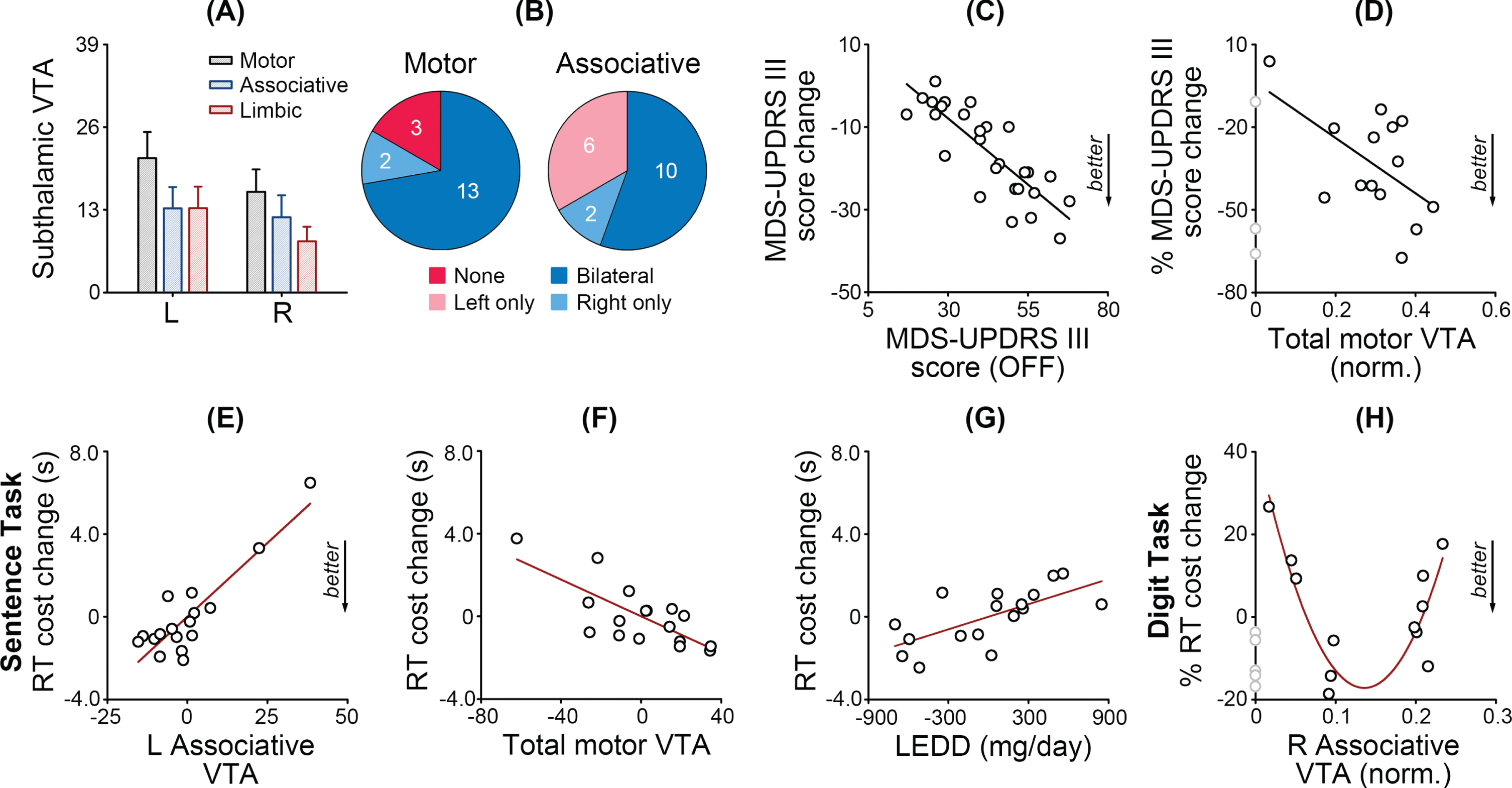
DBS effects correlated with the subthalamic volume of tissue activated (VTA). A) Mean VTAs and standard errors for the motor, associative, and limbic parts of the subthalamic nucleus. L, left, R, right. B) Numbers of patients with bilateral, unilateral, or no motor or associative VTAs. C) The MDS-UPDRS III score change between ON and OFF sessions correlated with the MDS-UPDRS III score of the OFF session. D) The percent MDS-UPDRS III score change correlated with the normalized total motor VTA. Grey dots indicate patients with no motor VTA. In the ‘before/after’ sentence task, the reaction time cost change between ON and OFF sessions correlated with (E) the left associative VTA, (F) total motor VTA, and (G) levodopa equivalent daily dose. Values are demeaned in multiple linear regression. H) In the digit ordering task, the percent reaction time cost change between ON and OFF sessions is a quadratic function of the normalized right associative VTA. Grey dots indicate patients with no right associative VTA.
Patients with more severe motor symptoms in the OFF session showed larger MDS-UPDRS III score changes between ON and OFF sessions (r = –0.83, p < 0.001, Fig. 3C). Moreover, the step-wise regression model (F(1,13) = 5.19, p = 0.042, R2 = 0.30) revealed that the percent MDS-UPDRS III score change (relative to the MDS-UPDRS III score in the OFF session) correlated with the normalized total motor VTA (relative to the total VTA, t = –2.28, p = 0.042) but not the levodopa equivalent daily dose (p = 0.539). It further confirmed the DBS’ effectiveness in treating motor symptoms.
DBS effects on reaction time costs correlated with the associative and motor VTAs
Given increased reaction time costs in the ‘before/after’ sentence task between ON and OFF sessions, we examined whether the reaction time cost change correlated with the associative or motor VTA, levodopa equivalent daily dose, or disease duration. The step-wise regression model (F(3,16) = 17.89, p < 0.001, R2 = 0.81) included the left associative VTA (t = 6.82, p < 0.001, Fig. 3E), total motor VTA (t = –4.02, p = 0.001, Fig. 3F) and levodopa equivalent daily dose (t = 3.30, p = 0.006, Fig. 3G) but removed the right associative VTA and disease duration (ps > 0.13). Patients with a larger left associative VTA, a smaller motor VTA, and a higher levodopa equivalent daily dose tended to show larger reaction time cost increases under DBS. Although there was no group-level change of the reaction time cost in the digit ordering task, the percent reaction time cost change (relative to the baseline reaction time in the OFF session) could be fitted as a quadratic function of the normalized right associative VTA (Fig. 3H, F(2,9) = 14.62, p = 0.001, R2 = 0.77, c = 43.60, β1 = –994.22, β2 = 4066.24). It suggests that an optimized right associative VTA may reduce reaction time costs. Six patients with no right associative VTA were excluded from the model.
Although the baseline reaction times increased between ON and OFF sessions in both tasks, the baseline reaction time changes did not directly correlate with the associative or motor VTA (ps > 0.28). We also explored the effect of education, general cognition, and mood status but found no significant contribution of these variables.
DISCUSSION
Previous studies have shown a lower accuracy in understanding ‘before’ sentences and sorting random numbers in medicated and unmedicated patients with PD than healthy adults [4, 15]. However, STN DBS did not improve the task accuracy. Instead, it caused a further impairment in reaction time. In the ‘before/after’ sentence task and digit ordering task, the baseline reaction times (‘after’/ordered) significantly increased with DBS ON versus OFF. In the sentence task, the ordering-related reaction time cost (‘before’ > ‘after’) tended to increase under DBS, and the increase correlated with the associative and motor VTAs and levodopa equivalent daily dose. Patients with a larger left associative VTA, a smaller motor VTA, and a higher levodopa equivalent daily dose exhibited larger reaction time cost increases under DBS. In the digit ordering task, the reaction time cost change was a quadratic function of the right associative VTA. Patients with a too large or too small right associative VTA (relative to an optimized value) exhibited larger reaction time cost increases under DBS.
Cognitive mechanisms of sequence processing
Sequence processing in language and memory might rely on the prefrontal competitive queuing mechanism and basal ganglia gating mechanism. The competitive queuing mechanism explains how the prefrontal cortex encodes and retrieves sequential items in working memory (Fig. 4) [45–49]. It comprises a parallel planning layer, which represents the serial position of items as the relative strength of node activations, and a competitive choice layer, which forms reciprocal connections with the parallel planning layer and selects the node with the strongest activation.
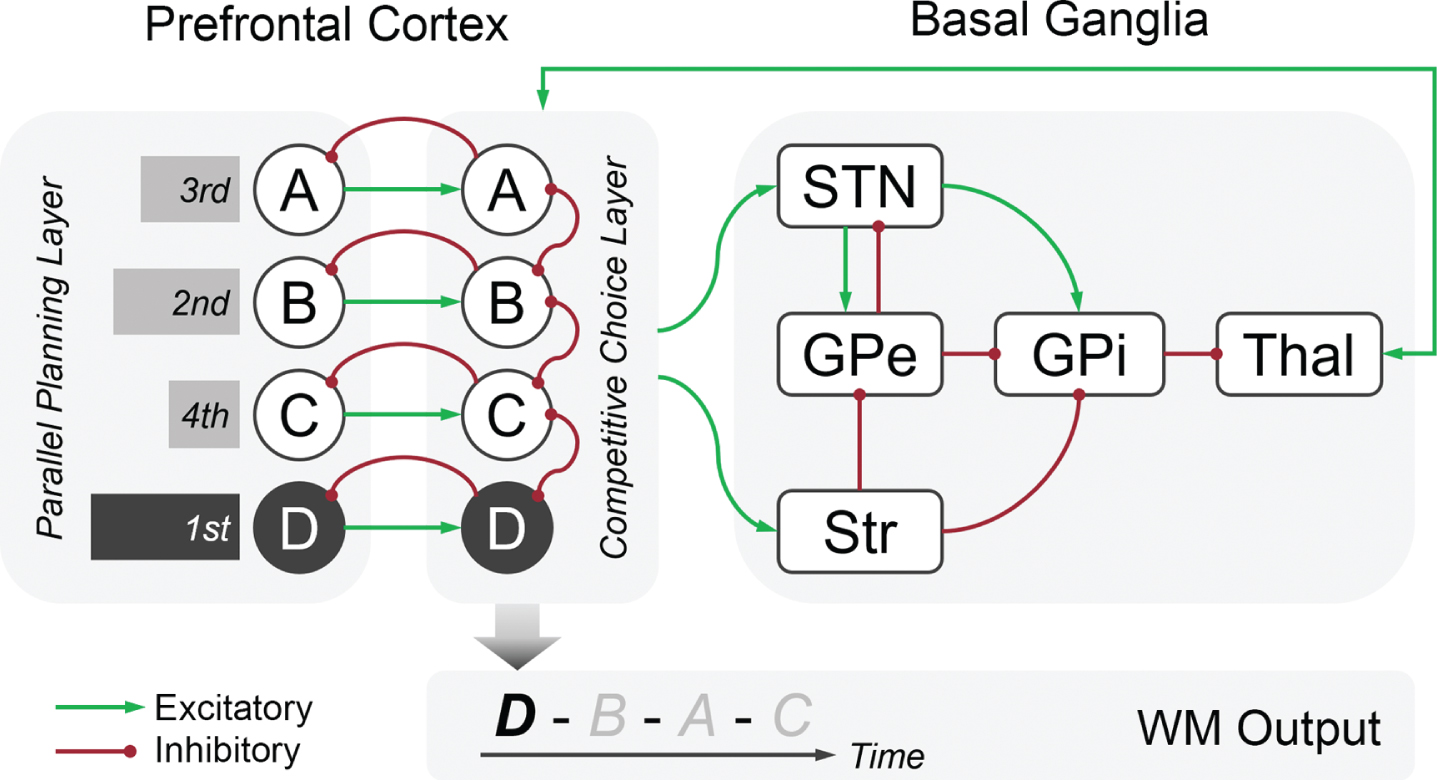
Schematic diagram of the prefrontal competitive queuing mechanism and basal ganglia gating mechanism for sequence processing. The prefrontal competitive queuing mechanism comprises a parallel planning layer and a competitive choice layer. In the parallel planning layer, the serial position of items (e.g., A, B, C, D) is represented as the relative strength of node activations (bars). In the competitive choice layer, each node receives a one-to-one input from the corresponding node in the parallel planning layer and projects back via a feedback signal. The nodes inhibit each other node in the same layer. The competitive choice layer selects the node with the strongest activation as the output (e.g., D). The basal ganglia interacts with the competitive choice layer to update the node activations in the parallel planning layer. This process may be promoted by the cortico-subthalamic hyperdirect pathway or cortico-striatal indirect pathway. STN, subthalamic nucleus; Str, striatum; GPe, globus pallidus externus; GPi, globus pallidus internus; Thal, thalamus; WM, working memory.
The gating mechanism explains how the basal ganglia balance two competing processes in working memory: robust maintenance versus dynamic updating [50, 51]. The gate is open, and incoming relevant information is processed when working memory contents need to be updated. The gate is closed, and distracting information is suppressed when maintenance demands are relatively high. The basal ganglia may interact with the competitive choice layer to update the node activation in the parallel planning layer [15] (Fig. 4). This process might be promoted by the cortico-subthalamic hyperdirect pathway or cortico-striatal indirect pathway.
The STN receives monosynaptic projections mainly from the ipsilateral cortex [52, 53]. The conduction velocity of cortico-subthalamic axons is ∼7 m/s, much faster than the transmission through cortico-striatal axons in rats (∼1.5 m/s) [54], making the STN a strong candidate for inhibiting an initiated response in hundreds of milliseconds and quickly updating multiple working memory representations. It is an open question for future research whether working memory updating shares certain neural bases with response inhibition.
DBS effects on sequence processing
STN DBS impaired sequence processing in both language and memory. The baseline reaction time reflects multiple cognitive and motor processes at the probe stage of each task. DBS may affect cognitive and motor processes common to the tasks, such as sequence retrieval, response selection, and motor programming and execution. The baseline reaction time increase most likely arose from cognitive processes (e.g., sequence retrieval, response selection) as motor functions were significantly improved under DBS.
One possibility is that STN DBS slows down information processing. Previous studies reported that six months to two years after surgery, patients receiving STN DBS showed slower information processing speeds than patients receiving the best medical therapy or globus pallidus internus DBS [55–57]. However, these studies often used working memory tests to measure information processing speed (e.g., digit span test, symbol digit modalities test, trail making test A), raising the question whether such impairment is specific to working memory. It is unclear whether an acute STN stimulation could also change information processing speed and whether the change is working memory specific or domain-general. This study cannot answer these questions because both tasks relied on working memory. Future research should study the DBS effect at the level of latent variables (e.g., what is unique or universal to multiple cognitive tasks) rather than manifest variables from individual tasks.
The reaction time cost was derived from the contrast of ‘before’ versus ‘after’ sentences in the sentence task and random versus ordered in the digit task. It is assumed to reflect the processing of serial order, regardless of the item type. Our primary finding is that the reaction time cost increase correlated with the associative STN. However, the left and right associative STN responded differently to DBS (linear versus quadratic functions).
This is analogous to the apparent difference between the right versus left STN in response inhibition. Response inhibition is assumed to rely upon a right-lateralized hyperdirect pathway connecting the right inferior frontal gyrus and pre-supplementary motor area with the right STN [58]. The right but not the left STN is more activated for suppressing inappropriate actions in healthy adults [59]. However, stimulating the left STN increased stop-signal reaction times more than stimulating the right STN [60]. On the other hand, left subthalamotomy, i.e., a targeted destructive surgical intervention, reduced stop-signal reaction times but not Go reaction times, whereas right subthalamotomy reduced Go reaction times but not stop-signal reaction times [61]. Future research is needed to address the functional difference between the left and right STN.
Interaction between DBS and dopaminergic drugs
We observed an additional impact of dopaminergic drugs on the processing of event sequences, although the medication state was controlled in this study. Most patients (26/29) received levodopa, and about 58%of patients (17/29) received dopamine D2/3 receptor agonists. Levodopa is more likely affecting the striatum rather than the STN, given the distribution of D1 and D2 receptors in primate brains [62, 63].
It is unclear whether and how STN DBS interacts with dopaminergic drugs. In 6-OHDA-induced rat models of PD, high-frequency subthalamic stimulation (130 Hz) exacerbated the levodopa effect on striatal neurons in terms of FosB/ΔFosB immunoreactivity, preprodynorphin mRNA expression, preprotachykinin mRNA expression, and GLT1 mRNA expression [64]. However, the interaction does not show up consistently in patients with PD at the behavioral level. Some studies found that STN DBS improved facial expression recognition with levodopa ON but impaired facial expression recognition with levodopa OFF [65]. Other studies found the DBS effect on decision-making without a contribution of levodopa [66].
Limitations
This study has limitations. First, this study used an atlas-based parcellation of the STN. Functional segmentation of the STN may vary from patient to patient. Second, this study treated bipolar stimulation as a first-approximation sphere. Better computational models are needed to optimize the estimation of bipolar stimulation.
CONCLUSIONS
In conclusion, STN DBS impaired sequence processing in language and memory in patients with PD. Patients were slower with DBS ON versus OFF in both ‘before/after’ sentence task and digit ordering task, even though their motor symptoms were significantly improved under DBS. In the sentence task, patients showed higher ordering-related reaction time costs with DBS ON versus OFF, particularly patients with a larger left associative VTA, a smaller motor VTA, and more daily exposure to dopaminergic drugs. In the digit task, patients with a too large or too small right associative VTA tended to show larger reaction time cost increases under DBS. Our findings suggested that the associative STN may play a role in sequence processing in language and memory.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the National Natural Science Foundation of China [31961133025 to ZY and TFM] and the German Research Foundation [MU1311/20-1 to TFM, GRK1957 to JS and NB]. ZY was supported by the Alexander von Humboldt Foundation. We would like to thank Dr. Marcus Heldmann for his assistance in data acquisition.
CONFLICT OF INTEREST
NB served as a consultant for BridgeBio and Centogene and is a member of the scientific advisory board of BridgeBio. Other authors declared no conflict of interest.
